Abstract
Background:
Although migraine is the second most prevalent form of headache, its preventive treatment has some contraindications and complications. It has been postulated that lacosamide reacts with collapsin response mediator protein 2 and prevents its phosphorylation, inhibiting calcitonin gene-related peptide release in the trigeminal system, which might have a role in migraine management.
Objective:
Our study aimed mainly to evaluate the efficacy and safety of lacosamide as an alternative medication to topiramate for the prevention of migraine, especially in patients who had contraindications to topiramate and other approved antiseizure medications used for migraine prevention.
Design:
Our study included two parallel groups: the lacosamide and the topiramate groups.
Methods:
We recruited episodic migraine patients between the ages of 18 and 65; the lacosamide group received lacosamide 50 mg once daily for 1 week, then twice daily from the 8th day till the 90th day); while the topiramate group received topiramate 50 mg once daily for 1 week, then 50 mg twice daily from the 8th day till the 90th day.
Results:
There was not a statistically significant difference between the lacosamide and topiramate in the absolute change in monthly migraine days (MMD) at 90 days with p-value 0.34, there was no significant difference between lacosamide and topiramate groups regarding the percentage of patients with ⩾50% reduction in the baseline migraine days frequency in the last 4 weeks of the treatment period with a p-value 0.11. In total, 14.0 (4.7%) patients in the lacosamide group and 24.0 (8.0%) in the topiramate group stopped treatment prematurely due to intolerance to drug-related adverse effects, hazard ratio 2.83, 95% confidence interval (1.34–4.72), p-value 0.03.
Conclusion:
In episodic migraine patients, the regular use of lacosamide 50 mg Bid for 3 months yielded reductions in the MMD, migraine days that required acute medications, and Headache Impact Test-6 score comparable to those achieved using topiramate 50 mg Bid. Lacosamide was more tolerable than topiramate in episodic migraine patients.
Trial registration:
Prospectively registered on clinicaltrials.gov, NCT06243692—January 29, 2024; https://clinicaltrials.gov/study/NCT06243692.
Plain language summary
It has been postulated that lacosamide reacts with CRMP-2 and prevents its phosphorylation, inhibiting CGRP release in the trigeminal system, which might have a role in migraine management; our study aimed mainly to evaluate the efficacy and safety of lacosamide as an alternative medication to topiramate for prevention of migraine, especially in patients who had contraindications to topiramate and other approved antiseizure medications used for migraine prevention. We recruited migraine patients between the ages of 18 and 65; the study was double-blinded randomized trial and had two groups: lacosamide group who received lacosamide 50mg once daily for one week, then twice daily from 8th day till 90th day); topiramate group who received topiramate 50 mg once daily for one week then 50 mg twice daily from 8th day till 90th day. We concluded that, in episodic migraine patients, the regular use of lacosamide 50mg Bid for three months yielded reductions in the monthly migraine days, migraine days that required acute medications, and HIT6 score comparable to those achieved using topiramate 50 mg Bid. Lacosamide was more tolerable than topiramate in episodic migraine patients.
Introduction
Migraine is a common disorder associated with recurrent attacks of headache that persist from 4 to 72 h, leading to clinical and socioeconomic dysfunctions.1,2
Although many studies demonstrated the important role of using appropriate preventive treatment in migraine patients to achieve both clinical and socioeconomic improvement, 3 many epidemiological studies showed that only around 5% of migraine patients received their preventive treatment regularly.4,5
Many randomized controlled trials (RCTs) showed that using calcitonin gene-related peptide monoclonal antibodies (CGRP-mAbs), including eptinezumab, erenumab, galcanezumab, and fremanezumab, led to a significant reduction in episodic migraine attack frequency compared to placebo without significant adverse effects.6–10
The European Headache Federation recommends using anti-CGRP therapies for migraine prevention due to their long-term efficacy and safety. 11
Due to economic and logistical difficulties, anti-CGRP medications are still not widely available in developing countries, so there is a need to evaluate other oral medications to be used as migraine preventive therapy. 12
Topiramate is widely used as migraine preventive therapy in middle- and low-income countries. Topiramate blocks glutamate’s excitatory effect. It has a sodium channel blocking effect and a carbonic anhydrase inhibiting effect. 13
Although topiramate is an approved antiseizure medication for migraine prevention, it has many contraindications and could produce many side effects. 12
Lacosamide, a third-generation antiseizure amino acid molecule with the chemical formula R-2-acetamido-N-benzyl-3-methoxypropionamide, was synthesized as an anticonvulsive drug candidate and has demonstrated antinociceptive effects in animal models, including a rat model for painful diabetic neuropathy. 14
It shows rapid absorption and almost complete oral bioavailability, and about 95% of the oral lacosamide is renally excreted. Lacosamide binds minimally (15%) to plasma proteins, limiting the drug-drug interactions in poly-medicated patients. 15
It has a novel molecular mode of action, which involves the delay or blockage of voltage-gated sodium channels (VGSCs). In contrast, the second mechanism involves interacting with collapsin response mediator protein 2 (CRMP2), preventing phosphorylation, and inhibiting CGRP release in the trigeminal system. 16 CGRP is a peptide that produces vasodilation and inflammation in leptomeningeal and extracranial vessels, causing the pulsating pain associated with migraines. 17
Our study aimed to evaluate the safety and efficacy of lacosamide as an antiseizure medication that may be a new alternative to topiramate in episodic migraine prevention.
Methods
Study design
We executed our double-blinded randomized controlled study and screened all episodic migraine patients diagnosed following The International Classification of Headache Disorders 3 (ICHD-3) 18 and sought medical advice in Kafr-Elsheikh University Hospital, Kafr-Elsheikh General Hospital, and Al Obour Hospital in the time from February 1, 2024 to July 1, 2025, we enrolled the last patient in our trial on March 10, 2025. We followed up with patients for 3 months.
We randomly assign our patients to receive either lacosamide or topiramate in a one-to-one ratio.
Participants
Our study included two parallel groups: the (A) group, which included 300 patients who received lacosamide, and the (B) group, which included 300 patients who received topiramate.
Eligibility criteria
The study recruited participants between the ages of 18 and 65 who were diagnosed with episodic migraine following ICHD-3. 18
The detailed inclusion and exclusion criteria are available in the Supplemental Material.
Interventions
All of our patients underwent randomization and routine laboratory tests, and magnetic resonance imaging of the brain.
The study had two parallel groups: the (A) group, which consisted of 300 patients who received (lacosamide 50 mg once daily for 1 week, then twice daily from the 8th day till 90th day) 19 ; the (B) group which consisted of 300 patients who received (topiramate 50 mg once daily for 1 week then 50 mg twice daily from 8th day till 90th day). 12
The detailed study interventions are available in the Supplemental Material.
Outcome assessment
Primary outcome
The absolute change in migraine days in each group’s last 4 weeks of the treatment period (migraine day was defined as a calendar day when the patient reported four continuous hours of headache meeting ICHD-3 criteria for migraine).
Secondary outcomes
Our trial involved three efficacy endpoints including; the percentage of patients who achieved ⩾50% reduction in the baseline headache days frequency in the last 4 weeks of the treatment period, 20 the absolute change in the number of migraine days that required receiving acute headache medications in the last 4 weeks of the treatment period compared to baseline, the absolute change in the Headache Impact Test-6 (HIT-6) score in the last 4 weeks of the treatment period compared to baseline.
Our trial included secondary safety and tolerability outcomes: the safety outcome was evaluated by assessing the rate of treatment-emergent adverse events in each group via regular follow-up, while the tolerability was assessed by monitoring the number of patients who prematurely discontinued treatment due to treatment-emergent adverse effects in each group.
Sample size
After using Power Analysis & Sample Size System (PASS, V12; NCSS, Kaysville, Utah, USA), we determined that a total of 560 migraine patients would provide 80% power to detect a mean difference of 1.5 migraine days per 4 weeks (primary outcome) in the lacosamide group as compared with the topiramate group, with a final two-sided significance level of 95%, alpha error of 5%, assuming the reduction in migraine days per 4 weeks 4.5 ± 0.77 migraine days in the topiramate group 21 and an overall dropout rate of 5%. The final size of our trial was 600 patients, 300 patients in each group.
Randomization and blinding
Our study was double-blinded to the investigators and the patients; an independent statistician generated a blocked randomization sequence using computer-generated random numbers with a block size of 4, and all the investigators included in the study did not know the block size. In a one-to-one ratio, participants were randomly assigned to receive either topiramate or lacosamide by a specially trained and qualified nurse. All the investigators included in the study did not know the patients’ assignments. We prepared sequentially numbered opaque Pill boxes and 600 labels for each drug, drug A or B. According to the randomization chart, we attached the number to the box. Patients were recruited sequentially and were given enrollment numbers starting from 1, which were mentioned in their files. Files carrying the same number as the patient enrollment number were then opened, and the patients were assigned to receive drugs A or B based on their randomization. Drug A included lacosamide 50 mg tablets, and Drug B included topiramate 50 mg tablets.
Statistical analysis of the data
We used the IBM SPSS software package, version 29.0 (IBM Corp., Armonk, NY, USA), to analyze our data and base all efficacy and safety analyses on the intention-to-treat principle. Both the primary and secondary outcomes underwent separate statistical analyses. Depending on their distribution, as determined by the Shapiro–Wilk test, we described numerical data as means, standard deviation, or median and interquartile range (IQR). We also reported categorical data using numbers and percentages. The Mann–Whitney U test was used to compare the irregularly distributed numerical data, while Pearson’s Chi-square was utilized to correlate categorical data. All statistical analyses were two-sided, and differences with a p-value of <0.05 were considered statistically significant. To avoid type 1 statistical error in the analysis of secondary efficacy outcomes, we used correction for multiple comparisons, and secondary efficacy outcomes differences with an adjusted p-value of <0.02 were considered statistically significant.
Survival analysis was performed using the Kaplan–Meier test and log-rank method to assess the hazard of premature treatment discontinuation due to intolerable drug-related side effects in each group. The Cox regression method obtained the hazard ratio (HR) at a 95% confidence interval (CI). Statistical significance is established when the CI does not cross the null value.
Results
Six hundred (153 males and 447 females) patients underwent randomization and were divided into two parallel groups. The lacosamide group consisted of 300 patients, and the topiramate group consisted of 300 patients; 555 patients (131 males and 424 females) completed the study during the 3-month follow-up period, as shown in Figure 1.

Study flow diagram.
There were no statistically significant differences between the two study groups regarding the baseline characteristics, as shown in Table 1.
Baseline criteria of participants.

Median (IQR).
Percentage.
HIT, Headache Impact Test-6; IQR, interquartile range; MMD, monthly migraine days.
In our study, we found that at the 90-day mark, the lacosamide group showed a 2.56 ± 1.52-day reduction in monthly migraine days (MMD) relative to baseline with a p-value of 0.001, while the topiramate group showed a 2.65 ± 1.28-day reduction in MMD relative to baseline with a p-value of 0.001. There was no statistically significant difference between the lacosamide and topiramate in the absolute change in MMD at 90 days, with a p-value of 0.34, as shown in Table 2.
Analysis of efficacy outcomes in all patients.

Median (interquartile range: IQR).
Percentage.
IT-6, Headache Impact Test-6; IQR, interquartile range; MMD, monthly migraine days.
We also found that, at the 90-day mark, there was no significant difference between lacosamide and topiramate groups regarding the percentage of patients with ⩾50% reduction in the baseline migraine day frequency in the last 4 weeks of the treatment period, with a p-value of 0.11, as shown in Table 2.
Moreover, we found that at the 90-day mark, the lacosamide group showed a 2.58 ± 1.51-point reduction in HIT-6 scores relative to baseline, with a p-value of 0.001, while the topiramate group showed a 2.78 ± 1.84 point reduction in HIT-6 scores relative to baseline, with a p-value of 0.001. There was no statistically significant difference between the lacosamide and topiramate in the absolute reduction in HIT-6 score at 90 days, with a p-value of 0.34, as shown in Table 2.
Our study found that at the 90-day mark, the lacosamide group showed a 1.76 ± 1.1 days reduction in the MMD that required acute medications relative to baseline, with a p-value of 0.002, while topiramate showed a 1.75 ± 0.91 days reduction in MMD that required acute medications relative to baseline, with a p-value of 0.001. There was no statistically significant difference between the lacosamide and topiramate in the absolute change in the MMD requiring acute medications at 90 days, with a p-value of 0.17, as shown in Table 2.
Our study showed that the topiramate group had statistically significantly higher percentages of decreased appetite and numbness compared to lacosamide, with p-values of 0.007 and 0.03, respectively, while the lacosamide group had statistically significantly higher percentages of dizziness, with a p-value of 0.03, as shown in Table 3.
Analysis of the safety of each treatment.

Percentage.
Statistically significant at p-value < 0.05.
Our trial showed that 39.0 (13.0%) in the lacosamide group and 65.0 (21.7%) in the topiramate group had drug-related side effects, HR 1.89, 95% CI (1.27–2.83), p-value 0.008, also 14.0 (4.7%) patients in the lacosamide group and 24.0 (8.0%) in the topiramate group stopped treatment prematurely due to intolerance to drug-related adverse effects, HR 2.83, 95% CI (1.34–4.72), p-value 0.03, as shown in Table 4 and Figure 2.
Analysis of the tolerability of each treatment.
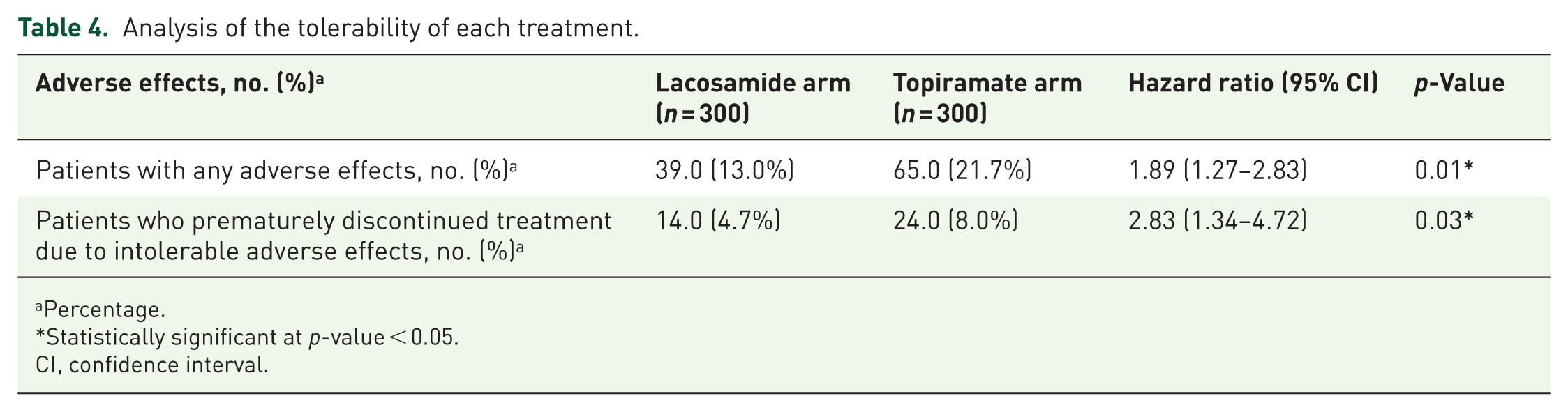
Percentage.
Statistically significant at p-value < 0.05.
CI, confidence interval.

Cumulative probability of treatment discontinuation due to drug-related side effects.
Discussion
Anti-CGRP MAbs showed significant effectiveness and tolerability, 22 compared to placebo and conventional oral preventive drugs, 23 and therefore they represent a valuable option for migraine prevention. 24 However, the use of anti-CGRP MAbs in daily clinical practice is limited due to economic, logistic difficulties, and the national reimbursement policies, which are driven by the thought that these treatment options are expensive. 25 The limitation of anti-CGRP MAbs is present not only in low-income African and Asian countries but also in European countries such as Spain, where anti-CGRP MAbs can be prescribed only after failure of three or more preventive treatments, one of them being onabotulinumtoxin A in case of chronic migraine. 25
The limited availability of anti-CGRP MAbs increases the need to assess the safety and efficacy of other new cheap oral medications that can be used as migraine preventive therapy, other than the traditional class I migraine preventives like propranolol and topiramate, which have many side effects and contraindications. 12
Lacosamide, a third-generation antiseizure amino acid molecule, is an anticonvulsive drug that has a novel molecular mode of action, which involves the delay of blockage of VGSCs, and interaction with CRMP2, preventing its phosphorylation and inhibiting CGRP release in the trigeminal system. 16
Our study aimed to evaluate the efficacy and safety of lacosamide as an alternative medication to topiramate for the prevention of migraine, especially in patients who cannot tolerate topiramate or other approved antiseizure medications for migraine prevention.
We followed up our patients for 3 months following the guidelines of the international headache society (IHS) for trials for migraine prevention which recommended the follow-up period to be at least 3 months, 26 moreover, many migraine prevention clinical trials followed up patients for 3 months such as PROMISE-1 trial where the primary efficacy analysis was based on data captured through week 12 7 and ARISE trial where the key assessments of the safety and efficacy outcomes were at week 12. 9
All of our patients had migraine for at least 1 year before enrollment in our trial following the guidelines of the IHS, 26 while the median duration of migraine in our study was form 2 to 3 years, around 25% of our patients had migraine for more than 4 years, moreover, our study involved only episodic migraine patients and it is known that older age and subsequently longer migraine duration were associated with increased risk of transformation of episodic migraine to chronic one.27,28
The patients in both groups reported a median of approximately 6–7 monthly (range 6–9) migraine days following the guidelines of the IHS which recommended including patients who had at least four migraine attacks per 4 weeks, 26 our study focused on episodic migraine patients, and many studies showed that the more frequent monthly migraine attacks, the higher the risk of chronification of episodic migraine.29,30
We used the HIT-6 score only, as the migraine disability assessment test (MIDAS) and HIT-6 scores are positively correlated. At the same time, the HIT-6 evaluated the patients’ subjective impressions concerning the effects of headaches on their daily life activities, the MIDAS, on the other hand, looked for more concrete responses of the number of days of work or school were missed in the last 3 months. The MIDAS score showed difficulty in evaluating the impact of headaches on household tasks and social affairs after work. Furthermore, a MIDAS score is influenced by the patient’s employment status. Those who are employed may get a score of 2 rather than 1 for each of questions 1 and 2, and 3 and 4, as they may have been disabled at work and home. Those who are unemployed and not attending school would only get a score of 1 for each of those questions since they cannot be disabled at work, only at home. The MIDAS may be somewhat confusing for patients. Specifically, as items #2 and #4 instruct patients not to include days that they counted in items #1 and #3, which can sometimes be overlooked by patients, yielding an exaggerated total score. 31
We found that the regular use of lacosamide for 3 months yielded significant reductions in the MMD at 90 days compared to baseline, HIT score at 90 days compared to baseline, the migraine days that required acute medications at 90 days compared to baseline, and a significant increase in patients who achieved more than 50% reduction in the MMD. Moreover, lacosamide’s efficacy results were comparable to those achieved in the topiramate group.
Lacosamide efficacy results were comparable to topiramate’s, as it shared with topiramate in blocking VGSCs and inhibiting cortical hyper-responsiveness to stimuli. Moreover, lacosamide interacted with CRMP2, inhibiting CGRP release in the trigeminal system and reducing the CGRP-mediated vasodilation and inflammation in leptomeningeal and extracranial vessels. 16
In addition, the ability of lacosamide to reduce the HIT score agreed with many trials,14,32–34 which showed that using lacosamide in patients with neuropathic pain or trigeminal neuralgia was associated with improvements in the quality of sleep, patient global impression of change, quality of life, and pain interference owing to its sodium channel blocking properties and sodium channels are known to play a major role in pain signaling pathways. 35
Our trial showed that lacosamide was more tolerable than topiramate, as significantly fewer patients prematurely discontinued treatment due to its side effects. Moreover, patients on topiramate experienced significantly higher rates of paresthesia and decreased appetite.
Our findings aligned with Yang et al., 16 Hou et al., 36 Shin et al., 37 and Luk et al. 38 who stated that lacosamide was well tolerated, especially in epileptic patients. Moreover, Brodie et al. 39 found that topiramate use significantly increases the incidence of paresthesia and weight loss compared to lacosamide use.
The patients in the lacosamide group did not experience significant cardiac adverse effects, as most of our patients aged from 18 to 30 years, received relatively low doses of oral lacosamide (50 mg Bid), and had no history of cardiac diseases; the cardiac adverse effects of lacosamide were dose dependent. The risk of cardiac adverse events is higher in elderly patients, those with a history of previous cardiac diseases, and patients who received rapid infusions of intravenous lacosamide.40,41 Moreover, many studies showed that the effects of lacosamide on cardiac rhythm and blood pressure were clinically insignificant.41,42
Study limitations and conclusions
Study limitations
Our trial had some advantages, as it was the first double-blinded RCT worldwide to compare lacosamide versus topiramate in migraine patients. However, it had some limitations: the relatively small sample; second, all the patients were Egyptian; third, the relatively short follow-up period; fourth, the study population was representative of the lower range of episodic migraine population, so we need to pursue a larger-scale, long-term, randomized trial powered for both safety and efficacy to establish the durability, validity, and generalizability of the results.
Conclusion
In episodic migraine patients, the regular use of lacosamide 50 mg Bid for 3 months yielded reductions in the MMD, migraine days that required acute medications, and HIT6 score comparable to those achieved using topiramate 50 mg Bid. Lacosamide was more tolerable than topiramate in episodic migraine patients.
Supplemental Material
sj-doc-2-tan-10.1177_17562864251396529 – Supplemental material for Lacosamide versus topiramate in episodic migraine: a randomized controlled double-blinded trial
Supplemental material, sj-doc-2-tan-10.1177_17562864251396529 for Lacosamide versus topiramate in episodic migraine: a randomized controlled double-blinded trial by Mohamed G. Zeinhom, Mohamed Fouad Elsayed Khalil, Mohamed Almoataz, Tarek Youssif Omar Youssif, Ahmed Mohamed Ali Daabis, Hossam Mohamed Refat, Ahmed Ahmed Mohamed Kamal Ebied, Shady S. Georgy, Ahmed Zaki Omar Akl, Mohamed Ismaiel, Salah Ibrahim Ahmed, Hesham Farouk Eissa, Asmaa Ibrahem Desouky Mostafa Ibrahem, Asmaa Mohammed Hassan, Mohamed Elshafei, Amir Ahmed Elsaeed Egila and Sherihan Rezk Ahmed in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-1-tan-10.1177_17562864251396529 – Supplemental material for Lacosamide versus topiramate in episodic migraine: a randomized controlled double-blinded trial
Supplemental material, sj-docx-1-tan-10.1177_17562864251396529 for Lacosamide versus topiramate in episodic migraine: a randomized controlled double-blinded trial by Mohamed G. Zeinhom, Mohamed Fouad Elsayed Khalil, Mohamed Almoataz, Tarek Youssif Omar Youssif, Ahmed Mohamed Ali Daabis, Hossam Mohamed Refat, Ahmed Ahmed Mohamed Kamal Ebied, Shady S. Georgy, Ahmed Zaki Omar Akl, Mohamed Ismaiel, Salah Ibrahim Ahmed, Hesham Farouk Eissa, Asmaa Ibrahem Desouky Mostafa Ibrahem, Asmaa Mohammed Hassan, Mohamed Elshafei, Amir Ahmed Elsaeed Egila and Sherihan Rezk Ahmed in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
