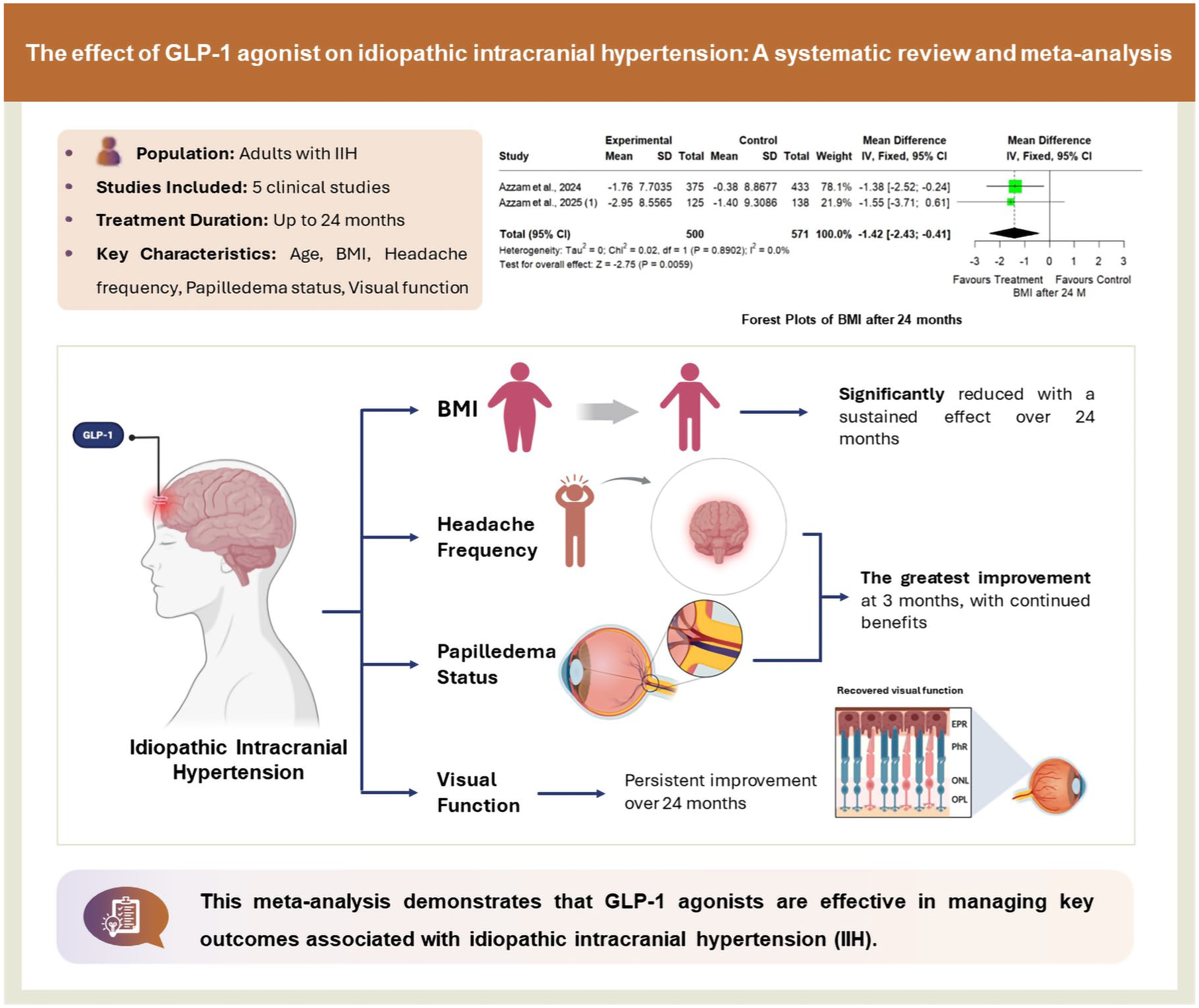
Abstract
Background:
Idiopathic intracranial hypertension (IIH) is a condition with elevated intracranial pressure, leading to headaches and vision issues. Current treatments offer limited relief. Glucagon-like peptide-1 (GLP-1) agonists, known for their established metabolic benefits including weight loss and an emerging potential to lower intracranial pressure, may provide a novel approach for managing IIH.
Objectives:
This systematic review and meta-analysis aimed to assess the effectiveness of GLP-1 agonists in improving IIH symptoms.
Design:
A systematic review and meta-analysis.
Data sources and methods:
A systematic search of PubMed, Scopus, Web of Science, and Cochrane was conducted until March 2025. Dichotomous outcomes were pooled using risk ratio (RR), while continuous outcomes were pooled using standardized mean difference. PROSPERO ID: CRD420251008614.
Results:
The meta-analysis of four studies assessed the efficacy of GLP-1 agonists across various outcomes. Results showed a significant reduction in Body Mass Index (BMI) over 24 months, with mean differences of −0.36, −1.08, −1.18, and −1.42 at 3, 6, 12, and 24 months, respectively, with 95% CI, and low heterogeneity (I² = 0.0%). GLP-1 agonists also reduced headache risk, with the most significant effect at 3 months (RR: 0.69), remaining statistically significant at 6, 12, and 24 months. The risk of papilledema was reduced across all time points, with the lowest RR at 3 months (RR: 0.69 (95% CI: 0.55, 0.86)), and the effect remained significant through 24 months. Additionally, GLP-1 agonists consistently reduced the risk of visual disturbances and refractory IIH, with statistically significant effects sustained up to 24 months, demonstrating a sustained benefit throughout the treatment period.
Conclusion:
This meta-analysis demonstrates that GLP-1 receptor agonists are a promising therapeutic option for patients with IIH, demonstrating significant efficacy in reducing intracranial pressure-related symptoms such as BMI, headache frequency, papilledema, and visual disturbances.
Plain language summary
This research reviewed several studies on using GLP-1 agonists, a class of drugs for weight loss and diabetes, to treat Idiopathic Intracranial Hypertension (IIH). IIH is a condition of high pressure in the skull that causes severe headaches and vision problems. The combined results showed that patients treated with GLP-1 agonists experienced significant benefits, including: Weight loss Fewer headaches Improved vision and reduced swelling of the optic nerve (papilledema) A lower need for surgery for treatment-resistant IIH The study concludes that GLP-1 agonists are a highly promising treatment for IIH. However, larger clinical trials are still needed to confirm these positive results.
Introduction
Idiopathic intracranial hypertension (IIH) is a neurological disorder characterized by elevated intracranial pressure (ICP) without an identifiable cause, mostly affecting obese women of childbearing age. 1 The clinical picture includes symptoms such as headache, papilledema, visual disturbances, and, in severe cases, irreversible vision loss. The etiology of IIH remains poorly understood, but it is mostly connected to metabolic imbalance and obesity. 2 Management flow charts mainly depend on weight loss and acetazolamide therapy. Refractory cases to the previously mentioned approaches would need surgical intervention, that is, cerebrospinal fluid (CSF) shunting or optic nerve sheath fenestration might be the answer. 3 Originally developed to treat type 2 diabetes and obesity, glucagon-like peptide (GLP)-1 receptor agonists have recently gained attention for their potential role in managing IIH. While their metabolic benefits are well known, their potential to directly influence intracranial pressure may be even more significant. These drugs simulate the GLP-1 incretin hormone released after eating, it stimulates insulin secretion, reduces glucagon levels, slows gastric emptying, and increases satiety ultimately improving blood sugar control and aiding weight loss. 4 The available approved agonists include tirzepatide, exenatide, liraglutide, dulaglutide, lixisenatide, and semaglutide. 5 GLP-1 RAs come in different doses with equally variable lasting effects and molecular structures, making it possible to formulate tailored treatment regimens based on patient needs and preferences. 6 Beyond their metabolic effects, recent studies suggest that GLP-1 RAs may have central nervous system activity and anti-inflammatory properties, making them a potential therapeutic option for conditions like IIH. 7
The direct effects on the central nervous system and the weight loss contribution to ICP reduction were a pleasant finding in a randomized, placebo-controlled, double-blind trial in which IIH patients in the exenatide group experienced a significant reduction in ICP both immediately and after 12 weeks of follow-up, along with nearly eight fewer headache days across the 12 weeks. 8 In another GLP-1 RA trial, results were associated with a similar effect on weight loss and ICP in IIH patients, indicating a tolerable and noninvasive alternative to older treatments. 9 Despite the promising preliminary results, significant research gaps remain regarding the use of GLP-1 RAs in IIH treatment. Most available data are derived from studies with small sample sizes and limited follow-up periods. Additionally, there is a lack of randomized controlled trials comparing GLP-1 RAs to standard IIH treatments such as acetazolamide or surgical interventions. Further research is needed to determine their long-term efficacy, safety, and potential role as a first-line treatment.8–10
This systematic review and meta-analysis aims to assess the efficacy of GLP-1 RAs in patients with IIH, by evaluating their impact on intracranial pressure, body mass index (BMI), headache burden, papilledema, and visual disturbances, compared to placebo or standard care.
Methodology
Protocol registration
This systematic review and meta-analysis followed the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) statement 11 and the Cochrane Handbook for Systematic Reviews and Meta-Analyses guidelines. 12 This review’s protocol has been published and registered in PROSPERO under the ID (CRD420251008614).
Data sources and search strategy
The Cochrane Central Register of Controlled Trials (CENTRAL), PubMed (MEDLINE), Web of Science, SCOPUS databases were all searched until March 2025. The results of each data base’s search terms and keywords are shown in Table S1.
Eligibility criteria
We used the Population, Intervention, Comparison, and Outcomes (PICO) criteria to select eligible randomized controlled trials and observational studies: population (patients diagnosed with idiopathic intracranial hypertension, aged ⩾ 18 years); intervention (Glucagon Like Peptide 1 RAs including: (Semaglutide, liraglutide, exenatide, or tirzepatide), alongside standard therapy); comparison (standard therapy); and outcomes: the primary outcome was the BMI reduction, mean days with headache, papilledema, visual disturbances and refractory or nonresponsive IIH.
Study selection
We conducted the review via the Rayyan online tool. After eliminating duplicates, two authors evaluated each record they retrieved separately. The same two authors reviewed the complete texts of the records for the first full-text screening for eligibility criteria. All differences were settled by consensus after consulting.
Data extraction
The baseline characteristics and outcomes data were extracted using a Microsoft Excel extraction sheet by two authors, and a senior author settled disagreements. These data were arranged as follows: (1) study characteristics, such as study ID, study design, country, population, main inclusion criteria, intervention and control measures (name, number in the group, Route of administration, and regimen), and primary endpoints; (2) baseline patient characteristics, including the number of patients in each group, age, sex, BMI, Race/Ethnicity, comorbidities, Frisén Grade (worst eye), disease duration, monthly headache days, ICP; and (3) the previously mentioned outcome measures.
Risk of bias
The risk of bias in the included randomized controlled trials (RCTs) was assessed using the revised Cochrane risk-of-bias tool (RoB2). 13 This tool evaluates five key domains: (1) randomization process, (2) deviations from intended interventions, (3) missing outcome data, (4) measurement of outcomes, and (5) selection of reported results. Each domain was rated as “low risk,” “high risk,” or “some concerns” for bias.
For cohort and case–control designs, study quality was assessed using the Newcastle-Ottawa Quality Assessment Scale (NOS). The NOS evaluates studies based on three criteria: (1) selection of study groups (representativeness, ascertainment of exposure, and demonstration that the outcome was not present at baseline), (2) comparability of groups (control for confounding factors), and (3) assessment of outcome (adequacy of follow-up and blinding of outcome assessment). Studies were awarded stars for each criterion, with a maximum of nine stars indicating the highest quality. Each domain was rated as “good quality,” “poor quality,” or “fair quality” for bias.
Two independent assessors evaluated study quality based on their respective designs. Discrepancies were resolved through discussion and consensus among the investigators.
Data synthesis and analysis
The main outcomes analyzed were the pooled mean differences in BMI reduction from baseline to posttreatment, and risk ratio (RR) for headache, visual disturbances, refractory IIH and papilledema between the GLP agonist and control groups. We calculated the log risk ratio (LogRR) and its corresponding standard error (SE) based on the reported risk ratios and 95% confidence intervals. The pooled effect estimates were expressed as RR with 95% confidence intervals (CIs) by exponentiating the weighted mean LogRR. To assess heterogeneity, Chi-square and I2 statistics were utilized, with significant heterogeneity defined as a p-value below 0.1 and an I2 exceeding 50%. The random-effects model, based on the DerSimonian and Laird methods, was chosen under the assumption that the true effect size may vary across studies due to clinical and methodological differences. Inverse variance method was used. Jackson method for confidence interval of tau^2 and tau. Calculation of I^2 based on Q.
None of the included studies provided the standard deviation of change scores (SD Change) for the analyzed outcomes. To address this, a correlation coefficient (r) of 0.5 was used to estimate the mean and SD change in BMI reduction outcome. Additionally, a sensitivity analysis was conducted using different correlation coefficients to evaluate the stability of the imputed values for such outcome (sensitivity analysis of (r) is provided in Table S6). For studies that reported medians and interquartile ranges (IQR) for changes from baseline, these values were converted into means and standard deviations using the formula proposed by Wan et al. 14 The converted values were then incorporated into the analysis alongside the imputed standard deviations. All formulas used for imputing the missing values were provided in the Cochrane Handbook for Systematic Reviews of Intervention (Version 6.5, Section 6.5.2.8). 12 All analyses were performed using R (version 4.3.3) for windows 10.
Results
Search results and study selection
By searching databases, we retrieved 67 records, and 18 references were excluded by endnote, leaving 53 references for primary screening by title and abstract. After screening by title and abstract, 16 articles were available to be assessed in full-text screening. Finally, we included six studies with 2133 patients in this systematic review and meta-analysis. The PRISMA flow chart of the selection process is shown in Figure 1.
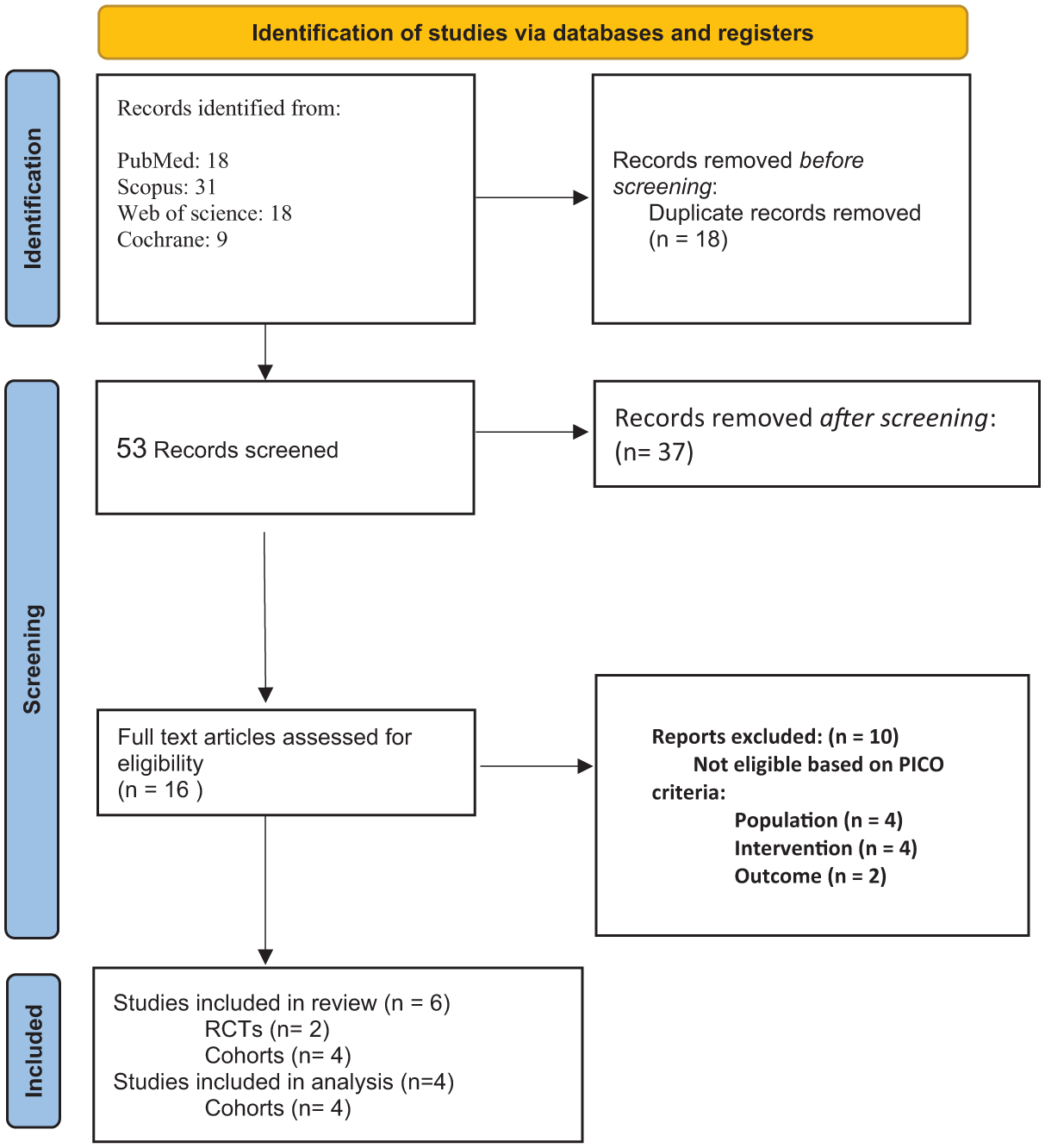
PRISMA flow chart of the screening process.
Characteristics of included studies
We included a RCT and the post hoc analysis of it, three cohorts and a case–control pilot study.8–10,15–17 Two studies were conducted in the United Kingdom, one in Austria, and three were conducted in multiple countries, and the USA was the predominant source of records.
All included studies investigated subcutaneously administered GLP-1 agonists. The intervention protocols differed by study design; the randomized trial investigated exenatide against a placebo control,8–10 while the cohort and case–control studies compared either semaglutide, liraglutide, or tirzepatide against usual care management.9,15–17 This usual care control included standard medications such as acetazolamide, topiramate, and diuretics.
Regarding the specific dosing regimens, exenatide was administered with a single 20 μg loading dose followed by fixed twice-daily injections of 10 μg for 12 weeks.8,10 Among the other drugs, liraglutide was administered daily, initiated at 0.6 mg and escalated to a target of 1.2–3.0 mg per day.9,16 The weekly dosed agonists included semaglutide, which started at 0.25 mg and titrated up to 2.0–2.4 mg, and tirzepatide, which began at 2.5 mg and escalated up to 15 mg weekly.9,15,17 Further details are highlighted in (Table 1) about the included characteristics and in (Table S9) about the included patients’ baseline data.
Summary characteristics of the included studies.

Analysis of cohorts
Our meta-analysis included four studies.9,15–17 The primary outcomes assessed were BMI, headache, papilledema, visual disturbances, and refractory idiopathic IIH between GLP-1 group and control. These outcomes were evaluated over multiple follow-up periods at 3, 6, 12, and 24 months to track changes and treatment effectiveness over time.
Regarding BMI, our results showed that GLP-1 agonists had a progressive impact on BMI reduction over time, with statistically significant effects emerging at 6 months and persisting up to 24 months, at 3 months, the mean difference (MD) was −0.36 (95% CI: −1.22, 0.50) with 0.0% heterogeneity (I2) and a p-value of 0.4102. At 6 months, the MD was −1.08 (95% CI: −1.81, −0.35) with 0.0% heterogeneity (I2) and a p-value of 0.0038. At 12 months, the MD was −1.18 (95% CI: −2.20, −0.16) with 0.0% heterogeneity (I2) and a p-value of 0.024. Finally, at 24 months, the MD was −1.42 (95% CI: −2.43, −0.41) with 54% heterogeneity (I2) and a p-value of 0.002. The consistency across studies, as evidenced by low heterogeneity (I2 = 0.0%), supports the reliability of these findings. While initial effects at 3 months were minimal, sustained reductions at later time points suggest a potentially durable effect on BMI (Figure 2).

Forest plots illustrating the reduced risk of headache with GLP-1 agonist compared to usual care group at 3, 6, 12, and 24 months.
For the risk of headache, we found that GLP-1 agonists consistently reduce the risk of headaches over time, with the strongest effect observed at 3 months (RR: 0.69 (95% CI: 0.55, 0.86); p = 0.0011). While the effect size decreases slightly at later time points including 6, 12, 24 months (RR: 0.78 (95% CI: 0.64, 0.95; p = 0.0113), RR: 0.81 (95% CI: 0.68, 0.95; p = 0.0121), RR: 0.80 (95% CI: 0.69, 0.92; p = 0.0023), respectively), the reduction remains statistically significant. However, we noticed moderate-to-high heterogeneity, peaking at 12 months (I2 = 77.1%), mostly due to the evident sample size difference in Azzam et al., 17 and other variabilities between studies (Figure 3).
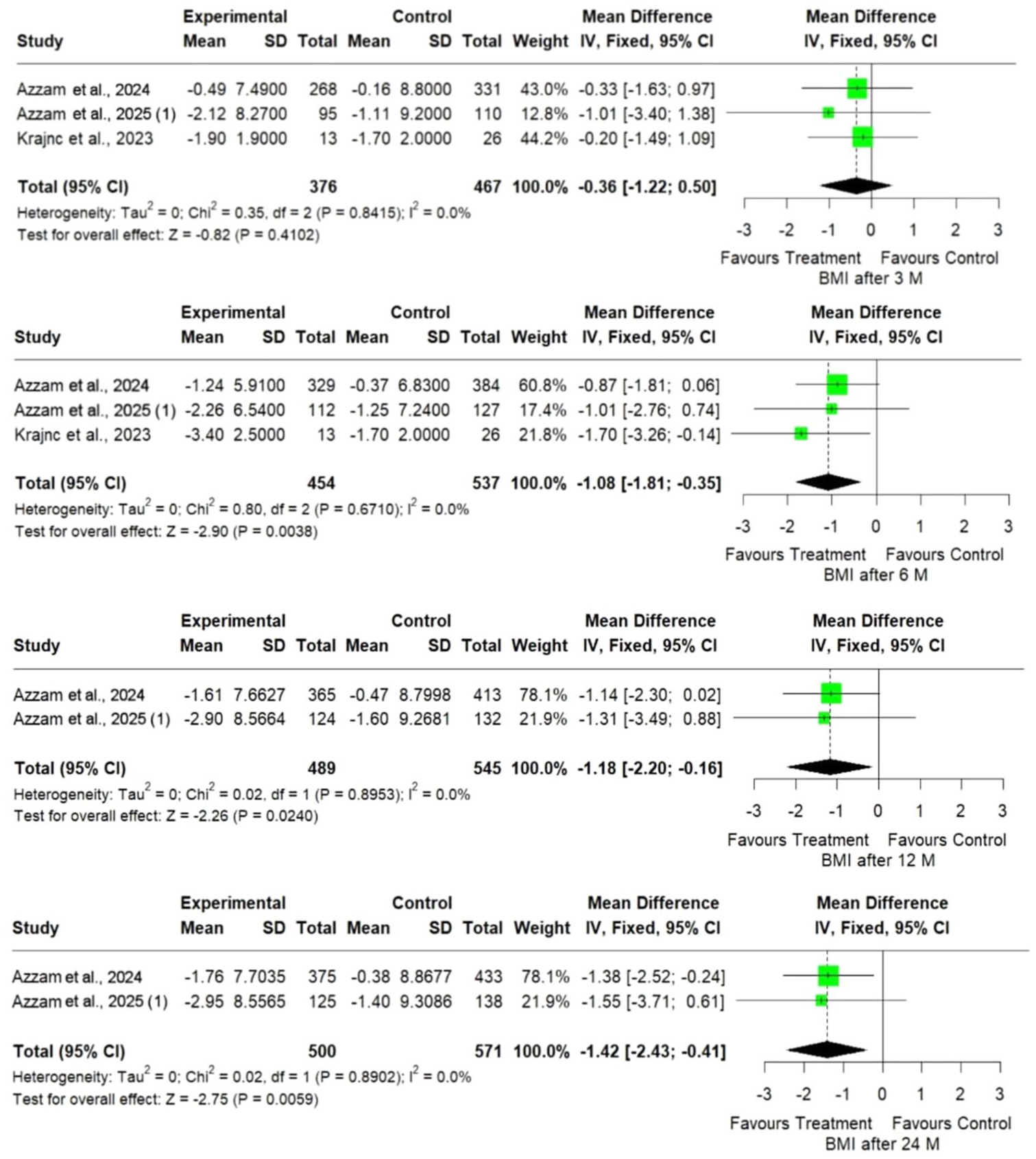
Forest plot illustrating the mean difference in between the GLP-1 agonist group compared to usual care group at 3, 6, 12, and 24 months.
Likewise, the analysis of papilledema revealed that the overall risk ratios consistently indicate a positive impact of GLP-1 agonists at all time points. The RR is lowest at 3 months 0.43 (95% CI: 0.28, 0.67; p = 0.0002), and there’s a slight increase in subsequent months (6 months: 0.47 (95% CI: 0.34, 0.64; p = 0.0001), 12 months: 0.47 (95% CI: 0.38, 0.58; p = 0.0001), and 24 months: 0.46 (95% CI: 0.36, 0.59; p = 0.0001)). Yet, heterogeneity (I2) decreases significantly from 66.1% at 3 months to 0.0% at 12 months, meaning more consistency in study results over time. Furthermore, the overall effect remains statistically significant (p < 0.0001) after 6 months, strengthening the reliability of findings (Figure 4).
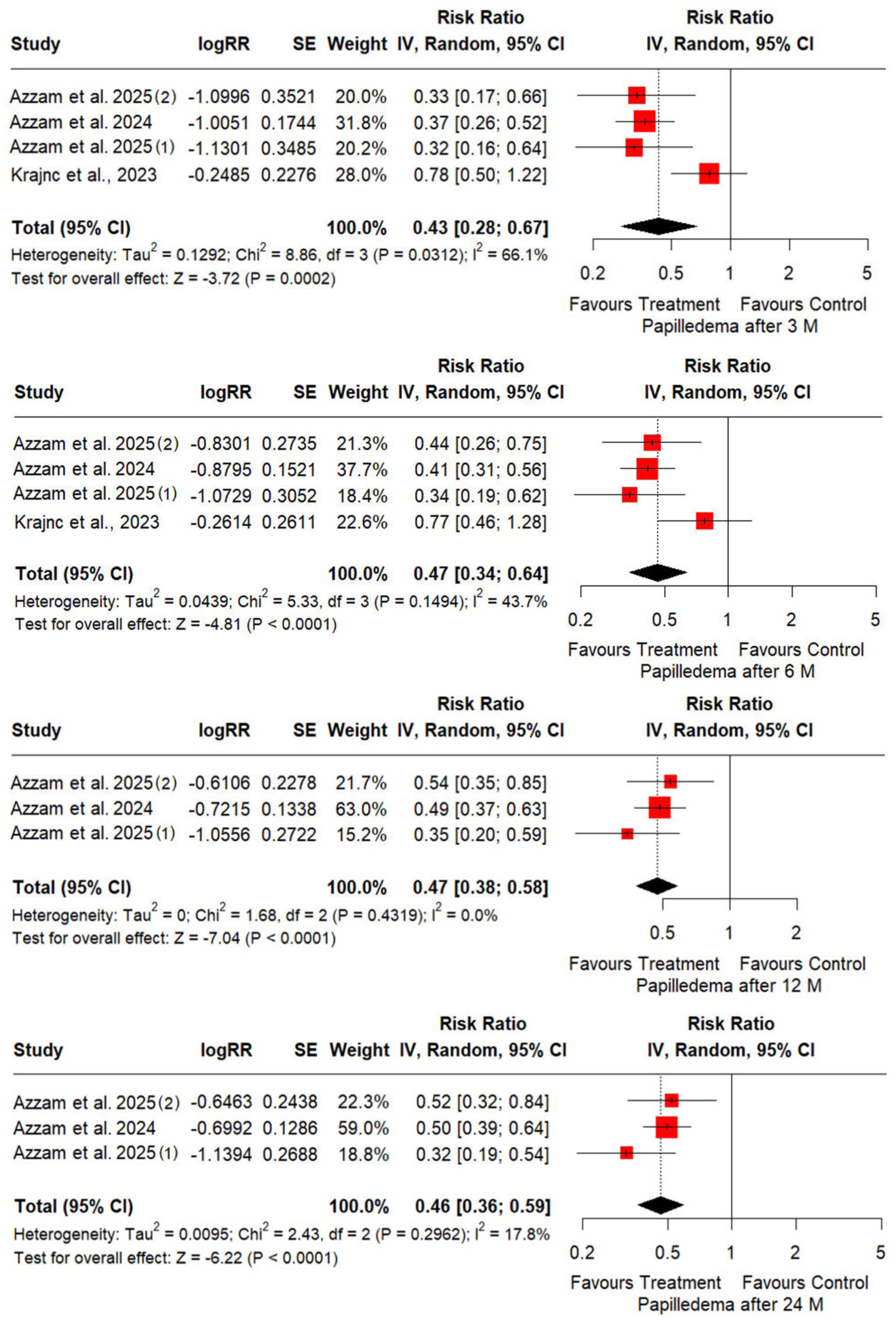
Forest plots illustrating the reduced risk of papilledema with GLP-1 agonist compared to usual care group at 3, 6, 12, and 24 months.
Similarly, across all time points, the RR constantly remains below 1, indicating a sustained risk reduction in visual disturbances associated with GLP-1 agonist use. The effect appears stable over time (RR ~0.42–0.50), suggesting sustained benefit of GLP agonists in reducing visual disturbances. Nevertheless, heterogeneity (I2) varies, with moderate values at 3 months (51.9%) and 12 months (66.4%) but high at 6 months (77.9%) and 24 months (78.9%), that can be attributed to having liraglutide as an intervention in combination with the presence of some other variabilities (Figure 5).
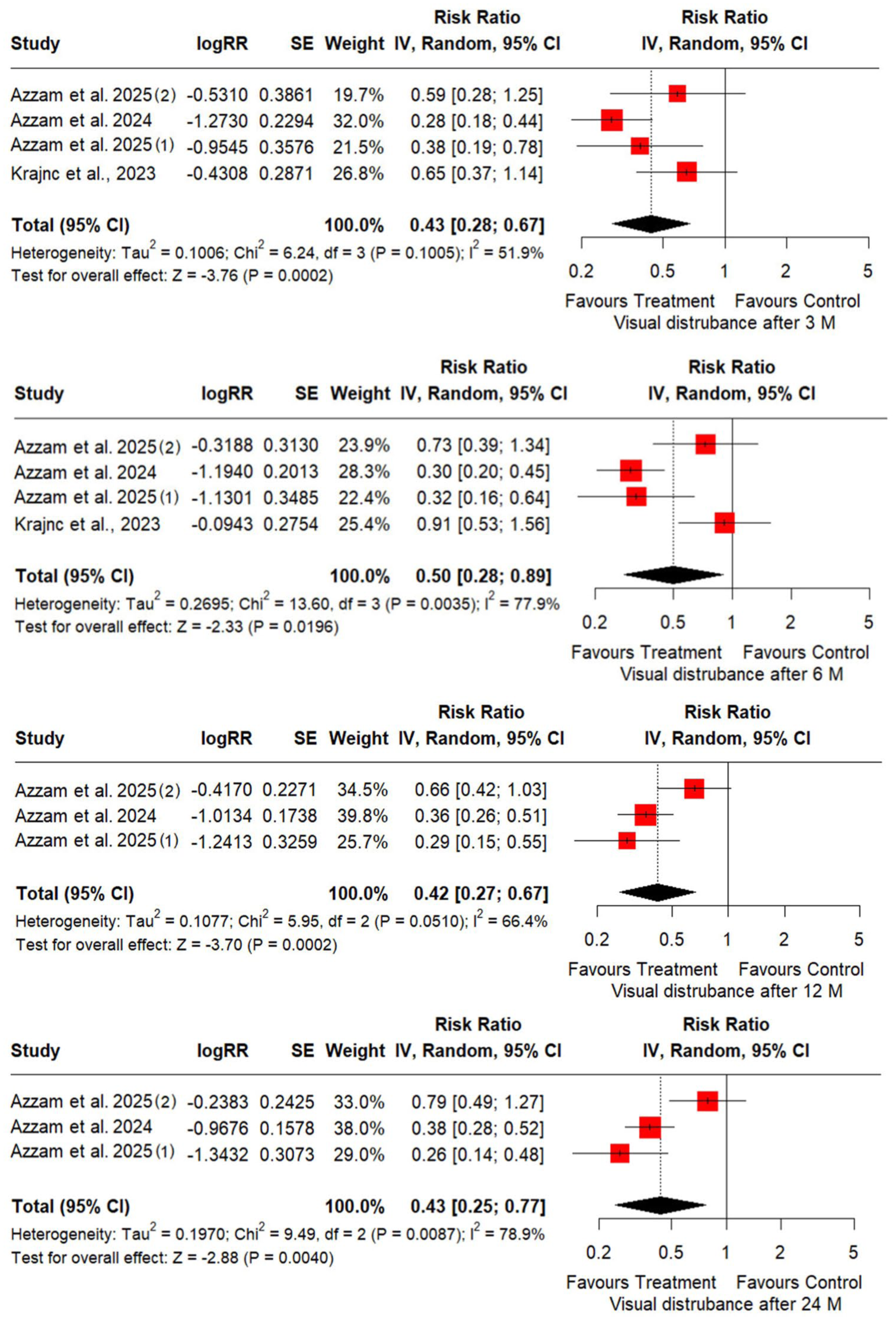
Forest plots illustrating the reduced risk of visual disturbances with GLP-1 agonist compared to usual care group at 3, 6, 12, and 24 months.
Finally, the pooled RR estimates at different time points suggest a progressive decrease in the risk of refractory IIH in the GLP-1 agonists group compared to the control group. At 3, 6, 12, 24 months, pooled RR was 0.69 (95% CI: 0.58–0.81, p < 0.0001), 0.77 (95% CI: 0.66–0.90, p = 0.0013), 0.84 (95% CI: 0.74–0.93, p = 0.0023), 0.81 (95% CI: 0.72–0.91, p = 0.0006) respectively. The findings suggest that the treatment significantly reduces the risk of refractory IIH, particularly in the short term (3–6 months), with a gradual decline in effect over time. The overall effect remains statistically significant for up to 24 months (Figure 6).
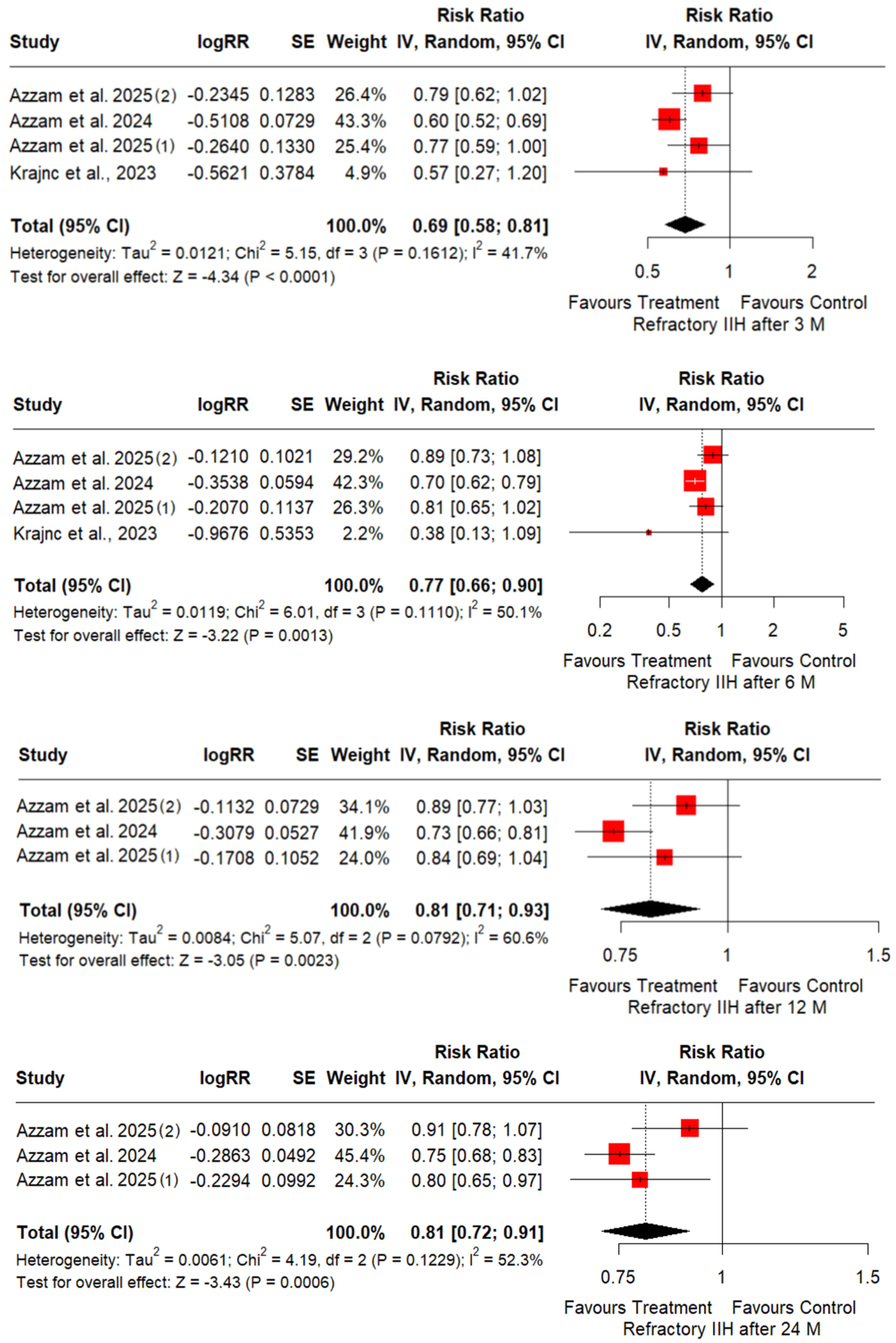
Forest plots illustrating the reduced risk of refractory IIH with GLP-1 agonist compared to usual care group at 3, 6, 12, and 24 months.
The safety outcome revealed that treatment with GLP-1 RAs was well tolerated in patients with idiopathic IIH, with no severe adverse events (AEs) or treatment discontinuations reported. Among the 13 participants, 69.2% experienced AEs, predominantly gastrointestinal in nature, with nausea being the most frequent (69.2%, of which 61.5% were moderate in severity). Other common AEs included decreased appetite (15.4%) and mild increases in lipase (23.1%, all < 1.5× ULN). Laboratory abnormalities were infrequent and mild, including isolated cases of elevated alanine aminotransferase (7.7%, <3.0× ULN) and mild hypoglycemia (7.7%, 55–70 mg/dL). The absence of severe AEs and the manageable nature of moderate AEs, particularly nausea, support the safety of GLP-1 RAs as a viable therapeutic option for weight management in this patient population. These findings align with the established safety profile of GLP-1 RAs in other indications, further reinforcing their potential utility in IIH management.
Results of clinical trials
Safety outcomes
Mitchell et al. 8 demonstrated that exenatide was safe and well-tolerated in patients with IIH, with no treatment withdrawals due to adverse effects. Twelve AEs were reported, with eight occurring in the exenatide arm (seven cases of nausea, all related to treatment initiation) and four in the placebo arm (including one unrelated serious AE, thyrotoxicosis). Nausea, a known class effect of GLP-1 RAs, was transient and resolved within the first week, likely attributable to the pharmacokinetic profile of twice-daily exenatide dosing. Notably, all participants remained compliant with treatment. 8
Efficacy outcomes
The efficacy of exenatide versus placebo was assessed by Grech et al. 10 and Mitchell et al., 8 evaluating improvements in intracranial pressure, headache frequency and severity, the need of analgesia, logMAR visual acuity, BMI, and cognitive function.
A significant reduction in the number of monthly headache days was observed among patients treated with exenatide, with a mean decrease of 7.7 days from baseline to 12 weeks. In contrast, patients in the placebo group exhibited no significant change, with a reduction of only 1.5 days. 8
Headache severity, measured by the Verbal Rating Scale (VRS 0–10), showed no significant differences between the exenatide and placebo arms at baseline (chi-squared p = 0.133) or at 12 weeks (chi-squared p = 0.084). At baseline, moderate headaches were more common in the exenatide group (86% vs 62%), while mild headaches were only reported in the placebo group (38% vs 0%). Severe headaches were present in 14% of the exenatide group at both time points, compared to 0% at baseline and 13% at 12 weeks in the placebo group. 8
Both treatment groups showed significant cognitive improvements:
Monthly analgesic use showed no significant changes within or between treatment groups. In the exenatide arm, frequency decreased slightly from 7.9 (4.5) to 7.0 (5.7) doses/month (mean difference −0.9 (5.2), p = 0.680). Conversely, the placebo group showed an increase from 3.4 (2.8) to 5.9 (5.2) doses/month (mean difference 2.4 (5.1), p = 0.254). The between-group difference in change from baseline was not statistically significant (1.1 (2.5), p = 0.648). 8
The exenatide arm demonstrated a statistically significant improvement in visual acuity compared to placebo (−0.1 (0.05) logMAR units, p = 0.036). At 12 weeks, this improvement corresponded to a clinically meaningful five-letter gain, equivalent to one line on the visual acuity chart. Previous studies have established that a five-letter change has >90% probability of representing a true clinical effect. 8
At baseline, mean BMI was 38.6 (4.7) kg/m2 in the placebo arm and 37.6 (7.9) kg/m2 in the exenatide arm. At 12 weeks, BMI remained stable in both groups (placebo: 38.1 (4.9) kg/m2; exenatide: 37.5 (7.4) kg/m2), with no significant between-group difference observed (p = 0.854). 8
Risk of bias assessment
The risk of bias in the included clinical trials was assessed using the revised Cochrane risk-of-bias tool for randomized trials (RoB2). 13 This tool evaluates key domains such as randomization process, deviation from intended intervention, missing outcome data, measurement of outcome, and selection of the reported results. The results of the risk of bias assessment are summarized in Figure 7. The risk of bias assessment indicated that all studies had a low risk of bias.
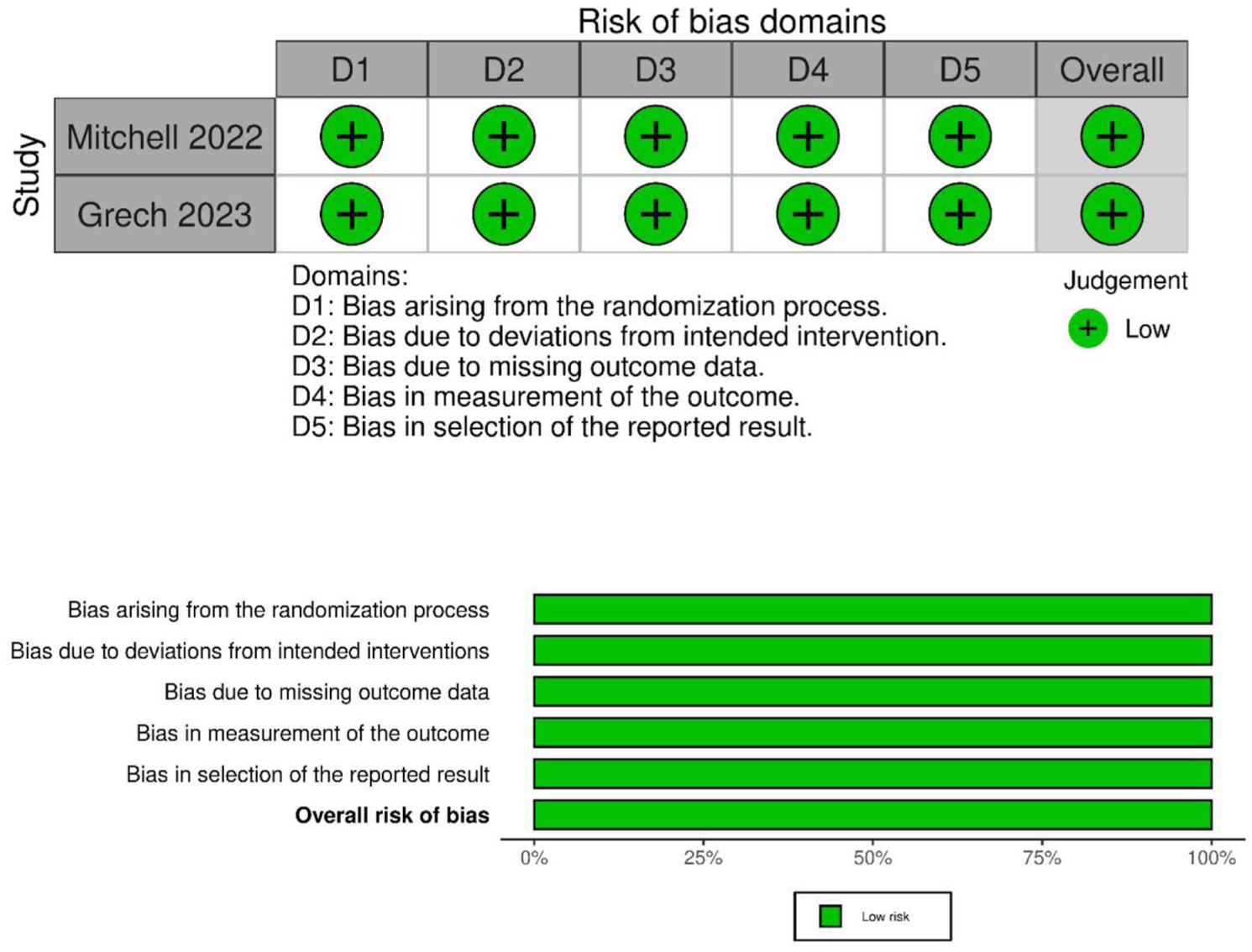
Quality assessment of risk of bias in the included Trials.
The risk of bias in the included cohort studies was assessed using Newcastle-Ottawa Quality Assessment Form for Cohort Studies (NOS). This tool evaluates key domains such as Selection, Comparability, and Outcome. The results of the risk of bias assessment are summarized in the Table S7. The risk of bias assessment indicated that all studies had a good quality.
The risk of bias in the included case–control study was assessed using Newcastle-Ottawa Quality Assessment Form for case–control studies. This tool evaluates key domains such as Selection, Comparability, and Exposure. The results of the risk of bias assessment are summarized in the Table S8. The risk of bias assessment indicated that the included study had a good quality.
Discussion
Summary of the results
Six studies—a RCT and the post-hoc analysis of it, three cohorts and a case–control study—were included in this study.8–10,15–17 Four outcomes were analyzed with five subgroups according to the time of follow-up: 3, 6, 12, and 24 months. We found that the GLP-1 agonist group was superior to the usual care group in all outcomes and all subgroups except in BMI outcome in the 3 months subgroup.
Outcomes
IIH is a neurological condition characterized by increased ICP without a known etiology. 7 The illness, primarily affecting obese women of reproductive age, causes severe symptoms such as recurrent headaches and papilledema, potentially causing irreversible vision impairment if ignored. 18 Incidence is rising due to global obesity trends, with even little weight gain increasing the chance of developing IIH or returning after remission. 19 A 10% or more significant reduction in body weight generally results in illness remission. 20 The etiology of IIH is complicated and diverse, with obesity-related processes such as increased intra-abdominal pressure impacting cerebral venous drainage and adipose tissue acting as an endocrine organ, releasing substances that alter ICP and neuroinflammation.21,22 Weight loss therapies are the most effective ways to influence the illness course of IIH, as body weight is the key modifiable factor linked to its development. 23 Adipose tissue dysfunction, especially in the setting of obesity, has been shown in recent studies to contribute to IIH via various pathways, such as changes in hormonal signaling and the generation of pro-inflammatory adipokines. 24
Medications often used in IIH have been shown to exacerbate cognitive deficits in patients, with acetazolamide and topiramate impairing fluid intelligence. Acetazolamide usage has been associated with memory impairment, and many investigations have shown deficits in language, working memory, verbal fluency, intelligence quotient, and verbal learning.25–28 Consequently, an unaddressed clinical need exists for medicines that successfully diminish intracranial pressure without adversely affecting cognitive function. 10
GLP-1, a peptide hormone, is generated by enteroendocrine cells at low baseline levels and by neurons in the caudal medulla. 29 It significantly affects blood glucose levels by increasing insulin release or suppressing glucagon secretion and inhibits appetite via receptors in the ventral tegmental region, nucleus accumbens, and hypothalamus.30–32 GLP-1-RAs were first created for type-2 diabetes but are now licensed for obesity management. Antiobesity drug therapy can be used in conjunction with a low-calorie diet and increased physical activity to manage weight in adults with a BMI of ⩾30 kg/m2 or 27–30 kg/m2 with weight-related comorbidity, such as dysglycemia, hypertension, dyslipidemia, or obstructive sleep apnea. 9
The advent of GLP-1 RAs has transformed the therapeutic framework for IIH, presenting an innovative strategy that concurrently targets the metabolic and neurological dimensions of the condition. 7 It has been shown that GLP-1 receptors in the choroid plexus alter Na+/K+-ATPase activity, which directly influences CSF production and may reduce ICP without exclusively depending on weight reduction.7,22 This technique confirms earlier research showing GLP-1 receptor activation regulates CSF in animal models.7,33
Our analysis revealed that papilledema was significantly improved in the GLP-1 agonist group compared to the usual care group. These results are aligned with Azzam et al.2,16 This study reported that liraglutide, a long-acting GLP-1 RA administered once daily, dramatically reduces the incidence of papilledema when used as a supplementary medication in managing idiopathic intracranial hypertension while demonstrating favorable trends in other clinical outcomes. The statistically significant impact on papilledema, mainly apparent at 3 months and maintained through 24 months, signifies considerable progress in treating IIH since existing therapeutic alternatives often encounter limitations related to tolerance and effectiveness.7,8,15 These results also align with Azzam et al. 17 and Krajnc et al. 9 These studies demonstrated that GLP-1 RAs significantly improved papilledema. Mitchell et al. emphasized the temporal impact of GLP-1 RAs, noting that intracranial pressure was significantly lowered by roughly 5.7 cm CSF at 2.5 h, with enduring benefits extending beyond 12 weeks. 8
Regarding visual disturbances, we found that GLP-1 RAs significantly reduce visual disturbances. These results are similar to those of Azzam et al., 17 who found that GLP-1 RAs contributed to a 72% decrease in blindness or visual abnormalities at 3 months with long-lasting advantages at 24 months. Azzam et al. demonstrated that the continuous improvement in visual outcomes, achieving a 73.9% decrease in risk at 24 months, has considerable therapeutic significance due to the potentially severe repercussions of visual decline in IIH. 15 These results are inconsistent with those of Azzam et al., who found no significant differences when comparing GLP-1 RAs with a usual care group. 16
Headaches are a significant debilitating symptom for people with IIH and are ranked among the top ten research priorities by patient organizations. 34 Headache is a crucial factor contributing to the markedly diminished quality of life in IIH. 35 We found that GLP-1 RAs significantly improved headache symptoms and frequencies. Our results show they reduce the risk of headache over time, with a notable risk reduction of 31% at 3 months (RR: 0.69) and a sustained, significant effect at 24 months (RR: 0.80). These results are in alignment with those of Mitchell et al. 8 and Azzam et al. 17 who revealed 42% headache symptoms decline. The findings presented here offer more support for the function that GLP-1 RAs play in the management of this exhausting feature of IIH. 17 To put these improvements in context, the current standard of care, acetazolamide, was studied in the Idiopathic Intracranial Hypertension Treatment Trial (IIHTT). 3 The trial established its efficacy against a backdrop of severe baseline symptoms, where patients reported an average headache severity of 6.3 out of 10 and over half experienced daily or constant headaches. 3 While the IIHTT confirmed acetazolamide’s role in reducing headache disability, our findings for GLP-1 agonists—showing a consistent and significant reduction in headache risk—suggest they represent a potent therapeutic pathway. 3
On the other hand, we found that Azzam et al. revealed a nonsignificant improvement in headache. 16 The patterns indicating improvement in headache and visual results, albeit not statistically significant, imply intricate underlying pathophysiological pathways that need additional exploration in prospective therapeutic trials. Symptoms of IIH often stem from elements other than increased intracranial pressure, which may explain the comparatively limited impact on these outcomes relative to papilledema. 36
BMI was found to be significantly reduced in the GLP-1 RA group. Azzam et al. results are aligned with ours revealing that BMI reductions that were seen (−1.147 kg/m2 at 24 months) indicate that GLP-1 RA weight loss effects contribute to therapeutic advantages by reducing intracranial venous pressure and decreasing the generation of pro-inflammatory adipokines.15,37
Refractory IIH was defined as either persistence of symptoms despite maximum medical therapy, progression to surgical intervention (identified through relevant CPT codes), or requirement for CSF diversion procedures or optic nerve sheath fenestration. 17 Our analysis shows that refractory IIH is improved in the GLP-1 RA group. These results are consistent with Azzam et al. (2024) who revealed 40% refractory reduction but inconsistent with Azzam et al. who found that no statistically significant difference between the two groups.16,17
Exenatide, a GLP-1 RA, shows significant promise for treating IIH by rapidly reducing intracranial pressure. 8 Mitchell et al. demonstrate it significantly lowers ICP both acutely, achieving a reduction of approximately 5.7 cmCSF within 2.5 h, and sustains this effect long-term, with a sustained reduction of −5.6 cmCSF noted at 12 weeks. This rapid onset of action within the first 24 h, not observed in studies of other off-label IIH treatments, presents a clinical advantage for managing potentially rapid patient deterioration and may also be relevant for other conditions involving raised ICP, such as traumatic brain injury or stroke. The magnitude of ICP reduction with exenatide at 12 weeks (−5.6 cmCSF) is comparable to that achieved through other interventions like acetazolamide combined with diet (approx. −5.9 cmCSF), bariatric surgery (approx. −6.0 cmCSF), or low-calorie diets (approx. −6.2 cmCSF). 8
Strengths and limitations
The meta-analysis included a variety of research formats (RCTs, cohorts, case–control studies), offering a comprehensive perspective on the subject. This extensive selection enhances the credibility of the results by integrating many approaches and viewpoints.
A major strength of this research is its comprehensive subgroup analysis categorized by follow-up durations (3, 6, 12, and 24 months). This methodology improves comprehension of the temporal impacts of GLP-1 RAs on IIH-related outcomes, allowing a more accurate assessment of therapy effectiveness across time.
Moreover, by synthesizing findings from many investigations (Azzam et al., Mitchell et al., Krajnc et al.), the meta-analysis fortifies the validity and applicability of its conclusions. The continuous correlation of data with previous research enhances the plausibility of the stated advantages of GLP-1 RAs in the therapy of IIH.
The limitations of this meta-analysis include the restricted number of included studies, characterized by small sample sizes and variability in research designs. The incorporation of several research designs (RCTs, cohorts, case–control studies) may add diversity in techniques, patient demographics, and data collecting methods, thereby impacting the consistency of findings. Participant attrition within (Azzam et al., 2025, 1 and Azzam et al. 2024) introduces potential methodological limitations. This decline in sample size may lead to attrition bias if nonrandom, reduce statistical power, and consequently affect the internal and external validity of the findings.15,17 The specific analytical strategy used to address missing data requires clarification. Future research should prioritize robust retention protocols, transparent reporting of attrition, and appropriate statistical methodologies to mitigate these concerns.
The limitations of this meta-analysis include the restricted number of included studies and their small sample sizes. With only four studies included in the quantitative synthesis, the power for subgroup analysis over multiple time points (3, 6, 12, and 24 months) is weakened. Furthermore, formal tests for publication bias, such as funnel plots or Egger’s test, were not performed. As recommended by methodological guidelines, these tests are unreliable and can be misleading when fewer than ten studies are analyzed. 12 Another limitation is the variability of the available literature, which includes different research designs (RCTs, cohorts, case–control studies). This underlying diversity in techniques, patient demographics, and data collecting methods, thereby impacting the consistency of findings and the generalizability of our results. Additionally, participant attrition within two of the included studies (Azzam et al. and Azzam et al., 2024) introduces potential methodological limitations. This decline in sample size may lead to attrition bias if nonrandom, reduce statistical power, and consequently affect the internal and external validity of the findings.15,17 The specific analytical strategy used to address missing data requires clarification. Future research should prioritize robust retention protocols, transparent reporting of attrition, and appropriate statistical methodologies to mitigate these concerns.
Strengths
A key strength of this meta-analysis lies in its comprehensive scope. By including a variety of research formats (RCTs, cohort studies, and case–control studies), this review offers a broad perspective on the topic. Furthermore, our detailed subgroup analysis, categorized by follow-up durations (3, 6, 12, and 24 months), was designed to provide a more nuanced comprehension of the temporal effects of GLP-1 RAs on IIH-related outcomes. The synthesis of findings from multiple investigations (Azzam et al., Mitchell et al., Krajnc et al.) fortifies the existing evidence base, enhancing the validity and applicability of the conclusions regarding the benefits of GLP-1 RAs in the management of IIH.
Limitations
Despite these strengths, several limitations must be acknowledged. The most significant is the restricted number of included studies.
We acknowledge that the inclusion of only four studies in the quantitative synthesis limits the statistical power of our subgroup analyses, particularly when examining outcomes over multiple time points (3, 6, 12, and 24 months). This small sample size may reduce the certainty of our findings and underscores the need for larger, more comprehensive studies in this area.
This limited number of studies also precluded a formal assessment for publication bias. As recommended by methodological guidelines, tests such as funnel plots or Egger’s test are unreliable and can be misleading when fewer than ten studies are included.
Another limitation is the heterogeneity of the included literature. While incorporating various research designs provided a broad overview, it also introduced diversity in study methodologies, patient demographics, and data collecting methods. This variability may impact the overall consistency of the findings and the generalizability of our results.
Finally, participant attrition within two of the included studies (Azzam et al., 2024; Azzam et al., 2025) introduces a potential methodological concern. This decline in sample size may lead to attrition bias if nonrandom, further reducing statistical power, and consequently affecting the internal and external validity of the findings.15,17 Future research should prioritize robust retention protocols and transparent reporting to mitigate these concerns.
Conclusion
The findings of this systematic review and meta-analysis suggest that treatment with GLP-1 RAs may be associated with improvements across various intervals, including in papilledema and Visual Disturbances, which appear to be maintained for up to 24 months. The observed decrease in BMI supports the significance of weight control in the treatment of IIH and the connection between obesity-related processes and elevated intracranial pressure. While these results are promising, they should be viewed as preliminary given the modest strength of the available evidence. The conclusions are drawn from a small number of studies with varied designs. Therefore, GLP-1 RAs represent a potential therapeutic strategy that targets both the metabolic and neurological aspects of IIH. Further large-scale, high-quality randomized controlled trials are essential to definitively establish the long-term efficacy and safety of this approach.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251378845 – Supplemental material for The effect of GLP-1 agonist on idiopathic intracranial hypertension: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tan-10.1177_17562864251378845 for The effect of GLP-1 agonist on idiopathic intracranial hypertension: a systematic review and meta-analysis by Jamal Ahmad, Ahmed Mohamed Hamdy, Bahaa Elfakharany, Muhammad M. Elsharkawy, Mohamed El-Samahy, Mazen Momtaz Shehata and Elsayed S. Moubarak in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864251378845 – Supplemental material for The effect of GLP-1 agonist on idiopathic intracranial hypertension: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tan-10.1177_17562864251378845 for The effect of GLP-1 agonist on idiopathic intracranial hypertension: a systematic review and meta-analysis by Jamal Ahmad, Ahmed Mohamed Hamdy, Bahaa Elfakharany, Muhammad M. Elsharkawy, Mohamed El-Samahy, Mazen Momtaz Shehata and Elsayed S. Moubarak in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-xlsx-3-tan-10.1177_17562864251378845 – Supplemental material for The effect of GLP-1 agonist on idiopathic intracranial hypertension: a systematic review and meta-analysis
Supplemental material, sj-xlsx-3-tan-10.1177_17562864251378845 for The effect of GLP-1 agonist on idiopathic intracranial hypertension: a systematic review and meta-analysis by Jamal Ahmad, Ahmed Mohamed Hamdy, Bahaa Elfakharany, Muhammad M. Elsharkawy, Mohamed El-Samahy, Mazen Momtaz Shehata and Elsayed S. Moubarak in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
