Abstract
Background:
Among the currently approved antiobesity medications, the glucagon-like-peptide-1 receptor-agonists (GLP-1 RAs) liraglutide and semaglutide, and the dual glucose-dependent-insulinotropic-polypeptide (GIP)/GLP-1 RA tirzepatide have been suggested to reduce cardiovascular-risk in overweight or obesity without diabetes.
Objectives:
The objective of this study was to evaluate the cardio- and neuroprotective potential of these novel agents in the nondiabetic overweight/obese adult population.
Data sources and methods:
A systematic review and meta-analysis of randomized-controlled clinical trials (RCTs) was performed to estimate the risk of major adverse cardiovascular events (MACE), all-cause and cardiovascular mortality in overweight or obese adults without diabetes treated with GLP-1 or GIP/GLP-1 RAs (vs placebo). Secondary outcomes included the risk of myocardial infarction (MI) and stroke.
Results:
Sixteen RCTs (13 and 3 on GLP-1 RAs and tirzepatide, respectively) comprising 28,168 participants were included. GLP-1 or GIP/GLP-1 RAs reduced MACE (odds ratio (OR): 0.79; 95% confidence interval (CI): 0.71–0.89; p < 0.01; I2 = 0) and all-cause mortality (OR: 0.80; 95% CI: 0.70–0.92; p < 0.01; I2 = 0), while there was a trend toward lower cardiovascular-mortality (OR: 0.84; 95% CI: 0.71–1.01; p = 0.06; I2 = 0%) compared to placebo. Additionally, GLP-1 or GIP/GLP-1 RAs reduced the odds of MI (OR: 0.72; 95% CI: 0.61–0.86; p < 0.01; I2 = 0%) and nonfatal-MI (OR: 0.72; 95% CI: 0.61–0.85; p < 0.01; I2 = 0%); while no associations between antiobesity treatment and fatal-MI, stroke, nonfatal, or fatal stroke were uncovered.
Conclusion:
GLP-1 and GIP/GLP-1 RAs reduce cardiovascular-risk and all-cause mortality in overweight or obese adults without diabetes. Additionally, GLP-1 RAs and GIP/GLP-1 RAs attenuate the risk of MI. Since data on stroke are still limited, future RCTs are warranted to evaluate the neuroprotective potential of these novel antiobesity agents.
Trial registration:
PROSPERO CRD42024515966.
Introduction
With the prevalence of overweight and adiposity rising exponentially worldwide, 1 emerging therapeutic strategies with novel antiobesity agents are gaining increasing traction. Overweight and obesity rank among the leading causes of death globally, conferring increased risk for disability and comorbid chronic diseases, most notably diabetes, metabolic syndrome, cardiovascular disease, and cancer.2,3 While geographic and racial/ethnic disparities still exist, epidemiological models forecast that by 2030, the global prevalence of overweight and obesity will amount to 2.16 and 1.12 billion cases, accounting for 38% and 20% of the world’s adult population. 1 By this time, if secular trends continue unabated, obesity and severe obesity in the United States are expected to affect nearly 1 in 2 and 1 in 4 adults, respectively. 4 The growing health and economic burden of obesity has rendered antiobesity interventions a top-priority on the global health agenda, 5 with antiobesity drugs currently racing from bench to bedside.
As per clinical practice guidelines, pharmacotherapy is recommended as an adjunct to lifestyle interventions for obese or overweight adults with a body mass index (BMI) ⩾ 30 kg/m2 or a BMI ⩾ 27 kg/m2 with at least one cardiovascular risk factor (e.g., prediabetes, diabetes, hypertension, dyslipidemia, elevated waist circumference) or obesity-related comorbidity (e.g., nonalcoholic fatty liver disease, obstructive sleep apnea).6–8 Among the currently Food Drug Administration approved antiobesity medications, two glucagon-like peptide-1 receptor agonists (GLP-1 RAs), liraglutide and semaglutide, and a novel dual glucose-dependent insulinotropic polypeptide (GIP) and GLP-1 receptor agonist, tirzepatide, are indicated for treatment of nonsyndromic obesity. 9 GLP-1 RAs exert antiobesity effects by targeting peripheral and central pathways that increase insulin and inhibit glucagon secretion, while inducing satiety.9,10 GIP/GLP-1 RAs additionally act on peripheral tissues and islets, improving pancreatic beta cell function and augmenting energy expenditure. 10
Experimental and clinical evidence from randomized-controlled clinical trials (RCT) have previously established that GLP-1 RAs and GIP/GLP-1 RAs exert pleiotropic cardioprotective effects, attenuating cardiovascular risk in type-2 diabetes (T2DM).11–13 Importantly, meta-analyses of RCTs have established that GLP-1 RAs exert highly potent glucose-regulating effects in T2DM, significantly reducing hemoglobin A1c (HbA1c) levels and body weight in a dose-dependent manner compared to placebo. 14 Additionally, tirzepatide has demonstrated even greater effects on glycemic control and weight loss, suggesting potentially superior efficacy compared to other antidiabetic agents. 15 Beyond diabetes, phase III RCTs have provided first evidence on the efficacy and safety of GLP-1 and GIP/GLP-1 RAs for treatment of overweight and obesity in nondiabetic individuals.16,17 This evidence was recently reinforced by the publication of the SELECT trial results, 18 the first cardiovascular outcome trial (CVOT) assessing the cardiovascular efficacy of semaglutide, which demonstrated significant reduction of major adverse cardiovascular events (MACE) in patients with preexisting cardiovascular disease and overweight or obesity without diabetes.
In light of the rapidly evolving landscape of obesity trials and the availability of new RCT data, the aim of the present systematic review and meta-analysis was to evaluate the cardio- and neuroprotective potential of GLP-1 RAs and GIP/GLP-1 RAs assessing the risk of MACE, including stroke, all-cause, and cardiovascular mortality in overweight or obese adults without diabetes.
Methods
Standard protocol approvals and registrations
Reporting adheres to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 19 No Ethical Committee approval was required as per study design (systematic review and meta-analysis). The study protocol, comprising predetermined PICOS (Population, Intervention, Comparison, Outcome, and Study) framework, was a priori designed and registered at the PROSPERO database (CRD42024515966). All supporting data are available within the article and its Supplemental Files.
Data sources and searches
Two independent reviewers (M.-I.S., L.P.) searched for published randomized placebo-controlled trials testing GLP-1 RAs or GIP/GLP-1 RA in adults with overweight or obesity without diabetes. Eligible RCTs were identified by systematic search in MEDLINE (via PubMed) and Scopus databases. The combination of search strings for all database queries included combined search terms: “glucagon-like peptide-1 receptor agonist,” “dual GLP-1/GIP receptor agonists,” “semaglutide,” “lixisenatide,” “exenatide,” “albiglutide,” “liraglutide,” “dulaglutide,” “tirzepatide,” “randomized controlled trial,” “placebo,” “MACE,” “major adverse cardiovascular events,” or “stroke.” The full search algorithms used in MEDLINE and SCOPUS databases are provided in the Supplemental Material. Our search was restricted to RCTs, while no language restrictions were applied. The search spanned from each electronic database’s inception to 10 February 2024. Manual search of bibliographies of articles meeting study inclusion criteria was additionally performed to ensure the comprehensiveness of the literature.
Placebo-controlled RCTs that reported on MACE in adults with overweight or obesity without diabetes treated with GLP-1 RAs or GIP/GLP-1 RAs were eligible for inclusion. Exclusion criteria comprised: (1) RCTs that were not placebo-controlled; (2) RCTs not including overweight or obese populations without diabetes; (3) RCTs investigating compounds of GLP-1 RAs or GIP/GLP-1 RA combined with other drugs; (4) study population of <300 participants12,20; (5) reported outcomes not aligned with our inclusion criteria; (6) observational studies, narrative, and systematic reviews, case-series or case-reports, commentaries, pre-prints or nonpeer reviewed studies, and conference abstracts. Given the fact that inclusion of individual studies that are underpowered to detect differences in rare outcomes (such as MACE or mortality) introduces significant sampling errors and statistical/methodological biases that cannot be mitigated using meta-analytical approaches, we excluded small RCTs with <300 participants in line with a previously published meta-analysis by our group. 12 In case of studies with overlapping data, the study with the largest dataset was retained. All retrieved studies were independently assessed by two reviewers (M.-I.S., L.P.) and disagreements were resolved by consensus after discussion with a third tie-breaking evaluator (G.T.).
Quality control, bias assessment, and data extraction
For relevant domains of each included study, the risk of bias was assessed using the Cochrane Collaboration risk of bias tool. 21 Three independent reviewers (M.-I.S., L.P., A.T.) performed quality control and bias assessment, and in case of disagreement consensus after discussion with the corresponding author (G.T.) was reached. Data including first author name, publication year, study design and duration, patient population, sample size, and event type (i.e., MACE, all-cause mortality, cardiovascular mortality, stroke, and myocardial infarction (MI)) were extracted from individual studies in structured reports.
Publication bias across individual studies was evaluated for all primary outcomes graphically using funnel plots, 22 while Egger’s linear regression test was used for funnel plot asymmetry assessment, 23 and the threshold of the statistical significance was set at p < 0.10.
Outcomes
An aggregate data meta-analysis was performed including all identified placebo-controlled RCTs. The predefined primary outcomes of interest were threefold: (i) the incidence of MACE; (ii) all-cause; and (iii) cardiovascular mortality. Secondary outcomes included the incidence of (i) stroke, (ii) fatal, and (iii) nonfatal stroke; (iv) MI, (v) fatal, and (vi) nonfatal MI. Subgroup analysis was performed based on type of treatment either with GLP-1 RAs or GIP/GLP-1 RAs.
Statistical analysis
R-software version 3.5.0 (packages: meta and metafor) was used for meta-analysis (R Foundation for Statistical Computing, Vienna, Austria; URL: http://www.R-project.org/). All intended outcomes of interest were handled as dichotomous variables, and all the associations evaluating the effect of GLP-1 RAs or GIP/GLP-1 RAs with different outcomes are reported as odds ratios (ORs) with their corresponding 95% confidence intervals (CIs). The random-effects model of meta-analysis (DerSimonian and Laird) was utilized for estimation of the pooled estimates. We used the Q test to assess subgroup differences. The I2 and Cochran Q statistics were employed for heterogeneity assessment. With respect to qualitative heterogeneity interpretation, I2 values >50% and values >75% were considered to represent substantial or considerable heterogeneity, respectively. The significance level was set at 0.1 for the Q statistic, 24 while the equivalent z test with a two-tailed p value < 0.05 was considered statistically significant for each pooled estimate.
Results
Literature search and included studies
The systematic database search yielded 2751 records from MEDLINE and 3460 records from SCOPUS databases. After exclusion of duplicates and articles that were out-of-scope, 451 records were considered eligible for inclusion and were assessed in full. After reading the full-text articles, 433 were further excluded (Supplemental Material). Finally, we identified 18 eligible studies for inclusion reporting on 16 RCTs (13 RCTs on GLP-1 RAs,18,25–36 1 study reporting on 3-year assessment of a previous RCT on GLP-1 RA, 37 1 post hoc analysis, 38 3 RCTs on tirzepatide16,17,39), comprising a total of 28,168 participants. All original studies were placebo-controlled RCTs, and Table 1 summarizes their main characteristics. In Figure 1, the PRISMA flowchart of the meta-analysis is presented.
Main characteristics of randomized-controlled trials (n = 16) included in the meta-analysis and reported outcomes of interest.

BMI, body mass index; CV, cardiovascular; GIP, glucose-dependent insulinotropic polypeptide; GLP-1 RAs, glucagon-like peptide-1 receptor agonists; MACE, major adverse cardiovascular event; MI, myocardial infarction; SC, subcutaneous; SD, standard deviation.

PRISMA flowchart diagram presenting the selection of eligible studies.
Quality control and publication bias of included studies
The risk of bias of studies included in the present meta-analysis is presented in Supplemental Figure S1. The risk of bias was considered low in all the included RCTs.
Funnel plot symmetry inspection and Egger statistical testing were performed for outcomes involving ⩾4 studies. 22 Accordingly, no asymmetry was revealed for assessment of publication bias among trials reporting MACE (p = 0.0795; Supplemental Figure S2), cardiovascular mortality (p = 0.939; Supplemental Figure S3), and all-cause mortality (p = 0.8691; Supplemental Figure S4) between treatment with GLP-1 RAs or GIP/GLP-1 RAs and placebo.
Primary and secondary outcomes
In overweight or obese adults without diabetes, treatment with GLP-1 RAs or GIP/GLP-1 RAs was associated with significant reduction of MACE (7 RCTs; OR: 0.79; 95% CI: 0.71–0.89; p < 0.01; I2 = 0%; Figure 2) and all-cause mortality (15 RCTs; OR: 0.80; 95% CI: 0.70–0.92; p < 0.01; I2 = 0%; Figure 3(a)). In addition, there was a trend toward reduced cardiovascular mortality (15 RCTs; OR: 0.84; 95% CI: 0.71–1.01; p = 0.06; I2 = 0%; Figure 3(b)), which did not reach statistical significance (summarized outcomes in Table 2).

Forest plot comparing the risk of MACE in overweight or obese adults without diabetes treated with GLP-1 or GIP/GLP-1 RAs versus placebo.

Forest plot comparing the risk of all-cause mortality (a) and cardiovascular mortality (b) in overweight or obese adults without diabetes treated with GLP-1 or GIP/GLP-1 RAs versus placebo.
Overview of vascular outcomes among overweight or obese adults without diabetes treated with GLP-1 or GIP/GLP-1 RAs versus placebo.

CI, confidence interval; GIP, glucose-dependent insulinotropic polypeptide; GLP-1 RAs, glucagon-like peptide-1 receptor agonists; MACE, major adverse cardiovascular event; MI, myocardial infarction; OR, odds ratio; RCTs, randomized controlled trials.
Concerning secondary outcomes, no association was uncovered between GLP-1 RA or GIP/GLP-1 RA treatment and the risk of stroke (14 RCTs; OR: 0.92; 95% CI: 0.74–1.14; p = 0.48; I2 = 0%; Figure 4); fatal (13 RCTs; OR: 0.34; 95% CI: 0.02–5.42; p = 0.44; I2 = NA; Supplemental Figure S5); or nonfatal stroke (3 RCTs; OR: 0.92; 95% CI: 0.74–1.15; p = 0.49; I2 = 0%; Supplemental Figure S6). Conversely, GLP-1 RA or GIP/GLP-1 RA treatment was associated with significant reduction of MI (15 RCTs; OR: 0.72; 95% CI: 0.61–0.86; p < 0.01; I2 = 0%; Supplemental Figure S7) and nonfatal MI (5 RCTs; OR: 0.72; 95% CI: 0.61–0.85; p < 0.01; I2 = 0%; Supplemental Figure S8); while no association between fatal MI and GLP-1 RA or GIP/GLP-1 RA treatment was disclosed (14 RCTs; OR: 1.62; 95% CI: 0.20–13.23; p = 0.65; I2 = 0%; Supplemental Figure S9). With respect to stroke subtypes, no meta-analysis could be performed for hemorrhagic stroke (only 1 RCT 17 reported 0 events in the treatment and placebo arms), while no association between ischemic stroke (2 RCTs; OR: 0.50; 95% CI: 0.02–12.31; p = 0.67; I2 = NA%; Supplemental Figure S10) and transient ischemic attack (2 RCTs; OR: 0.83; 95% CI: 0.04–17.44; p = 0.91; I2 = NA%; Supplemental Figure S11) and GLP-1 RA or GIP/GLP-1 RA treatment was uncovered. Subgroup analyses on GLP-1 RA versus GIP/GLP-1 RA treatment, revealed no significant subgroup effects on any of the primary or secondary outcomes.
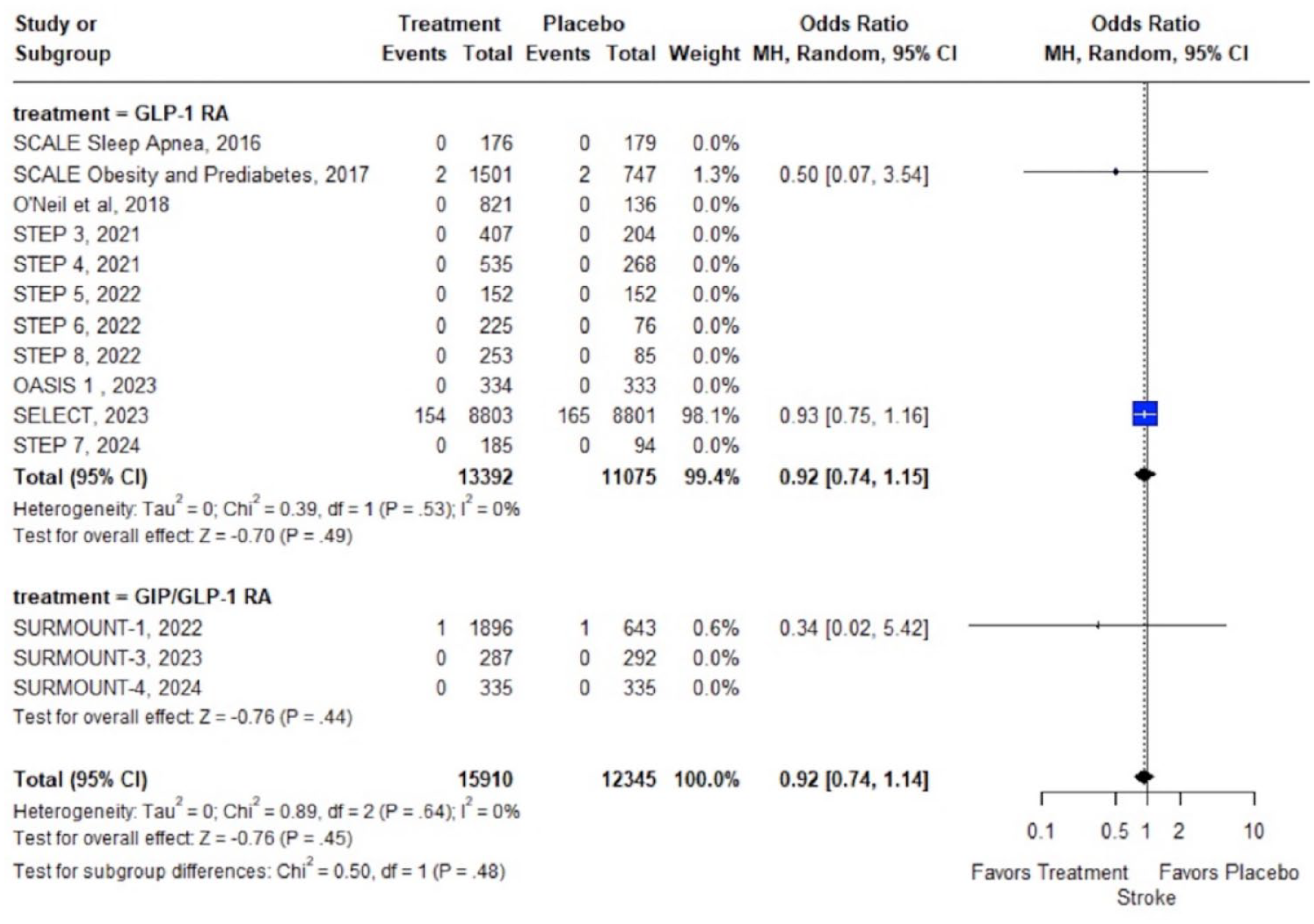
Forest plot comparing the risk of stroke in overweight or obese adults without diabetes treated with GLP-1 or GIP/GLP-1 RAs versus placebo.
Discussion
In the present systematic review and meta-analysis, treatment with GLP-1 RAs or GIP/GLP-1 RAs for overweight or obesity in the absence of diabetes was associated with significant reduction of MACE. In addition, GLP-1 RA or GIP/GLP-1 RA treatment was associated with significant reduction of all-cause mortality, while there was a trend toward reduced cardiovascular mortality, which did not reach statistical significance. Regarding secondary outcomes, treatment with GLP-1 or GIP/GLP-1 RAs was associated with significant reduction of MI and nonfatal MI compared to placebo, whereas no association between GLP-1 or GIP/GLP-1 RAs and fatal MI, stroke, fatal stroke, and nonfatal stroke was uncovered.
These findings align with results of prior meta-analyses from our group and others that have demonstrated potent cardioprotective effects of GLP-1 RAs or GIP/GLP-1 RAs in T2DM.12,13,40 In addition, the current meta-analysis expands on our previous findings, documenting consistent cardiovascular benefits in the non-diabetic overweight/obese adult population. Considering the global prevalence of overweight and obesity, these findings could have critical public health implications. Currently, the accruing evidence on GLP-1 RAs and GIP/GLP-1 RAs, particularly the compelling recent data from the SELECT trial, highlights a shift in the clinical approach to managing overweight and obesity. Notably, prior research on atherosclerotic cardiovascular disease (ASCVD) prevention primarily relied on observational data, which showed limited efficacy of pharmacological or bariatric-induced body weight loss for secondary ASCVD prevention. 41 It should thus be emphasized that the striking results of the SELECT trial, along with emerging evidence from ongoing RCTs on GLP-1 and GIP/GLP-1 RAs, are expected to reshape future treatment guidelines, especially for managing patients with obesity and preexisting ASCVD. 41
With respect to cardiovascular mortality, which showed a nonsignificant trend toward reduction with treatment, several reasons may have contributed to an underestimation of observed treatment effects, including the limited number of events and thus statistical power, adjudication biases, along with substantial heterogeneity in definitions of cardiovascular mortality and safety outcomes in included RCTs. Notably, several mechanisms have been implicated in GLP-1 and GIP/GLP-1 RAs-induced cardiovascular risk reduction, comprising (i) mechanisms directly related to weight loss effects, including improved glucose and arterial blood pressure regulation, lipid metabolism, myocardial, and endothelial function; and (ii) weight loss independent effects, including anti-inflammatory, 42 and nephroprotective 43 actions. In addition, GLP-1 and GIP/GLP-1 RAs have shown promising efficacy in reducing liver fat, improving hepatic enzymes, and promoting the resolution of metabolic dysfunction-associated steatotic liver disease and steatohepatitis through mechanisms such as enhanced insulin sensitivity, decreased lipogenesis, weight loss, and attenuation of hepatic inflammation. 44 Clinical trials have demonstrated these agents’ potential not only in improving liver histology but also in conferring cardiovascular and metabolic benefits.45,46 The pleiotropic effects of GLP-1 and GIP/GLP-1 RAs may synergistically account for the observed attenuation of all-cause mortality, extending beyond reduction of cardiovascular risk.
Certain methodological nuances must be considered for an accurate interpretation of the current findings. First, as evidenced by the individual study weights of the present meta-analyses, the pooled effects were mainly driven by the effects of GLP-1 RA treatment in the SELECT trial. 18 SELECT was a phase III RCT investigating the cardiovascular efficacy and safety of once-weekly subcutaneous semaglutide at a dose of 2.4 mg at a mean follow-up of 39.8 ± 9.4 months including 8803 and 8801 nondiabetic individuals with preexisting cardiovascular disease and a BMI ⩾27 kg/m2 allocated to treatment or placebo groups, respectively. As per CVOT design, outcome events in SELECT exceeded by far the events documented in other included RCTs, which largely excluded overweight or obese individuals at high cardiovascular risk, had limited sample sizes, and short follow-up periods. Although these trials were underpowered to detect cardiovascular efficacy, the included data from 10,564 overweight or obese participants (in addition to the SELECT trial population) from 15 phase III RCTs on GLP-1 RAs and GIP/GLP-1 RAs significantly strengthen the findings of our meta-analysis and expand on the safety profile of these novel agents, demonstrating lower all-cause mortality and tendentially lower cardiovascular mortality compared to placebo. Second, it should be noted that the incidence of fatal stroke and fatal MI was not reported in SELECT; thus, data were unavailable for meta-analysis. Third, regarding the nonsignificant association of GLP-1 or GIP/GLP-1 RA treatment with stroke, it should be noted that these results stand in contrast to the well-established reduction of stroke risk and particularly ischemic stroke with GLP-1 RAs in T2DM.12,13 Several concerns regarding SELECT’s design should be expressed at this point: (i) as per study design, only patients with preexisting cardiovascular disease were included; however, stroke was clearly underrepresented, with the ratio of prior MI to prior stroke being 4:1 (i.e., corresponding to 68% and 18% of included patients, respectively); (ii) the vast majority of patients were under treatment with lipid-lowering medications (90%) and platelet-aggregation inhibitors (86%); (iii) patients at high risk for stroke were excluded (i.e., patients could not be enrolled within 2 months after a cardiovascular or neurologic event or if they planned to undergo coronary, carotid, or peripheral revascularization). Notably, experimental evidence also suggests robust neuroprotective effects from GLP-1 RAs, particularly in ischemic stroke. 47 Given the limited number of outcome events in the present meta-analysis (326 cumulative stroke events) and the limited follow-up, type II errors cannot be excluded and larger well-designed RCTs with adequate sample sizes and trial periods are warranted to evaluate GLP-1 RAs and GIP/GLP-1 RAs efficacy for primary and secondary stroke prevention in overweight and obese adults without diabetes.
Concerning differential effects of GLP-1 RA versus GIP/GLP-1 RA treatment, subgroup analyses revealed no significant subgroup effects on any of the primary or secondary outcomes. Nonetheless, due to limited data availability on the novel dual GIP/GLP-1 RA tirzepatide and the lack of dedicated CVOTs, we caution that the lack of observed associations should not be interpreted as evidence of comparable efficacy of these agents in the overweight or obese nondiabetic population. The available evidence on tirzepatide primarily stems from the SURMOUNT clinical development program, which aimed to evaluate the safety and efficacy of tirzepatide as an adjunct to lifestyle intervention, compared to placebo, for chronic weight management in adults with a BMI ⩾27 kg/m², with or without T2DM. Notably, among the seminal SURMOUNT trials, SURMOUNT-2 has not been included in the present meta-analysis due to inclusion of patients with obesity and established T2DM. 48 SURMOUNT-1 was a phase III, double-blind RCT involving 2539 adults with a BMI ⩾30 kg/m² (or ⩾27 kg/m² with at least one weight-related complication, excluding T2DM), which demonstrated that weekly administration of tirzepatide at doses of 5, 10, and 15 mg significantly reduced body weight by up to 20.9% compared to 3.1% with placebo over 72 weeks. 39 This weight reduction was accompanied by improvements in cardiometabolic risk factors, including reductions in systolic and diastolic blood pressure, fasting insulin levels, waist circumference, and lipid levels. The most common adverse events associated with tirzepatide were gastrointestinal, primarily occurring during dose escalation. SURMOUNT-3 was a phase III, double-blind RCT that included 579 adults with a BMI of ⩾30, or ⩾27 kg/m² with at least one weight-related complication, including those with T2DM. Participants achieved a ⩾5.0% weight reduction following a 12-week intensive lifestyle intervention and were subsequently randomized to receive the maximum tolerated dose of tirzepatide (10 or 15 mg) or placebo once weekly for 72 weeks. 17 The tirzepatide group experienced significant, dose-dependent weight loss of up to 18.4%, while those in the placebo group experienced a 2.5% weight regain. Additionally, tirzepatide improved HbA1c levels, lipid profiles, waist circumference, fasting glucose, fasting insulin, and blood pressure, with a safety profile similar to that observed in SURMOUNT-1. SURMOUNT-4 was a phase III, double-blind RCT involving 670 adults with a BMI of ⩾30, or ⩾27 kg/m² with at least one weight-related complication, excluding those with T2DM. 16 The trial comprised a 36-week open-label lead-in period where all participants received tirzepatide (10 or 15 mg), followed by a 52-week double-blind phase, where participants were randomized to continue on tirzepatide or switch to placebo. Those continuing on tirzepatide achieved a total weight loss of 25%, with significant improvements in waist circumference, lipid profiles, and blood pressure. From week 36 to week 88, the mean percent weight change was −5.5% with tirzepatide versus a 14.0% weight regain in the placebo group, demonstrating a substantial regain of lost weight upon tirzepatide withdrawal.
In fact, emerging data indicate that the effects of tirzepatide on glycemic control and weight loss may supersede those of GLP-1 RAs,49,50 with the growing body of real-world evidence and upcoming head-to-head trials (e.g., SURMOUNT-5) expected to shed more light on potential differential effects of novel antiobesity medications in the near future. At present, it should be emphasized however, that based on the robust design of the SELECT trial, the so-far available data favor the use of the GLP-1 RA semaglutide for cardiovascular risk reduction in nondiabetic overweight or obese adults with established cardiovascular disease or at high cardiovascular risk.
The following limitations of the current meta-analysis need to be acknowledged. First, due to lack of individual participant data and the reliance on aggregate data, meta-analyses of participant characteristics in association with the risk of MACE, MI, or stroke could not be performed. Second, regarding stroke subtypes, the interpretability of our findings was significantly limited by the extremely low number of reported outcome events. Consequently, no reliable inferences regarding potential associations between GLP-1 RAs or GIP/GLP-1 RAs and stroke in overweight or obese populations can be drawn based on the so-far available evidence. Third, the generalizability of the current findings is limited by the fact that RCTs on GLP-1 RAs or GIP/GLP-1 RAs—with the exception of SELECT—included overweight or obese adults without cardiovascular comorbidities; thus, real-word data and future CVOTs are warranted to corroborate our findings. Despite these limitations, a significant number of RCTs comprising a total population of 34,575 participants contributed data to the pooled primary outcomes analyses, with the exceptionally low heterogeneity of reported cardiovascular outcomes from included RCTs supporting the robustness of the present results. Finally, to the best of our knowledge, this is the first to date meta-analysis evaluating the comparative efficacy of GLP-1 versus GIP/GLP-1 RAs and the largest assessing the cardiovascular safety and efficacy of these novel medications in the nondiabetic overweight or obese adult population.
Conclusion
In conclusion, the findings of the present systematic review and meta-analysis indicate that GLP-1 RAs and GIP/GLP-1 RAs for treatment of overweight and obesity in the absence of diabetes significantly attenuate the risk of MACE. Moreover, our findings suggest a favorable safety profile with significant survival benefits, reinforcing the utility of these agents in obesity management strategies. With respect to individual components of MACE, our meta-analysis demonstrates a significant reduction of the risk of MI with anti-obesity treatment, while further well-designed RCTs are needed to firmly ascertain the role of GLP-1 RAs and GIP/GLP-1 RAs for stroke prevention in overweight or obese individuals without diabetes.
Supplemental Material
sj-docx-1-tan-10.1177_17562864241281903 – Supplemental material for Risk of major adverse cardiovascular events and all-cause mortality under treatment with GLP-1 RAs or the dual GIP/GLP-1 receptor agonist tirzepatide in overweight or obese adults without diabetes: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tan-10.1177_17562864241281903 for Risk of major adverse cardiovascular events and all-cause mortality under treatment with GLP-1 RAs or the dual GIP/GLP-1 receptor agonist tirzepatide in overweight or obese adults without diabetes: a systematic review and meta-analysis by Maria-Ioanna Stefanou, Lina Palaiodimou, Aikaterini Theodorou, Apostolos Safouris, Urs Fischer, Peter J. Kelly, Jesse Dawson, Mira Katan, Aristeidis H. Katsanos, Vaia Lambadiari, Sotirios Giannopoulos, Andrei V. Alexandrov, Gerasimos Siasos and Georgios Tsivgoulis in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864241281903 – Supplemental material for Risk of major adverse cardiovascular events and all-cause mortality under treatment with GLP-1 RAs or the dual GIP/GLP-1 receptor agonist tirzepatide in overweight or obese adults without diabetes: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tan-10.1177_17562864241281903 for Risk of major adverse cardiovascular events and all-cause mortality under treatment with GLP-1 RAs or the dual GIP/GLP-1 receptor agonist tirzepatide in overweight or obese adults without diabetes: a systematic review and meta-analysis by Maria-Ioanna Stefanou, Lina Palaiodimou, Aikaterini Theodorou, Apostolos Safouris, Urs Fischer, Peter J. Kelly, Jesse Dawson, Mira Katan, Aristeidis H. Katsanos, Vaia Lambadiari, Sotirios Giannopoulos, Andrei V. Alexandrov, Gerasimos Siasos and Georgios Tsivgoulis in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
