Abstract
Migraine with brainstem aura (MBA) constitutes a rare subtype of migraine, characterized by aura symptoms including vertigo, dysarthria, diplopia, tinnitus, ataxia, and impaired consciousness. Patients with MBA have been reported to exhibit abnormal electroencephalograms (EEGs) featuring diffuse slow-wave activity and bilateral slowing of the posterior head activity. Notably, there have been no documented reports of abnormal discharges specifically localized in the anterior head. The presence of abnormal EEG discharges in MBA patients who experience loss of consciousness may lead to potential misdiagnosis, especially as epilepsy, in the early stages. This study describes three patients who were ultimately diagnosed with MBA, offering a retrospective analysis of their clinical features, electroencephalographic manifestations, and diagnostic procedures. In the three cases described, all patients were female, aged 16–21, and had been admitted to the hospital due to recurrent loss of consciousness. They exhibited a consistent EEG pattern, characterized by paroxysmal moderate-to-high amplitude theta activity in the anterior head, interspersed with spikes and sharp waves. Laboratory tests and imaging studies yielded unremarkable results. They all received a diagnosis of epilepsy and were treated with antiseizure medication, which proved ineffective. After evaluation by an epilepsy specialist, they received a final diagnosis of MBA. Following flunarizine administration, all three patients demonstrated improvement, with no subsequent occurrences of loss of consciousness during the follow-up period. This study describes the pattern of abnormal discharges that may be observed in the interictal EEGs of these MBA patients, which is characterized by a predominantly anterior head pattern. Recognizing this specific condition constitutes a crucial element in the differential diagnosis of epilepsy, with the aim of preventing misdiagnosis. Concurrently, we investigate their pathophysiological origins.
Keywords
Introduction
Migraine with brainstem aura (MBA) is a rare subtype of migraine, accounting for approximately 1.5% of headaches and between 6.6% and 10% of migraines accompanied by aura. 1 Initially, Bickerstaff 2 described it as “basilar-artery migraine” in 1961. Currently, the Headache Classification Committee of the International Headache Society redefines “basilar-artery migraine” as MBA. Yamani et al. 3 reported electroencephalogram (EEG) abnormalities in 56% of MBA patients, noting the most common as diffuse and bilateral posterior slowing, with additional findings including excessive beta activity, frontal intermittent rhythmic delta activity (FIRDA), and bilateral sharp and slow wave discharges.
Migraine and epilepsy are both paroxysmal disorders that can overlap in terms of triggers, genetic predisposition, transient neurological, autonomic, and psychological symptoms. 4 Notably, in rare clinical scenarios, these disorders may exhibit complete symptom overlap, and in that case, we can have as diagnosis the so-called “ictal epileptic headache” (IEH). This entity is characterized by headaches fulfilling physiological criteria of epileptic seizures (e.g., ictal EEG discharges), where cephalalgia itself constitutes the sole ictal manifestation of epilepsy.5,6 Furthermore, children/adolescents with migraine with aura may present “atypical aura,” such as complex visual aura, sensory deficits, and motor deficits, which may mimic focal epileptic activity, thereby complicating differential diagnosis. 7 Consequently, EEG recognition is crucial for the differential diagnosis of epilepsy and migraine. Although autonomic symptoms and signs (e.g., pallor, tachycardia, and nausea/vomiting) can be present in both epilepsy and migraine, their temporal profiles and phenotypic signatures diverge across migraine phases (premonitory, ictal, and postdromal) and epilepsy subtypes (focal vs generalized). 8 These discriminative features underscore the critical role of autonomic symptomatology in differentiating epilepsy from migraine, particularly in younger populations where phenotypic overlaps are prevalent yet diagnostically challenging.
Additionally, MBA is often misdiagnosed as epilepsy due to the complexity of its signs and symptoms, coupled with abnormal EEG discharges. This can have a serious impact on the lives of patients misdiagnosed with epilepsy, such as adverse effects from taking unnecessary antiseizure medications (ASMs), reduced quality of life, the development of psychological problems such as anxiety, depression, or sleep disorders, and financial burden. Therefore, an accurate differential diagnosis between MBA and epilepsy is critical to ensure proper treatment and management of the underlying condition.
In this context, we present three cases of MBA with impaired consciousness, in which EEG revealed abnormal discharges.
Patients and methods
This retrospective case series analyzed three patients derived from a cohort of 536 individuals evaluated for recurrent loss of consciousness at the Epilepsy Clinic of the Second Affiliated Hospital, Chongqing Medical University (2019–2023). Exclusions comprised 523 patients with confirmed epilepsy (including refractory cases) or syncope. Among the remaining patients, there were three patients who presented with recurrent loss of consciousness with interictal EEG abnormal discharges, previously diagnosed with epilepsy but ineffective on antiepileptic seizure medication. Following comprehensive reevaluation—including detailed history, EEG analysis, and diagnostic therapeutic trials—all three cases were reclassified as MBA. This study encompassed the collection and analysis of demographic data, clinical manifestations, laboratory and imaging tests, treatment courses, and prognoses throughout the disease progression. Upon admission, all patients underwent 24-h video EEG monitoring using a 16-lead configuration (Fp1, Fp2, F3, F4, C3, C4, P3, P4, O1, O2, F7, F8, T3, T4, T5, T6). Electrodes were placed using a 500-Hz electroencephalograph (solar6000N) according to the International 10–20 System (American Clinical Neurophysiology Society). Prognostic status was assessed through telephone interviews. No patients reported a family history of epilepsy. All patients and/or their parents were satisfied with their diagnosis and treatment and provided written informed consent. Our institution does not require ethical approval for reporting individual cases or case series.
Case reports
Patient 1
A girl aged 16 years first presented at the age of 13 with sudden onset of vertigo, which was described as seeing things spinning outside and feeling unsteady on her feet for about 5 min. She then fell to the ground and lost consciousness for 15 min. Her Glasgow Coma Scale (GCS) score was 5. Upon awakening, she experienced persistent throbbing temporal and occipital-parietal headache, accompanied by nausea, photophobia, and palpitations for 6 h. During the ictal period, she developed dysarthria, which manifested as a disturbance of voice production consistent with dysphonia. She had similar paroxysmal attacks 2–5 times per month for the next 3 years. Long-term EEG monitoring in the interictal period indicates abnormal discharges of intermittent medium–high amplitude (80–180 μV) θ activity in the frontal and temporal lobes bilaterally, interspersed with sharp waves, as well as occasional occurrences in the parietal lobes and central regions bilaterally (Figure 1(b)–(d)). Her interictal EEG recordings demonstrated normal background activity (Figure 1(a)).

(a) Background EEG during the waking period of patient 1. (b–d) The abnormal EEG discharges of patient 1 showed medium–high amplitude (80–180 μV) θ activity in the frontal and temporal lobes bilaterally, interspersed with sharp waves, as well as occasional occurrences in the parietal lobes and central regions bilaterally.
Previously, she was misdiagnosed with focal epilepsy, attributed to seizure loss of consciousness and abnormal EEG discharges, and received treatment with oral levetiracetam (LEV; 500 mg twice daily) for 1 year, which was followed by the addition of oral oxcarbazepine (300 mg twice a day) because her symptoms did not improve. These two ASMs were taken together for 1 year. However, the ASMs proved ineffective. Following re-evaluation by an epileptologist, she was diagnosed with MBA and underwent a 3-month treatment regimen of flunarizine (10 mg daily at bedtime) and 300 mg of ibuprofen during acute attacks, which resulted in the complete cessation of migraines. During the follow-up, she did not encounter any subsequent episodes of loss of consciousness.
Patient 2
A 21-year-old female, who had been experiencing spontaneous vertigo attacks since the age of 17, which manifested as a sensation of imbalance in the body. Each attack lasted 5–8 min and was followed by unconsciousness. Coma lasted about 20 min. Documentation from the patient’s initial evaluation at an external hospital indicated a GCS score of 8 during the acute phase. Upon awakening, she experienced persistent throbbing pain, nausea, vomiting, and photophobia. These symptoms would completely resolve within approximately 24 h. Over the past 4 years, she has continued to have attacks at intervals of 3–6 months. On the initial day of her admission, she underwent 24-h video EEG monitoring. The abnormal interictal EEG revealed frequent intermittent full-lead medium–high amplitude (120–240 μV) θ activity during full wakefulness. This activity was interspersed with a few spikes and sharp waves, primarily in the anterior head (Figure 2(b)–(d)). The interictal EEG recordings revealed normal background activity (Figure 2(a)).

(a) Background EEG during the waking period of patient 2. (b–d) Patient 2 experienced frequent intermittent occurrences of medium–high amplitude (120–240 μV) θ activity in full leads, interspersed with a few spikes and sharp waves. These activities were characterized by the anterior head.
Similarly to Patient 1, she was misdiagnosed with epilepsy and prescribed oxcarbazepine (300 mg twice daily) for 1 year. Nevertheless, the treatment proved ineffective. She was ultimately diagnosed with MBA and was treated with oral flunarizine 10 mg for 3 months. Follow-up continued until January 2024, during which she experienced no further loss of consciousness. However, she still experienced occasional headaches, which were noticeably less severe than before.
Patient 3
A 16-year-old girl presented with a sudden onset of bilateral visual blurring 2 months earlier, manifested by an initial halo before the eyes, followed by a blurring of everything in front of her eyes and a gray tinge of everything she saw, which lasted for about a few minutes. Vertigo soon followed, manifested by a feeling of spinning and the need to hold on to a wall in order to stand, lasting about 10 min, with a severe, generalized throbbing headache, accompanied by photophobia and a state of confusion lasting 30 min. According to the patient’s mother’s description, the patient had a GCS score of 10. She had similar paroxysmal attacks once a week. Following a 24-h video EEG, findings indicated abnormal interictal discharges. The abnormal EEG manifestations were characterized by paroxysmal medium-to-high amplitude (50–130 µV) θ activity across all leads during full wakefulness, with more pronounced activity in the anterior head region. In addition, several spike-wave discharges were observed intermittently in the bilateral frontal regions (Figure 3(b)–(d)). Interictal EEG recordings revealed normal background activity (Figure 3(a)).

(a) Background EEG during the waking period of patient 3. (b–d) Patient 3, while fully awake, had paroxysmal occurrences of medium-to-high amplitude (50–130 μV) θ activity in the full leads, during which a few spike-wave discharges were interspersed in the frontal regions bilaterally.
Initially, owing to the patient’s recurrent loss of consciousness and EEG abnormalities, she was considered to have seizures and was prescribed oral oxcarbazepine (300 mg twice daily) for 3 weeks, which was ineffective. Upon reevaluation of the patient’s EEG and symptoms by an epileptologist, she was correctly diagnosed with MBA. Treatment with oral flunarizine (10 mg) was administered for 2 months. Upon follow-up, the patient has not experienced any subsequent loss of consciousness; however, she still suffers from occasional headaches.
Table 1 delineates the primary clinical characteristics of the three patients.
The primary clinical characteristics of the three patients.

Summary of inspection results
During the interictal period, neurological examinations of all patients revealed no abnormalities, and none of them had comorbidities such as anxiety and depression. Routine blood tests and cerebrospinal fluid analyses yielded normal results for all patients. Comprehensive evaluations, including prolonged video-electroencephalography (EEG), brain magnetic resonance imaging, computed tomography angiography, echocardiography, and 24-h Holter monitoring, yielded normal results without detectable abnormalities. Continuous cardiac monitoring, passive standing blood pressure testing, and tilt-table testing were administered to all patients; these investigations revealed no abnormalities. Collectively, these findings exclude syncope as a diagnostic consideration.
We summarize the primary clinical characteristics of the three patients at the time of the seizure (as shown in Table 1). In all three cases, EEG patterns exhibited relative similarity, featuring moderate-to-high amplitude θ activity in the anterior head, interspersed with spikes and sharp waves. Each occurrence lasted for 2–10 s. Furthermore, this EEG pattern is observable during both wakefulness and the early stages of drowsiness.
Discussion
Each of our patients experienced loss of consciousness promptly following approximately 5 minutes of vertigo, lasting for about 15–30 min. They exhibited a significant throbbing headache, which could occur before or after loss of consciousness. In a small percentage of patients, the headache may begin simultaneously with the aura, while in the majority of patients, the headache begins after the onset of the aura. 9 They all met the ICHD-3 criteria for MBA diagnosis (criterion 1.2.2). 10 However, this condition could easily be misdiagnosed as epilepsy since it usually manifests as a young individual suddenly loses consciousness and the EEG reveals abnormal discharges. At the same time, clinicians must consider the possibility of IEH. Parisi et al. 5 specify that the diagnostic criteria require headache attacks to be the sole clinical manifestation of epileptic seizures the EEG must demonstrate synchronization between epileptiform discharges and headache activity, alongside resolution of symptoms post-antiepileptic treatment. This distinction is critical, as overlapping symptoms between migraine and epilepsy frequently lead to misdiagnosis, as noted in a case review by Parisi et al. 6 In all three reported cases, patients exhibited neither responsiveness to antiepileptic drugs nor EEG epileptiform discharges synchronized with headache activity. Thus, a diagnosis of IEH can be confidently excluded.
However, after taking a thorough history and EEG patterns, we can identify some features that distinguish MBA from epilepsy. First, each attack has significant throbbing headaches, which is a core symptom of migraine, and the brainstem aura (including vertigo and impairment of consciousness) could last roughly between 15 and 30 min, which is significantly longer than the duration of epileptic aura (usually lasts less than 5 min). 11 Second, the patient’s attack performance cannot be explained by any of the International League Against Epilepsy (ILAE)-defined seizure types. 12 For instance, the symptoms do not exhibit the tonic and convulsive features typical of generalized tonic-clonic seizures, nor do they exhibit the hyperkinetic or postural seizures associated with frontal lobe epilepsy, nor the automatisms characteristic of temporal lobe epilepsy. Third, vertigo aura occurs infrequently in epilepsy. 13 Fourth, treatments with ASMs not targeting migraine prevention have proven ineffective. The administration of oxcarbazepine and levetiracetam to these three patients was ineffective in preventing migraines. If valproic acid, lamotrigine, and topiramate had been used, achieving the therapeutic objective might have been possible, given the migraine-preventive effects of these antiepileptic drugs. 14 Last but not least, we can distinguish them based on the EEG pattern. The EEGs of these three patients lacked typical interictal epileptiform discharges, such as typical spiking slow waves and spiking slow complex waves. 15 Instead, we found that positive EEG findings in MBA usually reveal a pattern of bilateral anterior head-dominant paroxysmal θ activity of moderate-to-high amplitude, interspersed with spikes and sharp waves.
This interictal discharge pattern is sometimes easily recognized as immature spike and waves, occurring in the early stages of childhood somnolence. Immature spike and waves, prevalent among children aged 3–10 years and observed in cases of febrile convulsions, is not inherently epileptic. This waveform is characterized by a low amplitude of spike, unstable waveform, and is observed solely in paroxysmal slow waves at the beginning of the drowsiness period. 16 In contrast, the pattern of abnormal discharges in the EEGs of our patients can manifest not only in the early stages of drowsiness but also during wakefulness. Furthermore, all our patients were aged between 16 and 21 years.
In the past, there have been many case reports and studies on abnormal EEG in migraine with aura.17,18 Reported abnormal EEGs include focal slow waves, peaked slow waves, posterior quadrant slow wave abnormalities, global slow waves, low-amplitude recordings, and hyperventilation response. Abnormal EEG findings in MBA have previously also been reported; reversible transient synchronous bilateral slow EEG activity with occipital predominance is the most characteristic.19,20 Investigations into MBA have identified EEG abnormalities potentially associated with brainstem structural dysfunction or ischemia in the posterior cerebral artery region, and irregular slow-wave activity in the posterior occipital region has been suggested to indicate ischemia.21–23 Among MBA patients with impaired consciousness, EEG manifestations often comprise diffuse slow delta activity or FIRDA, which is primarily recorded during episodes of impaired consciousness. 21 However, the interictal EEG appears normal. Their EEG abnormalities and impaired consciousness are hypothesized to stem from reduced cortical activation due to ischemia in the brainstem reticular formation, attributed to vascular dysfunction in the anastomotic lateral portion of the basilar artery and its branches.21,24
In our patients, the EEG patterns showed abnormal discharges during the interictal period, predominantly in the anterior head region. We posit that this abnormal discharge is potentially linked to the cortical origins in MBA patients. During the interictal period, patients with migraine exhibit cortical hyperexcitability, characterized by lower cortical excitability thresholds compared to healthy individuals.25–27 And, dysregulation of the excitatory/inhibitory (E/I) balance allows for the induction of cortical hyperactivation by internal or external factors, creating conditions for initiating the positive feedback loop of Cortical spreading depression (CSD). 28 Furthermore, it has been proposed that brainstem aura symptoms might have their origin in the cerebral cortex, with their pathophysiological mechanisms explicable through CSD.3,29 Therefore, we suggest that the paroxysmal medium–high amplitude θ oscillations observed on the EEG during the interictal period in patients with MBA may originate from cortical hyperexcitability. In our three patients, the aura manifested as several minutes of vertigo followed by loss of consciousness. Vertigo may stem from vestibular cortex dysfunction, particularly within the parietal-insular vestibular cortex, a core region of the vestibular cortex.30,31 Impaired consciousness might result from abnormal neural activation within specific networks in the prefrontal and posterior parietal cortex, 29 rather than stemming from dysfunction in the brainstem’s reticularis superior. Verrotti et al. 26 proposed that key pathophysiological alterations in headache may arise at any cortical or subcortical site, highlighting that migraine is not solely a brainstem disorder but also involves cortico-subcortical network dysfunction. This might explain the observed abnormal EEG discharges concentrated in the anterior head region in our patients. Additionally, a few spike-wave discharges were seen in all of our patients, which may be related to enhanced synaptic transmission in the thalamocortical network and dysregulation of the E/I balance. Tottene et al. 32 found that enhanced thalamocortical synaptic transmission and dysregulation of the E/I balance may be present in brains with migraine with aura, and that these mechanisms may lead to headaches, increased sensory gain, and dysfunction of sensory processing in migraine. Vinogradova 28 also concluded that episodic disruption of the E/I balance triggers epileptiform discharges in the epileptic cortex, which in patients with migraine causes CSD without resulting in seizures. This can be explained by the fact that the initiation mechanisms of CSD and seizure are similar, but the evolution is different. 33
In the past, it has been widely recognized that migraine is a phenomenon linked to only hypoexcitability, whereas epilepsy is linked to phenomena of hyperexcitability. This view has hampered the recognition of a possible “overlap” between these two “ictal” events, particularly in cases of IEH. 34 New evidence supports the “dysexcitability concept,” whereby both conditions arise from dynamic changes in cortical excitability states, rather than static hypoexcitability/hyperexcitability. 33 Genetic susceptibility may confer a predisposition to dysregulation of the E/I balance in migraine patients, resulting in transient hyperexcitability or hypoexcitability within the same individual.25,33,35
Ion channel dysfunction, resulting from certain genetic mutations such as the CACNA1A mutation affecting the voltage-dependent P/Q-type calcium channel, leads to increased Ca2+ influx and subsequent Glu release into the synaptic gap; mutations in the ATP1A2 gene impact the α2 isoform of Na+-K+-ATPase, which is predominantly expressed in astrocytes. These cells are essential for maintaining ion homeostasis and the reabsorption of glutamate (Glu). Astrocytes play a crucial role in the initiation of CSD. The primary role of astrocytes is to use the Na+-K+-ATPase and the Glu transporters excitatory amino acid transporter 1 and 2 (EAAT1 and EAAT2), to clear K+ and Glu from the synaptic gap.36,37 Dysfunctional Na+-K+-ATPase leads to a diminished capacity in astrocytes to clear K+ and glutamate, thereby elevating extracellular K+ and glutamate levels,25,38 increasing susceptibility to CSD-induced effects. 39 It has been proposed that the initial accumulation of extracellular K+ results from the repeated depolarization and repolarization of overexcited neurons in the cerebral cortex, and this accumulation then further depolarizes the neurons responsible for its release.40,41 Our theory and its potential mechanisms are depicted in Figure 4.
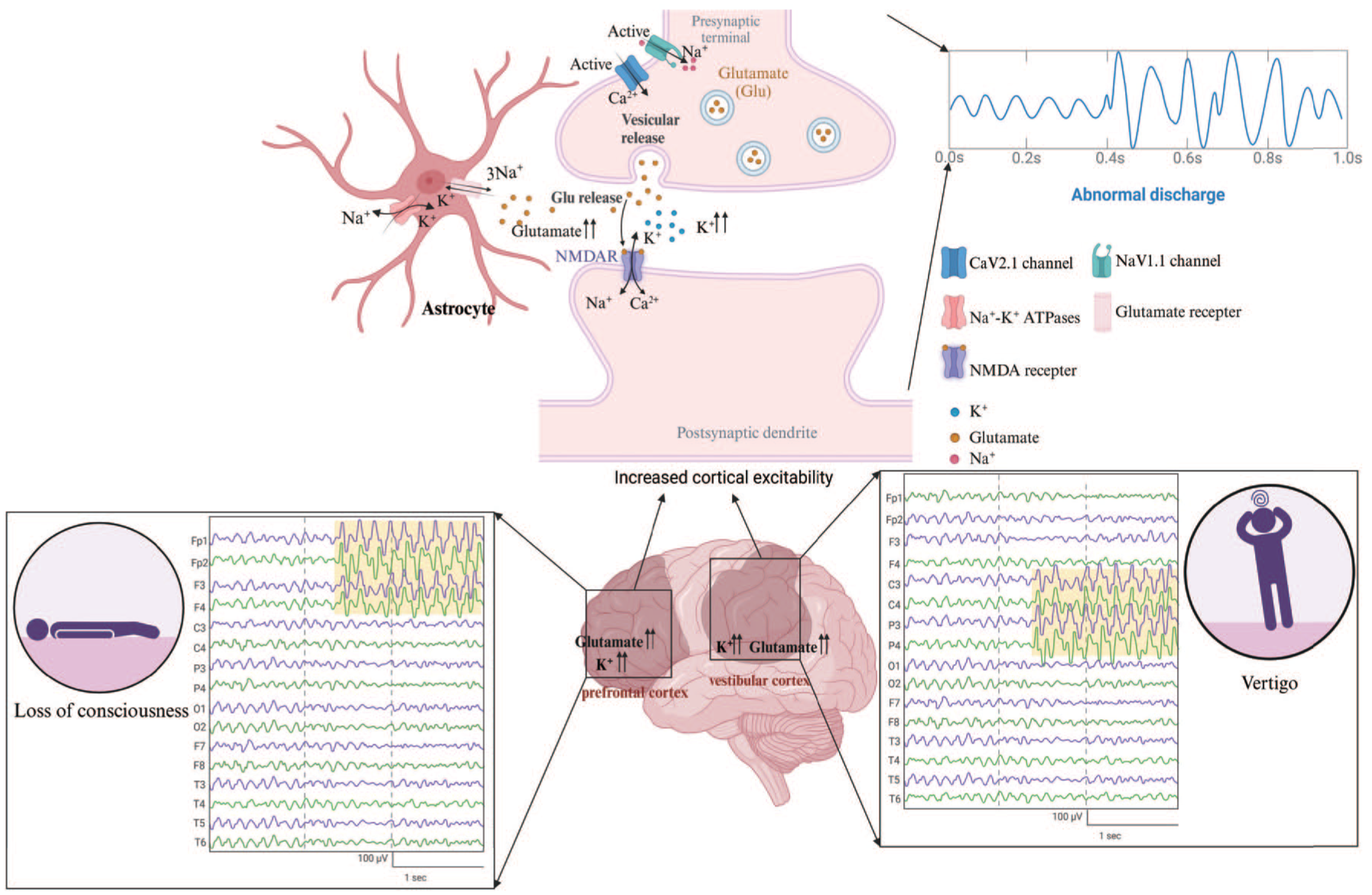
Hypothesis of mechanisms of impaired consciousness, vertigo, and abnormal EEG discharges in MBA patient. Due to ion channel dysfunctions, leading to a significant rise in extracellular K+ and glutamate levels in neurons, the cortical excitability during the interictal period of migraine patients is significantly elevated, consequently resulting in abnormal discharges within the interictal cortex. Due to the enhanced excitability of the PIVC, prefrontal and posterior parietal cortices, this hyperexcitability evolves into CSD, manifesting as auras of vertigo and impaired consciousness. The EEG findings are characterized by localized abnormal discharges in the parietal, central, and frontal lobes.
In our study, all three patients were treated with ibuprofen during headache attacks and were given oral flunarizine as prophylactic treatment due to their frequent headache attacks. Ibuprofen and flunarizine were chosen because they were readily available in our ward and effective. Additionally, flunarizine is a first-line prophylactic drug recommended by migraine management guidelines.42,43 The observation that our patient was unresponsive to LEV and oxcarbazepine, yet demonstrated a positive response to flunarizine, indirectly supports the ion channel mechanism hypothesis for cortical hyperexcitability in MBA patients. The importance of calcium channels in CSD has been elucidated, with P/Q-type calcium channels playing a predominant role. 44 We speculate that, similar to familial hemiplegic migraine, P/Q-type calcium channels also play a major role in initiating CSD in MBA patients.45,46 Flunarizine is a non-selective calcium channel antagonist that effectively blocks Ca2+ inward flow through P/Q-type calcium channels, thereby reducing cortical excitability. In contrast, LEV is an agent that further inhibits neuronal N-type calcium channels by binding to synaptic vesicle proteins, especially SV2A. 47 Therefore, LEV cannot effectively prevent the occurrence of CSD. Some studies have found that LEV has no significant inhibitory effect on SD-induced epileptiform activity, 48 indicating that LEV is not sensitive to CSD. There is conflicting evidence about the efficacy of oral LEV for migraine prevention, 49 so more studies are needed to further explore this in the future. Oxcarbazepine has been shown to be ineffective in preventing migraine, and animal studies have demonstrated that oxcarbazepine does not inhibit diffuse cortical suppression.50,51
A distinctive feature of the ictal semiology of migraine aura is their gradual onset and progression and their long duration (Symptoms evolve over 5–20 min), in contrast to the sudden onset and short duration of epilepsy. 52 In addition, positive, visual, and sensory symptoms (e.g., flashes of light and paresthesias) are more common than negative symptoms in migraine aura. These reflect the fact that migraine aura is an underlying physiological phenomenon of slow onset and slow spread (CSD). 7 Previous studies suggested that almost all CSDs associated with migraine aura originate in the occipital lobe and usually present with visual aura, followed by sensory, motor, and speech symptoms.53–57 However, a recent review by Bolay et al. 58 summarized studies on CSD associated with migraine aura and concluded that the complex clinical features of migraine aura are difficult to explain by a single occipital lobe origin of CSD, suggesting that CSD likely originates in multiple cortical regions. Furthermore, recent clinical findings indicate that some migraine patients with “atypical aura” may exhibit co-occurring multiple aura symptoms or non-sequential symptom progression. 7 This study led to the development of an important theory, namely the “Multiple, Synchronous and Asynchronous, Cortical and Subcortical Spreading Depression.” This theory suggests that, unlike classical CSD, which is confined to the occipital cortex, simultaneous waves of depolarization may propagate to multiple cortical regions (e.g., parietal lobe, temporal lobe), or even subcortical structures (e.g., thalamus, basal ganglia), leading to atypical aura manifestations. Therefore, we propose that the presence of brainstem aura symptoms could be explained by a cortical origin.
It is critical to acknowledge that lifestyle modifications exert a more substantial influence on migraine prevention and symptom management compared to epilepsy. Recent studies indicate that structured interventions targeting regular sleep and meal, adequate hydration, limited consumption of caffeine, and stress mitigation demonstrate greater efficacy in reducing the frequency and severity of pediatric migraine.59,60 This phenomenon is likely attributable to migraine-specific neurovascular and neuroinflammatory pathways, which exhibit heightened sensitivity to behavioral and environmental modifications. In contrast, epilepsy management predominantly relies on ASMs, whereas lifestyle adjustments demonstrate more limited therapeutic impact in seizure reduction.61,62
Conclusion
We summarize three cases of adolescent female migraineurs experiencing clear impairment of consciousness and vertigo as an aura, which are classified as MBA according to the current ICHD-3 classification. Based on its clinical features, abnormal EEG patterns, and possible pathogenesis, we propose that this type of “migraine with vertigo and loss of consciousness” may be related to cortical hyperexcitability that is transformed into CSD, and may be not originated from brainstem dysfunction. It is the first time an EEG has been described in patients with MBA that shows paroxysmal medium–high amplitude θ activity interspersed with spikes and sharp waves, predominantly in the anterior head. This type of migraine, characterized by a specific age of onset, gender, symptoms, and abnormal EEG discharge patterns, may be misdiagnosed as epilepsy. The exact pathogenesis of migraine necessitates further exploration through animal or in vitro experiments.
Supplemental Material
sj-doc-1-tan-10.1177_17562864251378208 – Supplemental material for Migraine with brainstem aura with abnormal EEG discharges easily misdiagnosed as epilepsy: a case series study
Supplemental material, sj-doc-1-tan-10.1177_17562864251378208 for Migraine with brainstem aura with abnormal EEG discharges easily misdiagnosed as epilepsy: a case series study by Xianyun Liu, Jing Ran, Yu Tong, Wenqi Yang and Xi Peng in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
