Abstract
Background:
Sex differences in multiple sclerosis (MS) progression are poorly characterized, especially for disability worsening independent of relapses.
Objectives:
To assess sex-specific risks of progression independent of relapse activity (PIRA), relapse and magnetic resonance imaging (MRI) activity (PIRMA), and relapse-associated worsening (RAW) in a real-world MS cohort, quantify each event’s contribution to disability accumulation, and explore variation across clinical subgroups.
Design:
Inverse probability-weighted analysis of adults with relapsing-remitting MS ambispectively enrolled in a local registry who had their first recorded neurological evaluation within 12 months of MS onset.
Methods:
We used weighted conditional proportional hazard models for recurrent events, adjusted for visit/MRI frequency, to compare risks between sexes. We also tested the homogeneity of sex effects across prespecified subgroups: age at MS onset, symptom location at onset, presence of ⩾10 T2-hyperintense brain lesions, ⩾1 spinal T2 lesion, ⩾1 Gadolinium-enhancing brain lesion at baseline, initial MS treatment type, and percentage of follow-up time on disease-modifying therapy (DMT).
Results:
We included 492 people with MS (median age 44.0 years, interquartile range (IQR) 35.0–53.6; 68.9% women), followed for a median of 5.1 years (IQR 3.1–7.2). In the weighted cohort, women had higher hazards of PIRA (hazard ratio (HR) 2.44, 95% confidence interval (CI) 1.56–3.70; p < 0.001) and PIRMA (HR 2.13, 95% CI 1.25–3.70; p < 0.001), mainly postmenopausal (56.6% and 52.4%). RAW risk was similar (HR 1.07, 95% CI 0.54–2.11; p = 0.843), despite higher relapse rates in women (0.13 ± 0.18 vs 0.06 ± 0.16; p < 0.001). Men had greater Expanded Disability Status Scale worsening per PIRA (+0.29 ± 0.71 vs +0.16 ± 0.53; p = 0.023) and PIRMA (+0.25 ± 0.71 vs +0.09 ± 0.38; p = 0.001). Age ⩾50 at onset increased PIRA/PIRMA risk without sex interaction. Significant sex interaction was seen for onset symptom location (p < 0.001 for both outcomes) and initial DMT (p = 0.013 for PIRA; p = 0.022 for PIRMA).
Conclusion:
Women, especially postmenopausal, had higher PIRA/PIRMA risk, but disability worsening per event was greater in men, associations varied by onset phenotype and initial DMT strategy.
Keywords
Introduction
Multiple sclerosis (MS) is a chronic inflammatory and neurodegenerative disease of the central nervous system that disproportionately affects women and typically manifests during early adulthood. 1 Sex differences in MS incidence and relapse rates had long been recognized, with women exhibiting a higher susceptibility to the disease.2,3 Emerging evidence suggested that sex may also play a critical role in the dynamics of disability progression. 4 While relapse-associated worsening (RAW) had traditionally been the focus of disease monitoring in MS, it became evident that a significant proportion of disability accrual occurs independently of relapses. Accordingly, progression independent of relapse activity (PIRA) gained increasing attention as a key driver of sustained disability in MS, contributing to most of the disability accumulation even in patients diagnosed with relapsing-remitting MS (RRMS). 5 Recently, the concept of progression independent of relapse and magnetic resonance imaging (MRI) activity (PIRMA) further refined this paradigm by excluding subclinical disease activity. 6
Real-world cohort studies 7 and post-hoc analyses of clinical trials8,9 generally reported no consistent sex-based disparities in the risk of PIRA. While men were often described as having a more aggressive disease course and worse long-term disability outcomes7,10 this may have reflected a greater susceptibility to neurodegenerative processes despite a lower relapse rate. In contrast, women typically exhibited higher inflammatory activity, manifesting as increased relapse frequency and MRI lesion burden.10,11 However, evidence on sex differences in RAW is scarce; available studies suggested that, despite higher relapse frequency in women, the associated disability worsening per relapse may be greater in men.10,11 Overall, real-world data exploring sex-specific risks of PIRA, PIRMA, and RAW, as well as their respective contributions to cumulative disability, remain limited. Understanding these sex-specific patterns is clinically relevant, as they may inform personalized treatment strategies, guide monitoring priorities, and improve prognostic counseling for people with MS (pwMS). Identifying whether men and women differ in their risk of PIRA, PIRMA, RAW, and in the disability impact of these events could help clinicians optimize the timing and choice of disease-modifying therapies and tailor follow-up schedules according to individual risk profiles.
Hence, we aimed to assess sex-based differences in the risk of PIRA, PIRMA, and RAW in a well-characterized real-world cohort of pwMS. We further explored the sex-specific contributions of each PIRA/PIRMA event to overall disability accumulation and examined whether the risk of both outcomes varied across key clinical subgroups.
Methods
Data extraction
This study was based on data extracted from the Ravenna Multiple Sclerosis Registry (RMSR). The RMSR is an observational registry incepted in November 2021, including all individuals diagnosed with MS who are followed at the MS Center of Ravenna, S. Maria delle Croci Hospital, Italy. PwMS who were already under care prior to October 2021 were retrospectively enrolled, with data collected from existing medical records, including clinical documentation, treatment history, and neuroimaging reports. For these cases, data were systematically extracted and entered into the registry using a standardized data collection protocol to ensure consistency and accuracy. From October 2021 onward, all newly diagnosed or referred pwMS have been prospectively included in the registry. Data were continuously collected during routine clinical practice, through face-to-face visits conducted by Expanded Disability Status Scale (EDSS) by the same certified neurologists (M.F., A.S., I.P., M.G.P.). During these visits, detailed information was recorded on disease phenotypes, relapses, disability progression, treatment exposure, and MRI findings. Clinical data were entered into the registry in real time or shortly after the visit, ensuring a comprehensive and up-to-date dataset. For this analysis, data extraction was executed on May 20, 2025.
Study population
We included people with a diagnosis of RRMS established according to the 2017 revision of the McDonald Criteria, as per international consensus. 12 Inclusion criteria were as follows: (1) age at MS onset ⩾18 years; (2) availability of a baseline EDSS score within 12 months of MS onset; (3) a minimum follow-up duration of 12 months; (4) ⩾1 EDSS assessment every 6 ± 3 months through face-to-face visits throughout the follow-up; (5) ⩾1 brain MRI with gadolinium and a complete spinal MRI within 6 months of the initial clinical evaluation; and (6) ⩾1 brain MRI every 12 ± 6 months throughout the follow-up. Additional visits and MRI exams were conducted in case of suspected relapses.
Non-inclusion criteria were: (1) a progressive (primary or secondary) disease course at baseline; (2) MS onset occurring ⩾12 months prior to the first available EDSS evaluation; and (4) pregnancy during follow-up, due to its potential confounding impact on disease course and disability progression. 13
Baseline EDSS was defined as the score recorded at the first neurological evaluation and confirmed 6 months later, following a roving EDSS approach. 14 If the initial evaluation occurred ⩽30 days of a relapse, including the initial clinical event, baseline EDSS was redefined as the first score assessed ⩾6 months after the relapse (or ⩾3 months if it was not the first relapse), and confirmed after 6 months. 14
Menopausal status and age at menopause were recorded at baseline based on self-reported information provided by the patients during clinical evaluations. Women were classified as postmenopausal if they reported having stopped menstruating (i.e., no menstrual bleeding for ⩾12 consecutive months), while those reporting ongoing menstrual cycles were classified as premenopausal.
Study outcomes
Confirmed disability accrual (CDA) was defined as a confirmed 6-month disability increase from the study baseline, measured by the EDSS (⩾1.5-point increase for baseline EDSS = 0, ⩾1.0-point increase for baseline EDSS between 1.0 and 5.0, and ⩾0.5-point increase for baseline EDSS >6.0). 14 Date of CDA was assigned at the first visit when the EDSS increase was recorded.
PIRA was defined as a CDA that occurred in the absence of a relapse that occurred within 90 days after or within 30 days before. 15
PIRMA was defined as a PIRA event in the absence of new or enlarging T2-hyperintense lesions and/or Gd-enhancing lesions on brain MRI in the 12 months preceding the event. 16
RAW was defined as a CDA in which the EDSS worsening occurred within 90 days after or within 30 days before the onset of a relapse. 15
In the case of multiple CDAs, the EDSS was re-baselined after each event. Specifically, the EDSS score at the time of each 6-month disability increase was considered the new baseline for evaluating subsequent events. 15
Statistical analysis
Inverse probability weighting
To achieve balanced baseline characteristics between women and men, we applied Inverse Probability Weighting (IPW) to generate a pseudo-weighted cohort. Propensity scores were estimated using a logistic regression model including the following predefined baseline variables: age at MS onset, baseline EDSS score, symptom location at onset (supratentorial, optic neuritis, brainstem-cerebellum, or myelitis), and baseline MRI findings (⩾1 T1 gadolinium-enhancing brain lesion, ⩾10 T2-hyperintense brain lesions, and ⩾1 T2-hyperintense spinal lesion). Initial MS treatment was also considered and categorized as either high-efficacy disease-modifying therapies (HE-DMTs), including monoclonal antibodies, sphingosine-1-phosphate receptor modulators, cladribine, or platform DMTs, including teriflunomide, dimethyl fumarate, and interferons. Among other immunosuppressants, mitoxantrone and cyclophosphamide were assimilated to HE-DMTs, whereas azathioprine was assimilated to platform DMTs. Lastly, we included the percentage of follow-up time on HE or platform DMTs as a matching variable. This metric accounted for treatment switches or temporary discontinuations. Gaps ⩽3 months were not classified as therapy interruptions. For DMTs with recognized prolonged pharmacological effects, an estimated duration of action was applied. Specifically, an effect duration of 10 months was assumed for rituximab and ocrelizumab; 5 years for alemtuzumab and autologous hematopoietic stem cell transplantation; 6 months for mitoxantrone and cyclophosphamide; 2 months for natalizumab, siponimod, and ozanimod; 3 months for ofatumumab; and 12 months for cladribine.17,18
Propensity scores were then used to assign inverse probability weights to each case. To test the balance between groups, standardized mean differences (MD) were calculated for each covariate. We also checked the overlap of propensity score distributions between groups to ensure sufficient comparability and applied stabilized weights to reduce the impact of extreme values and improve the reliability of the model.
Outcomes analysis
Cumulative hazards of PIRA, PIRMA, and RAW were estimated using weighted conditional proportional hazard models for recurrent events (Andersen-Gill models) adjusted for visit frequency (all outcomes) and brain MRI frequency (PIRMA only). Kaplan-Meier survival curves were used to visualize the overall risk for each outcome, with the log-rank test applied to determine the statistical significance of differences between sexes. Additionally, we calculated the total EDSS increase for each individual by summing the EDSS changes associated with all documented PIRA and PIRMA events and compared these values between sexes to assess their sex-specific contributions to overall disability accumulation. We used the two-tailed Welch’s t test to assess statistical significance. Lastly, we compared the mean number of PIRA, PIRMA, and RAW events during follow-up between postmenopausal and premenopausal women using the Mann–Whitney U test.
Subgroup analysis
Subgroup analysis for the risk of PIRA and PIRMA in the weighted cohort was performed in 7 prespecified subgroups: age at MS onset <50 or ⩾50 years, MS onset symptom location (supratentorial, optic neuritis, brainstem-cerebellum, or myelitis), presence of ⩾10 T2-hyperintense lesions at baseline brain MRI, presence of ⩾1 T2-hyperintense lesion at baseline spine MRI, presence of ⩾1 Gd-enhancing lesion at baseline brain MRI, first MS treatment type (DMT platform, HE-DMT, or not treated), percentage of follow-up time on any DMT (<50% or ⩾50%). Odds ratios (ORs) with 95% confidence intervals (CIs) were calculated for women versus men within each subgroup, along with the corresponding p-value. We used a generalized linear model with sex, subgroup variable, and their interaction term as independent variables to determine the homogeneity in the effect of sex by each binary subgroup variable. Interaction for multi-level subgroup variables was tested using the likelihood ratio test. The p-value was presented for the interaction term.
Given the observational nature of the study, a prespecified sample size calculation was not required, as this analysis was based on all eligible patients available in the registry during the study period. All statistical analyses were performed using R software (R Core Team, R Foundation, Wien, Austria), version 4.2.2. Statistical significance was set at a p-value <0.05.
Results
Of the 1003 pwMS in the RMSR, 492 were included in the analysis (median age at baseline: 44.0 years, IQR 35.0–53.6 years). Among these, 339 (68.9%) were women and 153 (31.1%) were men (Figure 1). In the unweighted cohort, the median follow-up time was 6.2 years (3.7–8.7 years) in women and 5.1 years (IQR 3.1–7.2 years) in men.

Study flow-chart.
Inverse probability weighting
Weighted and unweighted baseline characteristics are summarized in Table 1. The weighted pseudo-population consisted of 260 women (52.8%) and 232 men (47.2%). No missing data were recorded in the variables used to calculate the propensity scores. Overall, a satisfactory balance was achieved for all baseline variables, which showed a standardized difference (SD) in propensity scores below 0.10. Balance diagnostics indicated that the samples were well-weighted, with a mean SD in propensity scores between groups of 0.015 (acceptable balance <0.25) and a variance ratio of propensity scores of 1.51 (acceptable range: 0.5–2). 19 Graphical representations of covariate balance distributions further confirmed the overall quality of the weighting process (Figure S1).
Baseline characteristics.
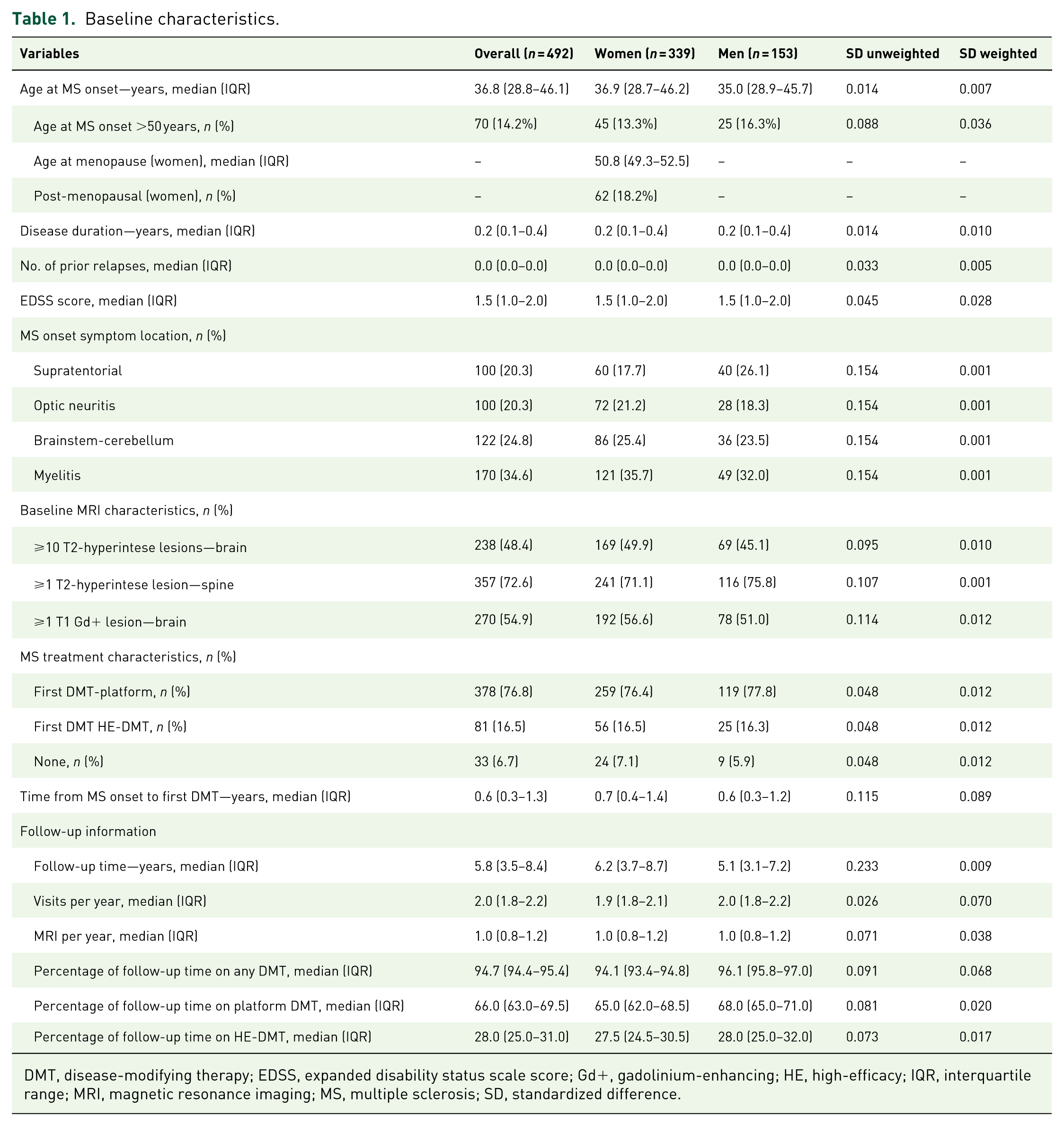
DMT, disease-modifying therapy; EDSS, expanded disability status scale score; Gd+, gadolinium-enhancing; HE, high-efficacy; IQR, interquartile range; MRI, magnetic resonance imaging; MS, multiple sclerosis; SD, standardized difference.
Outcomes analysis in the weighted cohort
Overall, CDA was recorded in 85 (32.7%) women and 35 (15.1%) men (risk difference (RD) + 17.6% (95% CI 10.3%–24.9%); p < 0.001). The median number of CDA events was slightly higher in women (1.0, IQR 0.0–1.0) versus men (0.0, IQR 0.0–1.0; p = 0.055). The median EDSS score at last follow-up visit was significantly higher in men (2.0, IQR 1.5–2.5) compared to women (1.5, IQR 1.0–2.0; p < 0.001).
Progression independent of relapse activity
At least a single PIRA event was recorded in 61 (23.4%) women and 22 (9.5%) men in the weighted cohort (RD, +13.9% (95% CI 8.1%–20.7%); p < 0.001). Overall, women had a significantly higher mean number of PIRA events during the available follow-up compared to men (0.28 ± 0.57 vs 0.12 ± 0.46; MD +0.16 events; p = 0.001; Figure S2(A)). In the analysis of recurrent PIRA events using the Andersen-Gill model adjusted for visit frequency, women showed a significantly higher hazard of experiencing a PIRA compared to men (HR 2.44, 95% CI 1.56–3.70; p < 0.001). The corresponding PIRA-free survival curves are shown in Figure 2 (log-rank test p-value <0.001). Notably, when comparing cumulative disability worsening across sexes by summing the EDSS changes associated with each documented PIRA event, men showed a significantly greater total mean EDSS increase than women (+0.29 ± 0.71 vs +0.16 ± 0.53; MD +0.13 EDSS points; p = 0.023; Figure 3). Among women, the majority of PIRA events occurred in postmenopausal individuals (n = 43/76 total PIRA events, 56.6%). Postmenopausal women experienced a significantly higher mean number of PIRA events compared with premenopausal women (0.35 ± 0.60 vs 0.19 ± 0.40; MD +0.16 events, p = 0.014).

PIRA-free survival curves stratified by sex in the weighted cohort.
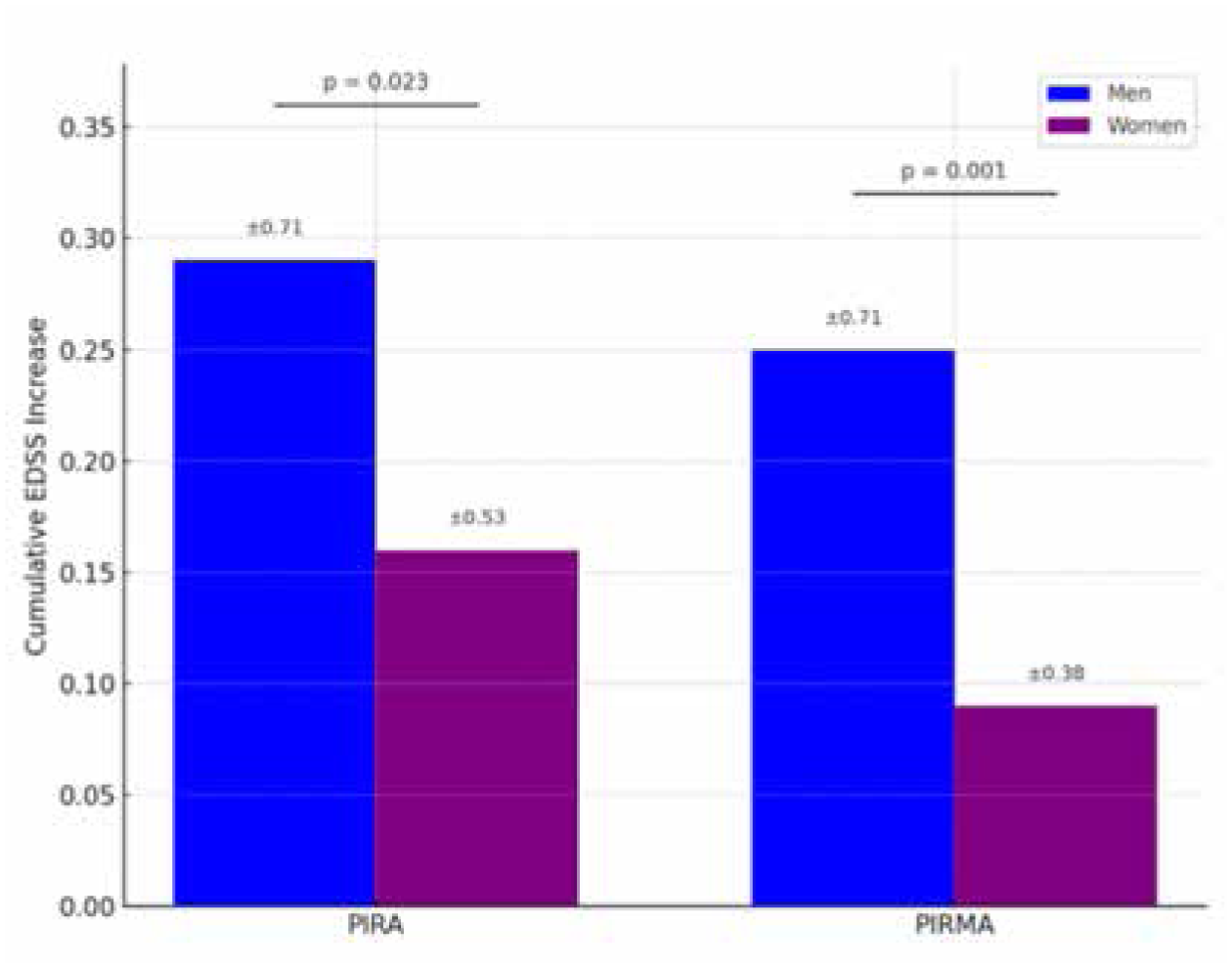
Comparison of cumulative EDSS changes due to PIRA and PIRMA events between sexes.
Progression independent of relapse and MRI activity
PIRMA occurred in 56 (21.5%) women and 16 (6.9%) men in the weighted cohort (RD +14.6% (95% CI 8.7%–20.6%); p < 0.001). PIRMA events accounted for most PIRA events in both sexes, and this proportion did not significantly differ between women and men (91.8% vs 72.7%; p = 0.058). Overall, the mean number of PIRMA events was significantly higher in women compared to men (0.27 ± 0.62 vs 0.09 ± 0.43; MD +0.18 events, p < 0.001; Figure S2(B)). The Andersen-Gill model adjusted for visit and MRI frequency showed that the hazard of experiencing a PIRMA event was significantly higher in women than in men (HR 2.13 (95% CI 1.25–3.70); p < 0.001), as confirmed by the corresponding survival curves (log-rank test p < 0.001; Figure 4). Similarly to PIRA, for PIRMA events, the comparison of cumulative disability worsening between sexes revealed a significantly greater accrual of disability in men compared to women (+0.25 ± 0.71 vs 0.09 ± 0.38; MD +0.16 EDSS points, p = 0.001; Figure 3). Among women, PIRMA events occurred mostly in postmenopausal individuals (n = 33/63 total PIRMA events, 52.4%). Postmenopausal women had a significantly higher mean number of PIRMA events compared with premenopausal women (0.30 ± 0.65 vs 0.23 ± 0.55; MD +0.07 events, p = 0.021).

PIRMA-free survival curves stratified by sex in the weighted cohort.
Relapse-associated worsening
RAW occurred in 24 (9.2%) women and 13 (5.6%) men (RD +3.6% (95% CI −1.0% to 8.2%); p = 0.170). Mean number of RAW events was similar (0.10 ± 0.33 vs 0.06 ± 0.28; MD +0.04 events, p = 0.196; Figure S2(C)). Women and men showed similar hazards for RAW events (HR 1.07, 95% CI 0.54–2.11; p = 0.843; Figure 5). Notably, the mean annualized relapse rate during the available follow-up was significantly higher among women than in men (0.13 ± 0.18 vs 0.06 ± 0.16; MD +0.07, p < 0.001). RAW events were more frequent in premenopausal women (n = 27/39, 69.2%), who had a significantly higher mean number of events compared with postmenopausal women (0.15 ± 0.35 vs 0.05 ± 0.20; MD +0.10 events, p = 0.015).

RAW-free survival curves stratified by sex in the weighted cohort.
Subgroup analysis for the risk of PIRA and PIRMA in the weighted cohort
Subgroup analyses revealed consistent sex-related differences in the risk of PIRA and PIRMA, with women showing a significantly higher risk compared to men across several clinical and radiological categories. The association was robust among persons with disease onset ⩾50 years (OR 8.33, 95% CI 2.50–25.00; p = 0.001 for PIRA and OR 11.11, 95% CI 3.03–50.00 for PIRMA; Table 2). While several subgroup-specific associations reached statistical significance in both models, interaction tests confirmed significant heterogeneity for both outcomes only in relation to MS onset symptom location (p for interaction <0.001 for both PIRA and PIRMA) and first treatment type (p for interaction = 0.013 for PIRA and 0.022 for PIRMA). In particular, the risk of both PIRA and PIRMA was more pronounced in women with supratentorial or optic neuritis onset, and in those who were untreated or initially treated with platform DMT (Table 2 and Figure S3).
Subgroup analysis for PIRA and PIRMA in the weighted cohort.

CI, confidence interval; DMT, disease-modifying therapy; Gd, gadolinium; HE, high-efficacy; MRI, magnetic resonance imaging; MS, multiple sclerosis; OR, odds ratio; PIRA, progression independent of relapse activity; PIRMA, progression independent of relapse and magnetic resonance imaging activity.
Statistical significant p-values are reported in bold.
Discussion
This inverse-probability weighted analysis from a monocentric MS registry showed that women with RRMS had a significantly higher risk of PIRA and PIRMA than men over a median follow-up of 5 years. Among women, these events were more frequent postmenopausally, while RAW was more common premenopausally, suggesting a role of menopausal status in modulating both the risk and clinical expression of progression. Despite their lower incidence, PIRA and PIRMA events in men were associated with greater disability accrual, and the median EDSS at last evaluation was significantly higher in men, pointing to a more severe or less compensable form of progression once clinically manifest. The risk of RAW did not differ significantly between sexes. Sex-related differences in PIRA and PIRMA varied across subgroups, particularly by onset phenotype and treatment strategy, with a higher risk in women with supratentorial or optic neuritis onset and in those untreated or started on platform DMTs. PIRA/PIRMA were also strongly associated with late-onset MS (⩾50 years) in both sexes, underscoring the central role of age at onset in shaping progression risk.
Previous observational studies10,20 and post-hoc analyses of trials such as EXPAND 8 and OPERA 9 reported no clear sex differences or slightly higher progression in men early in the disease, with faster male progression typically emerging after 10–15 years or in progressive forms. 7 Our findings suggest that women may develop earlier non-relapse-related progression, possibly influenced by hormonal, immune, and neurobiological factors.21–24 Menopause may further increase vulnerability, as declining estrogen levels reduce neuroprotection and anti-inflammatory effects 25 consistent with the higher PIRA/PIRMA frequency post menopause in our cohort.
Although PIRA and PIRMA were less frequent in men, they produced greater disability accrual, supporting the hypothesis of a more severe neurodegenerative process with reduced compensatory capacity.2,4,10 For RAW, our findings align with previous reports of similar sex-related risk despite women’s higher relapse frequency,10,22,26 confirmed in our weighted analysis. This dissociation may reflect the mitigating effects of early and sustained high-efficacy DMT use. 27 The higher RAW frequency in premenopausal women suggests that menopausal status may also influence relapse expression.
The pronounced PIRA/PIRMA risk in women with supratentorial or optic neuritis onset may reflect the interplay between sex and lesion topography. These phenotypes, more common in women and linked to inflammatory courses,28,29 paradoxically showed greater silent progression, possibly due to more frequent initiation on lower-efficacy DMTs in patients with features perceived as favorable. 7 Higher risk in untreated or platform-treated patients supports evidence that these strategies may be insufficient to prevent early disability accrual,30,31 particularly in active disease, an effect more evident in women. From a therapeutic perspective, our findings suggest that sex-specific risks of progression should inform treatment strategies. The higher PIRA/PIRMA incidence in women, postmenopausally, supports early use of high-efficacy DMTs to limit relapse-independent disability accrual. While our study was not designed to directly compare treatment effects, these sex-specific patterns support an individualized therapeutic approach that considers menopausal status, age at onset, and disease phenotype when selecting and sequencing DMTs, balancing efficacy with long-term safety. Finally, the strong link between PIRA/PIRMA and late-onset MS, with no sex-related heterogeneity, supports age at onset as a robust predictor of early progression,32,33 likely reflecting reduced neuroprotective and reparative capacity in older patients.
Strengths and limitations
The main strength of this study lies in the rigorous selection of participants, ensuring complete data for all baseline and follow-up variables. We adopted a strict follow-up protocol with scheduled face-to-face visits (every 6 ± 3 months) and brain MRI (every 12 ± 6 months), enabling consistent and thorough monitoring. We also applied a rigorous IPW approach to reduce the influence of potential confounding variables, creating a pseudo-population with balanced baseline covariates and improving the validity of causal inferences from observational data. Limitations include the retrospective nature of data collection for patients enrolled up to October 2021, which may have introduced inconsistencies. Residual confounding from unmeasured variables cannot be excluded, as we did not systematically collect information on risk factors such as smoking or comorbidities, which may influence disability progression.34,35 Likewise, genetic and environmental determinants such as HLA-DRB1 status or recent Epstein-Barr virus infection/reactivation at baseline, which may have a relevant impact on both clinical and MRI outcomes, were not available. Additionally, menopausal status was based on self-reported menstrual history, which may be subject to recall bias and further complicated by the irregularity of menstrual bleedings in the years preceding menopause. No biochemical confirmation was available, as such testing is not performed in the routine clinical monitoring of pwMS. Moreover, menopausal classification was determined at baseline and transitions from pre- to post-menopause during follow-up were not accounted for in the analysis, potentially resulting in misclassification for some individuals. Another limitation is the relatively short follow-up (median 5 years), which may have underestimated late-onset PIRA and PIRMA incidence and reduced the likelihood of detecting multiple events in the same patient, limiting the assessment of long-term disability dynamics. Furthermore, disability progression was assessed exclusively using the EDSS, which, although widely adopted in both clinical trials and real-world studies, only partially captures the complexity of MS-related functional decline. Additional measures such as the 9-Hole Peg Test, Timed 25-Foot Walk, Symbol Digit Modalities Test, and fluid biomarkers like neurofilament light chain could have provided a more comprehensive assessment of motor, cognitive, and subclinical changes. However, these measures are not routinely collected in standard clinical practice in our setting. Finally, as this is a monocentric cohort consisting almost entirely of individuals of European ancestry, external validity is limited. Ethnic and geographic variability can influence MS presentation and progression, and sex-related differences may manifest differently across populations with distinct genetic, environmental, and socioeconomic profiles.
Conclusion
In conclusion, our study highlights the multifaceted role of sex in the trajectory of disability progression in RRMS. Women showed a higher risk of relapse-independent worsening (PIRA/PIRMA), particularly postmenopausally, whereas men experienced greater disability accrual per event. These results support the view that MS progression is both heterogeneous and sex-specific, with distinct temporal and phenotypic patterns. The influence of sex also varied across key clinical subgroups, suggesting an interplay with disease onset features and initial treatment. Overall, these findings underscore the need for personalized MS care that considers sex, age at onset, menopausal status, clinical presentation, and DMT history in shaping disease trajectories.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251376807 – Supplemental material for Sex differences in relapse-independent and relapse-associated disability progression in relapsing-remitting multiple sclerosis: a real-world inverse-probability weighted study
Supplemental material, sj-docx-1-tan-10.1177_17562864251376807 for Sex differences in relapse-independent and relapse-associated disability progression in relapsing-remitting multiple sclerosis: a real-world inverse-probability weighted study by Matteo Foschi, Damiano Marastoni, Ivan Panzera, Luca Mancinelli, Cristiana Ganino, Gianmarco Abbadessa, Lucio D’Anna, Francesca Gabriele, Simona Sacco, Elisabetta Signoriello, Alessandra Lugaresi, Elena Tsantes, Maria Grazia Piscaglia and Andrea Surcinelli in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864251376807 – Supplemental material for Sex differences in relapse-independent and relapse-associated disability progression in relapsing-remitting multiple sclerosis: a real-world inverse-probability weighted study
Supplemental material, sj-docx-2-tan-10.1177_17562864251376807 for Sex differences in relapse-independent and relapse-associated disability progression in relapsing-remitting multiple sclerosis: a real-world inverse-probability weighted study by Matteo Foschi, Damiano Marastoni, Ivan Panzera, Luca Mancinelli, Cristiana Ganino, Gianmarco Abbadessa, Lucio D’Anna, Francesca Gabriele, Simona Sacco, Elisabetta Signoriello, Alessandra Lugaresi, Elena Tsantes, Maria Grazia Piscaglia and Andrea Surcinelli in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
