Abstract
Background:
Pregnancy in patients with multiple sclerosis (MS) is accompanied by a decline of relapse activity with increased risk of relapses 3 months post-partum, for unknown reasons. Eomesodermin+ T-helper cells (Eomes+ Th cells) are known to mediate neuroinflammation and disease progression in MS and are induced by prolactin-secreting cells.
Objectives:
Here, investigated immune cell alterations and the pathophysiological role of Eomes+ Th cells for disease activity during pregnancy and post-partum in MS.
Methods:
We enrolled n = 81 pregnant patients with relapsing-remitting MS (RRMS), n = 27 post-partum RRMS and n = 26 female RRMS control patients under the umbrella of the German Multiple Sclerosis and Pregnancy Registry. Clinical data were collected and immune cell alterations were analysed using flow cytometry.
Results:
While CD3+CD4+ Th cells were unaffected, CD3+CD8+ cytotoxic T-cells were elevated post-partum (p = 0.02) with reduced B-cell frequencies (p = 0.01) compared to non-pregnant RRMS patients. NK cells were elevated during first trimester (p = 0.02) compared to the third trimester. Frequencies of Eomes+ Th and Eomes+ Tc cells did not differ. There was no correlation of prolactin release and expression of Eomes+ Th cells. However, Eomes+ Th cells correlated with lower frequencies of regulatory T-cells during second (r = −0.42; p < 0.05) and third trimester (r = −0.37; p < 0.05). Moreover, Eomes+ Th cells correlated with frequencies of B-cells during third trimester (r = 0.54; p = 0.02). Frequencies of Eomes+ Th cells were not associated with the number of relapses before pregnancy, during pregnancy or post-partum. However, Eomes+ Th cells strongly correlated with disability post-partum as assessed using the EDSS (r = 0.52; p = 0.009).
Discussion:
Pregnancy in MS is associated with robust immunological alterations. Eomes+ Th cells are capable of inducing immune cell alterations during the course of pregnancy, most evident during the second and third trimester as shown with a correlation of reduced Treg cells and a significant increase of B-cells. Importantly, Eomes+ Th cells correlate with disability post-partum. In summary, during late pregnancy in MS an inflammatory, cytotoxic and dysregulated immunological environment is primed gaining function post-delivery. This may be responsible for post-partum disability accumulation.

Keywords
Introduction
Multiple sclerosis (MS) is a chronic inflammatory disease of the central nervous system (CNS) characterized by demyelination, axonal damage and neurodegeneration. 1 As the disease affects increasingly women during early adulthood, a phase often associated with the wish to build a family and become pregnant,2,3 knowledge about the interaction of pregnancy with MS is crucial. The relapse rate decreases during pregnancy with the lowest risk during the third trimester, increasing frequency during the post-partum period and normalization to pre-pregnancy rates approximately 3 months after delivery. 4 Patients planning pregnancy need to be individually adjusted to disease-modifying therapies (DMTs) in order to decrease the risk of relapses during and after pregnancy.5,6 Treatment with DMTs and low annualized relapse rates before pregnancy have a positive impact on the frequency of post-partum relapses. 7 Besides, pregnancy in MS has a normal course and is not associated with foetal abnormalities or miscarriage 8 and pregnancy does not influence secondary progression of MS, especially in patients without significant increase in post-partum clinical and MRI disease activity. 9 Breast feeding was shown to be slightly protective against post-partum relapses. 10 However, the protective effect of breast feeding for post-partum disease activity is controversially discussed. At the moment, there is still lacking knowledge about immunological pathomechanisms driving disease activity during pregnancy and the post-partum period. Identifying patients with an increased risk of relapses during or after pregnancy using biomarkers could potentially be an important approach towards future personalized MS therapy during and after pregnancy. The expression of cytotoxic Th cells expressing Eomesodermin (Eomes) were associated with a higher risk of disease progression. 11 B-cells and non-B cell antigen-presenting cells (APCs) upregulate the hormone prolactin in the progressive phase of experimental autoimmune encephalomyelitis (EAE), a mouse model for MS. 12 Those prolactin producing APCs can induce the expression of Eomes on Th cells ex vivo. 12 Prolactin in general is mainly produced by the anterior pituitary gland and regulates, in particular, lactation in pregnant women. Additionally, prolactin elicits an important role for immune system regulation, for example, through the generation of autoreactive lymphocytes.13,14 The role of prolactin in MS has been investigated in several studies with divergent results. Prolactin was found in higher concentration in female MS patients compared to healthy control persons but did not correlate with disease activity, disease duration or active lesions in the CNS. 15 Furthermore, prolactin is associated with the generation of autoreactivity via B-cells in MS. 16 Contrarily, prolactin promotes proliferation of oligodendrocyte precursor cells and white matter repair and is therefore discussed as therapeutic target for remyelination. 17
We hypothesized that prolactin release during pregnancy and post-partum might induce the expression of Eomes+ Th-cells, potentially associated with an increased risk of disease activity. We assessed disease activity of pregnant MS patients in relation to alterations of immune cell subsets and prolactin concentration.
Materials and methods
Enrolment of patients
Participants were recruited from February 2021 to September 2022 in a prospective study design at the outpatient clinic at the Department for Neurology, Ruhr-University Bochum, St. Josef Hospital. Included patients were enrolled in the German Multiple Sclerosis and Pregnancy Registry (DMSKW), founded in 2006. 18 Written informed consent was signed by the subjects before study inclusion. Patients had a diagnosis of definite MS according to McDonald criteria 2017. 19 The study included in total n = 26 female relapsing-remitting MS (RRMS) patients without current pregnancy, n = 25 RRMS patients during the first trimester of pregnancy, n = 24 patients during the second trimester of pregnancy, n = 32 patients during the third trimester of pregnancy and n = 27 patients during the post-partum period (Figure 1). Exclusion criteria were competing diagnoses such as active cancerous disease or acute inflammatory diseases.
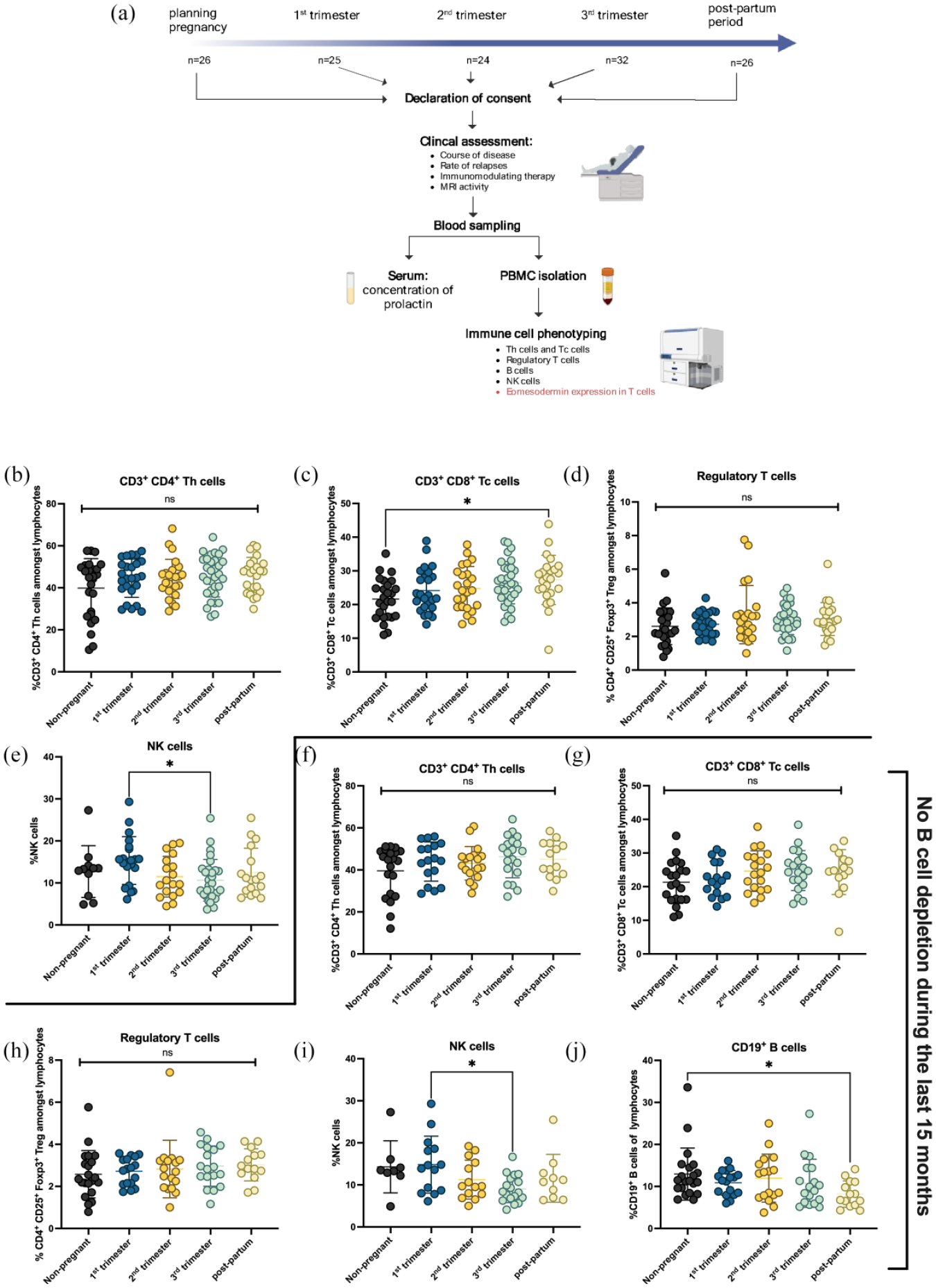
Immune cell subsets during pregnancy in MS. (a) RRMS patients were included at different time points before, during or after pregnancy. After written informed consent, demographic clinical data were collected. Blood was drawn in order to analyse immune cell alterations via flow cytometry and concentration of prolactin in serum. Patients were clinically assessed during the course of pregnancy and post-partum. (b) CD4+ Th cells did not differ in pregnant women while (c) CD8+ Tc cells were elevated during post-partum period compared with non-pregnant RRMS patients. (d) Proportions of CD4+CD25+Foxp3+ regulatory T-cells amongst lymphocytes did not differ. (b–d) Data are derived from pregnant RRMS patients (first trimester: n = 25, second trimester: n = 24, third trimester: n = 32), RRMS patients during post-partum period (n = 24) and RRMS patients with childbearing preferences (n = 26). (e) Patients during first trimester expressed higher levels of NK cells than patients during third trimester (first trimester: n = 21, second trimester: n = 18, third trimester: n = 29, post-partum period: n = 17, non-pregnant RRMS patients: n = 11). (f–j) Immune cell subsets were shown for patients who had not received B-cell depleting therapy during the last 15 months [first trimester (n = 17), second trimester (n = 17), third trimester (n = 18), post-partum period (n = 14), non-pregnant RRMS patients (n = 21)]. (j) B-cells were reduced during post-partum period compared to non-pregnant RRMS patients (p < 0.05). Shown is a mean ± SD for each patient group. Data were analysed using non-parametric Kruskal-Wallis test with post hoc Dunn’s multiple comparison test. *p < 0.05.
Clinical parameters
Neurological examination was performed by attending consultant Neurologists. Demographic, disease-specific data and information about immunomodulating therapies were collected from all patients [Table 1(a)]. Disability was assessed using the Expanded Disability Status Score (EDSS). Recent relapse was defined as relapse within the last 8 weeks. Patients ‘in remission’ were clinically stable without recent exacerbations. Clinical disease activity post-partum was routinely assessed under the umbrella of the DMSKW in telephone interviews. 18
Demographic and clinical characteristics of recruited pregnant and non-pregnant RRMS patients.

Age, disease duration, age onset (in years) and EDSS are presented as mean. Recent relapse is defined as an episode that has occurred no longer than 8 weeks ago.
EDSS, Expanded Disability Status Score; IQR, interquartile range; RRMS, relapsing-remitting MS; SD, standard deviation; SEM, standard error of mean.
Overview of therapies according to different pregnancy periods including all follow ups.

DMF, dimethyl fumarate; GA, glatiramer acetate; IFN-ß1a, interferon-beta-1a.
Blood collection and PBMC isolation
Serum and EDTA (ethylenediaminetetraacetic acid) whole-blood were collected before intravenous administration of medications. Serum was used for the analysis of prolactin concentration. EDTA blood was used for flow cytometric analysis of peripheral blood mononuclear cells (PBMCs). 20 PBMCs were isolated with density gradient centrifugation using ROTI®Sep 1077 human lymphocyte separation medium.
Flow cytometry
PBMCs were incubated with fluorochrome-conjugated antibodies against surface antigens (eBioscience™ and BioLegend®, Supplemental Table S1). For intracellular staining we permeabilized and fixed cells using eBioscience™ Foxp3/Transcription Factor Staining Buffer Set according to manufacture’s instructions. Flow cytometry data were obtained using a FACSCanto™ II (BD Biosciences, Franklin Lakes, NJ, USA) (gating: Supplemental Figure S2). Cells were analysed directly after staining. Data analysis was performed with FlowJo v.10.7.2 (FlowJo, Becton, Dickinson and Company, Ashland, OR, USA).
Prolactin concentration
Concentration of prolactin in serum was measured using electrochemiluminescence immunoassay (Roche, Elecsys Prolactin II; range 4.79–23.3 ng/mL).
Statistical analysis
Data were analysed using Prism software V.9 (GraphPad Software, San Diego, CA, USA). Normality tests were applied for all data sets using Kolmogorov-Smirnov test. Comparison of two groups with non-parametric data was performed with Mann-Whitney U test. More than two non-parametric data sets were compared using Kruskal-Wallis test with Dunn’s multiple comparison. Correlation was measured with non-parametric Spearman correlation. Data are presented as mean values with standard deviation (SD). Correlations are depicted with linear regressions. A p-value <0.05 was considered as significant.
Results
Study population
The study included n = 134 samples of which 59 patients were recruited at different time points during pregnancy. 29.6% (n = 8) of post-partum participants were breast feeding at sampling. Non-pregnant RRMS patients, used as controls, matched regarding age (mean ± SD: 30.7 ± 5.1) and EDSS (2.0 ± 1.1) [Table 1(a)]. Patients received different disease modifying therapies (DMTs) before or during pregnancy. Most patients (59%) paused DMT intake before pregnancy. Ten percent of the patients did not receive DMTs before pregnancy. Six patients (10%) had received a cycle of cladribine at least 6 months prior to pregnancy. Thirty-one percent were treated with DMTs during pregnancy (Supplemental Table S2). Therapies applied during pregnancy include glatiramer acetate (n = 2, 2%), azathioprine (n = 1, 1%), natalizumab (n = 9, 11%), interferon-beta (n = 7, 9%), dimethyl fumarate (n = 1, 1%) and ocrelizumab (n = 1, 1%) [Table 1(b)].
Immune cell alterations during pregnancy
We analysed different immune cell subsets before, during and after pregnancy (Figure 1). CD3+CD4+ T-helper cells did not differ between non-pregnant RRMS patients, during different trimesters of pregnancy and during the post-partum period, respectively [Figure 1(b)]. CD3+CD8+ cytotoxic T-cells were elevated during the post-partum period (p = 0.02) compared to non-pregnant RRMS [Figure 1(c)]. Frequencies of regulatory T-cells did not change during or after pregnancy [Figure 1(d)]. We analysed B-cells in a proportion of patients who had not received B-cell depleting therapies so far. Interestingly, B-cell frequencies declined after pregnancy compared to non-pregnant RRMS [p < 0.05; Figure 1(j)]. Tc cells did not change in patients without B-cell depleting therapies in the last 15 months [Figure 1(g)]. NK cells were elevated during first trimester (p = 0.02) compared to the third trimester [Figure 1(i)]. Since most patients were untreated during pregnancy, there were only minor treatment-related effects on immune cells subsets detectable. Frequencies of B-cells were significantly higher in natalizumab treated patients compared to patients without therapy, while frequencies of CD4+ T-cells were lower in natalizumab treated patients compared to interferon β1a treated patients (Supplemental Figure S3).
Eomes expressing Th cells
Analysis of Eomes+ Th cells showed no differences between non-pregnant RRMS, pregnant RRMS or post-partum RRMS patients [non-pregnant RRMS: 2.4 ± 1.9 mean ± SD; first trimester: 3.8 ± 4.1; second trimester: 3.6 ± 3.2; third trimester: 2.8 ± 2.2; post-partum: 3.1 ± 2.5; Figure 2(a)]. Since CD3+CD8+ cytotoxic T-cells were elevated in post-partum RRMS compared to non-pregnant RRMS patients we analysed the expression of Eomes expressing CD3+CD8+ cells. Eomes+ Tc cells did not differ between respective groups (non-pregnant RRMS: 30.9 ± 16.0; first trimester: 35.3 ± 13.7; second trimester: 35.9 ± 11.6; third trimester: 30.8 ± 12.0; post-partum: 31.6 ± 12.4). The majority of patients did not receive therapies during pregnancy. However, immunomodulating therapies applied during pregnancy did not influence the frequency of Eomes+ Th cells in contrast to patients with halted DMTs during pregnancy (Supplemental Figure S3).
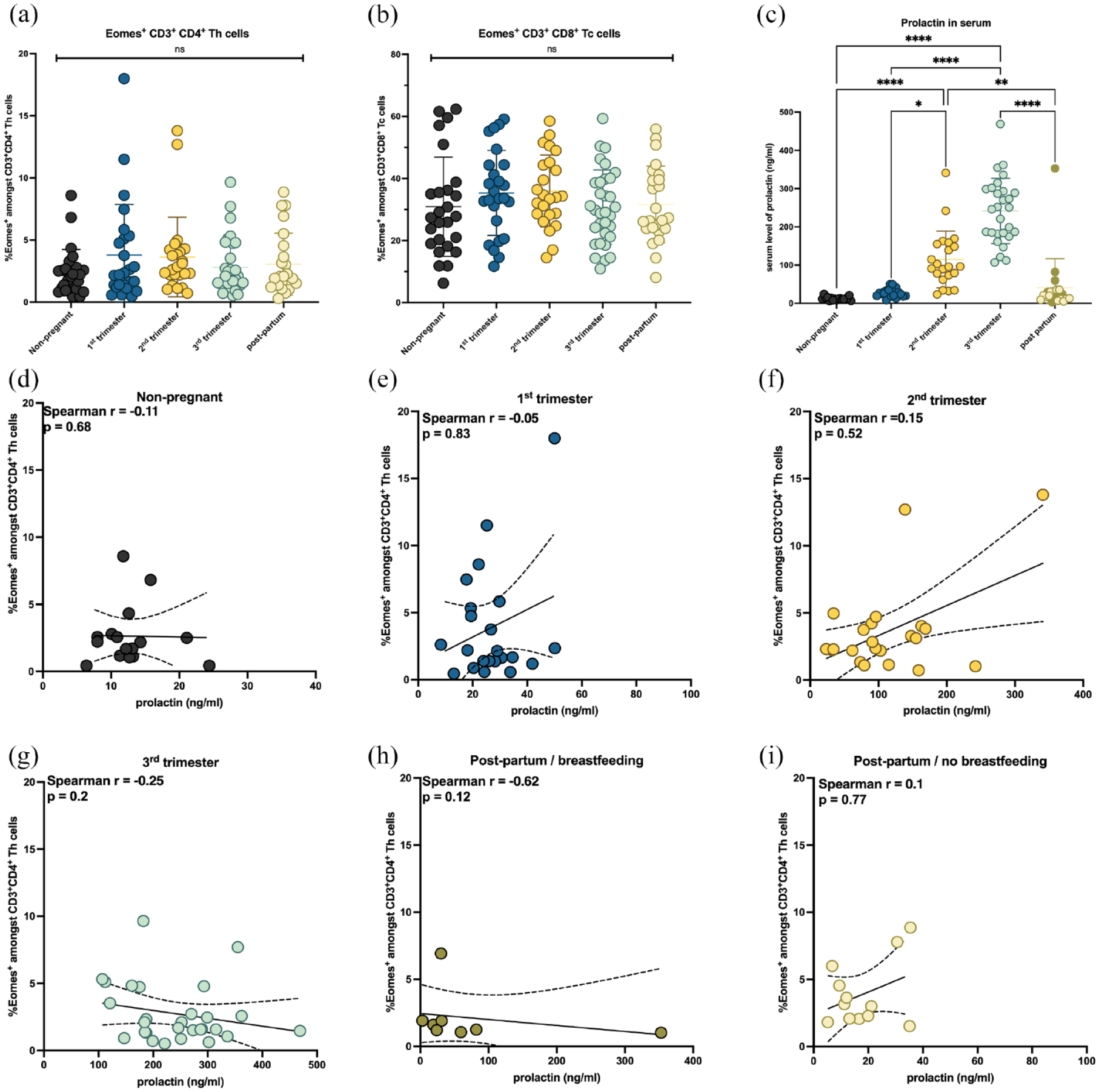
Expression of Eomesodermin (Eomes) in Th cells and correlation with prolactin in serum. Frequencies of (a) Eomes+ Th cells amongst CD3+CD4+ Th cells and (b) Eomes+ Tc cells amongst CD3+CD8+ Tc cells did not differ. (c) Prolactin in serum rose continuously during the course of pregnancy and decreased after delivery. Breastfeeding patients are marked in green colour. There was no correlation between prolactin in serum and Eomes+ Th cells in (d) non-pregnant RRMS patients, (e) during first trimester, (f) second trimester, (g) third trimester, (h) in breastfeeding patients and (i) non-breastfeeding patients. (a, b) Data are derived from pregnant RRMS patients (first trimester: n = 25, second trimester: n = 24, third trimester: n = 32, RRMS patients during post-partum period: n = 24 and non-pregnant RRMS patients: n = 26). (c–i) Data are derived from pregnant RRMS patients [first trimester: n = 22, second trimester: n = 22, third trimester: n = 28, RRMS patients during post-partum period (breastfeeding: n = 8; no breastfeeding: n = 12) and non-pregnant RRMS patients (n = 17)]. Shown is (a–c) mean ± SD and (d–i) linear regression with 95% confidence interval. Data were analysed using (a–c) non-parametric Kruskal-Wallis test and (d–i) non-parametric Spearman test.
No association of prolactin levels and Eomes+ Th cells
In order to analyse an effect of prolactin release during pregnancy on immune cell alterations, we investigated whether there was an association of prolactin and Eomes+ Th cells. As expected, the concentration of prolactin in serum rose continuously during the course of pregnancy and dropped post-partum [p < 0.0001; Figure 2(c)]. There was no evident correlation of prolactin release and expression of Eomes+ Th cells in non-pregnant patients and during the different trimesters of pregnancy as well as in post-partum patients [Figure 2(d)–(i)]. Breastfeeding with consequently high concentrations of prolactin showed no influence on the expression of Eomes in Th cells [Figure 2(h)]. The expression of Eomes+ Tc cells was not associated with the levels of prolactin (Supplemental Figure S4).
Eomes+ Th cells correlate with reduction of regulatory T-cells during second and third trimester
We then set out to investigate how Eomes+ Th cells and regulatory T-cells (Tregs) interact during pregnancy. In non-pregnant RRMS patients there was no association of Eomes+ Th cells and Tregs [Figure 3(a)]. This held also true during the first trimester of pregnancy (r = −0.08; p = 0.71). There was, however, a negative correlation between % of Tregs and the level of Eomes+ Th cells during both second (r = −0.42; p < 0.05) and third trimester [r = −0.37; p < 0.05; Figure 3(c) and (d)]. During the post-partum period, the trend was similar, but missed significance [r = −0.42; p = 0.07; Figure 3(e)].

Correlation of Eomes+ Th cells and regulatory T-cells and B-cells. The frequency of Eomes+ Th cells and frequencies of regulatory T-cells did neither correlate in (a) non-pregnant RRMS patients nor in (b) pregnant RRMS patients during first trimester and (e) patients during post-partum period. The frequency of Eomes+ Th cells and regulatory T-cells correlated negatively in pregnant RRMS patients during (c) second trimester (r = −0.42; p = 0.05) and (d) third trimester (r = −0.37; p = 0.05). Expression of Eomes in Th cells and frequencies of B-cells did not correlate in (f) non-pregnant RRMS patients, pregnant RRMS patients during (g) first trimester, (h) second trimester and (j) patients during post-partum period. Eomes+ Th cells and B-cells correlated positively in pregnant RRMS patients during (i) third trimester. (k) Pregnant and post-partum patients with B-cell depleting therapy before pregnancy (n = 32) showed a trend towards lower frequencies of Eomes+ Th cells compared to patients with no B-cell depleting therapy before pregnancy (n = 64); p = 0.12. (a–e) Data are derived from pregnant RRMS patients (first trimester: n = 25, second trimester: n = 22, third trimester: n = 30, RRMS patients during post-partum period: n = 20 and RRMS patients with childbearing preferences: n = 26). (f–k) Data are derived from pregnant RRMS patients (first trimester: n = 19, second trimester: n = 18, third trimester: n = 18), RRMS patients during post-partum period: n=15 and RRMS patients with childbearing preferences: n = 21. Patients did not receive B-cell depleting therapy within the last 12 months. Shown is (a–j) linear regression with 95% confidence interval and (k) mean ± SD. Data were analysed using (a–j) non-parametric Spearman test and (k) non-parametric Mann-Whitney U test.
Eomes+ Th cells are associated with higher B-cell frequencies during third trimester
B-cells are strongly involved in the pathophysiology of MS by providing an inflammatory environment through the activation of T-cells and the production of antibodies and pro-inflammatory cytokines. 21 Since B-cell frequencies were lower in post-partum RRMS patients compared to non-pregnant RRMS patients, we assessed whether there was an association between Eomes+ Th cells and B-cells. B-cells showed no correlation with Eomes+ Th cells in non-pregnant RRMS [Figure 3(f)]. There was, however, a positive trend of Eomes+ Th cells and B-cells during second trimester (r = 0.42; p = 0.08), which reached significance during third trimester (r = 0.54; p = 0.02) [Figure 3(h) and (i)]. Since there was a substantial amount of patients having been treated with B-cell depleting therapies prior to pregnancy (29.6%), we assessed whether frequencies of Eomes+ Th cells might differ depending on prior anti-CD20 therapy. There was a trend of lower Eomes+ Th cells in anti-CD20 treated patients, which however did not reach significance [Eomes+ Th cells mean ± SD: with B-cell depletion: 2.8 ± 2.4, no B-cell depletion 3.7 ± 3.5, p = 0.12; Figure 3(k)].
Eomes+ Th cells and disease activity during and after pregnancy
We then set out to analyse whether there might be an association between frequencies of Eomes+ Th cells and disease activity in order to characterize their role during pregnancy. The number of relapses during the course of the disease before pregnancy did not correlate with the frequencies of Eomes+ Th cells (number of relapses during the course of the disease before pregnancy: non-pregnant RRMS patients: 3.8 ± 3.0, p = 0.35; first trimester: 3.1 ± 2.6, p = 0.69; second trimester: 3.5 ± 2.8, p = 0.73; third trimester: 3.3 ± 2.4, p = 0.84; post-partum: 3.6 ± 2.2, p = 0.15) (Supplemental Figure S5). We then analysed if Eomes+ Th cells might be associated with relapse activity during or after pregnancy. We documented n = 13 relapses during pregnancy of which n = 6 were in the first trimester (46%), n = 6 were in the second trimester (46%) and n = 1 was in the third trimester (8%) [Table 1(a)].
Patients with a relapse during pregnancy did not show higher levels of Eomes+ Th cells than patients without relapse activity [Eomes+ Th cells: relapse during pregnancy: 2.9 ± 1.9; no relapse during pregnancy: 3.3 ± 3.2; Figure 4(a)]. Because of the relation between Eomes+ Th cells and Treg cells, we analysed changes of Treg cells during relapses as well, which did not differ [Treg cells: relapse during pregnancy: 2.7 ± 0.9; no relapse during pregnancy: 3.0 ± 1.4; Figure 4(c)]. We documented n = 7 relapses after pregnancy. Patients with a relapse after pregnancy also showed no differences in the expression of Eomes+ Th cells [relapse after pregnancy: 4.9 ± 4.6; no relapse after pregnancy: 3.2 ± 3.0; Figure 4(b)] or Treg cells [relapse after pregnancy: 3.3 ± 2.1; no relapse after pregnancy: 2.9 ± 1.1; Figure 4(d)] in contrast to patients without relapse activity.
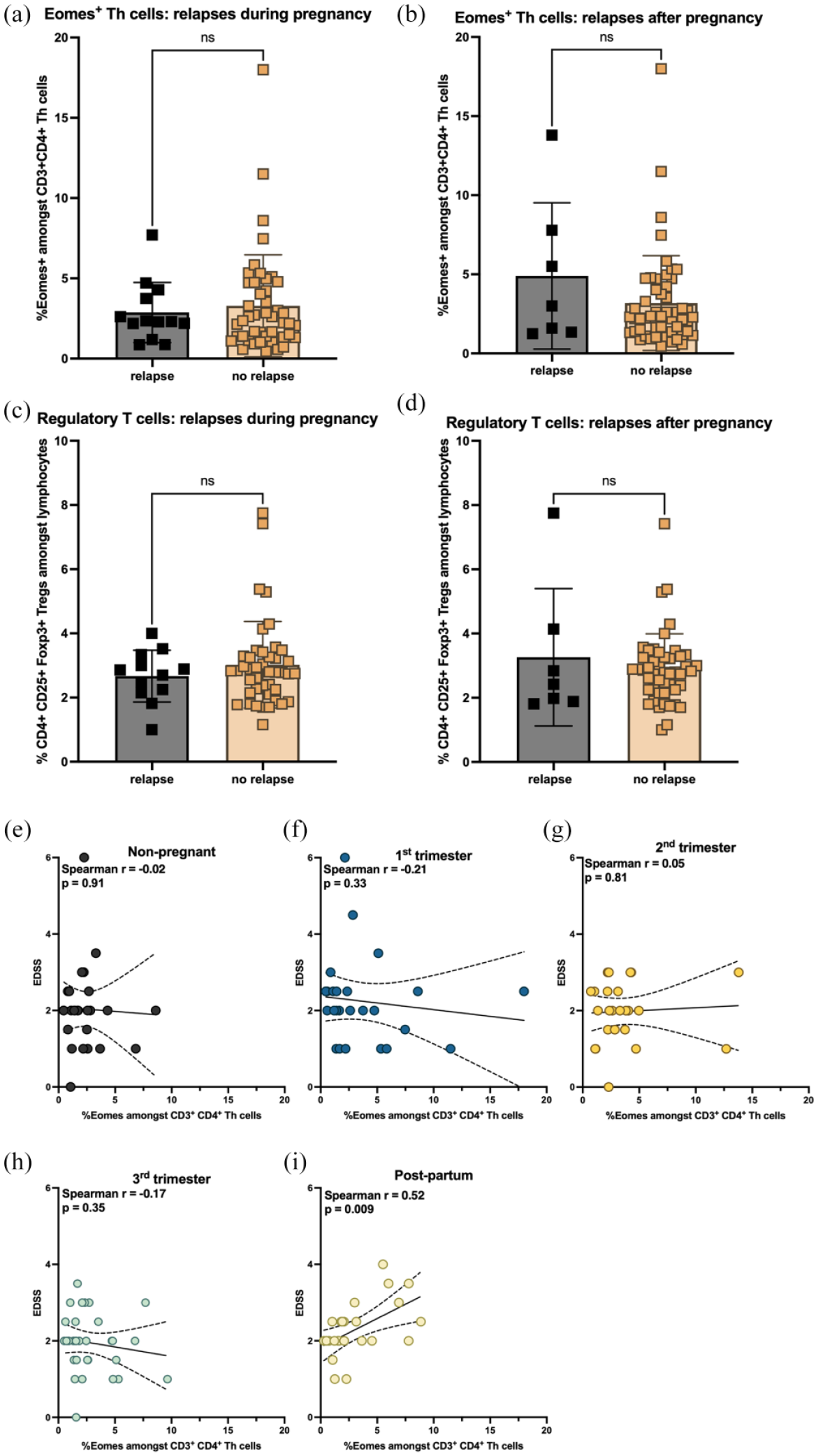
Frequencies of Eomes+ Th cells correlate with disability post-partum. (a, b) Eomes+ Th cells and (c, d) regulatory T-cells did neither differ between pregnant patients with current relapse activity and pregnant patients without relapse activity nor between pregnant patients with relapse activity after pregnancy and pregnant patients with stable disease activity. Frequencies of Eomes+ Th cells and EDSS did neither correlate in (e) non-pregnant RRMS patients nor in pregnant RRMS patients during (f) first trimester, (g) second trimester or (h) third trimester. The frequency of Eomes+ Th cells and EDSS strongly positively correlated, however, in (i) post-partum patients (r = 0.52; p = 0.009). (a, c) Date are derived from n = 10 patients with relapse during pregnancy and n = 41 patients without relapse during pregnancy. (b, d) Data are derived from n = 7 patients with relapse post-partum and n = 52 patients without relapse post-partum. (e–i) Data are derived from pregnant RRMS patients [first trimester (n = 25), second trimester (n = 24), third trimester (n = 33)], RRMS patients during post-partum period (n = 24) and RRMS patients with childbearing preferences (n = 26). (a–d) Shown is a mean ± SD for each patient group and (e–i) linear regression with 95% confidence interval. Data were analysed using (a–d) non-parametric Mann-Whitney test and (e–i) non-parametric Spearman test.
The neurologic function of patients was characterized using the EDSS [Table 1(a)]. The EDSS did not correlate with frequencies of Eomes+ Th cells in non-pregnant RRMS patients or during the course of pregnancy [Figure 4(e)–(h)]. However, we observed a highly significant positive correlation between Eomes+ Th cells and EDSS in post-partum patients (r = 0.51, p = 0.009) [Figure 4(i)].
Discussion
The interactions between MS and pregnancy are driven by complex immunological pathways. Here, we investigated immune cell alteration in pregnant RRMS patients with particular focus on Eomes expressing T-cells. We discovered immune cell alterations especially at the end of pregnancy and post-partum. Cytotoxic T-cells were elevated during third trimester and the post-partum period. Additionally, frequencies of B-cells were lower after pregnancy compared to non-pregnant patients. Mean frequencies of Eomes+ Th cells and cytotoxic T-cells did not differ between groups. There was, however, a correlation of Eomes+ Th cells with lower frequencies of regulatory T-cells in the second and third trimester. The effect was also present during the post-partum period but less pronounced.
It is well established that, in general, pregnancy outcome is not negatively influenced by MS. 6 Pregnancy reduces the risk of relapses with yet higher activity post-partum. 4 This protective effect is, however, not sufficient to protect women with highly active disease who withdraw from DMTs such as natalizumab or fingolimod.22,23 Of note, modern MS therapy since 2010 has led to a reduction of the post-partum relapse rate from 31% to 14% within 3 months post-partum. 6 This is in line with the data shown in our cohort with 22% of the patients with a relapse post-partum, potentially related to the good disease management in post-partum patients.
During pregnancy, the maternal immune system is reprogrammed to create tolerance towards the embryo. This leads to a shift in the immunological balance of CD4+ T-cells with an increase of Th2 cells and a decrease of Th1 cells. 24 Th2 cells secrete anti-inflammatory cytokines like interleukin (IL)-4 and IL-10, which support stability of pregnancy, while inflammatory cytokines tumour necrosis factor (TNF)-α, interferon (IFN)-γ and IL-2 released by Th1 cells have a negative effect on the foetus development both in mouse and in human. 25 These immunological changes are considered as a possible explanation for the stabilization of MS during pregnancy. 25 Another influencing factor are changes in the hormonal balance during pregnancy. In fact, the decrease of relapse rate during late pregnancy correlates with the increase of the oestrogens estradiol and estriol. 26 Estriol in combination with glatiramer acetate compared to monotherapy has a protective effect on the relapse rate with a risk reduction of 37% over 24 months. 27 However, the link between immunological changes and hormonal changes remains largely unknown to date.
To understand dynamics and potential associations of prolactin release and immune cell alterations we performed a conclusive immunological analysis at different time points of pregnancy. Tregs were reduced with rising Eomes+ Th cells. Foxp3 is an important transcription factor of regulatory T-cells and is therefore essential in regulatory pathways of the immune system. We found a strong negative correlation of Eomes+ Th cells and Tregs during the second and third trimester of pregnancy and a trend post-partum. The negative feedback between induction of Eomes+ Th cells and Tregs might hence be responsible for the termination of an anti-inflammatory milieu established during pregnancy to protect the unborn baby. In a murine model of colitis, Eomes+CD4+ T-cells accumulated in old mice and deficiency of Eomes favoured the accumulation of Foxp3+ cells. 28 Moreover, Eomes-deficient CD4+ T-cells have a protective effect in accordance with enhanced frequencies of Foxp3+ cells in EAE, a mouse model of MS. 28 This is in line with the data presented here, showing a suppression of Tregs by Eomes+ Th cells, most evident during the final phase of pregnancy.
While frequencies of Eomes+ Th cells were not associated with relapse activity during pregnancy and post-partum, there was a strong correlation between frequencies of Eomes+ Th cells and disability. This is in line with findings in patients with SPMS having shown that frequencies of Eomes+ Th cells are responsible for progression and the transition from RRMS to secondary-progressive multiple sclerosis (SPMS). 11 The underlying mechanisms in the CNS driving neurotoxicity and disability progression remain so far elusive. One potential mechanism might be related to progression independent of relapse activity (PIRA) which can be found in 25% following a first demyelinating event as shown in a longitudinal cohort of 1128 patients and which is associated with unfavourable long-term outcome. 29 RMS patients with PIRA exhibit accelerated brain atrophy especially in the cerebral cortex. 30 Of note, the post-mortem analysis in a SPMS patient showed, that Eomes+ Th cells are present in cortical lesions but most abundantly in frontal and occipital normal appearing white matter. 11 Magnet resonance tomography studies showed that women with MS have higher T2 lesion volume, greater annualized T2 lesion volume increase, lower brain parenchymal fraction and greater annualized brain volume loss post-partum compared to the pre-pregnancy period. 31 It might hence be speculated that Eomes+ Th cells, characterized by a cytotoxic phenotype, might support the formation of a neurotoxic environment associated with neuronal degeneration and disability progression, also post-partum. The initial hypothesis that pituitary prolactin might be linked to relapse activity could not be supported by the data presented here. This might, however, also be related to the sample size included in this study.
B-cells were lower in post-partum patients compared to non-pregnant patients, yet positively correlated with Eomes+ Th cells. B-cells are important for humoral immunity and may elicit both pro-inflammatory and regulatory functions.32,33 In MS, B-cells have more pro-inflammatory functions including increased release of pro-inflammatory cytokines such as IL-6 and TNF-α, reduced release of anti-inflammatory IL-10, 34 the stimulation of T-cell proliferation35,36 and the formation of lymphoid follicular structures in the meninges. 37 Since B-cell activation has been linked to disability accumulation in MS, the altered Eomes+ Th cell–B-cell axis might be a potential contributor to the enhanced risk of disability post-partum.
Limitations of the data presented here include the sample size and the missing power calculation prior to conducting the study, since the possible effect size of immunological changes was unclear. Moreover, recruitment of patients was conducted at different time points during pregnancy. Most patients could not be followed longitudinally, mostly related to the fact that patients usually presented only once or twice for a specialized consultation. 18 A strength of the dataset presented here is the inclusion of both a pregnant RRMS cohort and a non-pregnant cohort, conclusive immunophenotyping at different time points and the assessment of standardized clinical and immunological data with high accuracy.
Conclusion
In summary, we provide substantial evidence of immunological alterations during pregnancy in MS and show that a subgroup of Th cells with cytotoxic properties expressing Eomesodermin are associated with disability post-partum. Eomes+ Th cells might serve as surrogate for disease activity and could inform about the importance to optimize treatment in MS. Future work should focus on the functional characterization of those immune cell subsets and plasma cytokine levels to better understand pathogenic mechanisms.
Supplemental Material
sj-docx-1-tan-10.1177_17562864241229321 – Supplemental material for Eomesodermin-expressing CD4+ Th cells and association with pregnancy in multiple sclerosis
Supplemental material, sj-docx-1-tan-10.1177_17562864241229321 for Eomesodermin-expressing CD4+ Th cells and association with pregnancy in multiple sclerosis by Simon Faissner, Marielena Bongert, Paulina Trendelenburg, Sandra Thiel, Takashi Yamamura, Kerstin Hellwig and Ralf Gold in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
