Abstract
Twenty years on from its initial approval as the first monoclonal antibody for the treatment of multiple sclerosis (MS), natalizumab remains a valuable high-efficacy treatment option for people with relapsing-remitting MS, with robust real-world evidence supporting its long-term efficacy and well-characterized safety profile, provided that the risk of progressive multifocal leukoencephalopathy (PML) is monitored and mitigated. This review explores the long-term clinical impact of natalizumab. It draws on two decades of experience to guide treatment strategies with natalizumab, including its use early in the disease course, switching to natalizumab, its use during vaccination, and PML risk management and exit strategies. Guidance on the use of natalizumab in pregnant and breastfeeding women with MS, children with MS, and people with comorbidities is discussed, along with reflections on what has been learned from 20 years with natalizumab, and what the future holds for this impactful treatment in MS and beyond.
Introduction
In 2004, natalizumab became the first monoclonal antibody approved to treat multiple sclerosis (MS),1,2 marking the second major milestone in the MS treatment landscape following the introduction of the first interferon (IFN) treatment in 1993.1,3 Twenty years on, the long-term efficacy and well-characterized safety profile of natalizumab is supported by robust real-world evidence (RWE).4,5 Natalizumab has a unique mode of action, 6 selectively compromising the immune response in the central nervous system (CNS) and gut. 7 This results in low rates of opportunistic infections, 4 provided that the risk of progressive multifocal leukoencephalopathy (PML) is monitored and mitigated. 8 This, combined with the recent approval of biosimilar natalizumab by the European Medicines Agency (EMA) and U.S. Food and Drug Administration (FDA),9–11 which could see natalizumab become more accessible at a lower cost to healthcare systems, 12 means that it remains a valuable treatment option for MS and may hold promise for people with unmet medical need. 13
MS is a chronic, progressive, autoimmune disease affecting the brain and spinal cord and is characterized by inflammatory and neurodegenerative processes.14,15 Symptoms include those affecting motor and sensory function (e.g., weakness, spasticity, ataxia, and altered sensation),16,17 vision, 18 speech/swallowing, 19 bladder, and bowel.20,21 MS can also cause fatigue, cognitive, and mood symptoms. 14 MS affects around 2.3 million people worldwide and is usually diagnosed between the ages of 20 and 50 22 ; however, between 3% and 10% of cases are diagnosed in children under the age of 16. 23 The 2024 updates to the McDonald criteria for diagnosis of MS are expected to help clinicians make accurate diagnosis at an earlier stage of the disease. 24
Relapsing-remitting MS (RRMS) is the most common clinical phenotype, occurring in approximately 85% of people with MS, and it is characterized by alternating periods of distinct relapse and full or partial recovery of neurological function, with relapse-associated worsening of disability. 25 However, even from the early phases of RRMS, the majority of disability progression is progression independent of relapse activity (PIRA), which is the predominant driver of disability accumulation.26,27
Current treatment strategies aim to reduce the risk of relapses, and potentially, disability progression, with treatment response assessed via clinical measures and magnetic resonance imaging (MRI) activity. 3 Treatment objectives for people with MS include the prevention of novel disease activity based on clinical and imaging features, which is termed “no evidence of disease activity (NEDA)” as well as “minimal evidence of disease activity.” 28 In this line, a retrospective analysis from the natalizumab AFFIRM clinical trial was the first to use freedom from disease activity as a measure of treatment efficacy and showed that disease remission could be an attainable treatment goal in MS.29,30
Over the past three decades, there has been a large increase in the number of therapeutic options available for MS, 3 with around 20 disease-modifying therapies (DMTs) currently approved by the FDA and/or the EMA.31–33 The European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS) and European Academy of Neurology (EAN) guidelines recommend early treatment with DMTs for people with active RRMS, choosing between the wide range of available drugs from those that are “moderately effective” to “highly effective.” 3
Two distinct treatment strategies are often used in MS: (1) escalation, which involves starting treatment with limited efficacy, low side effect treatment options, and then switching to a high-efficacy treatment (HET), with potentially more side effects, if necessary, and (2) the early initiation of HET.34,35 Early use of DMTs is associated with better long-term treatment outcomes (Figure 1(a)); however, early use of HET, including alemtuzumab, natalizumab, ocrelizumab, ofatumumab, and off-label rituximab, has not only been demonstrated to provide a greater reduction in relapse rates than lower-efficacy DMTs, such as IFN beta and glatiramer acetate, but more recent evidence shows that treating early with an HET also results in better long-term outcomes in terms of disability accumulation.34,36–39 Additionally, RWE supports the use of natalizumab early in the disease course (Figure 1(b)), as a study of people who initiated treatment with either natalizumab or a lower-efficacy DMT after their MS diagnosis found that those initiating natalizumab were less likely to experience a relapse during the follow-up period. 40 Early use of natalizumab is discussed further in the section of this article, “Practical guidance on the clinical use of natalizumab.”
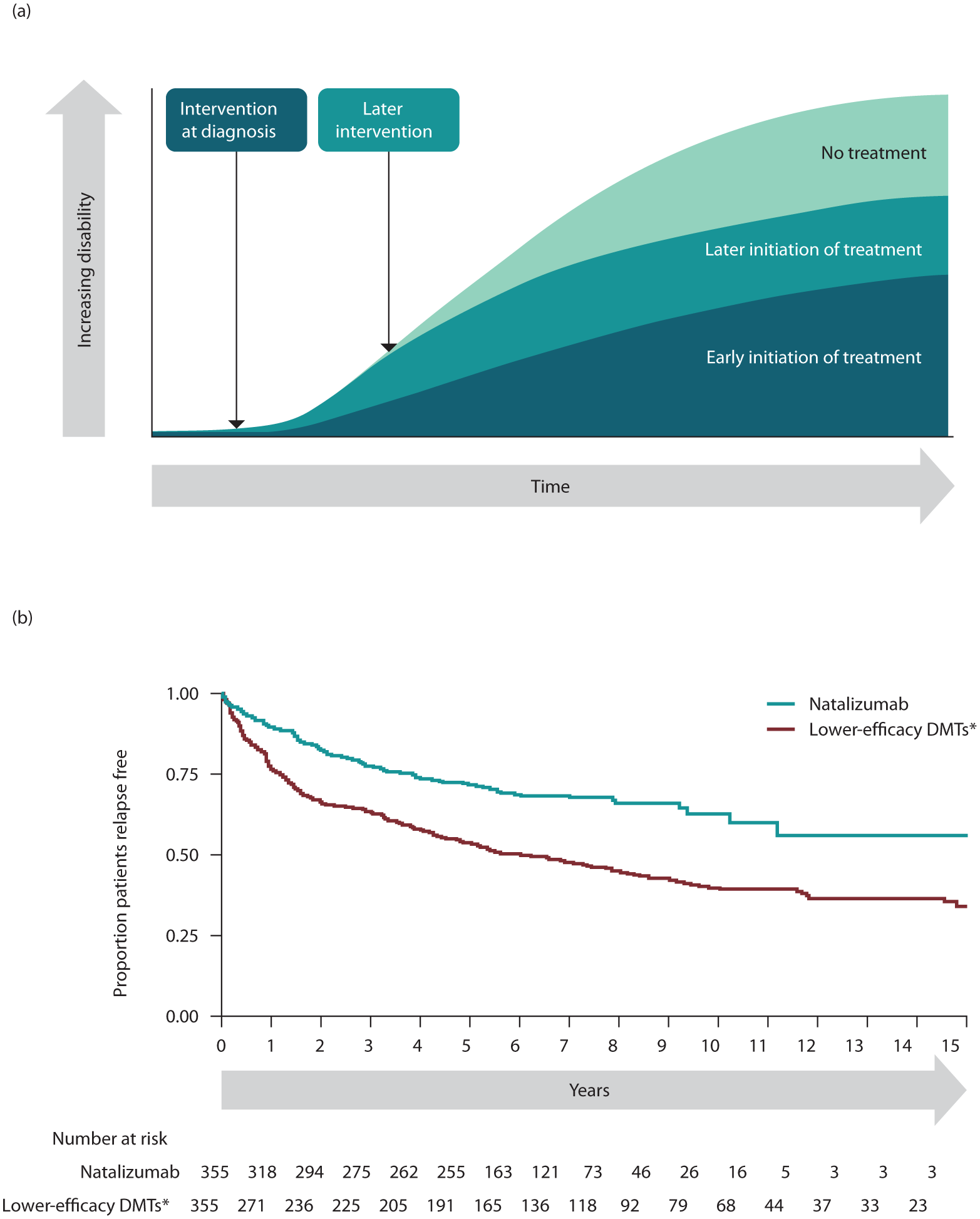
Long-term prognosis with (a) earlier versus later initiation of treatment and (b) early initiation of natalizumab versus lower-efficacy DMTs.
Natalizumab: Mode of action
Natalizumab is a humanized anti-α4β1 integrin monoclonal antibody 42 that was approved by the FDA in 2004 2 for the treatment of adults with relapsing forms of MS, 43 and by the EMA in 2006 for the treatment of adults with highly active RRMS. 44 A biosimilar, a biological medicine that has no clinically meaningful differences in quality, safety, and efficacy versus an already licensed reference biological medicine, has been developed for natalizumab and was approved for these same indications by the FDA and EMA in 2023.9–11,45 Natalizumab and its biosimilar are also approved by the FDA to treat Crohn’s disease.10,11,43
Figure 2 shows the mode of action of a range of high-efficacy DMTs used to treat MS. Natalizumab has a distinct and unique mode of action from other HET used in MS 6 (Figure 2(a) and (b)).
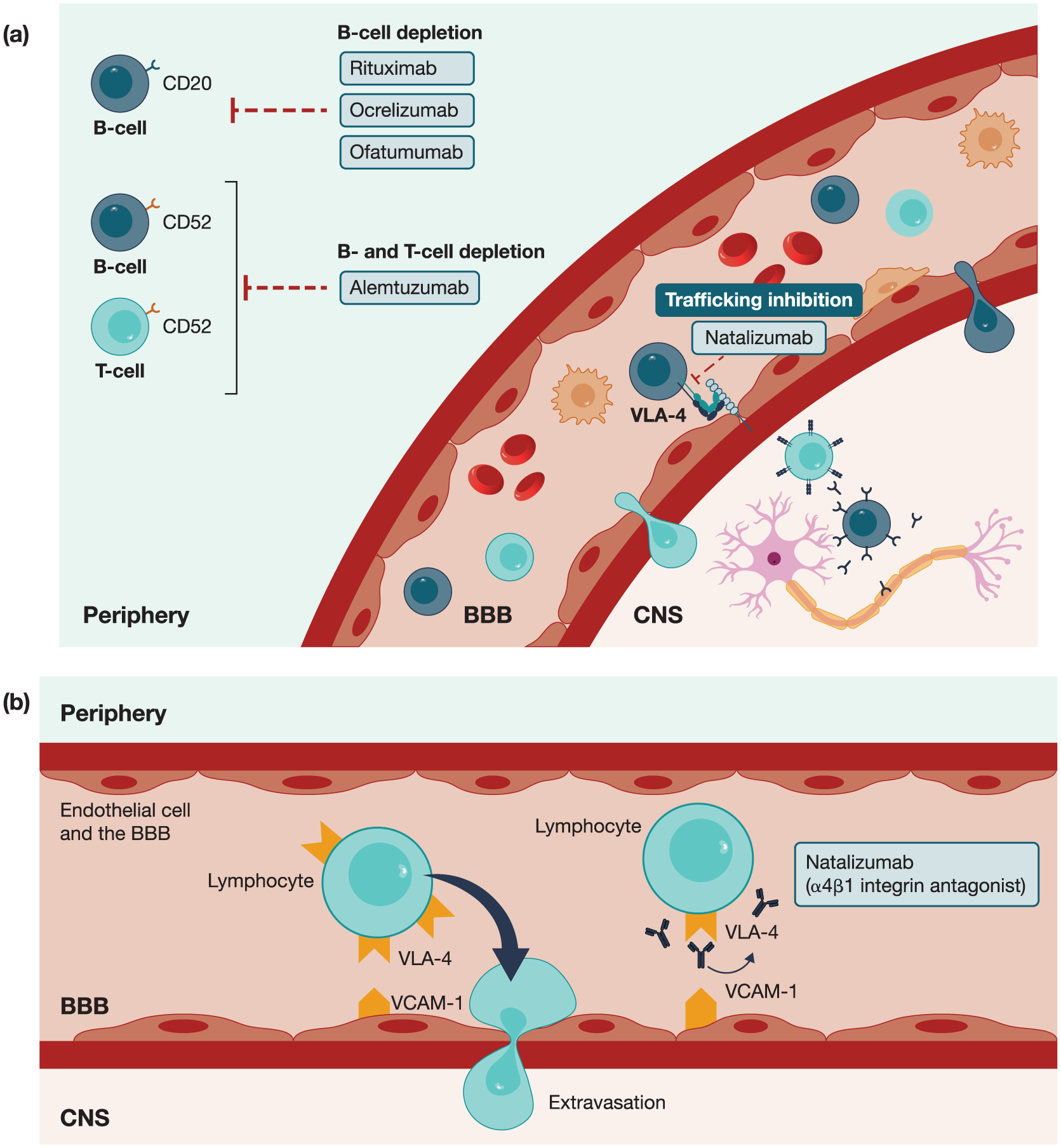
Mode of action for (a) high-efficacy DMTs for MS and (b) natalizumab specifically.
Unlike treatments that cause long-term B-cell depletion, natalizumab prevents leukocytes from entering the brain and spinal cord by blocking the interaction between α4β1 integrin and VCAM-1, preventing leukocytes from crossing the blood-brain barrier (BBB). 42 This inhibition of leukocyte migration into the CNS reduces new MS lesion formation.42,46,47 Natalizumab may also inhibit ongoing inflammation by disrupting the interactions between inflammatory leukocytes and extracellular matrix proteins, 48 or by inducing apoptosis of activated T cells. 49 Additionally, natalizumab treatment has been shown to indirectly impact microglial activation in the brain, a process considered to be associated with MS disease progression; a recent study of 21 people with MS found that microglial activation was reduced following 1 year of treatment with natalizumab. 50
Furthermore, natalizumab may have other immunological effects beyond VLA-4 interaction: an observational longitudinal cohort study of 22 people with RRMS found an increase in anti-inflammatory cytokines just a few hours to days after the first intravenous (IV) infusion of natalizumab, 51 and a recent meta-anlaysis has shown that natalizumab can lead to a reduction in intrathecal oligoclonal bands in cerebrospinal fluid, suggesting a possible effect on memory plasma cells. 52
Unique properties and clinical impact of natalizumab
The efficacy of natalizumab in people with RRMS was demonstrated in the phase III AFFIRM trial, with a significant reduction in relapse rate, disease activity, and disability progression versus placebo. 53 The phase III SENTINEL trial also confirmed that natalizumab in combination with IFN beta-1a was significantly more effective than IFN beta-1a alone; however, natalizumab was only approved as a monotherapy.43,54 With almost two decades of experience, natalizumab is now a well-established and well-characterized treatment, 55 which offers a range of benefits to people with RRMS. Not only does natalizumab provide long-term clinical effectiveness, but also, because of its distinct mode of action, natalizumab exhibits unique properties compared with other MS treatments, in particular, rapid onset of action and reversibility.
Rapid onset of action
Natalizumab treatment has a rapid onset of action. In a study of the effect of a single dose of natalizumab administered soon after the onset of an MS relapse, changes in MRI activity were noted as early as 1 week after dosing. 56 In AFFIRM, a significant reduction in the cumulative risk of relapse was observed early, within 8 weeks, and a significant reduction in the annualized relapse rate (ARR) was observed within 3 months. 57 The rapid action of natalizumab may be explained by its unique mode of action, with the inhibition of leukocyte migration into the CNS preventing lesion formation. 57
Comparing HET options, the phase IV REVEAL study, which investigated natalizumab versus fingolimod in people with RRMS, found that the effect on disease activity was more rapid with natalizumab than with fingolimod and that T1 gadolinium-enhancing lesion accumulation was lower with natalizumab than with fingolimod at 4 weeks (63% lower at 4 weeks (p = 0.353), ⩾70% lower at 12 weeks (p = 0.030), which was maintained at 24 weeks (p = 0.008)). 58 Additionally, a study assessing the effect of natalizumab versus ocrelizumab on quality-of-life (QoL) outcomes in people with MS found that those treated with natalizumab experienced a shorter time to clinically meaningful improvement in several QoL domains, including cognitive function, compared with ocrelizumab (event time ratio (95% confidence interval (CI)): 0.37 (0.24–0.57); p < 0.001). 59 In two other studies, the effect of ocrelizumab on disability progression 60 and ofatumumab on disability progression and ARR 61 were not measured until 12 weeks after the first dose,60,61 although a phase II study of ofatumumab showed rapid B-cell depletion within 14 days. 62 Rituximab was found to reduce the number of gadolinium-enhancing lesions from 12 weeks after the first dose, compared with placebo. 63
Reversibility
Given the wide range of therapies available to people with RRMS, clinicians should consider the temporal nature and reversibility of a treatment’s biological effects, as this could impact treatment sequencing should someone need to switch to a new treatment. 64
The effects of natalizumab on peripheral immune cells and pharmacodynamic markers are reversible, with both peripheral immune cell count and pharmacodynamic marker levels starting to decline 8–12 weeks after the final natalizumab dose, returning to pre-treatment levels by 16 weeks. 64 The risk of rebound MS disease upon cessation of natalizumab and guidance on exit strategies is discussed in the section on “Exit strategies in people with high PML risk.”
The reversibility of natalizumab treatment is in contrast to other HET immunotherapies available for RRMS that act to deplete specific cell populations, such as alemtuzumab and ocrelizumab, where reconstitution of depleted B- or T-cell populations can take 9 months or longer. 64 This can present an issue for treatment sequencing, as long-lasting effects on the immune system could present safety concerns when using sequential treatments. 65 However, after discontinuation of natalizumab, the rapid reconstitution of the peripheral immune system may allow increased flexibility in treatment sequencing. 64
Long-term clinical effectiveness
As the first monoclonal antibody to be approved for MS, 1 natalizumab benefits from the longest period of RWE of any HET for MS, with several studies demonstrating the long-term clinical effectiveness of natalizumab. The Tysabri Observational Program (TOP) 10-year interim analysis included 6148 people with RRMS and found that the reduction in ARR seen after 1 year was sustained over 10 years of natalizumab treatment. 4 Additionally, an Austrian study of 1596 people found that ARR reduction on natalizumab was sustained over a follow-up time of up to 14 years. 66
Confirmed disability improvement (CDI) was first described in a post hoc analysis of the AFFIRM trial where natalizumab increased the probability of 3-month CDI over 2 years compared with placebo.67,68 A separate TOP analysis including 5384 people with RRMS found that disability improvement on natalizumab could be sustained over 8 years. 69 Furthermore, the TOP 10-year interim analysis found that at 10 years, the cumulative probability of 24-week CDI was 33.1%, and Expanded Disability Status Scale (EDSS) scores remained stable over 10 years of treatment with natalizumab. 4
A study from Denmark, which followed 2424 people treated with natalizumab across a 13-year time period, found strong RWE for efficient suppression of clinical relapses, EDSS worsening, and radiological activity. 70 Notably in this study, MRI disease activity (defined as any new/enlarging T2 lesions or any gadolinium-enhancing lesions) was rare in people treated with natalizumab, with just 2.7% of people showing signs of MRI activity between 26 and 38 months after treatment initiation. 70 An extensive observational study of people with MS treated with natalizumab in Spain found that, among the 825 people treated with natalizumab for at least a year, there was a significant decrease in EDSS score, with the mean EDSS score decreasing from 3.71 (95% CI 3.60–3.82) at baseline to 3.37 (95% CI 3.25–3.49) after 12 months of treatment. 5 The effect of natalizumab on disability improvement may be linked, according to the author’s opinion, to its suppression of the evolution of new gadolinium-enhancing to T1-hypointense lesions and a reduction of axonal degeneration in relapsing forms of MS.71,72
Health-related QoL improvements
In addition to the long-term efficacy data on the prevention of relapse, disability accumulation, and disability improvement described above, natalizumab has been shown to improve health-related quality of life (HRQoL) significantly in both clinical trial73,74 and real-world75,76 settings. In particular, a study of 195 people who had been prescribed natalizumab highlighted the value of patient-reported outcomes in assessing the potential benefits of natalizumab and found that the majority of people (>92%) had stable or improved total, motor, and cognitive fatigue after 1 year of treatment. 77
Comparisons with other HETs
The efficacy of natalizumab has been compared to other HET options in several studies. In the BEST-MS prospective study of 223 people with RRMS, significantly more people treated with natalizumab had NEDA after 1 year versus those treated with fingolimod. 78 Another real-world study of 174 people comparing natalizumab with fingolimod found that a significantly greater proportion of people treated with natalizumab had NEDA after 4 years compared to those treated with fingolimod. 79 In a retrospective observational cohort study, which included 740 people with RRMS, natalizumab had a similar effect to anti-CD20 therapy (rituximab or ocrelizumab) on relapse rate and disability progression in people switching from fingolimod. 80 Furthermore, a study of the MSBase cohort comparing cladribine to other immunotherapies for MS found that cladribine was associated with a higher ARR than natalizumab, and the risk of disability worsening was lower in people treated with natalizumab than in those treated with cladribine. 81 Taken together, these studies indicate that natalizumab has improved efficacy with respect to the outcomes studied versus fingolimod or cladribine and comparable efficacy versus anti-CD20 therapies.
Safety and tolerability of natalizumab
Long-term safety and tolerability
Natalizumab demonstrated a favorable safety and tolerability profile within the 2-year evaluation period of the phase III AFFIRM study, with both serious adverse events (SAEs) and overall infection rates similar between people receiving natalizumab and those receiving placebo. 53 Real-world, long-term safety data are consistent with the results of the AFFIRM clinical trial: in the 10-year analysis of TOP, over 85% of people who received natalizumab did not experience any SAEs. 4 The most common SAEs were infections and infestations (4.1%); malignancy rates were 1.1%, and while breast cancer was the most frequent malignancy, it occurred at a rate of 86.7 per 100,000 patient-years, which is lower than the expected European age-standardized incidence rate of 94.2 per 100,000. 4 The incidence of PML in the TOP cohort was 0.9%, and the incidence of non-PML opportunistic infections was 0.2%, 4 which included herpes zoster, herpes meningitis, encephalitis, herpes simplex encephalitis, herpes zoster disseminated, listeria meningitis (sepsis), pneumonia, and pulmonary Mycobacterium kansasii infection. 4 In rare cases, natalizumab may increase the risk of herpes simplex virus reactivation in the CNS, leading to herpes encephalitis; however, these infections may present with atypical clinical features.82–84
Anti-natalizumab antibodies
Because natalizumab is a biologic treatment, some people develop anti-natalizumab antibodies, a fraction of which are persistent and have the potential to neutralize the drug’s effect. 85 In the AFFIRM trial, 57 people receiving natalizumab (9%) had detectable anti-natalizumab binding antibodies at some time during the study. 53 Of these, persistent antibodies developed in 37 people (6%), and these people had an increase in infusion-related adverse events (AEs) and a loss of efficacy of natalizumab. 53 A retrospective observational study of 1251 people with MS who had experienced either an infusion-related event (IRE) or disease exacerbation while receiving natalizumab found that 12.3% of this cohort had detectable anti-natalizumab antibodies. 85 These antibodies were typically detected earlier in the treatment course, with anti-natalizumab antibodies more frequently detected during the first six infusions. 85 Anti-natalizumab antibodies were more frequently detected among people with IREs compared to those with disease exacerbation (21.6% vs 10.8%), suggesting that anti-natalizumab antibodies may have an impact on the development of IRE and, to a lesser extent, disease exacerbation. 85 When a drug is released to the market, as in the case of natalizumab, anti-drug antibody testing may add to a risk-minimization plan (for IRE and anaphylactic reactions), shorten the time that people stay on treatment with a drug that is not effective for them, and consequently promote beneficial therapeutic switches. 86 However, persistent disease activity while taking natalizumab would result in a change of treatment strategy. This would make anti-natalizumab antibody testing unnecessary, although testing for anti-drug antibodies may indicate loss of treatment function in people with stable disease, thus enabling an early treatment change.
In the authors’ experience, anti-drug antibody testing strategies in clinical practice vary significantly by geographic region and are not always available. For example, in Denmark anti-drug antibodies are routinely tested at 3, 6, and 12 months, and if possible, re-analyzed 1–3 months later, while authors from Austria, Germany, and the UK only routinely test at 12 and 24 months into treatment if the person with MS experiences infusion reactions, and if there is an early lack of efficacy. In Sweden, anti-drug antibodies are tested only in those with an infusion-related reaction and in cases of treatment failure. However, many healthcare professionals (HCPs) are not regularly testing, as when adverse reactions or a lack of efficacy is experienced, a switch in treatment strategy would be considered regardless.
PML risk management
PML infection is an important safety concern related to natalizumab treatment. 87 It is an opportunistic CNS infection caused by the John Cunningham (JC) virus that can be fatal or result in severe disability.9,44,88 PML most commonly occurs in people with HIV infection, lymphoproliferative disease, and in people who are receiving natalizumab, and can also occur in association with other drugs, such as those used for organ transplantation. 89 Other drugs have been associated with a risk of PML, including other HETs for MS 89 ; however, the prevalence of PML in people with MS taking other DMTs is much lower than that associated with natalizumab. 88 Proposed mechanisms of action underlying the increased risk of PML in people taking natalizumab include reduction of JC virus-related CNS immunosurveillance by prevention of JC virus-specific cytotoxic T-cell entry into CNS tissue or other sites where JC virus may be dormant and blocking of α4β1 integrin receptors, leading to the release of B cells from bone marrow and spleen, which is the common site of JC virus latency. 90 In either case, reactivation of latent JC virus by the action of natalizumab then triggers the development of PML. 90 For diagnosis of PML, an evaluation including a gadolinium-enhanced MRI scan of the brain and, for confirmation, cerebrospinal fluid analysis for JC virus DNA is required.43,91,92
Following the approval of natalizumab by the FDA in November 2004, two cases of PML in people who had received natalizumab in the SENTINEL trial (which evaluated the safety and efficacy of natalizumab in combination with IFN beta-1a) led to the withdrawal of natalizumab from the market in February 2005.54,93 However, in June 2006, the FDA approved the reintroduction of natalizumab with revised labeling and improved safety warnings for PML risk management.93,94 In the TOP cohort (n = 6148), only 53 (0.9%) people had developed confirmed PML over 10 years, which equates to an incidence rate of 2.034 per 1000 patient-years. 4
The main risk factors for PML are JC virus antibody serum positivity, prior immunosuppressive treatment, and natalizumab treatment duration beyond 24 months. 95 PML risk stratification based on JC virus serologic test results, prior immunosuppressant use, and duration of natalizumab treatment, coupled with close monitoring and education, have reduced the incidence of natalizumab-associated PML in people with MS over recent years; for example, in Sweden, cumulative PML incidence fell from a high of 1.49 events per 1000 person years in 2012 to 0.72 in 2018.8,95 Figure 3 shows a guide to mitigating PML risk.
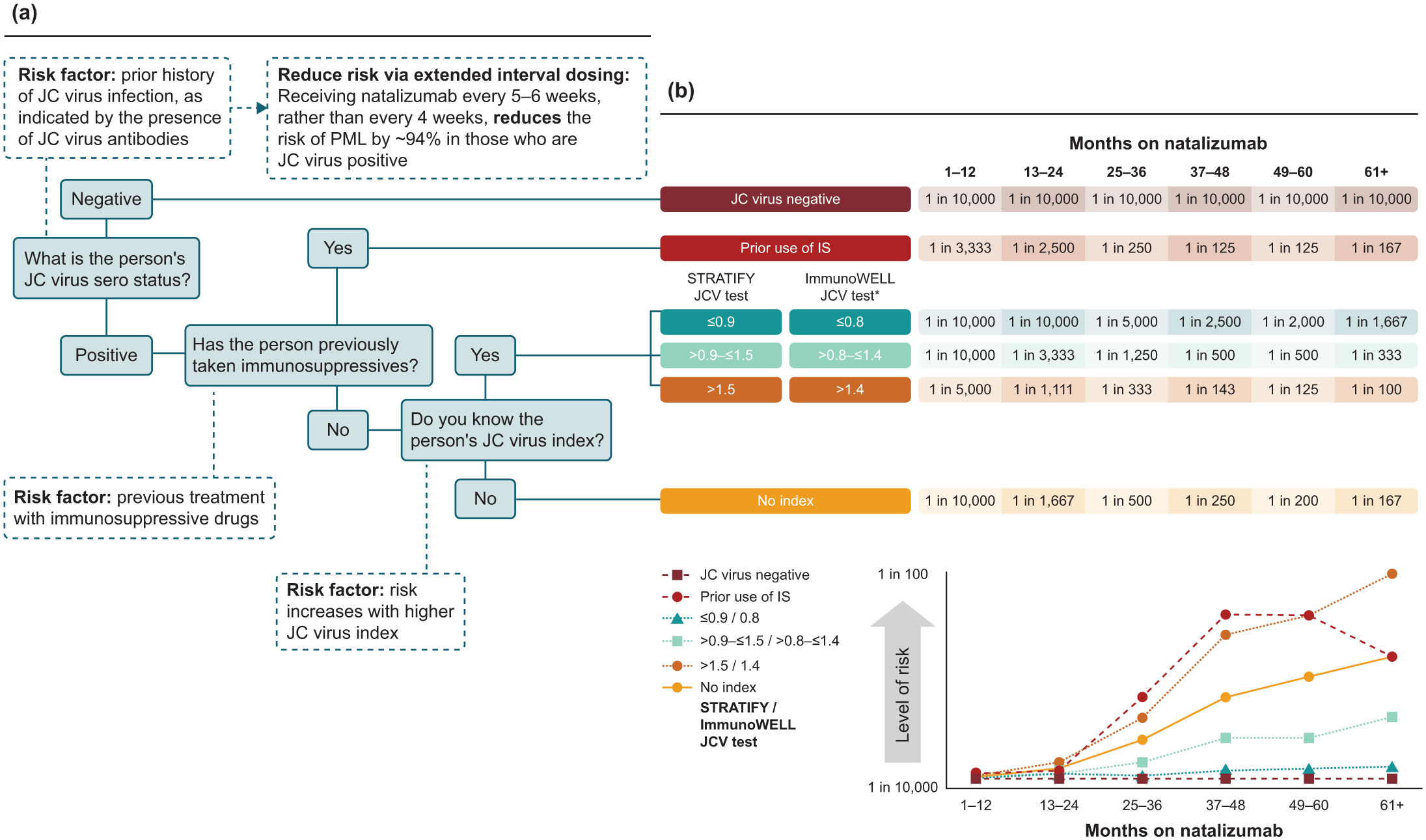
PML risk explained: (a) how to assess PML risk in people with MS and (b) PML risk over time.
Extended interval dosing, where natalizumab is given every 6 weeks (QW6) rather than every 4 weeks (QW4; standard interval dosing), is an emerging strategy in PML risk management, as the partial desaturation of α integrin receptors may enhance immune surveillance within the CNS and subsequently reduce the risk of PML. 99 A retrospective cohort study using a large dataset, the Tysabri Outreach: Unified Commitment to Health (TOUCH) program, has shown that extended interval dosing lowers PML risk compared with standard interval dosing. 100 When moving to extended interval dosing, several studies have shown no reduction in the efficacy of natalizumab when moving to extended interval dosing in relation to MRI activity and relapses.101,102 In particular, the phase III NOVA study comprising 499 people with RRMS who had been treated with natalizumab Q4W without relapse for 1 year or more and randomized to switch to natalizumab Q6W or remain on natalizumab Q4W concluded that the majority of people stable on natalizumab Q4W dosing can switch to Q6W without a clinically meaningful loss of efficacy. 103 Furthermore, another study of 73 people with RRMS demonstrated that extended interval dosing in people with RRMS did not increase axonal damage, as determined by serum neurofilament light chain concentrations. 104 However, the efficacy of natalizumab when administered by extended interval dosing still needs to be fully determined. Therefore, the benefit/risk balance of extended interval dosing is unknown. 44
Exit strategies in people with high PML risk
A switch to an alternative treatment in people with MS who respond to natalizumab but are exposed to a higher PML risk based on the anti-JC virus index level represents an important therapeutic challenge in clinical practice.41,105 In cases where natalizumab should be discontinued, it is important to have a clear exit strategy, as there is a risk of reactivation of MS disease activity in people who have stopped natalizumab treatment, particularly in those with a longer treatment gap after its withdrawal. 106 This risk of rebound can be mitigated by timely initiation of another HET after cessation of natalizumab.106,107 In the experience of the authors, the washout period should be no greater than 8 weeks, and as early as 4–6 weeks, or even sooner as long as the person has immunocompetence. Furthermore, the risk of carryover PML (PML that develops a few months after stopping natalizumab treatment and starting an alternative DMT) should also be thought of. 41 To lower this risk, lower-efficacy oral maintenance therapies may occasionally be considered to bridge patients before switching to another treatment option. 41
A systematic review indicated that anti-CD20 monoclonal antibody therapy is a suitable option after natalizumab treatment, with the demonstration of low rates of relapse and carryover PML. 108 The at-risk period for carryover PML is probably less than 12 months after switching from natalizumab. 41 Furthermore, two observational, retrospective studies have shown ocrelizumab specifically to be an effective exit strategy after natalizumab discontinuation.105,109 The first study demonstrated ocrelizumab to be superior to fingolimod, with a 70% lower relapse rate at 1 year 109 ; the second study showed a lower risk of relapses and new MRI activity for people switching from natalizumab to ocrelizumab versus cladribine. 105 The reversibility of natalizumab allows for increased flexibility in treatment sequencing, 64 as discussed in the section on “Reversibility.”
Practical guidance on the clinical use of natalizumab
Treatment strategies
IV and subcutaneous administrations
Natalizumab was first approved in 2004 as 300 mg IV infusion Q4W, 2 with subcutaneous (SC) administration approved by the EMA in 202144,110 following the pivotal DELIVER and REFINE randomized controlled trials,111,112 where the FDA has yet to approve. 43 Both DELIVER and REFINE demonstrated SC-natalizumab to be reasonably comparable to IV-natalizumab in regards to pharmacokinetics/dynamics, with REFINE also showing comparable safety and efficacy between the two formulations,111,112 and a post hoc analysis of REFINE has further demonstrated comparable efficacy with IV-natalizumab by reporting non-inferiority of natalizumab SC on ARR. 113 However, there have been some concerns regarding low drug concentrations with SC administration, potentially affecting those on extended interval dosing or those with a higher body mass index (BMI),114,115 suggesting that SC administration may not be the right application for these individuals. Regarding extended interval dosing, a retrospective study comparing the performance of extended interval dosing versus standard interval dosing found no difference in the primary analysis, though an exploratory BMI analysis revealed a potential interaction between high BMI and disease activity in those treated with extended interval dosing. 116
SC-natalizumab offers the potential to reduce the impact of time and healthcare costs for people with MS, 117 with a recent trend toward patient and medical staff preference for the SC formulation, largely due to its convenience and time-saving benefits. 110 The different formulations can offer pros and cons depending on the individual, and given the preference-sensitive nature of treatment choice, the decision between IV and SC administration should be made through shared decision-making between the person with MS and their HCP. 110
Early use
DMTs are more effective when the inflammatory stage is prominent, such as in early disease. 118 In highly active MS, HETs such as natalizumab and ocrelizumab are needed early in the disease course to inhibit inflammation and prevent disability accrual resulting from multiple relapses. 118 Several studies have demonstrated the benefit of early use of natalizumab. A retrospective study of DMT-naïve people showed that early initiation of natalizumab was associated with a significantly reduced relapse rate and lower risk of confirmed disability worsening compared to a first-line treatment with a lower-efficacy DMT (Figure 1(b)). 40 Another retrospective study, including 281 people with highly active MS starting HET with either natalizumab or ocrelizumab as first-line therapy within 1 year of diagnosis, found that the probability of achieving NEDA-3 was 66% with ocrelizumab and 68% with natalizumab. 118 Additionally, a cross-sectional study from Argentina reported that people with MS starting HET, including natalizumab, early in the disease course had a significantly lower burden of treatment and higher QoL than people receiving escalation therapy. 119 In an Austrian observational cohort study, 743 treatment-naïve people who started early intensive treatment, including 283 people receiving natalizumab, had decreased probability of relapse and EDSS progression compared with those who were treated with an escalation approach. 120 Furthermore, research into the potential benefits of very early use of natalizumab is ongoing with the AttackMS clinical trial, which is investigating the safety and efficacy of initiating natalizumab treatment within 14 days of MS symptom onset (ClinicalTrials.gov identifier: NCT05418010).
Switching to natalizumab from lower-efficacy therapies or other HETs
The ECTRIMS/EAN guidelines for the treatment of MS recommend switching people treated with IFN or glatiramer acetate who show evidence of disease activity to a more efficacious drug, with studies demonstrating benefit in switching to alemtuzumab, fingolimod, or natalizumab. 3 A real-world study of 2000 people with RRMS from the MSBase registry found that people demonstrated better outcomes in relapse rate and disability improvement with natalizumab than with fingolimod when switching from lower-efficacy DMTs. 121 Another real-world study using data from the MSBase registry compared the effectiveness of switching to ocrelizumab, cladribine, or natalizumab in 1045 people with RRMS who had previously been taking fingolimod and found that after fingolimod cessation, ocrelizumab and natalizumab were more effective in reducing relapses than cladribine. 122 Furthermore, in the authors’ opinion, a switch to natalizumab may be appropriate for people receiving anti-CD20 therapies who have an increased incidence of infection.
Vaccination and bridging therapy
Immunosuppressive therapy for MS, particularly anti-CD20 therapies that exert their effects via lymphocyte depletion, may compromise the desired immune response of vaccination. 123 Consequently, clinicians are faced with a choice to either delay treatment with these agents until vaccination schedules are completed and risk adversely affecting MS prognosis or to rapidly start anti-CD20 therapy without prior vaccination, which could potentially expose people to severe, vaccine-preventable infections. 123 Furthermore, unlike natalizumab, treatments that cause long-term B-cell depletion are associated with severe side effects such as hypogammaglobulinemia and infections. However, for ocrelizumab, this may be mitigated by extended interval dosing. 124
Results of a cohort study suggest that immunization with inactivated vaccines against hepatitis A, hepatitis B, and mRNA COVID-19 is safe and immunogenic in people with highly active MS receiving treatment with natalizumab. 123 Other studies have found that natalizumab treatment did not affect immunization with influenza or tetanus vaccines. 125 In contrast, while people treated with B-cell depleting ocrelizumab can mount an attenuated humoral response to inactivated vaccines, 126 available data indicate these people have a reduced humoral response to SARS-CoV-2 vaccines. 127
Regarding live vaccines, while generally not recommended for those on immunosuppressive/immunomodulating therapies, recent reports have shown that for people with MS treated with natalizumab, vaccination against varicella-zoster virus in people with highly active MS was not associated with AEs or increased disease activity, 128 and vaccination against yellow fever was not associated with worsening of MS disease activity or relapse rate in French and Swiss populations, respectively.129,130 However, there is one report of vaccine-associated measles in a person with MS treated with natalizumab. 131
For people with highly active MS who require both immunization and HET, natalizumab treatment may be used as a bridging therapy for a short-term period (3–6 months) while vaccination schedules have been completed, in order to prevent treatment delays. 123 Both the rapid onset of action and the reversibility of natalizumab treatment may make it suitable for use as a bridging therapy; however, this should be done with caution as a short exposure to natalizumab with a gap before redosing has been associated with higher incidences of anti-natalizumab antibodies and hypersensitivity reactions. 132 Nonetheless, the use of natalizumab during vaccination is supported by a study which demonstrated that switching DMTs from sphingosine 1-phosphate receptor modulators to natalizumab to restore lymphocyte counts prior to SARS-CoV-2 mRNA vaccination may result in a robust immune response after vaccination and avoid the risk of relapse associated with DMT interruption. 133
Use of natalizumab in specific populations
Pregnancy and breastfeeding
Treating pregnant women with RRMS needs careful consideration of the risks and benefits to maternal and fetal health. 134 The natalizumab U.S. FDA prescribing information recommends use during pregnancy only if the potential benefit justifies the potential risk to the fetus, 43 while the natalizumab EMA summary of product characteristics recommends that discontinuation should be considered if a woman becomes pregnant while taking natalizumab, following a risk/benefit evaluation.9,44 However, due to the risk of reactivation or rebound of disease activity upon cessation of natalizumab treatment, there is a rationale for continuing natalizumab at least until pregnancy is confirmed, or even during pregnancy in people with higher disease activity, as antibodies, including natalizumab, only minimally cross the placenta during the first trimester. 125
An observational cohort study including 350 pregnant women with MS exposed to natalizumab, who either stopped treatment during their first trimester or continued beyond, showed that maintaining treatment beyond the first trimester may protect against disability worsening; although there was a higher fetal risk for hematological abnormalities, most did not need treatment. 134 Additionally, an Italian study of 170 pregnancies found that in women with MS treated with natalizumab before conception, continuation of natalizumab throughout pregnancy and its early resumption after delivery mitigated the risk of clinical and radiological disease reactivation and that this approach had no major impact on newborns’ outcomes. 135
Recently, a Delphi consensus panel reached agreement that when pregnancy is planned in people with high MS disease activity, in selected cases, either immune reconstitution therapy or natalizumab should be used. 136 Furthermore, consensus was reached that natalizumab during pregnancy should be continued in people with highly active disease, 136 given the risk of rebound disease activity with natalizumab discontinuation. 137 For people with historically highly active disease who decide to continue natalizumab during pregnancy, it may be beneficial to extend the dosing interval to every 6–8 weeks until 34-week gestation, but it should not be continued after this point.137,138 Following delivery, it is recommended to resume natalizumab treatment in the first 4 weeks after birth to minimize the risk of return of disease activity. 138
More data are needed on breastfeeding while undergoing treatment with natalizumab; however, it has been shown that natalizumab transfer to human breast milk is low, with a mean relative infant dose (a metric comparing the infant with maternal drug exposure) of 0.04% (standard deviation 0.03), which is far below the 10% threshold of concern for this metric. 139 Interdisciplinary care is vital for people with MS who are pregnant and/or breastfeeding; MS teams should work closely with specialized obstetrics and neonatology teams for off-label decisions, particularly if immunotherapy continues during pregnancy. 137
Pediatric-onset MS
There is a need for early diagnosis and treatment of MS in children, as, although the incidence and prevalence of MS is lower for children than for adults, it is the most common cause of non-traumatic neurological disability in young adults. 140 Furthermore, pediatric-onset MS also has a higher relapse rate than adult-onset MS, which is thought to relate to a more inflammatory disease course in pediatric MS. 141 Historically, initiation of IFN beta or glatiramer acetate was recommended, with the use of HET reserved for people with more active disease. 142 While there are no recent and specific guidelines for the treatment of pediatric-onset MS, there is increasing evidence for a strong effect of HET on reducing relapse rate, disability, and MRI activity in this population, and as such, early use of HET has been suggested as a new therapeutic approach for children with MS. 142
Several studies have demonstrated the safety and efficacy of natalizumab in pediatric-onset MS, with more real-world data available on natalizumab in this population than for anti-CD20 therapies. 142 A post-marketing meta-analysis conducted using data from 621 children with MS treated with natalizumab, 158 of whom had data available prior to treatment, demonstrated a reduction in ARR during treatment. 44 A small Spanish study of nine children with MS found that safety and efficacy of natalizumab in children was in line with the experience published in adult populations. 143 Another study of 101 children with MS from an Italian cohort found that natalizumab was well tolerated, and efficacious in most cases. 144 The TyPed study, which included 21 children with MS treated with natalizumab in Portugal, also demonstrated that natalizumab may be an effective and well-tolerated DMT for pediatric MS, with results in line with the adult population. 145 Furthermore, a retrospective study evaluating the efficacy and safety of natalizumab in both adults and children with MS found that clinical and radiological outcomes were similar in both populations, with fewer PIRA events observed in children with MS compared with adults. 146 Taken together, these data support the potential use of natalizumab as a well-tolerated and effective treatment for pediatric-onset MS.
Only a few reports of PML exist in the pediatric population with MS. 147 As PML is the result of reactivation of a primary, usually asymptomatic infection with the JC virus, children may be at lower risk for this condition, as long as JC virus infection has not yet occurred. 147 Another reason may be that the number of children with MS treated with natalizumab is possibly too low to draw any significant risk estimates, as the safety and efficacy of natalizumab in children and adolescents has not been established. 145
Comorbidities
Comorbidities are common in people with MS and are associated with higher relapse rates, greater physical and cognitive impairments, lower HRQoL, and increased mortality. 148 Autoimmune comorbidities may pose particular challenges for people with MS, especially when a DMT does not successfully treat both the MS and the comorbid disease, or may even exacerbate the latter, as seen with psoriasis. 149 However, natalizumab may be a viable treatment option. In a retrospective analysis of 18 people with MS and comorbid psoriasis, who were switched from other DMTs due to lack of treatment efficacy for MS or worsening or development of psoriasis, improvement in both MS and psoriasis was observed after switching to natalizumab from other DMTs. 149
Another autoimmune disease that shares comorbidity with MS is inflammatory bowel disease, including Crohn’s disease and ulcerative colitis, due to their common epidemiological and immunological patterns.150,151 In the U.S., natalizumab is indicated for the treatment of Crohn’s disease as well as MS, so it could be used to treat individuals with both conditions. 43 People with Crohn’s disease have shown improvements in randomized controlled trials.152,153
A comorbidity that can complicate treatment decisions in MS is liver disease due to the risk of drug-induced liver injury. 154 However, a case report has described natalizumab with ursodeoxycholic acid as being used successfully in a person with comorbid progressive familial intrahepatic cholestasis type-3 after other DMTs were tried and associated with drug-induced liver injury. 154
Cardiovascular comorbidities are associated with the risk of MS progression. An observational, prospective cohort study in people with RRMS suggested that natalizumab treatment had a beneficial effect on lipid profile and increased uric acid levels during 12.9 ± 6.2 months of treatment; subsequent association with disease outcomes is unknown but of interest, 155 though increased uric acid levels have previously been associated with reduced risk of relapse and disease disability. 156 Taken together, natalizumab is approved as an effective treatment for MS and Crohn’s disease and may show beneficial effects in people with MS who have other comorbidities.
What have we learned from natalizumab and what does the future hold?
As the first monoclonal antibody to be approved for the treatment of MS, 20 years of natalizumab use has brought much to the MS community. 1 It has a unique mode of action 6 that, although likely to contribute to the increased risk of PML, 88 is also responsible for its efficacy and unique properties of rapid onset of action and reversibility, the latter of which avoids long-term immunosuppression.57,64 These properties, and natalizumab’s attractive overall risk/benefit profile, may make it an option for people who are complex to treat, including those in need of rapid onset of treatment effects, those who are pregnant or breastfeeding, children with MS, and people with MS with comorbidities. Over 20 years of experience with natalizumab has not only increased our understanding of the different MS treatment strategies involving the use of this agent and the types of people that are likely to derive the most benefit, but it also increased our understanding of the immunology of the disease itself, along with some of the first robust risk management strategies in MS. As the diagnostic criteria for MS evolves to improve early diagnosis, early use of HET, including natalizumab, will become increasingly important in managing the disease.
The definition of “rapidly evolving severe MS” resulted from post hoc analyses of large clinical trials to identify people who might benefit from early treatment with moderately high-efficacy drugs or HET. 157 As well as the introduction of a new therapeutic strategy for the treatment of MS, the modulation of immune cell trafficking across the BBB is an example of reverse translational medicine. 158 Data from clinical trials of natalizumab and fingolimod have revealed the involvement of different compartments in relapsing versus non-relapsing MS immune biology and contributed to our understanding of CNS immune surveillance. 158
In addition to paving the way for new therapeutic approaches for the management of MS as the first high-efficacy monoclonal antibody therapy, 159 natalizumab was also “ahead of the curve” in terms of the management of AEs. The risk of PML associated with natalizumab led to the development of the first structured risk management plan in MS treatment, allowing for individualization and risk stratification, which has been successful in reducing the incidence of PML over time.8,95 Looking forward, further progress is expected in understanding why some at-risk people do not develop PML, particularly the role of genetics in individual PML risk. 160
Finally, natalizumab may have potential in other indications beyond MS. For example, natalizumab has been tested as a treatment for metastatic osteosarcoma (ClinicalTrials.gov ID NCT03811886). As metastatic cancers use the VLA-4–VCAM-1 interaction to spread to the lung and other sites, anti-α4 integrin inhibitors, such as natalizumab, may reduce the risk of these cancers spreading. 161 Moreover, in the authors’ opinion, natalizumab may have the potential to treat CNS complications associated with cancer immunotherapies. First, checkpoint inhibitors can be associated with neurological immune-related AEs due to upregulated activity of the immune system, where these people are generally responsive to immune-modulating therapy. 162 Second, chimeric antigen receptor (CAR) T-cell therapy can result in immune effector cell-associated neurotoxicity syndrome due to the diffusion of cytokines and transmigration of CAR T cells, endogenous T cells, and monocytes into the cerebrospinal fluid and CNS. 163 In addition, the authors also suggest that natalizumab may have application in other CNS complications such as tuberculosis meningitis, which is underpinned by BBB dysfunction and the influx of leukocytes into the CNS. 164 Taken together, there is the potential to bring 20 years of experience with natalizumab in MS to new therapy areas.
Footnotes
Acknowledgements
Medical writing support, under the guidance of the authors, was provided by Holly Oates, PhD, CMC Connect, a division of IPG Health Medical Communications, and was funded by Sandoz, in accordance with Good Publication Practice (GPP 2022) guidelines.
