Abstract
Background:
Aggressive disease control soon after multiple sclerosis (MS) diagnosis may prevent irreversible neurological damage, and therefore early initiation of a high-efficacy disease-modifying therapy (DMT) is of clinical relevance.
Objectives:
Evaluate long-term clinical outcomes in patients with MS who initiated treatment with either natalizumab or a BRACETD therapy (interferon beta, glatiramer acetate, teriflunomide, or dimethyl fumarate).
Design:
This retrospective analysis utilized data from MSBase to create a matched population allowing comparison of first-line natalizumab to first-line BRACETD.
Methods:
This study included patients who initiated treatment either with natalizumab or a BRACETD DMT within 1 year of MS diagnosis and continued treatment for ⩾6 months, after which patients could switch DMTs or discontinue treatment. Patients had a minimum follow-up time of ⩾60 months from initiation. A subgroup analysis compared the natalizumab group to patients in the BRACETD group who escalated therapy after 6 months. Outcomes included unadjusted annualized relapse rates (ARRs), time-to-first relapse, time-to-first confirmed disability improvement (CDI), and time-to-first confirmed disability worsening (CDW).
Results:
After 1:1 propensity score matching, 355 BRACETD patients were matched to 355 natalizumab patients. Patients initiating natalizumab were less likely to experience a relapse over the duration of follow-up, with ARRs [95% confidence interval (CI)] of 0.080 (0.070–0.092) for natalizumab patients and 0.191 (0.178–0.205) for BRACETD patients (p < 0.0001). A Cox regression model of time-to-first relapse showed a reduced risk of relapse for natalizumab patients [hazard ratio (95% CI) of 0.52 (0.42–0.65); p < 0.001] and a more favorable time-to-first CDI. The risk of CDW was similar between groups. The subgroup analysis showed an increased relapse risk as well as a significantly higher risk of CDW for BRACETD patients.
Conclusion:
Early initiation of natalizumab produced long-term benefits in relapse outcomes in comparison with BRACETD, regardless of a subsequent escalation in therapy.
Introduction
Multiple sclerosis (MS) is a chronic inflammatory disease of the central nervous system (CNS), which can result in disability accumulation from neurological damage related to relapse and disease progression independent of relapse activity.1,2 The initiation of treatment with a disease-modifying therapy (DMT) at the time of first presentation of symptoms consistent with or suggestive of MS provides benefits over delaying treatment.3–8 Long-term follow-up of patients from randomized trials has shown a greater benefit for patients originally assigned to treatment groups as opposed to those originally assigned to placebo groups.9–14
The choice of which DMT is used as the first-line treatment option may be based on a number of factors including a patient’s disease presentation and prognosis 6 as well as the accessibility of DMTs within the given healthcare/payer system. 15 Health authorities and/or payers often require that first-line therapy for MS is a relatively safe DMT with low to moderate efficacy, and escalation of therapy to high-efficacy DMT is reserved for patients who fail the initial DMT.16–18 First-line therapy is often one of the BRACETD group of DMTs, consisting of interferon beta, glatiramer acetate, teriflunomide, or dimethyl fumarate.
An alternative strategy is the use of high-efficacy DMTs as a first-line treatment option. This is already an established treatment strategy in oncology and rheumatology practice.19,20 Early use of high-efficacy drugs, including the ‘induction’ strategy (e.g. early and brief use of a high-efficacy DMT followed by maintenance with a lower-efficacy DMT with less safety risk), has traditionally been applied only to individuals who present with a highly aggressive course of early MS.16–18,21 However, early MS is characterized in many patients by accumulation of inflammatory lesions that are associated with a greater risk of progressive MS after a period of time. Early MS is now considered by many as a ‘therapeutic window’, that is, an opportunity for early and complete control of lesion development and relapses to prevent the initiation of neurodegenerative processes and irreversible CNS damage.15,18,21,22 To take advantage of this therapeutic window, the use of high-efficacy drugs in early MS is growing.17,18,23,24 A growing body of evidence from observational studies and subgroup analyses from randomized trials now supports the early use of high-efficacy DMTs in patients without highly active early MS, showing short- and long-term benefits for disease progression and disability accumulation.15,18,25,26
Natalizumab (TYSABRI®) is a high-efficacy DMT approved for the treatment of relapsing forms of MS on the basis of randomized clinical trials27,28 and real-world studies.29–39 Natalizumab has shown a high degree of disease control in patients as an early treatment option.23,40–42 However, outcome data on long-term clinical benefits of natalizumab as a first-line therapy in comparison with lower-efficacy DMTs are still limited. Natalizumab is associated with a risk of progressive multifocal leukoencephalopathy (PML), a rare but potentially lethal disease caused by infection of the CNS by the John Cunningham virus (JCV).43,44 Risk stratification and patient monitoring guidelines have been developed to inform the decision to initiate or discontinue natalizumab. 45
We evaluated long-term clinical outcomes in patients with a first diagnosis of clinically definite MS who initiated treatment with natalizumab in comparison with those who initiated treatment with a BRACETD therapy, regardless of whether patients discontinued or switched therapy after the initial treatment period. This analysis utilizes retrospective data from MSBase to create a weighted population, balanced across baseline covariates, which allows comparison of first-line natalizumab DMT to first-line BRACETD DMT. A subgroup analysis investigated treatment effect differences that might arise from the common scenario of patients in the BRACETD group who escalated therapy after 6 months of treatment with their index DMT.
Methods
Study design and patients
Data were collected from patients initiating therapy since the initiation of the MSBase registry in 2001, with data extraction on 2 November 2020. To meet the criteria for inclusion, patients had a diagnosis of clinically definite MS 46 ; had initiated (index) treatment either with natalizumab (natalizumab patients) or with interferon beta-1b (BETAFERON®/BETASERON [US]®/EXTAVIA®), subcutaneous interferon beta-1a (Rebif®), intramuscular interferon beta-1a (AVONEX®), glatiramer acetate (COPAXONE®), teriflunomide (AUBAGIO®), or dimethyl fumarate (TECFIDERA®), collectively termed BRACETD, within 1 year of MS diagnosis; were ⩾18 years of age at the initiation of index therapy; had a treatment duration of the index therapy for ⩾6 months, after which patients could switch to an alternative DMT or discontinue treatment; and had a minimum follow-up time of ⩾60 months from initiation of index therapy to the last available clinical assessment or discontinuation of last therapy. Patients were excluded if they had received prior treatment with any MS DMT (including during participation in randomized clinical trials), or if they had <6 months of index therapy treatment.
The baseline of each patient was defined at the time of initiation of first-line treatment (natalizumab or BRACETD). Exposure to index therapy (administration of first-line treatment) is defined as time between index-therapy initiation and discontinuation of first-line treatment. Total follow-up time is defined as time between index-therapy initiation and last available Expanded Disability Status Scale (EDSS) measurement date or discontinuation of last therapy, whichever occurred last (regardless of index-therapy administration). Follow-up time after discontinuation was defined as time between index-therapy discontinuation and last available EDSS measurement date or discontinuation of last therapy, whichever occurred last (regardless of index-therapy administration).
Outcomes and assessments
Study outcomes included relapse, assessed as unadjusted annualized relapse rates (ARRs); confirmed disability improvement (CDI), defined as a decrease of ⩾1 point from a baseline EDSS score of ⩾2.0 and confirmed after ⩾24 weeks; and confirmed disability worsening (CDW), defined as an increase of ⩾0.5 point from a baseline EDSS score of >5.5, ⩾1.0 point for those with a baseline EDSS score between 1.0 and 5.5, inclusive, and ⩾1.5 points for those with a baseline EDSS score of 0, confirmed after ⩾24 weeks. All outcomes were calculated from the initiation of first-line treatment (natalizumab or BRACETD) to the end of follow-up.
Statistical analysis
Patients who received natalizumab or BRACETD index therapy were 1:1 propensity score-matched for age, MS disease duration, relapses in the prior year, relapses in the prior year requiring steroid treatment or hospitalization, and EDSS score (all at the time of index-therapy initiation), as well as sex until pairwise standardized differences were acceptably low (Supplemental Table 1).
Times to first relapse, first CDI, and first CDW were estimated using Kaplan-Meier methods. Hazard ratios (HRs) for natalizumab versus BRACETD patients were based on a Cox regression model with treatment as the classification variable.
The primary analysis population was defined as all patients receiving at least 6 months of either natalizumab or BRACETD and having an additional minimal follow-up time of 60 months from initiation of first-line treatment to end of follow-up. A subgroup included patients in the BRACETD group with 60 months of follow-up and at least 6 months of BRACETD with an escalation therapy taken before 36 months.
Results
Patients
As of 2 November 2020, 12,033 patients who initiated treatment with BRACETD and 360 patients who initiated treatment with natalizumab met the inclusion criteria for this study. Baseline characteristics for the entire unmatched population are presented in Supplemental Table 2. After 1:1 propensity score matching, 355 BRACETD patients were matched to 355 natalizumab patients. The baseline characteristics of the two matched groups did not display significant differences (Table 1). The mean age at initiation of index treatment was 34 years, and 61.4% of this population were female. Patients on average had about 2.5–2.8 years of disease duration and a median EDSS score of 2.5. In the previous year, the mean (SD) numbers of relapses were 1.27 (1.10) for the natalizumab group and 1.20 (1.01) for the BRACETD group. Mean (SD) follow-up was 4.48 (2.68) years for the natalizumab patients and 5.12 (3.99) years for the BRACETD patients.
Baseline characteristics of 1:1 propensity score-matched cohorts.

BRACETD, interferon beta, glatiramer acetate, teriflunomide, or dimethyl fumarate; EDSS, Expanded Disability Status Scale; IQR, interquartile range.
Outcomes
Relapses
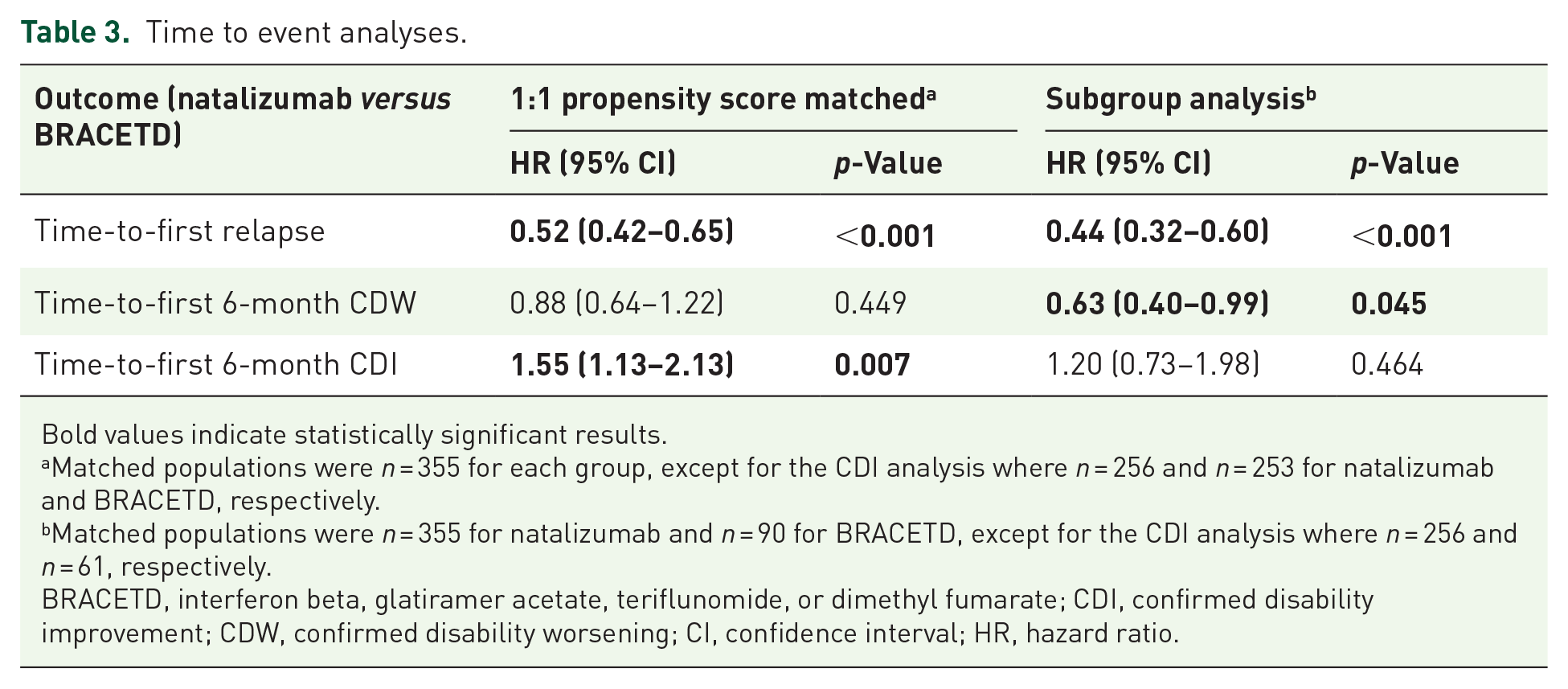
Patients initiating natalizumab in the intent-to-treat population were less likely to experience a relapse over the duration of follow-up. Natalizumab patients experienced 215 relapses over a cumulative 2685 on-treatment follow-up years, whereas BRACETD patients experienced 788 relapses over 4128 years of on-treatment follow-up. The resulting ARRs [95% confidence interval (CI)] were 0.080 (0.070–0.092) for natalizumab patients and 0.191 (0.178–0.205) for BRACETD patients (p < 0.0001; Table 2). The Cox regression model for the calculated time-to-first relapse showed a reduced risk of relapse for natalizumab patients, with a HR (95% CI) of 0.52 (0.42–0.65; p < 0.001) for natalizumab patients (Table 3). Kaplan-Meier estimates for the estimated proportion of relapse-free patients over 15 years are shown in Figure 1.
Relapse outcomes.

Versus natalizumab.
Subgroup analysis of BRACETD patients who escalated to a high-efficacy DMT. Time on treatment refers to time on BRACETD DMT and does not include time on subsequent DMT.
ARR, annualized relapse rate; BRACETD, interferon beta, glatiramer acetate, teriflunomide, or dimethyl fumarate; CI, confidence interval.
Time to event analyses.
Bold values indicate statistically significant results.
Matched populations were n = 355 for each group, except for the CDI analysis where n = 256 and n = 253 for natalizumab and BRACETD, respectively.
Matched populations were n = 355 for natalizumab and n = 90 for BRACETD, except for the CDI analysis where n = 256 and n = 61, respectively.
BRACETD, interferon beta, glatiramer acetate, teriflunomide, or dimethyl fumarate; CDI, confirmed disability improvement; CDW, confirmed disability worsening; CI, confidence interval; HR, hazard ratio.

Estimated proportion of relapse-free patients.
Disability outcomes
The 6-month CDW outcomes were similar between treatment groups, but favored natalizumab numerically [HR for natalizumab (n = 355) versus BRACETD (n = 355) = 0.88; 95% CI: 0.64–1.22; p = 0.449; Table 3]. The time to 6-month CDI, however, significantly favored natalizumab [HR for natalizumab (n = 256) versus BRACETD (n = 253) = 1.55; 95% CI: 1.13–2.13; p = 0.007]. Kaplan-Meier estimates for the estimated proportion of patients experiencing a progression event or improvement event over 15 years are shown in Figures 2 and 3.

Estimated proportion of progression-free patients.
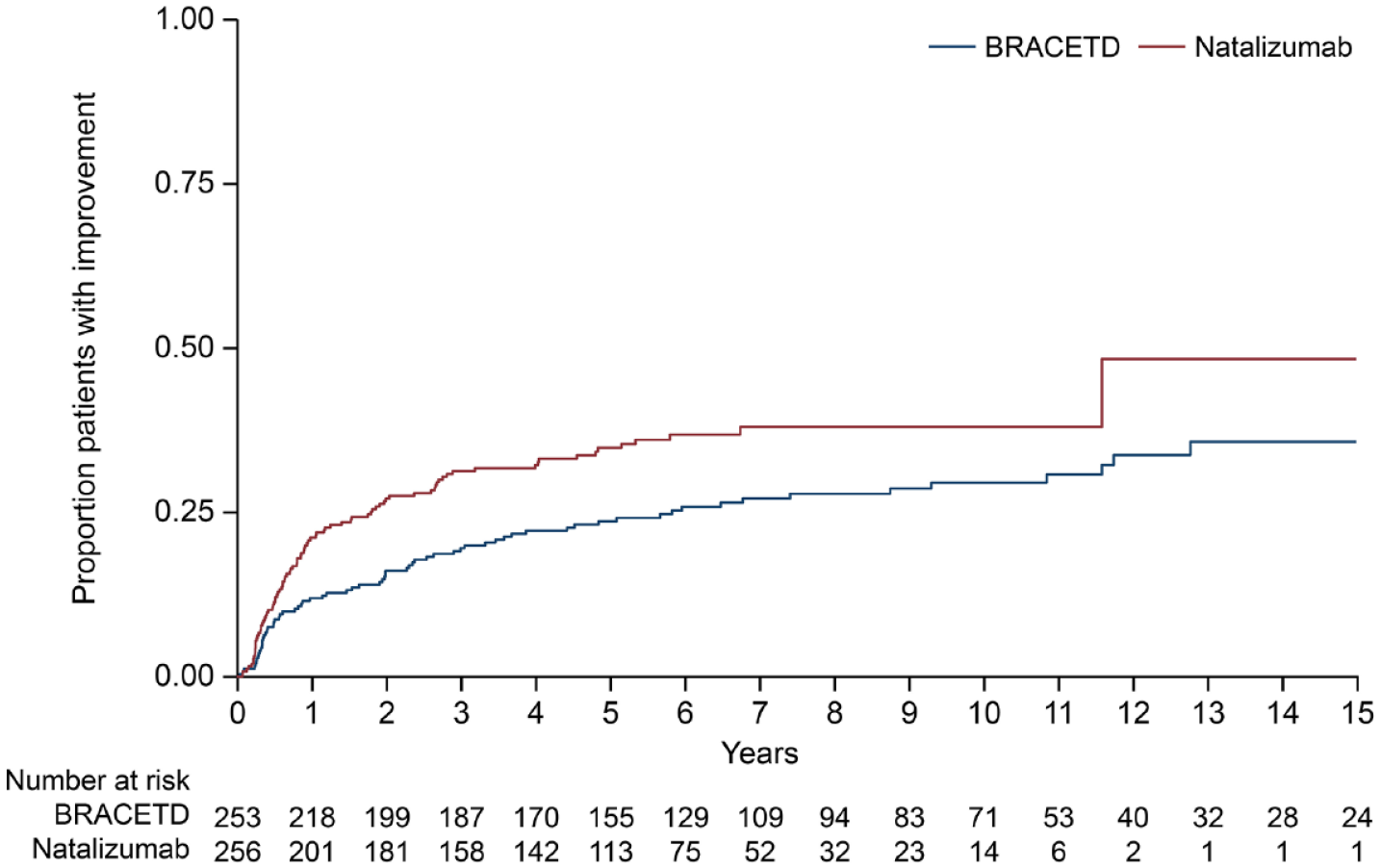
Estimated proportion of patients with improvement.
Treatment discontinuation and switching
In the overall population, 67.8% (244/360) of natalizumab users and 82.3% (9911/12,033) of the BRACETD group reported discontinuing treatment within the follow-up period. The reason for discontinuation was not reported in 33.2% and 43.2% of cases, respectively. However, among all discontinuations, the most common reason for discontinuing natalizumab was a scheduled stop (33.2) and for discontinuing BRACETD, lack of improvement (15.9%; Supplemental Table 3). The timing and reasons for scheduled stops were at the discretion of the treating physician.
DMT switches were recorded in the matched groups to know which therapies patients from each group may have switched to after at least 6 months on their index DMT (Tables 4 and 5). Within the matched groups, the natalizumab group was less likely to report a switch in treatment during follow-up (56.9% of patients did not have a recorded switch from the natalizumab group, in comparison with 30.7% from the BRACETD group). The BRACETD group was more likely to laterally switch to another moderate-efficacy DMT than to escalate treatment (57.7% versus 42.3%, respectively). The percentage of those who switched with an escalation increased during the second switch to 55.7%. The natalizumab group was more likely to switch to another high-efficacy DMT for both the first switch (75.8% of first switches) and the second switch (83.8% of second switches). It should be noted that for both index-therapy groups, the most popular first-switch DMT was fingolimod (22.8% for the BRACETD group and 43.8% for the natalizumab group).
Post-index DMT switch matrices for the BRACETD group (n = 355).
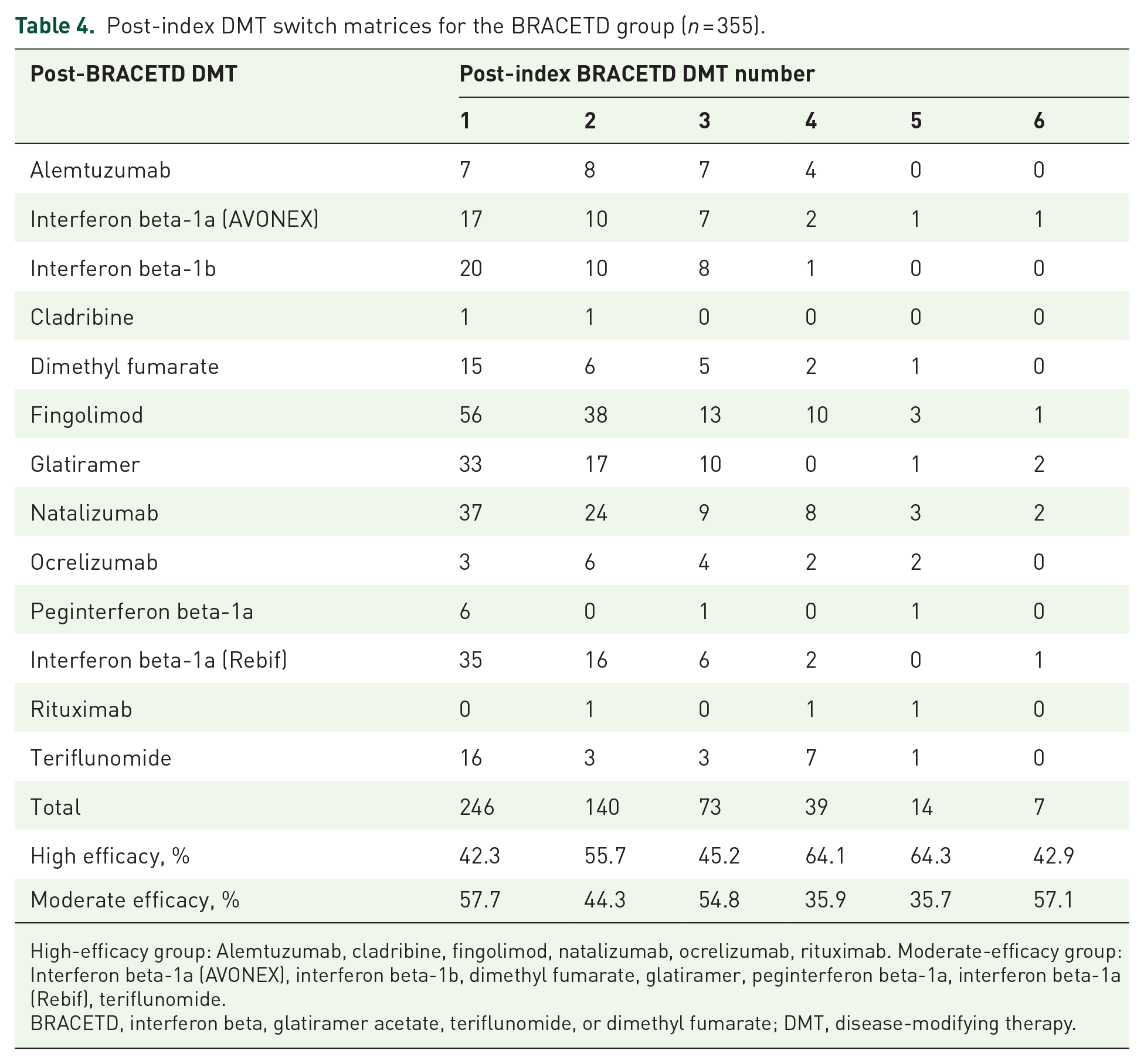
High-efficacy group: Alemtuzumab, cladribine, fingolimod, natalizumab, ocrelizumab, rituximab. Moderate-efficacy group: Interferon beta-1a (AVONEX), interferon beta-1b, dimethyl fumarate, glatiramer, peginterferon beta-1a, interferon beta-1a (Rebif), teriflunomide.
BRACETD, interferon beta, glatiramer acetate, teriflunomide, or dimethyl fumarate; DMT, disease-modifying therapy.
Post-index DMT switch matrices for the natalizumab group (n = 355).

High-efficacy group: Alemtuzumab, cladribine, daclizumab, fingolimod, natalizumab, ocrelizumab, rituximab. Moderate-efficacy group: Interferon beta-1a (AVONEX), interferon beta-1b, dimethyl fumarate, glatiramer, peginterferon beta-1a, interferon beta-1a (Rebif), teriflunomide.
DMT, disease-modifying therapy.
Subgroup analyses
To investigate the possible treatment outcome differences in common discontinuation scenarios, we compared a BRACETD patient subgroup switching from index therapy within 36 months. In this analysis, the 355 natalizumab patients were compared to a subgroup of 90 BRACETD patients with at least 6 months of BRACETD, who then escalated therapy within 36 months. Baseline characteristics for these groups were similar (Supplemental Table 4). Relapse outcomes for the BRACETD subgroup that escalated therapy within 36 months were worse than those for the whole BRACETD group and significantly worse in comparison with the natalizumab group, with an ARR of 0.239 (95% CI: 0.208–0.272; p < 0.001 in comparison with the natalizumab group) (Tables 2 and 3). The time-to-first relapse analysis showed a significant risk for the BRACETD subgroup in comparison with the natalizumab group, with an HR = 0.44 (95% CI: 0.32–0.60; p < 0.001). Although the BRACETD subgroup was more likely to experience a first 6-month CDW (HR = 0.63 for natalizumab versus BRACETD; 95% CI: 0.40–0.99; p = 0.045), there was no significant difference in achieving CDI (HR = 1.20; 95% CI: 0.73–1.98; p = 0.464).
Discussion
This retrospective observational analysis of propensity score-matched patients from the MSBase registry showed that patients who initiate natalizumab treatment within 6 months of their MS diagnosis saw long-term benefits over at least 60 months of follow-up when compared with patients who initiated a BRACETD therapy. The ARR in the natalizumab cohort was lower over the duration of follow-up, and Cox proportional hazard modeling showed a reduced risk of relapse and a shorter time to CDI. Disability worsening (CDW), however, was similar between groups over 15 years.
In the escalation approach to choosing an initial therapy, low/moderate-efficacy DMTs are likely to be the initial treatment option for most patients, whereas high-efficacy DMTs are reserved as first-line treatments for patients with a highly active disease presentation. Through the process of 1:1 propensity score matching, the BRACETD group was likely enriched with patients with higher disease activity, as evidenced by an increase in the baseline mean relapses in the prior year from 1.05 before matching to 1.20 after matching and an increase in median EDSS score from 2.0 to 2.5. In the context of this matched population, the patients in the BRACETD group could have been candidates for treatment initiation with a high-efficacy DMT instead. The difference in relapse outcomes observed for these matched populations highlights benefit of high-efficacy DMT options as first-line therapy for patients newly diagnosed with MS.
By not censoring patients after a therapy switch, this analysis investigated long-term benefits of early initiation of natalizumab in controlling disease, regardless of subsequent DMT choices. Although there may be a variety of reasons for switching therapies after 6 months of treatment, historical observations suggest common scenarios for each group. For instance, patients from the BRACETD group would only be expected to switch or escalate therapies if their index therapy proved to be ineffective. In the matched BRACETD group, the majority (57.7%) of patients switched laterally to another low/moderate-efficacy DMT, indicating that disease activity did not warrant escalation or was not the primary concern. This is supported by data from this study, because even though the most commonly reported reason for index-therapy discontinuation within the overall BRACETD was ‘lack of improvement’, it only represented 27.9% (1572/5633) of cases with a known discontinuation reason. If in addition to ‘lack of improvement’, one considers ‘MRI (magnetic resonance imaging) activity’, ‘persistence of relapse’, and ‘progression of disease’ as evidence of a lack of effectiveness, then 42.3% (2385/5633) of BRACETD patients, but only 11.7% (19/163) of natalizumab patients, with a known discontinuation reason could be considered to have discontinued their index therapy because of ineffective treatment. Interestingly, 42.3% of BRACETD patients from the matched group switched to high-efficacy therapy after discontinuing their index therapy.
Within the overall natalizumab group, 49.7% (81/163) of the discontinuations with a known reason were attributed to a ‘scheduled stop’. Although the timing and reasons for scheduled stops were at the discretion of the treating physician, concern related to the development of PML has been the most common reason for natalizumab discontinuation in recent observational studies.29–31 Data for the natalizumab group were collected starting from 2004, when natalizumab was approved for use in treating MS, but it was not until 2012 when assays became available for evaluating JCV serostatus, an important marker for PML risk. Prior to treatment guidelines that became available in 2016 that considered both natalizumab exposure and quantitative JCV index, 44 a common strategy for PML prevention was to simply limit exposure to natalizumab, hence a scheduled stop. Therefore, it is likely that the high prevalence of this discontinuation reason in this study is related to management of PML risk for patients treated before updated guidelines and JCV antibody assays were available.
To investigate the possible differences resulting from a strategy of BRACETD group treatment switch to high-efficacy DMT versus first-line natalizumab, we compared the entire matched natalizumab population with 90 patients in the matched BRACETD cohort who were treated with at least 6 months of their index DMT but escalated within 36 months of initiation to a high-efficacy DMT. This scenario likely enriches the matched BRACETD subgroup with patients who switched because of the ineffectiveness of their index DMT. Regardless of which high-efficacy DMT the patients switched to, or how many subsequent switches were made, patients who initiated therapy with natalizumab saw benefits in relapse control overall, and HRs indicated that CDW was more likely in the BRACETD subgroup than in the natalizumab group, different to the primary analysis.
Relapse outcomes for either the overall BRACETD group, and especially the BRACETD escalation group, were worse than for the overall natalizumab population. Similar findings have previously been demonstrated for comparisons of early versus delayed high-efficacy DMT escalation (as a group comprising fingolimod, natalizumab, and alemtuzumab) in two seminal papers assessing either 6-month CDW 23 or time to secondary progressive MS onset. 24
A limitation to the overall analysis is the lack of alternative disease activity measures on CNS inflammation and neurodegeneration that can be used to compare long-term effects of early versus late DMT initiation paradigms. Although the DMTs studied may reduce relapses, the comparative physical protection from CNS damage offered by these DMTs remains unmeasured. As a clinical event, relapses reflect disease activity, and long-term disability may be correlated with relapse rate and severity. However, changes in brain volume and other MRI measures, along with the accumulation of disability and the rate of transition into progressive MS, could offer even more insight into the long-term advantages of early, aggressive treatment,26,47 and could also be used to improve baseline matching as baseline MRI lesion load in both brain and spinal cord is a likely variable in DMT choice decision-making. An additional limitation of the analysis is the reduction in sample size due to propensity score matching. For example, although a large number of patients from Turkey and Kuwait were included in the unmatched population (2157 and 473, respectively), only 33 patients from Turkey and 33 from Kuwait were included following propensity score matching; this was likely due to the unevenness of the contribution of these countries to the two subgroups, with only one patient from Turkey in the unmatched natalizumab cohort compared with 2156 in the BRACETD cohort.
Although this study reports an improvement in MS relapse outcomes, it is also important to consider the accumulation of disability in MS that occurs independent of relapse activity. There is a growing body of evidence which suggests that most disability accumulation in relapsing-remitting MS (RRMS) is not associated with relapses, demonstrating an underlying worsening of disease in RRMS patients even when relapse activity appears well controlled.2,48–50 Although shortened time to CDI was seen in the early natalizumab cohort, CDW was similar in both groups over 15 years. This study reports disability progression through changes in overall EDSS, however assessing composite confirmed disability progression through upper and lower extremity function may provide more information on disability progression than EDSS alone. 50
The same logic that led to an early intervention paradigm with frequent monitoring for treatment escalation, in which ‘time is brain’, also supports the early use of high-efficacy DMTs for treating RRMS. The benefit-risk profile of high-efficacy therapies should always be considered when making treatment decisions for newly diagnosed patients. Data showing the advantage of initiating high-efficacy therapies at diagnosis in terms of long-term disability progression will always be limited to observational studies, and longitudinal studies investigating the long-term risk profiles of many new, high-efficacy DMTs are still underway. To help bridge this data gap, two large randomized clinical trials have been initiated to test the impact of early high-efficacy versus escalation approaches (ClinicalTrials.gov NCT03535298 [DELIVER-MS] and NCT03500328 [TREAT-MS]). These randomized prospective studies include several disease measures, in addition to relapse outcomes, including MRI and various disability measurements, but remain short-term in nature. Studies spanning more than 5 years will always require real-world data sources, preferably prospectively acquired registry data with agreed common datasets.
Natalizumab is a highly effective choice as an initial DMT following a diagnosis of MS. Specifically, natalizumab has a rapid and reversible onset, demonstrates high efficacy in patients with early MS, and has a relatively low risk of PML within the first few years of use.23,40,41,43,44 Although the main safety concern for natalizumab is the risk of PML, risk stratification guidelines 45 and extended interval dosing regimens 51 reduce this risk and extend the amount of time an individual can safely utilize this highly efficacious DMT, in particular for JCV antibody negative populations. Long-term safety data now extends to over 10 years in the Tysabri Observational Program (TOP) study, and no safety concerns beyond PML have emerged. 29 Furthermore, natalizumab can be initiated without delay, because effective vaccination can be achieved subsequent to initiating natalizumab treatment, whereas there can be a need to delay the initiation of other high-efficacy DMTs for a month or more owing to their impact on vaccination efficacy once initiated. 52
Conclusion
In conclusion, this observational study shows that a treatment strategy with early initiation of natalizumab produced long-term benefits in comparison with early initiation of BRACETD, regardless of subsequent potential switches in therapy. Patients who initiated natalizumab within 6 months of their MS diagnosis had lower ARR over the duration of follow-up than patients who initiated a BRACETD therapy, with Cox proportional hazard modeling showing a reduced risk of relapse and shorter time to CDI in the natalizumab cohort. Though some patients may not be good candidates for natalizumab treatment because of safety concerns, the introduction of other high-efficacy DMTs to the MS treatment landscape has expanded treatment options, and a shift away from the escalation paradigm is increasingly adopted. 17
Supplemental Material
sj-docx-1-tan-10.1177_17562864231221331 – Supplemental material for Long-term clinical outcomes in patients with multiple sclerosis who are initiating disease-modifying therapy with natalizumab compared with BRACETD first-line therapies
Supplemental material, sj-docx-1-tan-10.1177_17562864231221331 for Long-term clinical outcomes in patients with multiple sclerosis who are initiating disease-modifying therapy with natalizumab compared with BRACETD first-line therapies by Helmut Butzkueven, Tomas Kalincik, Francesco Patti, Mark Slee, Bianca Weinstock-Guttman, Katherine Buzzard, Olga Skibina, Raed Alroughani, Alexandre Prat, Marc Girard, Dana Horakova, Eva Kubala Havrdova, Anneke Van der Walt, Sara Eichau, Robert Hyde, Nolan Campbell, Karthik Bodhinathan and Tim Spelman in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
This manuscript is dedicated to our valued colleague Dr. Yara Fragoso, who sadly passed away during the development of this manuscript. Luke Ward, PhD, of Ashfield MedComms (Middletown, CT, USA), an Inizio Company, assisted with drafting the manuscript. Celia Nelson of Ashfield MedComms edited and styled the manuscript per journal requirements and submitted the manuscript on behalf of the authors. Biogen funded medical writing and editorial support in the development of this manuscript. The authors had full editorial control and provided final approval of all content.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
