Abstract
Myasthenia gravis is a chronic autoimmune disorder caused by antibodies directed against the neuromuscular junction. Some patients may have an associated thymoma, which confers a worse prognosis. Eculizumab, a monoclonal antibody that inhibits the activation of terminal complement, has recently been approved for the treatment of refractory generalized myasthenia gravis. This is an early case report of thymoma-associated refractory myasthenia gravis successfully treated with eculizumab in a real-world setting.
Introduction
Myasthenia gravis (MG) is a chronic autoimmune disorder caused by antibodies against receptors and proteins at the neuromuscular junction. Acetylcholine receptor antibodies are found in 85% of patients with MG, 1 typically of immunoglobulin subclasses IgG1 and IgG3, and induce a complement-mediated attack. This, in turn, causes morphological and functional changes in the postsynaptic membrane, and impairs neuromuscular synaptic transmission leading to fatigable muscle weakness. 2 MG can be classified into subgroups by pathogenic antibody specificity, thymus histology, and clinical features (e.g. extent of involvement and age of onset). 3 Moreover, about 15% of patients have an associated thymoma and about 30% of patients with thymoma develop MG, making it the most frequent autoimmune disorder linked to thymoma. These cases of thymoma-associated MG have a worse prognosis than other subgroups and more often become refractory. 4 In most cases, MG can be treated successfully with an individualized combination of immunosuppressive therapy, acetylcholinesterase inhibitors, and thymectomy, plus treatment of acute exacerbations; however, issues persist with the small group of patients that have treatment-refractory MG. 5 Their substantial disease burden has motivated the search for new therapeutic targets. 6
Eculizumab is a recombinant, humanized monoclonal antibody that inhibits terminal complement activation, thereby preventing damage to the neuromuscular junction. It was only recently approved for the treatment of refractory MG. 7 We report a case of refractory thymoma-associated MG treated with eculizumab.
Case report
A 25-year-old woman was transferred to our hospital with a 2-month history of binocular diplopia, severe dysphagia, nasal voice, and weakness of her proximal arm. Examination of the orbicularis oculi with single-fiber electromyography showed increased jitter, blood testing revealed acetylcholine receptor antibody positivity (>20.0 nmol/L; reference range: 0.00–1.00) with negative results for anti-muscle-specific receptor tyrosine kinase, anti-lipoprotein receptor-related protein-4 (LRP4), anti-titin and anti-ryanodine antibodies, and chest computed tomography showed a large anterior mediastinal mass suggestive of thymoma. Treatment was therefore started with pyridostigmine (360 mg/day), oral prednisone (50 mg/day with progressive decrease to 10 mg/day), intravenous immunoglobulins (2 g/kg each time; two rounds), and plasma exchange (two rounds). However, due to the persistence of bulbar symptoms and the suspected thymoma, she was referred to our MG clinic. Examination at the clinic revealed mild ptosis of the right eye, but normal ocular movement without diplopia. Her voice was nasal and became completely unintelligible after 45 s. She also had difficulty chewing and fatigable weakness in her cervical and proximal arm and leg muscles, corresponding to Myasthenia Gravis Foundation of America class IIIb. Other autoimmune diseases were excluded based on negative results for antinuclear antibodies, anti-extractable nuclear antigen panel, rheumatoid factor, antithyroid peroxidase, and antithyroglobulin antibodies, plus the presence of normal creatine kinase levels and normal needle electromyography results. Thymectomy was performed through a left anterolateral thoracotomy, and the subsequent histology showed a completely encapsulated stage I tumor (Masaoka–Koga classification), consistent with a type B1 thymoma with small areas of B2 (<10%).
Unfortunately, the symptoms of MG deteriorated after thymectomy and the patient required immunoglobulins (0.4 g/kg over 5 days) and plasma exchange, as well as an increased dose of prednisone (25 mg/day). We therefore started treatment with tacrolimus (4 mg daily with weekly monitoring of blood levels; reference range: 5.0–20.0 µg/L). She experienced a new exacerbation without a clear trigger 3 months later with tacrolimus levels of 7.7 µg/L. She then had persistent bulbar symptoms and generalized weakness of variable severity, and over the next 18 months, she needed regular intravenous immunoglobulin and plasma exchange almost every 2 weeks.
Due to lack of response, tacrolimus was discontinued after 6 months, and rituximab 375 mg/m2 was administered weekly for 4 weeks and then monthly for 2 months. Although the patient experienced a slight improvement with rituximab, she required admissions for septicemia due to methicillin-resistant Staphylococcus aureus and for large vessel thrombosis. Cyclophosphamide was then started at 1000 mg, and continued at 500 mg every 4 weeks for 6 months). Azathioprine was started 3 months later, with the daily dose gradually increased to 100 mg/day, with no related cytopenia or hepatic toxicity.
We then performed both 18 fluorodeoxyglucose positron emission tomography to identify any thymic remnants and a second thymectomy, but neither gave positive results. Finally, treatment with eculizumab (initial phase: 900 mg via intravenous infusion every week for 4 weeks, and then 1200 mg via intravenous infusion every 14 days) was approved in August 2018.
We used the quantitative myasthenia gravis (QMG) and the myasthenia gravis activities of daily living (MG-ADL) scales to document the response to treatment with eculizumab. The patient gave written informed consent for data collection and publication.
On initiating eculizumab her QMG and MG-ADL scores were 23 and 12, respectively. During the 18 months before starting eculizumab administration, her QMG scores had ranged between 15 and 32 (median: 22; SD 6.1) and her MG-ADL scores had ranged between 6 and 20 (median: 9; SD 1.5). Eculizumab notably improved her motor symptoms by week 8 of therapy, lowering both the QMG and the MG-ADL scores to 9 (median: 12; SD 4.5) and 3 (median: 3; SD 0.5), respectively (Figure 1). Improvement was particularly marked for those items related to bulbar symptoms.
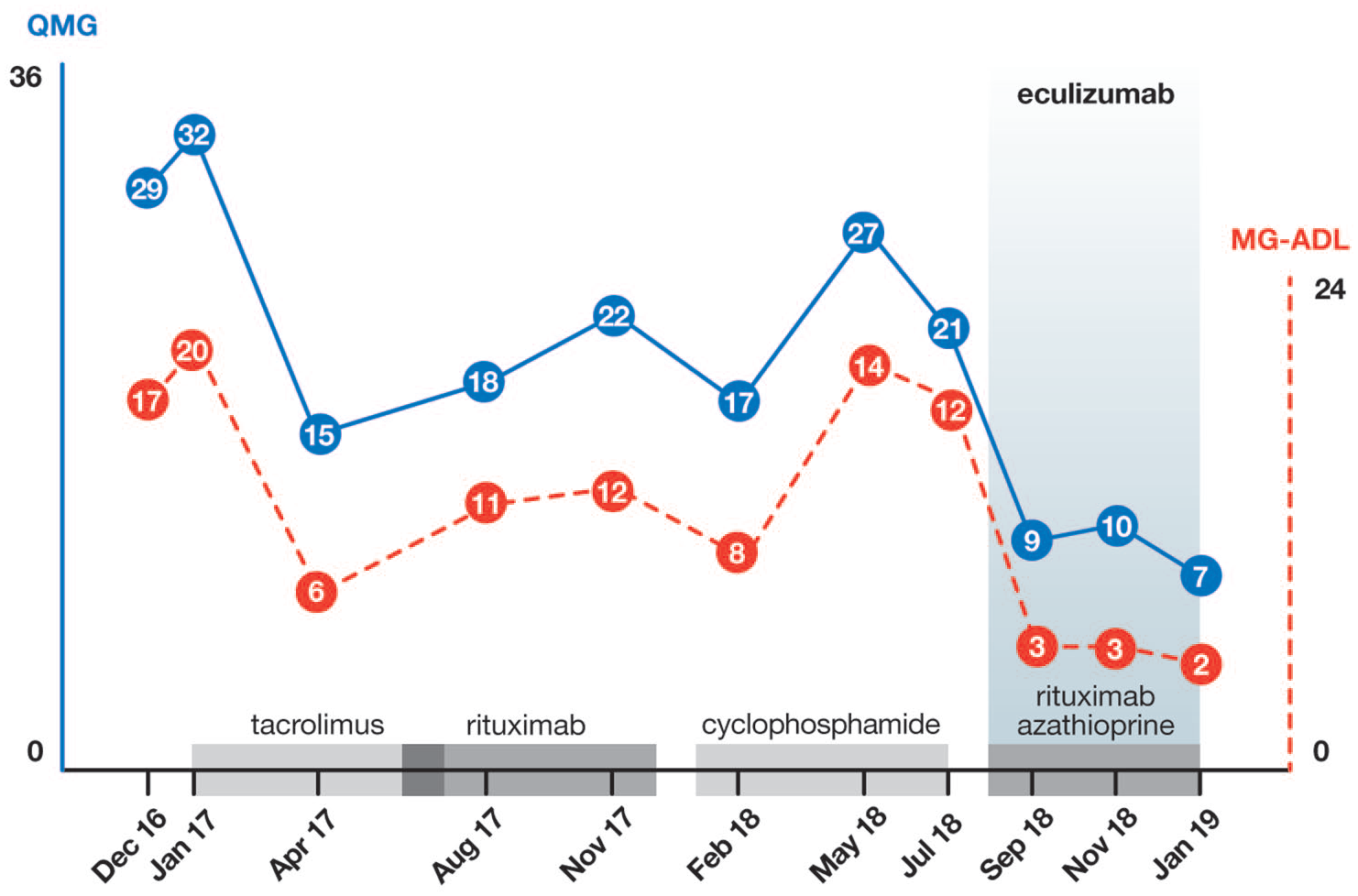
QMG (blue line) and MG-ADL (red line) scores under the different immunosuppressant treatments.
The patient continues to receive eculizumab every 2 weeks, prednisone was safely tapered, and the intravenous immunoglobulin infusions have been spaced. At week 48 after eculizumab initiation, the QMG score was 7 and the MG-ADL score was 2, with 1200 mg every 14 days eculizumab, 100 mg/day azathioprine, and 5 mg/day prednisone. Crucially, she has not presented with any serious side effects attributable to eculizumab.
Discussion
We present a case of thymoma-associated MG refractory to treatment with maximum safe and adequate doses of several immunosuppressive drugs. The patient had suffered frequent life-threatening MG crises and had needed frequent rescue therapies, resulting in severe side effects, including infection and thrombosis. Thus, her MG was considered highly refractory and suitable for eculizumab, which resulted in good clinical response.
Thymomas are strongly associated with refractory MG, conferring worse prognosis and often requiring more aggressive therapeutic strategies. This is partially due to certain well-recognized features of the thymus cytoarchitecture and environment that lead to a defective self-tolerance and an increase in mature T-cell escape into the circulation. These features include a disorganized cortex with no recognizable medulla, absent B or myoid cells, no major histocompatibility complex class II molecule expression, defective AIRE expression, and failure of FOXP3+ regulatory T-cell generation. The roles of autoantigen expression, autoimmunization, and T-cell selection remain unclear. 8
An optimized therapeutic strategy is essential for refractory MG, and it seems rational to use concomitant drugs that act on different pathways of the immune response. When eculizumab was started, besides the mandatory thymectomy, our patient was receiving combined treatment with corticoids and azathioprine (to inhibit T-cell proliferation and IL-2 production), the monoclonal antibody rituximab (to deplete B lymphocytes), and various short-term immunomodulatory therapies (to reduce circulating autoantibodies). 9 The complement pathway, specifically the activation and formation of the membrane attack complex, is an additional therapeutic target because of the role of complement in destroying the neuromuscular junction. 10
The efficacy of eculizumab in treating refractory generalized MG that is acetylcholine receptor antibody-positive was demonstrated in the phase III, randomized, double-blind, placebo-controlled REGAIN study. However, patients with a history of thymoma or thymic neoplasm were excluded from that trial and there are no data on its use in this subgroup. In our patient, administering eculizumab to act on a completely different target, while still treating with other immunosuppressants that act on ‘classic’ pathways, we achieved a clear improvement that has been maintained for a year. The patient has experienced no more crises requiring admission, her corticosteroid use has been reduced, the intervals between intravenous immunoglobulin doses have increased, and her quality of life has improved markedly. Furthermore, she has experienced no serious side effects due to eculizumab, which appears to be better tolerated than previous immunosuppressants.
In conclusion, although more data are required to determine the role of eculizumab in future therapeutic algorithms for MG, it is clear that new therapies such as this are urgently needed to remedy the considerable disease burden in these patients. This report supports that eculizumab is a promising treatment in thymoma-associated MG.
Footnotes
Authors’ Note
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CERCA Program/ Generalitat de Catalunya.
