Abstract
Background:
Standard management of generalized myasthenia gravis associated with anti-acetylcholine receptor autoantibodies (AChR-gMG) includes corticosteroids and nonsteroidal immunosuppressants. Complement inhibitors (CI) represent a more tailored therapeutic strategy. Real-world data on the steroid-sparing efficacy of CI remain limited.
Objective:
To investigate the steroid-sparing efficacy of CI in AChR-gMG.
Design:
We identified 69 AChR-gMG patients on corticosteroids treated with azathioprine (AZA), mycophenolate mofetil (MMF), or CI.
Methods:
Steroid tapering was assessed by comparing corticosteroid dosage at several time-points to baseline.
Results:
Steroids reductions were statistically significant for all therapies at every time point compared to baseline (all p < 0.001). Pairwise comparisons using the Mann–Whitney U test revealed significant differences between CI and MMF at 3 months (p = 0.0372), 6 months (p = 0.0193), and 9 months (p = 0.0321) and between CI and AZA at 6 months (p = 0.0415).
Conclusion:
CI rapidly and effectively reduced corticosteroid dosage in most AChR-gMG patients, suggesting their potential role as steroid-sparing therapeutic options.
Plain language summary
This study evaluated the effectiveness of complement inhibitor therapy in reducing steroid use in patients with generalized myasthenia gravis (gMG), an autoimmune disease associated with autoantibodies targeting the neuromuscular junction, leading to muscle weakness and fatigability. Standard treatment for gMG involves corticosteroids and non-steroidal immunosuppressants such as azathioprine and mycophenolate mofetil. However, long-term steroid use can lead to significant systemic adverse effects. Recently approved targeted therapies, including complement inhibitors such as eculizumab and ravulizumab, offer a promising approach by inhibiting the complement C5 protein. However, there is limited real-world data on their ability to reduce steroid use. In this study, we enrolled 69 gMG patients who were receiving steroids at baseline, in addition to either azathioprine, mycophenolate mofetil, or complement inhibitors. We assessed changes in steroid dosage at different time points. Our results showed a significant reduction in steroid use across all treatment groups. Notably, patients treated with complement inhibitors experienced a more significant reduction in steroid dosage at earlier time points, compared to those receiving azathioprine or mycophenolate mofetil. These findings suggest that complement inhibitors may be effective in reducing steroid use in gMG patients, making them a promising treatment option for minimizing steroid doses and their associated side effects.
Keywords
Introduction
Generalized myasthenia gravis (gMG) is a rare autoimmune disorder of the neuromuscular junction, caused by autoantibodies (Abs) targeting postsynaptic membrane proteins. 1 Symptoms include ptosis, diplopia, limb weakness, dysarthria, dysphagia, and dyspnea. 2 Standard therapy consists of acetylcholinesterase inhibitors, corticosteroids, and nonsteroidal immunosuppressants (ISTs), 3 yet approximately 10% of patients remain treatment-refractory. 4 Moreover, prolonged corticosteroids use causes significant systemic adverse effects, affecting patients’ quality of life.5,6 Consequently, a key therapeutic goal in gMG is sustained symptom control with minimized therapy side effects. 7 Most gMG cases involve Abs against the acetylcholine receptor (AChR), 8 which is pathogenic through three mechanisms: blocking of acetylcholine binding site; cross-linking of adjacent receptors, promoting internalization and degradation of AChRs; destruction of the postsynaptic membrane through complement activation and formation of membrane-attack complexes (MACs). 9 Complement inhibitors (CI) thus represent a targeted option for AChR-antibody-positive gMG (AChR-gMG). 10
Eculizumab and ravulizumab, approved for AChR-gMG, inhibit the complement C5 protein, preventing the formation of MACs.11,12 Although clinical trials have demonstrated their efficacy, real-world data on their steroid-sparing effects remain limited. This study aimed to assess the potential of CI as steroid-sparing agents.
Methods
Study population
We conducted a retrospective cohort study emulating a target trial to compare the steroid-sparing efficacy of CI, mycophenolate mofetil (MMF), and azathioprine (AZA) in gMG. We included patients ⩾18 years old with AChR-gMG, admitted to the outpatient clinic of Fondazione Policlinico Agostino Gemelli IRCCS between June 2020 and November 2024. At baseline, all received corticosteroids combined with AZA, MMF, or CI. Inclusion criteria included a confirmed diagnosis of AChR-gMG and a follow-up of at least 6 months.
Treatment followed standard guidelines. 13 Pyridostigmine was first-line medication. Thymectomy was performed in patients with thymoma or early-onset gMG. Prednisone (0.5–1 mg/kg daily) was started in patients unresponsive to pyridostigmine and progressively tapered to the minimum effective dose or withdrew, according to clinical response. Steroid-sparing agents, AZA as first-choice, were added in still symptomatic cases, requiring high maintenance steroid doses or at risk for adverse events. MMF was employed when AZA was ineffective or not tolerated. According to AIFA prescribing criteria, eculizumab was administrated to patients with Myasthenia Gravis-Activity Daily Living (MG-ADL) score ⩾6 and Myasthenia Gravis Foundation of America (MGFA) class ⩾III, with inadequate response to corticosteroids and ⩾2 ISTs. Ravulizumab was prescribed to patients unresponsive to one IST other than corticosteroids, with MG-ADL score ⩾6 and MGFA ⩾IIb.
To minimize confounding from nonrandom treatment assignment, we employed propensity score matching. Propensity scores were estimated using logistic regression models incorporating baseline covariates: age, sex, thymoma status, disease duration, disease severity (MGFA maximum class), baseline steroid dose, and comorbidities (hypertension, osteoporosis). Patients receiving MMF were matched 1:1 with those receiving CI using nearest-neighbor matching within a caliper of 0.2 standard deviations of the logit of the propensity score.
Recognizing that AZA is often prescribed as first-line IST and that baseline characteristics differed from those receiving MMF or CI, we randomly selected 30 patients on AZA for inclusion. Selection was performed by an investigator (S.F.) blinded to patient identities and steroid dosage data to reduce selection bias.
Steroid-tapering analysis
Steroid doses were recorded at baseline and at 3, 6, 9, and 12 months. The primary outcome was the percentage reduction in steroid dose from baseline at each time point, which was used as a measure of the steroid-sparing efficacy of each therapy.
Statistical analyses
The Shapiro–Wilk test was employed to verify the Gaussian distribution of the data. Categorical variables were presented as absolute frequencies and percentages, whereas continuous variables were summarized with means, medians, and interquartile ranges (IQR). Wilcoxon signed-rank test was used for pairwise comparisons between baseline and different time points (3, 6, 9, and 12 months) for each treatment group. Mann–Whitney U test was employed to perform pairwise comparisons between therapy groups at different time points.
To address missing data at 9- and 12-months follow-up, we applied two methods:
(1) Complete case analysis: Our primary analysis included only patients with data at all five time points (n = 62). The direction and significance of time-point comparisons remained unchanged.
(2) Multiple imputation: We used MICE in R to impute missing steroid doses at 9 and 12 months with different values based on baseline dose, therapy, MGFA maximum class, comorbidities, age, sex, disease duration. Post-imputation, Wilcoxon signed-rank, and Mann–Whitney U tests did not show significant differences.
To assess the longitudinal effect of different therapies on steroid reduction, multivariate linear regression models were conducted at 3, 6, 9, and 12 months, adjusting for relevant covariates including baseline dose, disease duration, age at onset, thymoma status, and treatment category (AZA, MMF, CI). Models were tested for linearity, homoscedasticity, and absence of collinearity using variance inflation factors, and residual diagnostics were assessed.
Statistical significance was set at p < 0.05.
The reporting of this study conforms to STROBE guidelines for cohort studies. 14
Results
A total of 69 AChR-gMG patients were enrolled: 30 treated with AZA, 20 with MMF, and 19 with CI (12 with eculizumab and 7 with ravulizumab). All 69 included patients had ⩾6 months follow-up. Data at 9 and 12 months were fully available for 68 and 62 patients, respectively. Missing 9-month data (n = 1) resulted from CI discontinuation due to inefficacy, whereas missing 12-month data (n = 7), were due to follow-up schedule deviations or transfers of care. Baseline demographics and clinical characteristics, including median age at therapy initiation and MGFA maximum class, showed no significant differences across groups (Figure 1(a)). In contrast, significant differences were observed between patients receiving CI and AZA-treated patients in terms of age at onset, disease duration, and thymoma status (p < 0.001). No significant differences were found between patients undergoing CI and those receiving MMF (Table 1).

(a) MGFA classification distribution by therapy group. (b) Comparison of steroid dose categories across therapy groups at baseline and at 6 months. (c) Density plots showing the distribution of steroid doses for patients receiving AZA, MMF, or CI at baseline, 3, 6, 9, and 12 months. CI therapy shows a marked shift toward lower steroid doses over time compared to MMF and AZA. Density reflects the probability distribution of doses in each group. (d) The colored lines represent the mean steroid reduction at each follow-up time point for each therapy group. The shaded bands around each line indicates the interquartile range (25th–75th percentile), illustrating the central spread of values. Different colors distinguish the therapy groups (CI in blue, AZA in green, MMF in red).
Demographic and clinical characteristics of MG population receiving CI, AZA, and MMF.
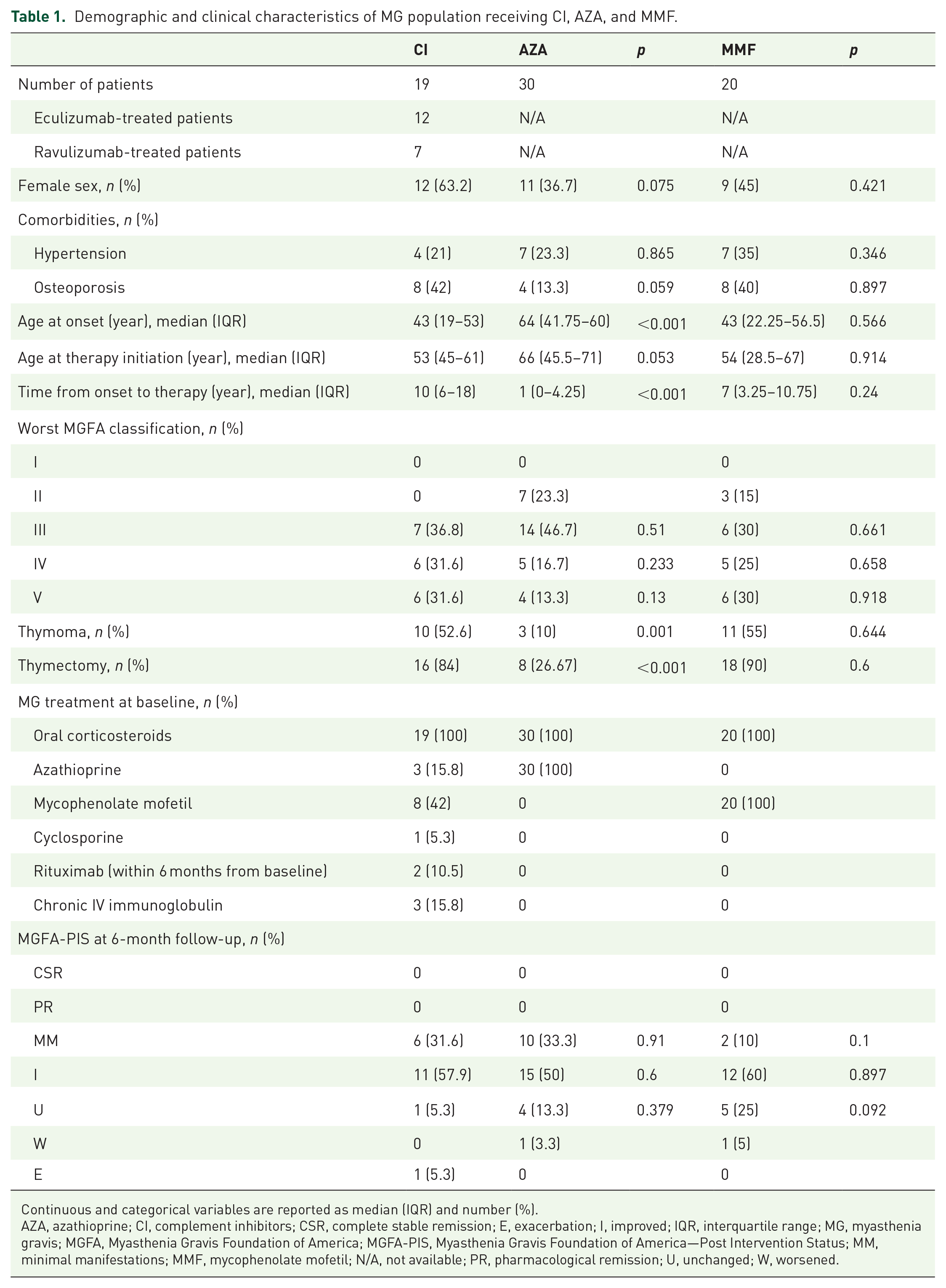
Continuous and categorical variables are reported as median (IQR) and number (%).
AZA, azathioprine; CI, complement inhibitors; CSR, complete stable remission; E, exacerbation; I, improved; IQR, interquartile range; MG, myasthenia gravis; MGFA, Myasthenia Gravis Foundation of America; MGFA-PIS, Myasthenia Gravis Foundation of America—Post Intervention Status; MM, minimal manifestations; MMF, mycophenolate mofetil; N/A, not available; PR, pharmacological remission; U, unchanged; W, worsened.
CI subgroup: Patient population and steroid-sparing effect
Median age at baseline was 53 (IQR: 45–61) years and 12/19 (63.2%) patients were female. All subjects underwent a 6-month follow-up, while 13/19 (68.4%) had a follow-up of 12 months.
Mean reduction (%) of steroid daily dose from baseline to 3-month follow-up was 5.6 (23.1%, p < 0.001) mg, increasing to 10 (41.2%, p < 0.001) mg at 6-month follow-up.
Steroid daily dose (median (IQR)) was reduced from 20 (15–27.5) mg at baseline, to 15 (10–16.25) mg at 6-month follow-up. Overall, 16/19 (84.2%) patients tapered their steroid dose, with 13/19 patients also receiving nonsteroidal ISTs (AZA, n = 3; MMF, n = 8; cyclosporine, n = 1; rituximab within 6 months, n = 2). No significant differences were observed in the percentage reduction of corticosteroids from baseline to 6-month follow-up between the two groups: 43.1% for patients under nonsteroidal ISTs compared to 37.3% for patients receiving only steroids (p = 0.86). Two patients (both on eculizumab) maintained stable steroid dosages of 5 and 16.25 mg/day, respectively, but they were able to discontinue chronic intravenous immunoglobulin. Due to poor response to eculizumab, one patient required an augmentation of her daily steroid prescription of 5 mg; after 6 months, she experienced an exacerbation that required intravenous immunoglobulins and withdrew CI.
At 12-month follow-up, 11/13 (84.6%) patients tapered their steroid dosages, with median daily dose decreasing to 12.5 (IQR: 6.88–14.38) mg and a mean reduction of 15.38 (57.54%, p < 0.001) mg; one patient withdrew steroids. Moreover, within 12 months from baseline, one eculizumab-treated patient discontinued AZA entirely.
Comparison of steroid-sparing effect among AZA, MMF, and CI
A total of 69 patients were included in the analysis. AZA was administered at a daily dose of 2 mg/kg, while the median daily dose of MMF was 2 g (IQR: 1.5–2). This dosing regimen remained stable throughout the entire follow-up. Steroid dose distributions across therapy groups at baseline and at 6-month follow-up are represented in Figure 1(b).
Steroid dose reductions were statistically significant for all therapies at every time point compared to baseline (AZA, MMF, and CI at 3, 6, 9, and 12 months: p < 0.001; Figure 1(c) and (d)).
CI demonstrated the most pronounced steroid-sparing effect across all time points, with significant reductions in steroid doses compared to MMF and AZA at earlier time points. Pairwise comparisons using the Mann–Whitney U test revealed significant differences between CI and MMF at 3 (p = 0.0372), 6 (p = 0.0193), and 9 months (p = 0.0321) and between CI and AZA at 6 months (p = 0.0415). Differences observed between CI and the other therapies at other time points were not statistically significant (Figure 1(d)).
Despite steroid tapering, most patients in each subgroup achieved clinical stability or improvement. At 6 months, MGFA-Post Intervention Status indicated Minimal Manifestation status in 31.6% of patients undergoing CI, 33.3% of those on AZA, and 10% on MMF. Improved status was observed in 57.9% of CI-treated patients, 50% of those on AZA, and 60% on MMF.
Multivariate regression models for steroid reduction at 3, 6, 9, and 12 months
CI consistently predicted strong steroid-sparing effects at 3 (p = 0.002), 9 (p < 0.001), and 12 months (p = 0.004) compared to AZA, and at 3 (p = 0.019) and 9 months (p < 0.021) compared to MMF, even after adjusting for baseline differences (thymoma, age, disease duration). In this model, baseline steroid dose was the strongest predictor of the percentage of steroid reduction while no significant differences were observed between AZA and MMF (all p > 0.1). Although thymoma status differed across groups, it was not a significant predictor of steroid reduction (all p > 0.4), while longer disease duration was associated with a reduced steroid-sparing effect, reaching statistical significance at 6 (p = 0.021) and 9 months (p = 0.044), but not at 3 months (p = 0.057; Supplemental Table).
Discussion
We analyzed corticosteroid dose reductions in AChR-gMG patients treated with CI at our outpatient clinic, comparing their steroid-sparing effects to those of AZA and MMF.
Notably, most patients receiving CI (16/19) successfully tapered their steroid dosages. Several patients demonstrated a faster initial taper (e.g., from a mean baseline dose of 30–50 mg to <15 mg by 6 months), followed by a stabilization or more gradual reduction from 6 to 12 months to avoid flare-ups.
Due to their recent approval, real-world data on the steroid-sparing effect of CI in AChR-gMG remain limited. Our findings provide further evidence of the steroid-sparing efficacy of eculizumab15–18 and ravulizumab, 19 suggesting that CI enable a safe and early reduction of steroid dosages in most patients.
Interestingly, no significant differences were observed between patients receiving nonsteroidal ISTs at baseline and those who were not. While the contribution of ISTs in maintaining clinical stabilization cannot be ruled out, our data suggest that steroid reduction was primarily obtained with the introduction of CI as an “add-on” therapy.
Moreover, CI demonstrated the higher steroid-sparing effects at 3, 6, and 9 months compared to MMF, and at 6 months compared to AZA, suggesting that these treatments may offer a rapid and valid strategy for reducing corticosteroids in gMG. Steroid tapering was comparable among the three therapies at 12 months, which are consistent with previous studies showing that both AZA and MMF are effective steroid-sparing agents, with a delayed onset of action. 20 Nonetheless, it is crucial to highlight the importance of slow steroid tapering to minimize exacerbation risk.
Multivariate regression models identified CI as strong steroid-sparing predictors at 3, 9, and 12 months compared to AZA, and at 3 and 9 months compared to MMF. Notably, longer disease duration showed a negative impact on steroid reduction at 6 and 9 months, suggesting greater difficulty in managing longstanding gMG.
This study has limitations. It is a single-center study with a relatively small sample size. The follow-up period was limited for some patients, due to the recent availability of CI, and 12-month follow-up data were unavailable for the entire study population. Steroid tapering was not standardized but left to the discretion of treating clinicians, sometimes influenced by comorbidities and patient preferences. However, the single-center design provides greater consistency compared to multicenter studies, where variability in steroid management by different clinicians may influence outcomes. Additionally, patients in the control groups received exclusively AZA or MMF, while in 13 patients undergoing CI, this treatment was an “add-on” to baseline immunosuppressive therapy. Further investigation, including larger patient cohorts and extended follow-up periods, is required to fully evaluate the steroid-sparing potential of CI in AChR-gMG.
Conclusion
CI therapy may represent a rapid and effective steroid-sparing option for gMG patients, reducing the burden of systemic adverse effects associated with prolonged corticosteroid use and improving overall disease management.
Supplemental Material
sj-docx-2-tan-10.1177_17562864251332037 – Supplemental material for Complement inhibitor therapy as a steroid-sparing strategy in generalized myasthenia gravis
Supplemental material, sj-docx-2-tan-10.1177_17562864251332037 for Complement inhibitor therapy as a steroid-sparing strategy in generalized myasthenia gravis by Sofia Marini, Silvia Falso, Federico Habetswallner, Martina Marini and Raffaele Iorio in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-pdf-1-tan-10.1177_17562864251332037 – Supplemental material for Complement inhibitor therapy as a steroid-sparing strategy in generalized myasthenia gravis
Supplemental material, sj-pdf-1-tan-10.1177_17562864251332037 for Complement inhibitor therapy as a steroid-sparing strategy in generalized myasthenia gravis by Sofia Marini, Silvia Falso, Federico Habetswallner, Martina Marini and Raffaele Iorio in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
