Abstract
Background:
Physical activity, particularly regular aerobic exercise, is effective in preventing dementia. However, such activities are less feasible for patients with Parkinson’s disease (PD) or other motor dysfunctions.
Objectives:
In this study, we investigated whether the minimal amount of exercise (MAE) through walking, which is practical for individuals with motor dysfunction, can reduce the risk of dementia in patients with PD.
Design:
For this retrospective longitudinal study, we enrolled 470 patients with PD without dementia from 3 centers in Taiwan.
Methods:
In total, 187 (39.8%) subsequently developed dementia, whereas 283 (60.2%) did not; the mean follow-up periods for these cohorts were 3.1 (range 0.3–6.1) and 2.4 (range 0.3–6.0) years, respectively. MAE was defined as walking approximately 1500–3000 steps or for 15–30 min. The patients were further stratified by the weekly frequency of MAE into MAE-no (frequency: 0), MAE-weekly (frequency: 1 or 2), and MAE-daily (frequency: ⩾3) groups, respectively. The incidence rates of dementia were compared among the three groups. Cox proportional-hazards analyses were performed to measure the effect of MAE on the incidence of dementia. The statistical model was adjusted for age, sex, education level, cognition level, activities of daily living, neuropsychiatric symptoms, vascular risk factors, and relevant medications.
Results:
The MAE-weekly and MAE-daily groups were 0.69 (95% confidence interval (CI): 0.41–1.17) and 0.59 (95% CI: 0.41–0.84) times, respectively, less likely to develop dementia than the MAE-no group. When the MAE-weekly and MAE-daily groups were combined, the hazard ratio for dementia was 0.62 (95% CI: 0.45–0.85). Cox regression revealed that older age, female sex, atrial fibrillation, antidiabetic drug use, and poor daily function were associated with an increased incidence of dementia.
Conclusion:
MAE may help prevent dementia in patients with PD. This finding highlights the benefits of walking for patients with PD and, potentially, older adults with motor dysfunction due to various disorders.
Plain language summary
Staying active is important for brain health, but for people with Parkinson’s disease (PD), regular intense exercise can be challenging due to movement difficulties. In this study, we explored whether even small amounts of walking could help reduce the risk of dementia in people with PD. We followed 470 people with PD in Taiwan over several years. They were divided into three groups based on how often they walked a minimal amount (defined as walking 1500–3000 steps or for 15–30 minutes): no walking (MAE-no), walking once or twice a week (MAE-weekly), and walking three or more times a week (MAE-daily). Our findings showed that those who walked once or twice a week were 31% less likely to develop dementia, and those who walked three or more times a week were 41% less likely, compared to those who did not walk. Factors such as older age, being female, having certain health conditions, and difficulty with daily activities were linked to a higher risk of dementia. This study highlights that even small amounts of walking can have significant brain health benefits for people with Parkinson’s disease. Walking may also help older adults with other movement challenges.
Introduction
Parkinson’s disease (PD), an age-related condition, is the second prevalent neurodegenerative disorder following Alzheimer’s disease. Epidemiological data suggest that the prevalence of PD is approximately 0.1%–0.2% in unselected populations and up to 1% in populations aged >60 years1–3; PD has affected more than 6 million patients worldwide. 4 Cognitive decline, a nonmotor progressive symptom of PD, markedly compromises patients’ safety and quality of life. 5 In the early stages of PD, mild cognitive impairment (MCI) occurs in 18.9%–38.2% of all patients. 6 Among patients with MCI at PD diagnosis, >25% develop PD dementia (PDD) within 3 years. 7 The global prevalence of PDD is 26.3%. 8 These data highlight that strategies for preventing PDD or decelerating its progression are urgently required. 9
Many nonpharmacological interventions have been explored for managing both motor and nonmotor symptoms in patients with PD. Notably, exercise has proven beneficial for these patients. 10 First, exercise may reduce the risk of PD. According to the National Institutes of Health–American Association for Retired Persons Diet and Health Study, moderate-to-severe exercise can reduce the risk of PD by 40%. 11 Second, exercise ameliorates motor symptoms such as bradykinesia, balance, gait, turning, and quality of life.12–14 It can also ameliorate nonmotor symptoms such as sleep problems, mood disorders, and pain.15–17 Third, exercise may influence the outcomes of PD treatment by reducing drug side effects and enhancing medication efficacy.18–20
Some small-scale randomized controlled trials have investigated the association of exercise with cognitive function in patients with PD. Treadmill training improved both cognitive and motor functions in these patients. 21 Tango improved balance, functional mobility, and cognitive outcomes.22,23 Training with Wii-based games improved balance and cognition. 24 Furthermore, programs involving anabolic and aerobic exercises improved executive function in patients with PD.25–27
Walking is an easy, simple, and low-cost exercise that does not require any special equipment or training. Regular walking offers numerous health benefits by reducing the risks of cardiovascular diseases, type 2 diabetes, obesity, depression, cancer, and all-cause mortality.28–34 Furthermore, a higher number of daily steps is associated with a lower risk of all-cause dementia. 35 However, few studies have investigated the dose–response association of weekly steps with dementia development in patients with PD. Thus, we conducted the present study to determine whether the minimal amount of exercise (MAE) through walking, which is easily achievable, can reduce the risk of dementia in patients with PD.
Methods
Study design
This was a retrospective, longitudinal, follow-up cohort study. Registration data were collected from the History-Based Artificial Intelligence Clinical Diagnosis of Dementia Syndrome (HAICDDS)36,37 (see the STROBE checklist in the Supplemental File). From these data, we identified consecutive patients from three centers in Taiwan.
Dementia diagnosis
The diagnostic criteria for PDD are according to the Movement Disorder Society (MDS) criteria in 2007.38,39 The diagnosis of PDD requires the following:
Core Features: Presence of PD.
Gradual onset and progressive cognitive decline, affecting daily life and independence.
Impairment in at least two cognitive domains, such as attention, executive function, memory, visuospatial abilities, or language. The Cognitive domains of the Clinical Dementia Rating (CDR) scale and the Cognitive Abilities Screening Instrument (CASI) were operationally used to indicate dementia.40–43 Activities of daily living (ADL) were evaluated using the new tool History-based Artificial Intelligent ADL (HAIADL). 44
The severity of cognitive impairment—MCI or dementia—was determined using the CDR scale. Patients with dementia typically have a global CDR score of 0.5 plus a CDR-sum of boxes score of ⩾4.5 or a global CDR score of ⩾1, with impairments in at least two CDR domains.40–42
Cognitive performance was evaluated using CASI. This instrument comprises the following nine cognitive domains: remote memory, recent memory, attention, mental manipulation, orientation, abstract thinking, language, visuospatial function, and verbal fluency. Adaptations of CASI in different languages have been validated and used globally. The cutoff score on CASI should be in the range for dementia after adjustments for patients’ age, education level, and sex. 43
The stage of cognitive impairment or dementia was determined using HAIADL, a 21-item questionnaire used in patients with both instrumental and basic impairments in ADL due to cognitive disorders. 44
The three study groups were compared also in terms of scores on the Clinical Frailty Scale (CFS), Montreal Cognitive Assessment (MoCA) tool, Unified Parkinson’s Disease Rating Scale, and Neuropsychiatric Inventory (NPI); each of these tools have undergone to validation and reliability tests.45–48
Minimal amount of exercise
MAE was defined as walking 1500–3000 steps for approximately 15–30 min. On the basis of the weekly frequency of MAE, the patients were divided into the following three groups: MAE-no (frequency: 0), MAE-weekly (frequency: 1 or 2), and MAE-daily (frequency: ⩾3).
Study cohort
The study groups were compared in terms of dementia incidence. Cox proportional-hazards analysis was performed to measure the effect of MAE on the conversion of idiopathic PD to dementia. These idiopathic PD patients were diagnosed according to the 2015 edition of the MDS’s diagnostic guidelines. 49 The statistical model was adjusted for age, sex, education level, cognition, ADL, neuropsychiatric symptoms, vascular risk factors, and relevant medications. All participants were recruited between 2015 and 2022. From the HAICDDS registration data, consecutive patients were evaluated against preset inclusion and exclusion criteria. A total of 12,348 patients who had visited our dementia clinic were registered in the HAICDDS database. Among them, 7300 patients without follow-up data were regarded as ineligible for this study, and the remaining 5048 patients with at least one follow-up assessment were deemed eligible. Patients with osteoarthritis or dementia before PD diagnosis were excluded from this study. The final study cohort comprised 470 patients with PD but without dementia. The inclusion and exclusion criteria are summarized in Figure 1, which outlines the process of patient selection.

Flowchart depicting patient selection.
Data analysis
Statistical analyses were performed using the Chinese version of SPSS (version 22.0; IBM Corporation, Armonk, NY, USA). The MAE groups were compared in terms of the following clinicodemographic characteristics: cognition measured using the CASI, HAIADL, CFS, MoCA, and NPI as well as clinical history of hypertension, diabetes, dyslipidemia, coronary artery disease (CAD), atrial fibrillation (AF), congestive heart failure (CHF), and smoking habit. Furthermore, the current use of various medications, such as antihypertensives, antidiabetic drugs, lipid-lowering drugs, antiplatelets, and antiparkinsonian drugs, was analyzed.
Cox proportional-hazards regression analysis was performed to identify the association of MAE frequency with dementia incidence among patients with PD. The study groups were compared in terms of hazard ratios (HRs) for dementia. The statistical model was adjusted for age, sex, education level, CFS score, CASI score, NPI score, HAIADL score, hypertension, diabetes, dyslipidemia, CAD, AF, CHF, and medications. Statistical significance was set at p < 0.05; all tests were two-tailed.
Results
This study included 470 patients. Among them, 187 (36.0%) developed dementia, whereas 283 (64.0%) did not; their mean follow-up durations were 3.1 (range 0.3–6.1) and 2.4 (range 0.3–6.0) years, respectively. Overall, 43.2% of all patients engaged in at least weekly MAE. The MAE-no, MAE-weekly, and MAE-daily groups comprised 267, 47, and 156 patients, and their conversion rates were 46.1%, 36.2%, and 30.1%, respectively. Figure 2 presents the results of the percentage frequency of conversion according to the frequency of MAE. Multiple logistic regression analysis was conducted for investigating the impact of MAE in non-dementia participants on conversion to dementia. HRs were adjusted for age, sex, education, follow-up years, cognition (CASI), ADL (HAIADL), neuropsychiatric symptoms (NPI), diabetes, hypertension, dyslipidemia, CADs, arrhythmia, CHF, and medications. Compared to the MAE-no group, HR was 0.55 and 0.53 in the MAE-weekly group and the MAE-daily group, respectively.

Percentage frequency of conversion according to the frequency of MAE. Multiple logistic regression analysis was conducted for investigating the impact of MAE in non-dementia participants on conversion to dementia. HRs were adjusted for age, sex, education, follow-up years, cognition (CASI), ADL (HAIADL), neuropsychiatric symptoms (NPI), diabetes, hypertension, dyslipidemia, CADs, arrhythmia, congestive heart failure, and medications. Compared to the MAE-No group, HRs were 0.55, and 0.53 in the MAE-weekly group, and the MAE-daily group, respectively.
A comparison of demographic characteristics of the three groups, without covariate adjustment, revealed significant differences in age (p = 0.004), CFS score (p = 0.001), CDR-sum of boxes score (p = 0.008), CASI score (p < 0.001), MoCA score (p < 0.001), HAIADL score (p < 0.001), and follow-up duration (years; p = 0.016). However, the groups did not differ significantly in terms of clinical history or current medications (Table 1).
Demographic characteristics of the different MAE groups.

AF, atrial fibrillation; CAD, coronary artery disease; CASI, Cognitive Abilities Screening Instrument; CDR-SB, Clinical Dementia Rating scale-sum of boxes; CFS, Clinical Frailty Scale; CHF, congestive heart failure; HAIADL, History-Based Artificial Intelligence Activities of Daily Living scale; LED, levodopa equivalent dose; LLD, lipid-lowering drug; MAE, minimal amount of exercise; MoCA, Montreal Cognitive Assessment; n, number of cases; NPI, Neuropsychiatric Inventory; ns, nonsignificant; SD, standard deviation.
Figure 3 presents the results of the Cox regression analysis performed to measure the effect of the MAE frequency on the conversion of PD to dementia. HRs were compared between the study groups in analyses adjusted for age, sex, education level, CFS score, CASI score, NPI score, HAIADL score, hypertension, diabetes, dyslipidemia, CAD, AF, CHF, and medications. After covariate adjustment, the HR for dementia incidence was 0.69 (95% confidence interval (CI): 0.41–1.17; p = 0.167) in the MAE-weekly group and 0.59 (95% CI: 0.41–0.84; p = 0.004) in the MAE-daily group.

Curves depicting the adjusted cumulative incidence of PDD in the study cohort. On the basis of the weekly frequency of MAE, the patients were divided into the following three groups: MAE-no (frequency: 0), MAE-weekly (frequency: 1 or 2), and MAE-daily (frequency: ⩾3). Cox proportional-hazards regression was performed to investigate the association of MAE frequency with dementia incidence among patients with PD without dementia. HRs were compared between the study groups; the statistical model was adjusted for age, sex, education level, CFS score, CASI score, NPI score, HAIADL score, hypertension, diabetes, dyslipidemia, CAD, AF, CHF, and medications.
Further analysis involved combining the MAE-weekly and MAE-daily groups (n = 167) and comparing the combined group with the MAE-no group in terms of HR; the statistical model was adjusted for the aforementioned variables. After covariate adjustment, the HR for dementia incidence among patients engaging in at least weekly MAE was 0.62 (95% CI: 0.45–0.85; p = 0.003).
Finally, Cox proportional-hazards regression was performed to investigate the effects of MAE and other factors on the conversion of PD to dementia. The study groups were compared in terms of HR; the statistical model was adjusted for age, sex, education level, CASI score, HAIADL score, NPI score, diabetes, hypertension, dyslipidemia, CAD, AF, CHF, and medications (Table 2). In addition to MAE, factors such as younger age, male sex, improved daily function (HAIADL), a history of diabetes, and a history of CAD were associated with a reduced incidence of dementia in patients with PD. By contrast, AF and current antidiabetic medication use were associated with an increased incidence of dementia in this patient population.
Results of Cox regression analysis for cognitive decline.
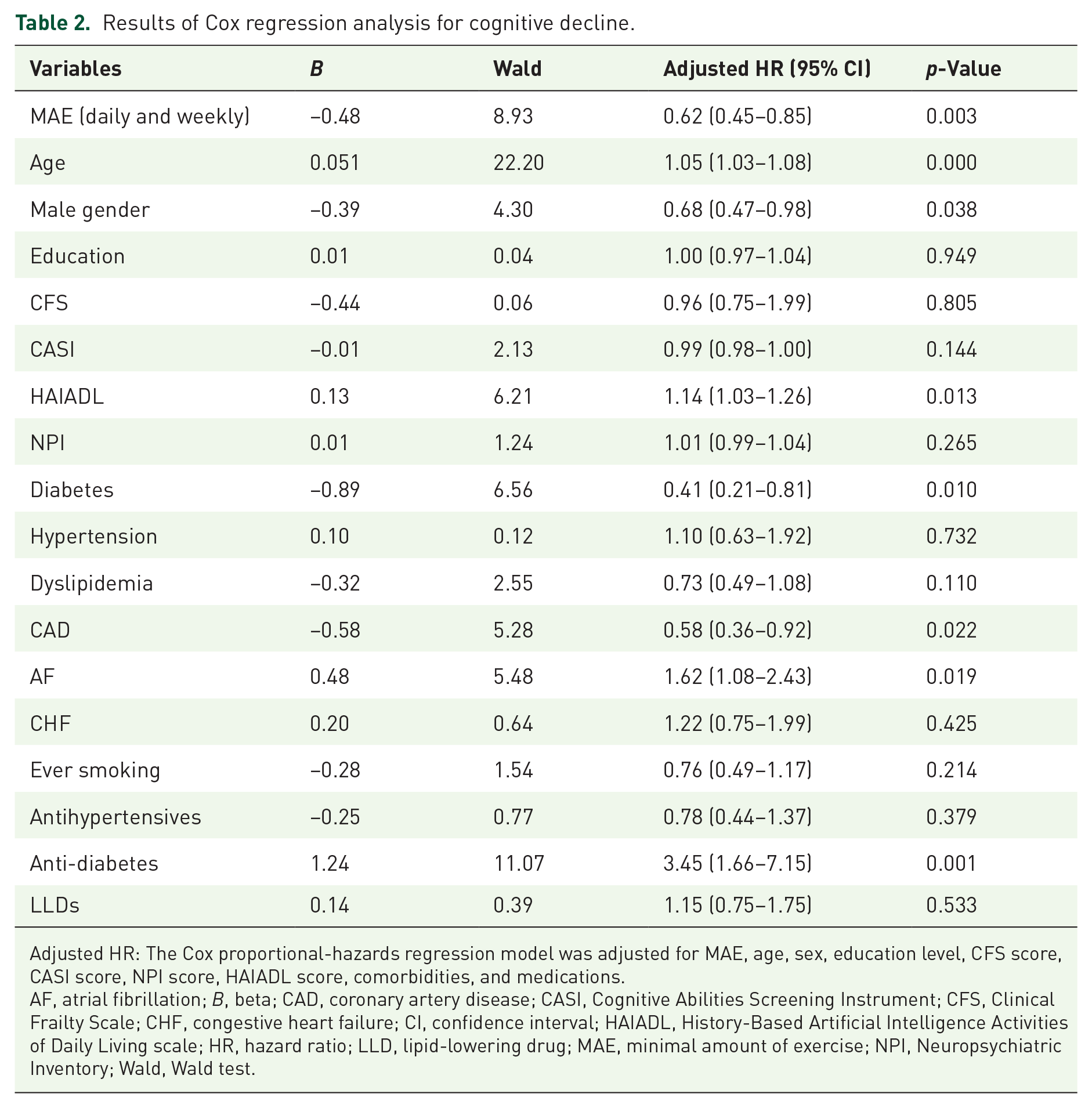
Adjusted HR: The Cox proportional-hazards regression model was adjusted for MAE, age, sex, education level, CFS score, CASI score, NPI score, HAIADL score, comorbidities, and medications.
AF, atrial fibrillation; B, beta; CAD, coronary artery disease; CASI, Cognitive Abilities Screening Instrument; CFS, Clinical Frailty Scale; CHF, congestive heart failure; CI, confidence interval; HAIADL, History-Based Artificial Intelligence Activities of Daily Living scale; HR, hazard ratio; LLD, lipid-lowering drug; MAE, minimal amount of exercise; NPI, Neuropsychiatric Inventory; Wald, Wald test.
Discussion
Our study indicated that MAE through walking independently reduced the risk of PDD, reinforcing the protective role of physical activity against cognitive decline. Patients who walked weekly or daily were 0.69- and 0.59-fold less likely, respectively, to develop PDD. This finding aligns with large-scale studies demonstrating that walking significantly lowers the risk of all-cause dementia, with benefits increasing in a dose-dependent manner. 35
Several mechanisms may explain this protective effect of walking. First, walking enhances cardiovascular health by improving brain vascularity. The higher blood flow delivers to the brain more oxygen and nutrients, which are essential for brain health and function. 50 Second, physical activities such as walking promote neurogenesis and synaptic plasticity, which are crucial for learning, memory, and overall cognitive function. 51 Third, exercise ameliorates mitochondrial dysfunction, thereby mitigating energy deficiency, calcium imbalance, oxidative stress, hyperphosphorylation, amyloid-β accumulation, and cognitive decline.52,53 Fourth, Alzheimer’s disease is associated with inflammation, which leads to neuronal loss and brain dysfunction. Exercise promotes the release of anti-inflammatory cytokines, such as interleukin-10, from skeletal muscles. 54 Fifth, leisure-time walking can reduce chronic stress and improve mood by releasing endorphins and reducing cortisol levels; however, walking to work does not have the same effect. Long-term stress and depression are associated with an increased risk of dementia.31,55,56 Sixth, regular walking improves sleep quality, which is essential for cognitive health. Poor sleep is associated with an increased risk of dementia. 57 Seventh, walking helps manage body weight, reduce blood pressure, and improve glucose metabolism, all of which are crucial for preventing cognitive decline.28–30 Metabolic syndrome and diabetes are prominent risk factors for dementia. 58 Finally, walking, particularly in group settings, promotes social interaction, which protects against dementia. Social engagement stimulates cognitive processes and emotional well-being. 59
Age is a key risk factor for neurodegenerative diseases, including PDD.60,61 In our study, the HR for age was 1.05 (95% CI: 1.03–1.08). Age-related neurodegenerative pathologies, such as amyloid-β deposition, may increase the risk of PDD in older adults. 62 We found that the risk of PDD was lower in men than in women; the adjusted HR for male sex was 0.68 (95% CI: 0.47–0.98). However, a meta-analysis indicated male sex was a positive predictor of PDD incidence without covariate adjustment. However, other studies have suggested no association between sex and PDD incidence after covariate adjustment. 61 Further research is needed to investigate the association of sex with the incidence of PDD in patients belonging to different races and regions. We found no association between education level and PDD incidence (adjusted HR: 1.00; 95% CI: 0.97–1.04). A systematic review reported that a lower education level was associated with a higher risk of dementia in many, but not all, studies; the correlation between education and PDD requires further studies with covariate adjustment.61,63
In our study, each patient underwent assessments performed using the following tools: CDR, CASI, HAIADL, CSF, MoCA, NPI, and Unified Parkinson’s Disease Rating Scale. Only the HAIADL score was positively associated with PDD incidence (adjusted HR: 1.14; 95% CI: 1.03–1.26). HAIADL, which was developed by modifying the Lawton Instrumental Activities of Daily Living and Barthel Index, can be used in patients with PD to determine cognitive stages and predict cognitive prognosis.44,64,65 This questionnaire, adapted for Parkinson’s patients, may effectively predict cognitive decline by encompassing motor, mood, motivation, and behavioral factors.
AF is strongly associated with dementia independent of stroke. 66 In the present study, AF was positively associated with PDD incidence (adjusted HR: 1.62; 95% CI: 1.08–2.43). However, CAD was negatively associated with PDD incidence (adjusted HR: 0.58; 95% CI: 0.36–0.92). A meta-analysis reported approximately 50% increases in the risks of MCI and dementia in patients with clinical CAD. 67 Further studies are needed to prevent cognitive decline in patients with PD.
Evidence suggests that type 2 diabetes increases the risk of PD and accelerates its progression. 68 However, the correlation between diabetes and PDD risk remains unclear. A systematic review and meta-analysis offered weak support for cognitive progression in patients with PD, whereas another systematic review and meta-analysis indicated a negative correlation between diabetes and PDD incidence.61,68 While our study found diabetes was associated with a lower risk of PDD (adjusted HR: 0.41; 95% CI: 0.21–0.81), the use of antidiabetic medication was linked to higher risk (adjusted HR: 3.45; 95% CI: 1.66–7.15), possibly due to hypoglycemia-related cognitive impairment. 69
A key strength of the present study lies in its sufficient sample size, which was larger than that of other studies assessing the correlation between exercise and cognitive function in patients with PD. However, our study also has some limitations. First, due to the retrospective longitudinal design of this study, we could not definitively establish causal relationships. Therefore, further large-scale randomized controlled trials are warranted to verify the effectiveness. Second, among the four domains of physical activity (work, transportation, household, and leisure time), only leisure-time walking was assessed in this study. Moreover, other forms of exercise—for example, strength, balance, flexibility, and endurance exercises—were not included. Nevertheless, among the four domains, leisure-time physical activity is the most promotable and healthful. 69 Third, this study was conducted in only two hospitals in central Taiwan and one in southern Taiwan, which might have led to potential selection bias. Participants should be recruited from multiple centers and countries to reduce selection bias related to ethnicity and region. Fourth, nutritional status and socioeconomic status should be adjusted for risk analyses. Finally, the study duration was relatively short; longer observation periods may reveal additional benefits.
Conclusion
MAE through walking may protect patients with PD from PDD. The strongest protective effect was exerted by walking >3 days a week, followed by walking 1–2 days a week. Therefore, physicians should encourage patients with PD to develop a habit of walking and exercising in a safe environment to prevent cognitive decline or dementia.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251330251 – Supplemental material for Walking reduces the risk of dementia in patients with Parkinson’s disease: a longitudinal follow-up study
Supplemental material, sj-docx-1-tan-10.1177_17562864251330251 for Walking reduces the risk of dementia in patients with Parkinson’s disease: a longitudinal follow-up study by Cheng-Yu Wei, Ray-Chang Tzeng, Hsu-Chih Tai, Chun-Hsien Su and Pai-Yi Chiu in Therapeutic Advances in Neurological Disorders
Footnotes
Appendix
Acknowledgements
We would like to thank Dr Hsin-Te Chang, Department of Psychology, Chung Yuan Christian University, for providing feedback on the study protocol and Dr Hsing-Ju Wu, Research Assistant Center, Show Chwan Memorial Hospital, for editing the manuscript.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
