Abstract
Cognitive stimulation therapy (CST) is widely used with people with dementia, but there is no evidence of its efficacy in mild cognitive impairment or dementia in Parkinson’s disease (PD-MCI; PDD) or dementia with Lewy bodies (DLB). We aimed to explore the impact of ‘CST-PD’, which is home-based, individualized CST adapted for this population. In a single-blind, randomized controlled exploratory pilot trial (RCT), we randomized 76 participant–dyads [PD-MCI (n = 15), PDD (n = 40), DLB (n = 21) and their care partners] to CST-PD or treatment as usual (TAU). CST-PD involves home-based cognitively stimulating and engaging activities delivered by a trained care partner. Exploratory outcomes at 12 weeks included cognition (Addenbrooke’s Cognitive Evaluation; ACE-III), neuropsychiatric symptoms and function. In care partners, we assessed burden, stress and general health status. Relationship quality and quality of life were assessed in both dyad members. At 12 weeks, the ACE-III showed a nonstatistically significant improvement in the CST-PD group compared with the TAU group, although neuropsychiatric symptoms increased significantly in the former. In contrast, care partners’ quality of life (d = 0.16) and relationship quality (‘satisfaction’, d = 0.01; ‘positive interaction’, d = 0.55) improved significantly in the CST-PD group, and care burden (d = 0.16) and stress (d = 0.05) were significantly lower. Qualitative findings in the CST-PD recipients revealed positive ‘in the moment’ responses to the intervention, supporting the quantitative results. In conclusion, care-partner-delivered CST-PD may improve a range of care-partner outcomes that are important in supporting home-based care. A full-scale follow-up RCT to evaluate clinical and cost effectiveness is warranted.
Keywords
Introduction
Disorders of cognitive impairment within the Lewy body spectrum of diseases include mild cognitive impairment in Parkinson’s disease (PD-MCI), dementia due to Parkinson’s disease (PDD) and dementia with Lewy bodies (DLB). Together, DLB and PDD constitute over 15% 1 of total dementias and PD-MCI occurs in about 25% of people with Parkinson’s disease (PD).2,3 PDD and DLB are associated with significant impairments in cognition, quality of life and high levels of disability and care-partner burden. 4 Treatment options focus on pharmacological approaches, which have modest effectiveness and may often not be tolerated by frail people with dementia in the context of a movement disorder. Thus, there is scope for nonpharmacological interventions that are specifically adapted for people with cognitive impairment or dementia within the Lewy body spectrum. 5 To date, only one other study of a psychosocial intervention in PDD has been conducted, and this study examined the impact of goal-oriented cognitive rehabilitation therapy. 6 This small study found positive outcomes, self-rated goal attainment, mood, and quality of life in those receiving the active intervention compared with relaxation therapy and ‘treatment as usual’.
Cognitive stimulation therapy (CST) is an evidence-based psychosocial intervention that involves engaging and cognitively stimulating activities and discussions based on principles of errorless learning and validation.7,8 As demonstrated by meta-analyses, CST improves cognition and quality of life in people with different forms of dementia, and improves outcomes, such as quality of life, for care partners.9,10 In people with PDD, only one study has evaluated CST. 11 This was a small pilot crossover trial (n = 12) of people living in a care-home setting. It found that group CST (offered for 8 weeks, twice weekly for 60 min), adapted for PDD, is feasible and potentially effective for cognitive and noncognitive outcomes in PDD, compared with ‘treatment as usual’.
We undertook an iterative development process to adapt the individualized form of CST (iCST) specifically for people with PD-MCI/PDD/DLB to be delivered by their care partners at home (PD-CST). 12 PD-CST differs from professionally delivered group-based CST, in that PD-CST can be delivered at home by a trained care partner and can be tailored more easily to the specific needs and capabilities of the recipient. Here, we report the results of an exploratory pilot study of the impact of PD-CST on recipients of the intervention and their care partners. In addition, we evaluated the acceptability of the intervention and the feasibility of conducting a full-scale randomized controlled trial (RCT). We found that PD-CST was well tolerated and acceptable, with certain modifications, by people with PD-MCI/PDD/DLB and their care partners, and that the trial design was feasible (reported elsewhere). 13
Methods
The full protocol is published in detail elsewhere. 14
Standard protocol approvals, registrations, and patient consents
The study received favourable ethical opinion from Yorkshire and Humber–Bradford Leeds Research Ethics Committee (reference: 15/YH/0531) and was performed in accordance with the Declaration of Helsinki and the principles of Good Clinical Practice. The study was registered at isrctn.com [registration number ISRCTN11455062]. All participants were volunteers and provided written informed consent or consultee agreement to participate in the study.
Study design and participants
The INVEST study was a single-blind, parallel-arm RCT, conducted at seven sites in the UK, to explore the impact of CST-PD compared with treatment as usual (TAU) on cognitive, behavioural, care partner and other dementia-related outcomes in people with PD-MCI/PDD/DLB and their care partners. Since this was the first time home-based CST adapted for this population had been tested, and we had no a priori evidence of how the intervention would be received, we specifically included participants with different levels of cognitive impairment, ranging from MCI to moderate-stage dementia.
We recruited people with PD-MCI/PDD/DLB and their care partners as participant–dyads if they met the following inclusion criteria: (a) diagnosis of PD-MCI (Level 1), PDD (probable or possible) or DLB (probable or possible) according to standard clinical diagnostic criteria;15–17 (b) willing and able to participate in the intervention; and (c) on stable medication for at least 4 weeks prior to study entry. Exclusion criteria were: (a) unwilling or unable to participate; (b) contact with a care partner three times or less per week; (c) no care partner or companion able to participate; (d) living in residential care; (e) unable to understand conversational English; and (f) neuropsychiatric complications too severe (i.e. depression, psychosis or fluctuating levels of cognitive impairment/delirium) to enable participation in the study (as per expert clinicians’ judgement). Care partners were included if they provided care or support for the participant with cognitive impairment and were well enough to be trained to deliver the intervention. Care partners were excluded if they were unable to understand conversational English, were nonliterate or had severe physical illness or dementia (as per self-report and performance on the Montreal Cognitive Assessment). 18 All PD-MCI/PDD/DLB participants (n = 76) had the capacity to consent to participation at the start of the trial. During the trial, four participants lost the capacity to consent, thus a nominated consultee was appointed, enabling all four to continue in the study.
Randomization and blinding
The Manchester Academic Health Science Centre Clinical Trials Unit, as an independent arbiter, applied a single-stratum, blocked randomization to CST-PD or TAU at a 1:1 level by participant–dyad. Due to the nature of the intervention, dyads were not blind to treatment allocation, but procedures were in place to conceal the allocation from the independent, blinded outcome raters. Following randomization, the distribution of the tree diagnostic subgroups (PD-MCI, PDD and DLB) was balanced across the two arms with respect to MCI and dementia.
Intervention
The details of the intervention and how we adapted it to the specific needs of people with PD-MCI, PDD or DLB is outlined in McCormick et al. 12 The adaptation process took account of several factors, including the cognitive profile (e.g. particular challenges with executive function, recall and visuospatial deficits), fatigue and apathy, fluctuating levels of attention, motor and general physical frailty of this population. The adapted intervention, CST-PD, entailed care-partner-delivered manual-based individualized CST-based therapy sessions, delivered at home for 30 min per session, two to three times per week. The activities varied in theme and complexity and could be tailored to suit individual needs. The adapted therapy manual comprised over 60 topics categorized into nine different themes, with each topic containing several cognitively stimulating activities such as discussion topics, word association games and creative tasks. Activities varied in complexity and were matched and adapted to suit the needs of the recipient. The manual itself was paper based, easy to handle and had large accessible print. It was indexed to enable ease of use. All care partners were trained to deliver the therapy as intended. The TAU group, which received no additional intervention, provided a comparison with the CST-PD group. Any additional nonpharmacological interventions that the participants in the TAU group might have received following randomization (e.g. physiotherapy, occupational therapy, speech therapy) were noted by the research team.
Procedure
Following consent, participant–dyads underwent a screening period to ensure eligibility. Participants meeting inclusion criteria received two to three care-partner-delivered CST-PD sessions of 30 min each or TAU for 12 weeks. Prior to the intervention being delivered, care partners in the intervention arm received a 2 h protocol-guided training session of CST-PD, delivered in their own homes by a member of the research team. Training included a researcher-guided therapy session with the person with PD-MCI/PDD/DLB. The researcher completed a protocol training checklist and provided additional training and telephone support as needed, based on a skills’ checklist. Assessments took place at baseline and 12 weeks. Participants who withdrew from the study before their scheduled assessment visits received an early termination assessment. Those experiencing a serious adverse event withdrew from the study. Feasibility, acceptability and tolerability evaluations were also undertaken (reported in McCormick et al.). 13 Assessments for people with PD-MCI/PDD/DLB included: cognition, neuropsychiatric symptoms, quality of life, functional ability, relationship satisfaction and resilience (Table 1). Care-partner assessments were: quality of life, health ratings, relationship satisfaction, burden and resilience (Table 1). Care partners in the CST-PD group used diaries to report adherence of sessions and 11 participant–dyads completed a semistructured interview to elicit their views and experiences of the intervention.
Outcome measure descriptions.
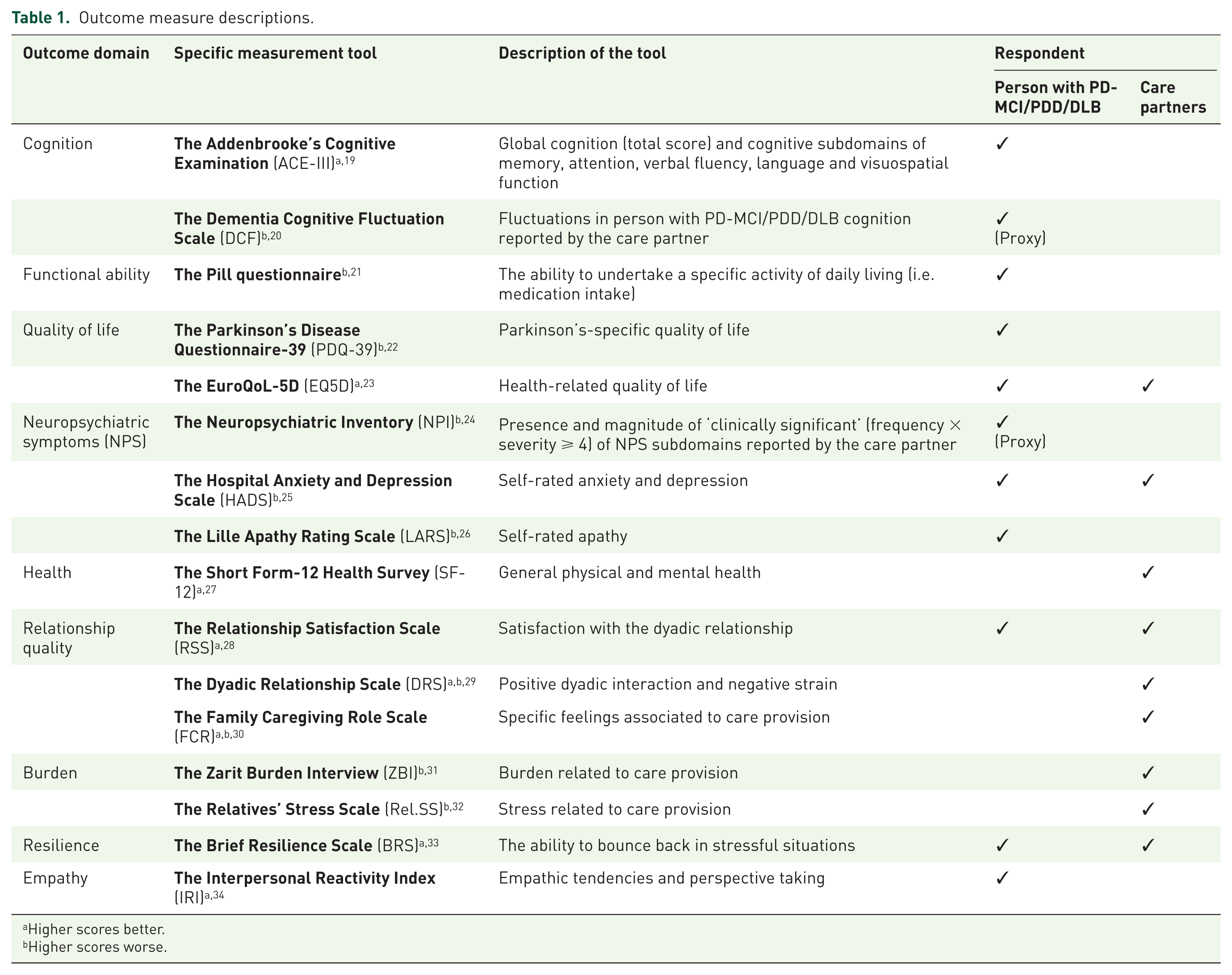
Higher scores better.
Higher scores worse.
Outcome measures
The primary outcomes of the overall INVEST study were tolerability, acceptability and feasibility (reported in McCormick et al.). 13 Here, we report a range of exploratory participant and care-partner outcomes (see Table 1) including cognition, behaviour, function, quality of life and care-partner burden and stress. We also examined aspects of the dyadic relationship, resilience and empathy. All rated outcomes were undertaken by highly trained research nurses with extensive experience in dementia and PD-related research. Additionally, qualitative outcomes using observational data from participant–dyad diaries, completed after each therapy session, and semistructured interviews, in a subsample of the CST-PD group were also included. The interview schedule was iteratively developed alongside the adaptation of the intervention (detailed in McCormick et al.) 13 and addressed three key areas: acceptability and usability of the intervention manual, adherence (including barriers and facilitators) and impact of the intervention. Probe questions under each of these themes facilitated the interview.
‘Feasibility’ included a detailed evaluation of eligibility, recruitment and retention rates, overall trial design (the degree to which the protocol balanced scientific and practical considerations), willingness to be randomized, blinding procedures and data collection (i.e. timing, quality, acceptability). ‘Acceptability’ was the extent to which the participant–dyads considered the intervention ‘appropriate’ (i.e. care-partner’s perceptions of the recipient’s interest, motivation and sense of achievement following each therapy session) and the ability of recipients endure the intervention (i.e. adverse event rate).
Sample size
We based our sample-size calculation on previous studies 35 and took a conservative approach, estimating the standardized effect size on cognition to be 0.4. As this was a pilot feasibility trial, we chose a one-sided test and a less stringent significance level of 0.2 to avoid missing a promising effect. Thus, assuming 80% power and a correlation coefficient of 0.5 between baseline and endpoint on cognitive outcomes, the required sample size was 27 completers per group. By enrolling 32 dyads per group, it allowed for a 15% attrition rate. For the secondary, exploratory outcomes, the proposed sample size of 27 per group was within the recommended guidelines (24–50 participants35,36) required to estimate the standard deviation (SD) for a sample-size calculation. Since the attrition rate was higher than expected during the first year of recruitment (28%), we obtained ethical approval to enrol 38 dyads per group to maintain the target number of completers.
Data analysis
Since this was an exploratory trial of a new complex intervention, we agreed a priori to interpret the results with caution. Thus, although we undertook initial inferential statistics and hypothesis testing, our goal was to uncover any important potential associations in the study variables. 37 For this reason, we evaluated statistical significance at the 0.2 level using a one-sided test. Specifically, we explored changes in measures between the two groups (CST-PD and TAU) over time using ANCOVA, controlling for baseline values. All analyses were conducted on an intention-to-treat basis, on complete case data. For the qualitative analysis, using NVivo 11 for Windows software (QSR International Pty Ltd.), 38 data from participant–dyad diaries, researcher field notes, and semistructured interviews were used. We triangulated the results of our quantitative findings with thematic analysis. 39 Using an inductive process, we systematically extracted codes from each data source to derive key themes; these were subsequently triangulated with the quantitative outcome to establish correspondence between the qualitative and quantitative data. We arrived at the final themes by consensus of five INVEST investigators (IL, SV, SM, SS and BK).
Results
The 76 recruited participant–dyads were randomized to either the CST-PD (n = 38) or the TAU group (n = 38) following randomization (Figure 1). Characteristics of participant–dyads are outlined in Table 2. A total of 21% (n = 16) participants with PD-MCI/PDD/DLB were female and all were native English speakers. Diagnoses included 19.8% (n = 15) PD-MCI, 52.6% (n = 40) PDD and 27.6% (n = 21) DLB. Of the care partners, 89% (n = 68) were female, 77.6% (n = 59) were spouses or live-in partners and 17.1% (n = 13) were adult children. The remaining four care partners included a grandchild, a friend, a live-in carer and a divorcee. Of those randomized, 72% completed the full study protocol.

CONSORT 2010 flow diagram.
Baseline demographic and clinical variables in the active intervention (CST-PD) and control (TAU) groups.

ACE-III, Addenbrooke’s Cognitive Evaluation; BRS, Brief Resilience Scale; CST-PD, cognitive stimulation therapy in Parkinson’s disease; DLB, Dementia with Lewy bodies; DRS, Dyadic Relationship Scale, positive interaction or negative strain subscale; EQ5D, EuroQoL-5D index or Visual Analogue Scale (VAS); FCR, Family Caregiving Role scale; HADS, Hospital Anxiety and Depression Scale; IQR, interquartile range; IRI, Interpersonal Reactivity Index; LARS, Lille Apathy Rating Scale; MoCA, Montreal Cognitive Assessment; NPI, Neuropsychiatric Inventory; PDD, Parkinson’s disease dementia; PD-MCI, Parkinson’s disease and mild cognitive impairment; PDQ-39, Parkinson’s Disease Questionnaire; PD-MCI/PDD/DLB, Parkinson’s-related dementia; Rel.SS, Relatives’ Stress Scale; RSS, Relationship Satisfaction Scale; SD, standard deviation; SF-12, Short Form 12 Health Survey; TAU, treatment as usual; UPDRS, Unified Parkinson’s Disease Rating Scale; ZBI, Zarit Burden Interview.
Baseline demographics revealed a relatively good case mix between the two arms, with only education and diagnosis seeing a slight imbalance (see Table 2). Descriptive statistics of the outcome measures at baseline are presented in Table 2. There were also some imbalances in baseline outcome scores between the two arms, suggesting randomization was not fully successful, possibly due to the small sample size. We avoided any potential bias by controlling for baseline scores in the analysis. No cognitive-enhancing medications were changed during the course of the study.
Preliminary analysis compared the effect of treatment allocation and baseline characteristics of subjects with and without complete data at follow up using a logistic model for each outcome. Differential missingness was observed in the treatment arms, with a higher proportion of missing data in the intervention arm. For the primary outcome, data were missing for 21 individuals; 6 (29%) in the control arm and 15 (71%) in the intervention arm. We found no differential missingness conditional on the participant characteristics; thus, we proceeded with the main analysis under the ‘missing at random’ assumption.
A total of 56 participant–dyads completed the study; 24 in the CST-PD group and 32 in the TAU group. Using ANCOVA to model group differences of change in cognition at 12 weeks by adjusting for baseline scores, global cognition (ACE-III) improved by 1.7 on average in the CST-PD group compared with the TAU group; however, this difference was not statistically significant [adjusted mean difference (AMD) = 1.7, Cohen’s d = 0.38, p = 0.227]. The results of the exploratory measures revealed a number of potential changes in outcomes for both intervention recipients and care-partner groups and are presented in Table 3. For intervention recipients, the CST-PD group had statistically lower scores on the verbal fluency subscale of the ACE-III (AMD = −0.74, d = 0.35, p = 0.134), higher scores on the Neuropsychiatric Inventory (NPI) total signalling greater symptoms (AMD = 4.46, d = 0.42, p = 0.049), as well as the proportion of ‘clinically significant’ [frequency × severity (F×S) score ⩾ 4] and ‘clinically present’ NPI (F×S score > 1) scores (AMD = 0.05, d = 0.35, p = 0.078; AMD = 0.05, d = 0.25, p = 0.173, respectively), the Brief Resilience Scale (AMD = −1.17, d = 0.12, p = 0.174), and the perspective-taking subscale of the Interpersonal Reactivity Index (AMD = −1.32, d = 0.03, p = 0.082). Conversely, for the care-partner sample, CST-PD resulted in statistically significant improvements compared with TAU on quality of life [EuroQol index (AMD = 0.08, d = 0.16, p = 0.048) and Visual Analogue Scale measures (AMD = 4.76, d = 0.07, p = 0.104)], burden and stress [Zarit Burden Interview (AMD = −2.24, d = 0.16, p = 0.193), and the Relatives’ Stress Scale (AMD = −1.75, d = 0.05, p = 0.160), respectively] and relationship quality [Relationship Satisfaction Scale (AMD = 3.46, d = 0.01, p = 0.020), and the Dyadic Relationship Scale positive-interaction subscale (AMD = 1.76, d = 0.55, p = 0.015)]. In contrast, care partners in the CST-PD group reported a significant increase in anxiety symptoms measured by the Hospital Anxiety and Depression Scale (AMD = 1.03, d = 0.30, p = 0.112). Adherence data, retention and integrity of blinding (for details, see elsewhere) 13 revealed that over two thirds of participants in the CST-PD group received the recommended dose of at least 60 min of therapy per week.
Exploratory results for participant–dyads in the two arms showing the difference between baseline and 12 weeks.

Adjusted for baseline outcome value.
One-sided confidence interval provides the (upper or lower) limit of range of plausible values of point estimate.
Significance level 0.2.
Higher scores better.
Higher scores worse.
ACE-III, Addenbrooke’s Cognitive Evaluation; BRS, Brief Resilience Scale; DRS, Dyadic Relationship Scale, positive interaction or negative strain subscale; EQ5D, EuroQoL-5D Index or Visual Analogue Scale (VAS); FCR, Family Caregiving Role scale; HADS, Hospital Anxiety and Depression Scale; IRI, Interpersonal Reactivity Index; LARS, Lille Apathy Rating Scale; MoCA, Montreal Cognitive Assessment; NPI, Neuropsychiatric Inventory; PDQ-39, Parkinson’s Disease Questionnaire; PD-MCI/PDD/DLB, Parkinson’s-related dementia; Rel.SS, Relatives’ Stress Scale; RSS, Relationship Satisfaction Scale; SD, standard deviation; SF-12, Short Form 12 Health Survey; ZBI, Zarit Burden Interview.
Qualitative evaluation
Synthesis of qualitative data elicited six themes, presented in Table 4, together with the corresponding outcome domain related to each theme, and example extracts supporting each theme. Overall findings suggest ‘in the moment’ enjoyment of CST-PD (Enjoyment/Fun). In addition, consistent with quantitative data regarding efficacy outcomes, the qualitative data suggest participants experienced improvements in cognition, with subsequent impact on communication and conversation with care partners (Communication/Cognition). The findings regarding physical and mental abilities suggested that while for some participants, CST-PD afforded the opportunity to demonstrate retained abilities, for others, the intervention highlighted changes and loss. The care-partner outcomes indicated that CST-PD provided opportunities for conversation and reminiscence that would not have otherwise have occurred (interpersonal relationships), although some degree of challenge and burden regarding the delivery of the CST sessions was reported by three care partners (care-partner aspects).
Key themes emerging from the semistructured interview with participant–dyads.

C, care partner; P, person with Parkinson’s-related dementia.
Discussion
This is the first randomized controlled feasibility trial of a CST-based intervention specifically adapted for people with Parkinson’s-related MCI or dementia (PDD/DLB) and is the largest study of its kind to date. It thus makes a valuable contribution to the emerging field of nonpharmacological interventions for cognition and other dementia-related outcomes in this population. The CST-PD programme retains the core principles of the already well-established CST but is specifically tailored to the needs of people with a complex form of dementia characterized by motor and other physical problems. The preserved CST features include positive discussion, enjoyable activities, affection, supportive feedback and a focus on opinions rather than facts. Critical modifications included removing motor-dependent activities, potentially hallucinogenic or unclear images and updating manual content by increasing the usability of the format. 12 The ability to tailor the intervention to specific needs and preferences of the participants enabled us to offer successfully the intervention to people with a wide range of cognitive abilities and interest, without the risk of the intervention not being challenging enough. Furthermore, we designed CST-PD to be a home-based, individualized, care-partner-delivered intervention, which strengthened the dyadic relationship of the person and their care partner, which is key in predicting positive outcomes of home-based care.40,41
We have already demonstrated the feasibility of conducting such a study in this population, as well as it being an acceptable and well-tolerated intervention in people with PD-MCI/PDD/DLB and their care partners. 13 Here, our exploratory analysis of potential efficacy outcomes in the intervention recipients indicated improvements for cognition overall, bu this did not reach statistical significance. This is consistent with a previous study of iCST in people with other forms of dementia, 8 but not group CST studies in non-PD groups, demonstrating significant improvement in cognition.7,42,43 It is possible that the imbalance in the education level of the two experimental arms may have impacted on this, as the participants in the CST-PD arm had a lower education level compared with those in the TAU group. A potential mechanism for improvement in cognition with this type of intervention could be the activation of compensatory mechanisms of synaptic plasticity. 44 As suggested by the original study of iCST in non-PD dementia, 8 it is possible that a higher dose of therapy is needed to impact cognition. In our study, the average dose was 1.76 (SD = 0.72) per week, 13 and this may not have been high enough. Higher intervention doses of cognitive rehabilitation have been shown as beneficial in very early-stage cognitive impairment in PD. 45 This is despite previous suggestions that in non-PD dementia, cognitive benefits can be seen with twice-weekly sessions of CST. 46 The added burden of physical disability and PD-associated fatigue and apathy may further hinder any potential benefit and necessitate an even higher dose.
Contrary to expectation, informant-rated behavioural outcomes assessed with the NPI appeared to worsen in the CST-PD group. This included the apathy domain, which has been shown to improve with intensive cognitive rehabilitation in early-stage cognitive impairment in PD. 45 It is likely that the additional time spent with the therapy recipient through undertaking the therapy may have highlighted previously unrecognized behavioural and psychiatric symptoms (BPSDs), resulting in higher informant ratings across a range of symptoms, or that the slight imbalance in diagnostic subtypes across the two treatment arms may have played a role. However, it is important to note that no participants were withdrawn due to worsening of BPSDs and BPSDs were not reported as adverse events in the qualitative data. 13 Although the direction of these results suggests a potentially harmful intervention, they should be interpreted with caution, since we purposely chose a high significance level to capture any potential effects increasing probability of type I errors. Combined with the exploratory multiple comparisons, we may be observing false positives. Furthermore, the behavioural outcomes were not mirrored in the qualitative reports, which revealed positive, ‘in the moment’ experiences in cognition, behaviour and function immediately following therapy sessions.
In contrast to the clinical outcomes in the participants with PD-MCI/PDD/DLB, care partners experienced improvements in several outcomes. This is crucial, considering that caregiving in this population is complex, and care burden is best described as a multidimensional construct 47 that has a significant negative effect on a care partners’ quality of life, health and relationships,48–50 in effect, creating ‘hidden or invisible patients’. 51 Specially, care partners in the CST-PD group reported reduced care burden and stress, improved quality of life, and enhanced relationships with the individual with PD-MCI/PDD/DLB. This finding is particularly striking, since in previous work, care partners rated relationship quality lower than people with dementia. 52 It also supports and extends the results of the original iCST study, which found that quality of life in care partners improved and that individuals with dementia regarded the care relationship more positively. 8 Maintaining a positive caring relationship and ensuring care-partner health and wellbeing is essential to delay or prevent long-term care for people with PD-MCI/PDD/DLB,39,53 slow progression of cognitive and functional decline 54 and lower care-partner burden. 55 It may also lower costs of providing care, and reduce length of hospitalization and rate of crisis interventions.56,57
The intervention had no observable effects on either ‘resilience’ or ‘empathy’; however, this was not surprising considering the relatively small sample size of this pilot study and that it was not powered to detect differences on these variables. Resilience, measured with the Brief Resilience Scale (BRS), assesses the ability to bounce back or recover from stress and consists of six items scored using a 5-point Likert scale. Empathy, measured with the Interpersonal Reactivity Index, encompasses two aspects: empathic concern and perspective taking, each measured on a Likert scale. These are important as they may reflect aspects of the apathy syndrome, which is closely linked with cognitive impairment/dementia in PD. We also saw no significant changes in apathy scores.
A potential limitation of our study is the heterogeneity of the diagnoses of the participant group. We purposefully included the three groups, as this feasibility study was an initial exploration of the appropriateness of the intervention across the range of cognitive impairment within the Lewy body spectrum. However, the heterogeneity renders the findings difficult to interpret and future trials should aim to limit inclusion to a single group or those with PDD/DLB only.
In conclusion, this study, although a pilot exploratory trial, has provided invaluable data to progress the emerging field of psychosocial interventions for PD-MCI/PDD/DLB, as well as contributed to the literature on dyadic psychosocial interventions. It strongly supports a role for care-partner-delivered interventions through the mechanism of supporting care-partner health and wellbeing, as well as strengthening relationship quality. A full-scale trial is now warranted to establish clinical effectiveness.
Footnotes
Acknowledgements
The authors would like to thank the participants and their care partners for their dedication and commitment to the project. We also acknowledge and thank Martin Orrell and the original CST and iCST development team for giving permission to adapt the therapy, the NIHR for funding the trial, the R&D personnel involved in study set-up and maintenance, the focus-group members who advised on the adaptation of the therapy and, not least, the NHS clinicians and nursing teams involved in referral of participants to the trial, including Patsy Cotton, Carol Miller and Ailish Fountain. We would also like to thank Sophie Baker for her committed work on the project, Benjamin Kwapong for conducting qualitative interviews, and the Clinical Research Network teams in England who helped with recruitment, data collection and trial procedures, including, but not limited to, Heena Mistry, Judith Brooke, Anita Davies, Audrey Williamson, Deborah Risorto, Caroline Cheetham, Emma Eccles, Joanne Woodward, Jane Burgess, Jacki Stansfeld, Katy Aros, Eanna Hanratty and Ritchard Ledgerd.
Roles of the authors were:
(1) Research project: (a) conception; (b) organization; (c) execution; (d) clinical oversight; (e) research oversight.
(2) Statistical analysis: (a) design; (b) execution; (c) review and critique.
(3) Qualitative analysis: (a) design; (b) execution; (c) review and critique.
(4) Manuscript: (a) writing of the first draft; (b) review and critique.
IL: (1) a–e; (2) c; (3) a–c; (4) a, b
SV: (1) b, c; (2) c; (3) b, c; (4) b
LAC: (2) a–c; (4) b
SJS: (1) e; (3) a–c; (4) b
VO: (1) e; (4) b
EP: (1) e; (4) b
MAS: (1) d; (4) b
JaR: (1) d; (4) b
DJA: (1) d; (4) b
CT: (1) d; (4) b
JoR: (1) d; (4) b
TAG: (1) d; (4) b
SAM: (1) b, c; (2) c; (3) b, c; (4) b
The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health.
Funding
This paper presents independent research funded by the National Institute for Health Research (NIHR) under its Research for Patient Benefit (RfPB) Programme (PB-PG-0613-31058). The trial was sponsored by the Greater Manchester Mental Health NHS Foundation Trust, UK.
Conflict of interest statement
VO has a patent: iCST Manual. No other authors have received any funding from any institution, including personal relationships, interests, grants, employment, affiliations, patents, inventions, honoraria, consultancies, royalties, stock options/ownership or expert testimony for the last 12 months.
VO has a patent: iCST Manual. The other authors declare that there is no conflict of interest.
Trial registration number
The trial is a psychosocial intervention with an allocated ISRCTN number 11455062.
