Abstract
Introduction:
Ozanimod is a new-generation sphingosine-1-phosphate (S1P) modulator, approved for the treatment of multiple sclerosis (MS), offering higher selectivity for S1P receptor 1 and 5 (SPR1-5), minimizing potential safety concerns related to S1P3 receptor activation, compared to fingolimod.
Objectives:
We aimed to compare the adherence and persistence on treatment in MS patients switched to ozanimod from fingolimod for safety reasons (mainly lymphopenia or liver enzymes increase).
Methods:
We retrospectively recruited patients treated with fingolimod who switched to ozanimod for safety reasons, with at least 12 months of follow-up. We collected demographic, clinical, biochemistry, and safety data during fingolimod and after switching to ozanimod to evaluate (1) lymphocytes and liver enzymes dynamics, (2) persistence on ozanimod over 6 months, (3) proportion of patients with no adverse events (NADE) on ozanimod and no evidence of disease activity (NEDA-3).
Results:
We recruited 60 relapsing–remitting MS patients (mean age of 42 ± 7.9 years) who were treated with fingolimod for an average of 5.7 years (61.6% female) and switched to ozanimod due to lymphopenia (70%) or hypertransaminasemia (21.6%). A total of 58/60 (96%) patients persisted on treatment with ozanimod for a mean of 1.50 ± 0.49 years; mean lymphocyte count increased from 0.39 to 0.56 (p = 0.025) in patients who switched due to lymphopenia; hypertransaminasemia decreased from 21.6% in fingolimod to 9.3% in ozanimod. NADE was recorded in 93% patients during ozanimod treatment and NEDA-3 in 88.3% of patients after 1 year. Overall, patients with complete control of disease (NEDA) in the absence of adverse events (NADE) were 83.7% (NEDA3/NADE).
Discussion and conclusion:
Our findings suggest that switching from fingolimod to ozanimod may mitigate lymphopenia or hypertransaminasemia and ameliorate effectiveness on disease activity.
Introduction
The treatment landscape of multiple sclerosis (MS) has rapidly expanded over the last years, 1 encompassing several therapeutic options for patients with MS (pwMS). These include different subtypes of sphingosine 1-phosphate receptor (S1PR)-modulating agents, such as fingolimod and ozanimod.
Fingolimod is an old-generation drug, nonselective S1PR modulator that binds to the receptor subtypes S1PR1, S1PR3, S1PR4, and S1PR5, and was the first oral disease modifying therapy approved for MS. After many years of experience, its cardiovascular, ophthalmologic, pulmonary, kidney, and hepatic safety concerns have been well characterized, encompassing bronchial tone and hyper-reactivity, natriuretic effect, hepatocyte proliferation, heart rate and vascular tone, macular edema. These effects are due to increased expression at the tissue level of receptor subtypes S1PR1, S1PR2, and S1PR3. 2
Ozanimod is a new molecule recently approved, designed to target selectively subtypes S1PR1 and S1PR5 3 minimizing potential safety concerns related to S1P3 receptor activation; the clinical efficacy, safety, and tolerability of ozanimod for the treatment of patients with relapsing multiple sclerosis (RMS) have been demonstrated in the multicenter randomized phase III trial (RADIANCE): and multicenter randomized phase III trial (SUNBEAM).4,5 Ozanimod therapy should be initiated with a 7-day titration period consisting of 0.23 mg once daily on days 1–4 and 0.46 mg once daily on days 5–7. From indirect comparative studies based on clinical trials emerges that ozanimod has a better safety profile, with lower risks of heart rate reduction, thanks also to 7-day titration period. Overall, patients receiving ozanimod had also a lower risk of any adverse event (AE), including severe lymphopenia and abnormal liver enzymes elevation than patients receiving fingolimod. This probably is attributed to the major selectivity of ozanimod to binding S1PR1 and S1PR5, thus reducing events related to S1PR3 and S1PR4 such as lymphopenia and endothelial regulation (mainly mediated by S1PR3 6 ). Sometimes treatment with fingolimod requires personalized dosage to counteract AEs such as lymphopenia and transaminase elevation. However, dosage reductions (i.e., administering every other day) are not part of the correct schedule, and studies also show reduced treatment efficacy. 7
The objective of our study was to evaluate the safety and effectiveness of switching from fingolimod to ozanimod in pwMS for safety reason (mainly lymphopenia or liver enzymes increase).
Methods
This was a multicenter retrospective observational study. We enrolled patients aged 18–60, who were enrolled from five different Italian MS centers. Inclusion criteria were (1) confirmed diagnosis of relapsing remitting multiple sclerosis (RRMS) according to the 2010 McDonald criteria 8 ; (2) ability to provide written informed consent; (3) switching from fingolimod to ozanimod for lymphopenia, increase in liver enzymes (for lymphopenia, patients who for more than one examination had lymphocytes below 150 units/mL and for transaminases a repeated value three times higher than the reference value), or other safety reasons (i.e., comorbidities, other AEs). Pregnancy or lactation in women were exclusion criteria. All patients had active MS, defined by at least one clinical relapse and/or radiological activity (new T2 lesions or gadolinium-enhancing (gd+) lesions) within the 2 years prior to initiating treatment with fingolimod, in accordance with the prescribing criteria for S1PR modulators in Italy.
Data collection
We collected blood cells count, lymphocytes, aspartate aminotransferase (AST), alanine aminotransferase (ALT), gamma glutamyl transferase (GGT), and total bilirubin during fingolimod and ozanimod treatment, scheduled as per clinical practice. Clinical and radiological data that were also recorded, including the number of relapses in the year before starting fingolimod, during fingolimod and during ozanimod; number of gd+ lesions before starting fingolimod, during fingolimod, and during ozanimod annually, as per clinical practice. Additionally, we recorded the expanded disability status scale (EDSS) score at the initiation and at the end of fingolimod treatment, at the initiation of ozanimod treatment, and at the most recent follow-up visit.
Main outcomes
We evaluated the time on treatment for both therapies and the percentage of patients who persisted in treatment over 6 months after switching from fingolimod to ozanimod. Lastly, we assessed the percentage of patients who remained on ozanimod treatment for over 1 year after switching from fingolimod, as well as those who discontinued due to lymphopenia or increased liver enzymes. We also evaluated the percentage of patients who were free of disease activity and progression (not evidence of disease activity (NEDA-3 9 )) and who were free of AEs (not evidence of adverse events (NADE)). All patients gave written informed consent to be included in the study.
Statistical analysis
Continuous variables were reported as mean ± standard deviation (SD) or median and range (minimum to maximum) according to their distribution. Categorical variables were presented as absolute number and percentage (%). For each patient, we compared the annualized relapse rate (ARR), the number of new T2 lesions, and gd+ lesions in the year before and after the initiation of ozanimod using the Wilcoxon signed-rank test for paired samples. Similarly, we used the same statistics to compare mean lymphocyte count and mean liver enzyme levels (AST, ALT, GGT), and bilirubin on fingolimod versus ozanimod treatment. Statistical analyses were performed using IBM SPSS Statistics 21. Results were considered statistically significant for p < 0.05.
Results
We enrolled 60 patients (61.6% female) who switched from fingolimod to ozanimod for lymphopenia (70%) or hypertransaminasemia (21.6%). Four patients (6.6%) switched due to other safety reasons (two patients for the diagnosis of ulcerative colitis and two for the diagnosis of Crohn’s disease).
The mean EDSS at diagnosis was 2.40 (sd 0.79), while the median number of treatments before Fingolimod was 1 (minimum 0, maximum 4). The mean duration of treatment with fingolimod was 5.72 years (sd 3.26). The mean follow-up in ozanimod treatment was 1.50 years (sd 0.49). Table 1 resumes the baseline demographic and clinical characteristics of our cohort.
Clinical and demographic characteristics of patients included in the study.

ARR, annualized relapse rate; EDSS, Expanded Disability Status Scale; gd+, gadolinium-enhancing.
The median time elapsed between the suspension of fingolimod and ozanimod initiation (median wash-out between treatments) was 23 days (minimum 1 day, maximum 180 days). The median time elapsed in patients with lymphopenia was slightly lower (24 days (minimum 1 day and maximum 180 days)) compared to patients who switched due to hypertransaminasemia (27 days (minimum 1 day and maximum 120)). Fingolimod was administered at an off-label dosing in 28.5% of patients, usually every other day, except for 2 patients who skipped 1 dose/week.
For treatment effectiveness, we observed a significant reduction in the ARR, decreasing from 0.26 (sd 0.44) in the year prior to switching to ozanimod to 0.02 (sd 0.15) after the switch (p = 0.004). There was also a reduction in the mean number of new T2 lesions, from 0.40 (sd 0.67) in the previous year to 0.09 (sd 0.17) on ozanimod, though this change was not statistically significant (p = 0.15). Additionally, the mean number of new gd+ lesions also decreased, but not significantly, from 0.11 (sd 0.21) to 0.02 (sd 0.04; p = 0.16).
After 1.5 years of follow-up, 58/60 patients (96%) continued treatment with ozanimod. One patient discontinued due to clinical relapses (1.7%), which occurred 30 days after initiating ozanimod and following a suboptimal response to fingolimod (administered off-label at every other day dosing). Another patient (1.7%) discontinued ozanimod due to the desire for pregnancy. Treatment effectiveness (fingolimod vs ozanimod) is compared in Table 2.
Comparison of treatment effectiveness, transaminase, and lymphocytes levels (fingolimod vs ozanimod).

ALT, alanine aminotransferase; ARR, annualized relapse rate; AST, aspartate aminotransferase; gd+, gadolinium-enhancing; GGT, gamma-glutamyl transferase.
Lymphopenia
None of our patients discontinued ozanimod due to lymphopenia occurring after the switch. In fact, we observed an overall increase in the mean lymphocyte counts within the overall study cohort (Table 2). Among patients who were lymphopenic prior to the switch, the mean lymphocyte count increased from 0.39 (sd 0.24) to 0.56 (sd 0.23; p = 0.025; Figure 1). A single patient (1.7%) developed grade 3 lymphopenia during ozanimod treatment.

Mean lymphocytes count before and after switching in patients who had lymphopenia.
Liver function
We observed a decrease in the percentage of patients with hypertransaminasemia from 21.6% during fingolimod to 9.3% during ozanimod (p = 0.03). Similarly, there was a significant decrease in the mean AST, ALT, and GGT levels in patients switching for hypertransaminasemia; bilirubin remains stable from fingolimod to ozanimod (Figure 2; Table 2 NEDA-3 and NADE).

Mean AST, ALT, and GGT levels before and after switching in patients who had hypertransaminasemia.
NADE was recorded in 93% patients during ozanimod treatment. After 1 year, no evidence of disease activity (NEDA-3) was achieved in 88.3% patients. Overall, patients with complete control of disease activity (NEDA) in the absence of adverse events (NADE) were 83.7% (Figure 3).
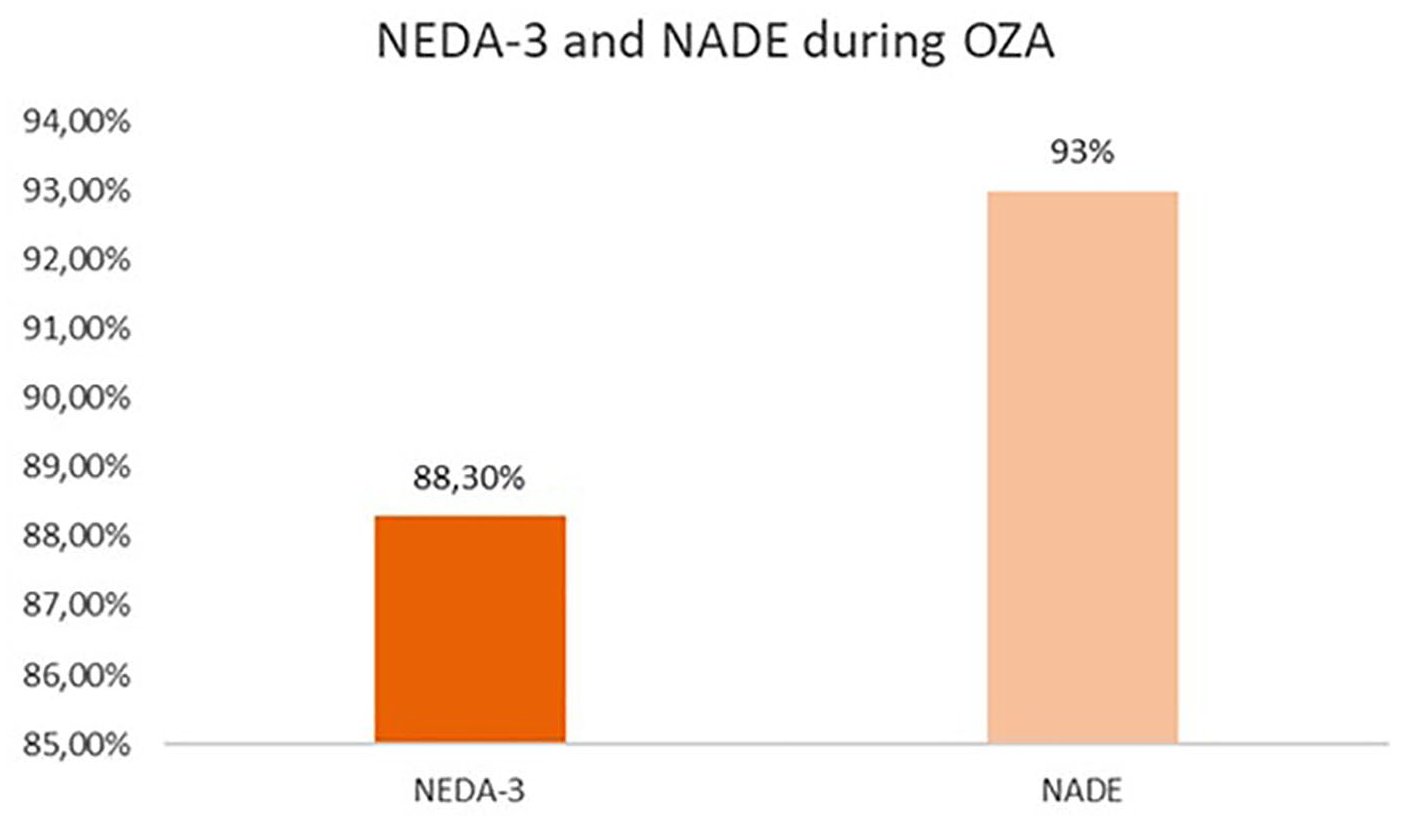
Percentage of patients with NEDA-3 and NADE on ozanimod.
Discussion
Ozanimod is a S1PR modulator that selectively interacts with S1PR-1 and S1PR-5, offering reduced interference with S1PR3 and S1PR4. This minimizes the risk of bradycardia, lymphopenia, and hepatic events. Clinical trials, including the extension study (DAYBREAK), have confirmed the favorable safety and tolerability profile. In a study involving 2494 participants, only 10.3% experienced lymphopenia, and 6.9% had increased GGT levels. 10 These findings prompted us to investigate whether patients experiencing safety issues, such as lymphopenia or elevated liver enzymes (hypertransaminasemia) during treatment with a less-selective S1PR modulator (fingolimod), might benefit from switching to ozanimod while maintaining the advantages of S1PR modulation.
Recent real-world evidence suggests that switching from fingolimod to ozanimod could be an effective strategy for patients with lymphopenia, particularly those taking fingolimod every other day. In a prior study involving 18 patients with RRMS, a significant increase in lymphocyte counts was observed 6 months after switching to ozanimod, compared to fingolimod discontinuation and to the prior 12 months. 11 Our study, with a larger sample size, confirmed improvements in lymphopenia in both patients who were treated with a reduced dosage of fingolimod and who received the standard daily schedule.
Regarding liver safety, we observed a lower incidence of hypertransaminasemia in patients who had elevated AST, ALT, and GGT levels during fingolimod and switched to ozanimod. This finding aligns with indirect comparisons of clinical trials for both treatments. For instance, Swallow et al. reported that ozanimod, with comparable efficacy to fingolimod, was associated with a significantly lower risk of AEs and abnormal liver enzyme elevations (−3.0%) compared to fingolimod. The differing impact on the liver function might be attributed to ozanimod’s metabolism via the CYP2C8 pathway, with less involvement of CYP3A4, compared to fingolimod. 12
In terms of efficacy, ozanimod demonstrated a significant reduction in the ARR and fewer new T2 or gd+ lesions compared to the prior year on fingolimod. This observation may potentially stem from the fact that 28.5% of patients were on an off-label dosing of fingolimod, which has been associated with reduced efficacy, as previously reported. Our study introduced the concept of “no adverse drug events” (NADE), with 93% of ozanimod-treated patients falling into this category. This measure complements traditional MS treatment goals, such as achieving NEDA-3, by also emphasizing safety and treatment adherence.
Our study has some relevant limitations. Given the small sample size, we could not identify predictors of favorable outcomes on ozanimod, and we doesn’t evaluate the adherence to treatment. Similarly, we cannot exclude the possibility of confounders when comparing the effectiveness of the two treatments or changes in laboratory parameters. However, to the best of our knowledge, this was the largest cohort of patients investigating the effects of switching from fingolimod to ozanimod for safety reasons. Furthermore, the observational study design may harbor risk of selection bias and limit the generalizability of our findings.
Conclusion
In conclusion, our findings suggest that switching from fingolimod to ozanimod may mitigate lymphopenia or hypertransaminasemia and ameliorate effectiveness on disease activity. Our findings need to be validated in larger prospective cohorts to determine whether switching from fingolimod to ozanimod is an effective option for patients experiencing safety issues. Additionally, future studies should aim to identify predictors of improved clinical outcomes following the switch, providing further insight into patient selection and optimizing treatment strategies.
