Abstract
Background:
Several oral disease-modifying therapies (DMTs) have been approved by the Food and Drug Administration for the treatment of relapsing-remitting multiple sclerosis (RRMS). In the absence of head-to-head randomized data, matching-adjusted indirect comparisons (MAICs) can evaluate the comparative effectiveness and safety of ozanimod versus other oral DMTs in RRMS.
Objectives:
To synthesize results from the published MAICs of ozanimod and other oral DMTs for 2-year outcomes in RRMS.
Methods:
Published MAICs involving ozanimod for the treatment of RRMS were identified. Extracted data elements included efficacy [annualized relapse rate (ARR), confirmed disability progression (CDP), and brain volume loss] and safety [adverse events (AEs), serious AEs (SAEs), AEs leading to discontinuation, and infection] outcomes.
Results:
The four MAIC studies identified compared ozanimod with fingolimod, teriflunomide, dimethyl fumarate (DMF), and ponesimod. All comparisons were adjusted for differences in age, sex, relapses within the previous year, Expanded Disability Status Scale score, and percentage of patients with prior DMTs. Outcomes at 2 years were analyzed based on comparisons that lacked a common comparator arm. Ozanimod was associated with significantly lower ARR versus teriflunomide [ARR ratio (95% CI) 0.73 (0.62, 0.84) and DMF 0.80 (0.67, 0.97)], with no significant difference versus fingolimod or ponesimod. The proportions of patients treated with ozanimod or fingolimod had similar 3- and 6-month CDP. Compared with teriflunomide and DMF, ozanimod was associated with a significantly lower risk of 3-month CDP; 6-month CDP was comparable. Ozanimod was associated with significantly lower rates of any AE and AEs leading to discontinuation compared with the other oral DMTs evaluated. Ozanimod also had significantly lower rates of SAEs versus teriflunomide and DMF and lower rates of reported infection outcomes versus fingolimod and ponesimod.
Conclusion:
Compared with the other oral DMTs evaluated in MAICs, ozanimod was associated with a favorable safety profile and improved or comparable efficacy outcomes.
Plain language summary
The many treatment options available for relapsing-remitting multiple sclerosis (RRMS) make treatment decisions difficult. While direct head-to-head treatment comparisons provide useful information, these studies are not available for every pair of treatments. Indirect comparisons of published study results can help fill that evidence gap. A technique called matching-adjusted indirect comparison (MAIC) offers a statistically robust way to compare safety/efficacy outcomes from different studies by accounting for important differences across the studies.
We collected data from four MAIC studies that compared 2-year treatment outcomes in patients treated with ozanimod versus those treated with fingolimod, teriflunomide, dimethyl fumarate (DMF), or ponesimod. Each study accounted for differences in age, sex, relapses within the previous year, disability status, and previous therapy use.
We found ozanimod was either better than or similar to other treatments based on the outcomes measured. The annual rate of RRMS relapse was lower for patients treated with ozanimod than for patients treated with teriflunomide or DMF and similar for patients treated with ponesimod or fingolimod. Ozanimod-treated patients saw their RRMS progress at rates similar to those treated with fingolimod at 3 and 6 months and teriflunomide and DMF at 6 months; RRMS was more likely to progress at 3 months in patients treated with teriflunomide and DMF versus those treated with ozanimod.
Our analyses also found that patients treated with ozanimod had lower rates of side effects, including those serious enough to cause treatment discontinuation, compared with patients receiving other treatments.
By comparing findings from existing MAIC studies, we found that patients with RRMS treated with ozanimod had fewer side effects and better or similar efficacy outcomes compared with patients who received other treatments for RRMS. These findings can potentially inform treatment decisions for patients with RRMS.
Keywords
Introduction
Multiple sclerosis (MS) is a chronic immune-mediated and neurodegenerative disease of the central nervous system that leads to debilitating symptoms such as fatigue, depressive symptoms, and cognitive impairment.1,2 MS may manifest as a clinically isolated syndrome, relapsing-remitting MS (RRMS), secondary progressive MS, or primary progressive MS.3,4 RRMS is the most prevalent type of MS, affecting about 85% of individuals with MS 5 and characterized by episodes of relapses followed by periods of remission. 6
Disease-modifying therapies (DMTs), administered either orally, as an injection, or an infusion, have been used to alter the disease course by reducing relapses, with the goal of preventing or slowing long-term disability.7–11 Patients with MS report higher patient satisfaction and medication adherence with oral DMTs than with injectables.12,13 At the time of this assessment, nine oral DMTs were approved by the Food and Drug Administration for the treatment of RRMS, all of which modulate the immune system in various ways. These include sphingosine 1-phosphate (S1P) receptor modulators [fingolimod (Gilenya), ozanimod (Zeposia), siponimod (Mayzent), and ponesimod (Ponvory)], fumarates [dimethyl fumarate (DMF; Tecfidera), monomethyl fumarate (Bafiertam), diroximel fumarate (DRF; Vumerity)], cladribine (Mavenclad), and teriflunomide (Aubagio).
A comparison of the efficacy and safety of treatments for RRMS is needed to better inform clinical decision makers. Due to a lack of head-to-head randomized trial data comparing oral DMTs, separate matching-adjusted indirect comparisons (MAICs) have been used to compare ozanimod with four of these oral treatments (fingolimod, 14 teriflunomide, 15 DMF, 16 and ponesimod 17 ) to adjust for differences in patient data across the clinical trials.
Some oral DMTs were not included in this review. Since no randomized controlled trial was conducted for DRF and, at the time of the ozanimod versus DRF MAIC, only 48 weeks of interim results from the EVOLVE-MS-1 trial were available, the MAIC between DRF and ozanimod was outside the scope of this review. 18 Moreover, no MAICs between ozanimod and cladribine, siponimod, or monomethyl fumarate have been conducted to date.
The objective of this study was to identify, review, and synthesize the methods and results from currently published MAICs of ozanimod for RRMS.
Methods
Brief description of MAIC methodology
MAIC is a propensity score weighting method that uses individual patient data (IPD) from one trial (or set of trials) and aggregate data (AD) from another trial (or set of trials) to conduct indirect treatment comparisons while accounting for between-trial imbalances in observed covariates that are suspected prognostic or predictive factors related to outcomes of interest.19–21 Individuals in the IPD trial population are weighted by the inverse of their propensity score to balance the covariate distribution with that of the target AD trial population. Under this approach, individual patients in the IPD trial(s) are assigned weights such that the weighted mean baseline characteristics in the IPD trial population(s) exactly match those reported in the AD trial population(s). Outcomes of interest are then compared across the balanced populations. When a common comparator arm (e.g. placebo) is not available in the IPD and AD trials, an unanchored MAIC approach is used to directly compare the outcomes from the reweighted IPD and the AD trial populations. The MAIC weights can also be used to calculate the effective sample size (ESS), which can be used to assess the statistical validity of the indirect comparisons: small ESSs are indicative of a lack of overlap between the trial populations with possibly unstable comparative effect estimates and wide confidence intervals (CIs).
Data sources
Published MAICs involving ozanimod for the treatment of RRMS were identified via a targeted literature review. The data sources for this study included the information in these publications, complemented as necessary with information extracted from the ClinicalTrials.gov site of each trial.
Data extraction elements
Table 1 presents the data elements that were extracted from each published MAIC.
Extracted data elements.

Based on data availability and the results of a feasibility assessment, the current study focused on outcomes compared at 2 years. Outcomes compared at 1 year were also considered. First-dose monitoring outcomes were not included in the current study as these data are available only for the MAIC comparing ozanimod and fingolimod.
MAIC, matching-adjusted indirect comparison.
Outcome measures across MAICs
Efficacy outcomes
Efficacy outcomes extracted as available from the data included annualized relapse rate (ARR), proportions of patients free of 3- and 6-month confirmed disability progression (CDP) at 2 years, and brain volume loss (BVL) expressed as the mean percent change from baseline at 2 years.
Safety outcomes
Safety outcomes extracted as available included rates of adverse events (AEs), of serious AEs (SAEs), of AEs leading to discontinuation, and of infection outcomes (appendicitis, herpes zoster infection, upper respiratory tract infections, and nasopharyngitis).
Synthesis approach
After the data extraction, key similarities and differences across MAICs in terms of the analytical approaches and limitations were summarized qualitatively. This study did not include meta-analyses or analytic calculations of data across studies.
Results
Overview of trials
Four published MAICs involving ozanimod for the treatment of RRMS were identified in the targeted literature review, including comparisons with fingolimod, 14 teriflunomide, 15 DMF, 16 and ponesimod. 17 Table 2 lists the trials included in each MAIC. Trial details are presented in Table A1 in the Appendix. All trials involved in these comparisons were phase III.
Ozanimod MAIC studies and the respective phase III clinical trials.

AD, aggregate data; IPD, individual patient data; MAIC, matching-adjusted indirect comparison.
Baseline characteristics used for adjustments
Table 3 shows the variables that were used for cross-trial adjustments in each MAIC. Age, sex, relapses within previous year, Expanded Disability Status Scale (EDSS) score, and percent of patients with prior DMTs were used for cross-trial adjustments in all MAICs. Some MAICs included additional characteristics, such as absence of gadolinium-enhancing lesions, resting heart rate, or lymphocyte count.
Baseline variables used for cross-trial adjustments in the identified MAICs.

Used for adjustment for first-dose monitoring outcomes only.
DMT, disease-modifying therapy; EDSS, Expanded Disability Status Scale; Gd, gadolinium; MS, multiple sclerosis.
Efficacy outcomes at 2 years
Due to a lack of common comparators across trials, all the 2-year outcomes were analyzed based on unanchored MAICs.
The ARR data presented in Figure 1 show that treatment with ozanimod was associated with a significantly lower adjusted ARR at 2 years versus both teriflunomide [ARR ratio 0.73 (95% CI: 0.62, 0.84)] 15 and DMF [0.80 (0.67, 0.97)], 16 a nonsignificantly lower ARR versus ponesimod [0.80 (0.57, 1.10)], 17 and a nonsignificantly higher ARR versus fingolimod [1.06 (0.70, 1.62)]. 14 With smaller proportions of patients experiencing relapse over 2 years, ozanimod was associated with significantly lower risk of relapse than teriflunomide and DMF.15,16

Forest plot of adjusted ARR ratios of ozanimod versus each oral DMT at 2 years.
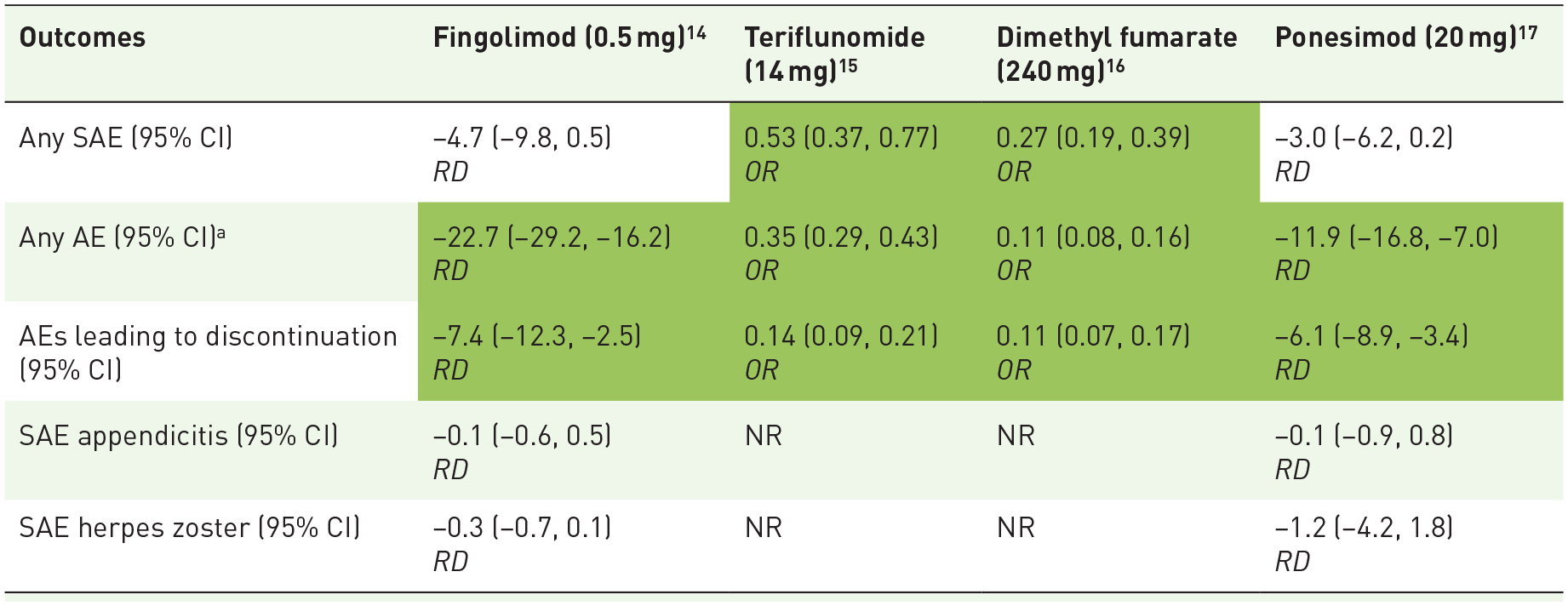
Table 4 presents MAIC-adjusted (i.e. after matching) results for key efficacy and safety outcomes (other than ARR) at 2 years. Note that there were differences across MAICs in the measures used to compare outcomes.
Adjusted efficacy outcomes (other than ARR) for ozanimod versus each comparator at 2 years for the identified MAICs

Cells in green represent statistically significant results in favor of ozanimod (95% CIs do not cross 0 or 1 depending on the type of outcome). Cells in white represent results for which ozanimod and the respective comparator are statistically comparable (95% CIs do cross 0 or 1 depending on the type of outcome).
ARR, annualized relapse rate; CD, change difference; CDP, confirmed disability progression; HR, hazard ratio; MAIC, matching-adjusted indirect comparison; NR, not reported; OR, odds ratio; RD, risk difference; RR, rate ratio.
The 3- and 6-month CDP data were compared in three of the four MAICs (the comparison involving ponesimod was not conducted due to cross-trial differences in outcome definitions). Compared with fingolimod, ozanimod had similar proportions of patients with 3- and 6-month CDP (no significant differences). 14 Compared with teriflunomide and DMF, ozanimod was associated with a significantly lower risk of 3-month CDP and a nonsignificantly lower risk of 6-month CDP.15,16
In the only MAIC to examine BVL, ozanimod was associated with significantly less BVL at 2 years than ponesimod. 17
Safety outcomes at 2 years
Adjusted safety outcomes data indicated that ozanimod had a consistently favorable safety profile compared with the other oral DMTs (Table 5). Ozanimod was associated with significantly lower rates of any AEs and AEs leading to discontinuation compared with the other oral DMTs evaluated. Ozanimod had lower rates of infection outcomes compared with fingolimod and ponesimod.14,17 Ozanimod had no significant differences in the rates of any SAE compared with fingolimod and ponesimod.14,17 Finally, comparative data on rates of nasopharyngitis were not reported in any of these MAICs.
Adjusted safety outcomes for ozanimod versus each comparator at 2 years for the identified MAICs.

Cells in green represent statistically significant results (95% CIs do not cross 0 or 1 depending on the type of outcome) in favor of ozanimod. Cells in white represent results for which ozanimod and the respective comparator are statistically comparable (95% CIs do cross 0 or 1 depending on the type of outcome).
Rates of any AEs should be interpreted with caution when comparing across trials because event ascertainment and reporting may differ, especially for lower-severity events.
AE, adverse event; NR, not reported; OR, odds ratio; RD, risk difference; SAE, serious adverse event.
Key differences across MAICs
Table 6 summarizes the key cross-trial differences noted by the authors of each MAIC. Common cross-trial differences included lack of common comparator arms, as well as differences before matching in baseline EDSS scores, in geographic regions, and in mean disease duration after diagnosis. See Table A2 in the Appendix for additional details.
Cross-trial differences commonly reported in the identified MAICs.

Countries in the OPTIMUM trial that were not included in RADIANCE Part B (e.g. Czech Republic) were classified as Eastern European based on the United Nations delineation of geographic regions.
DMF, dimethyl fumarate; EDSS, Expanded Disability Status Scale; MAIC, matching-adjusted indirect comparison; NR, not reported.
Discussion
Due to a lack of head-to-head randomized trial data between ozanimod and other oral DMTs in RRMS, indirect treatment comparisons have been conducted to further assess and contextualize the comparative effectiveness and safety of ozanimod versus available oral DMTs while explicitly adjusting for cross-trial differences in patient characteristics. In the current study, we evaluated and synthesized the evidence from four published MAICs comparing ozanimod with fingolimod, teriflunomide, DMF, and ponesimod.
Across the four MAICs, the results indicate that, compared with other oral DMTs, ozanimod is statistically better or comparable in terms of key efficacy outcomes and has a consistently superior safety profile; thus, ozanimod has a favorable risk-benefit profile compared with the oral DMTs evaluated. Specifically, compared with teriflunomide and DMF, ozanimod was associated with significantly lower rates of relapse and disability progression as well as key safety outcomes. Ozanimod also had a favorable safety profile versus fingolimod and ponesimod, with significantly lower risk of any AEs and AEs leading to discontinuation. The increased selectivity of S1P modulation with ozanimod compared with fingolimod may contribute to lower systemic AEs. 22 Finally, ozanimod had comparable effects on ARR versus fingolimod and ponesimod and significant reductions in BVL compared with ponesimod.
The results of these MAICs were aligned in terms of the directions of the pairwise treatment comparisons with the results from a previous network meta-analysis (NMA) 23 that assessed the relative efficacy (ARR) and safety (rates of AEs and SAEs) of ozanimod compared with other DMTs in RRMS (but did not include ponesimod owing to lack of data at the time of the NMA). As an established method of indirect treatment comparison, an NMA compares multiple treatments simultaneously; MAIC methods, a newer approach, can be conducted only between discrete paired comparisons. An NMA uses AD and assumes low heterogeneity between trial populations; by weighting patient-level data from one trial to make them more comparable to AD from another, MAICs reduce observable heterogeneity in trial populations, thereby limiting potential imbalances in effect modifiers across trials. 24 The overall alignment of the results from the MAICs examined here and the previous NMA point to the robustness of each of these indirect comparisons with different methodological specifications, suggesting that there may be little unobserved difference or heterogeneity between the populations.
With respect to ARR, the NMA and MAIC results were similar for fingolimod and teriflunomide and directionally aligned for DMF (the treatment effect differences were statistically significant in the MAICs for DMF but not in the NMA). For AEs, the results of the MAICs were also directionally the same as the NMA results, with treatment effect differences that were statistically significant in the MAICs but not in the NMA. For SAEs, results from the MAICs and NMA were similar for fingolimod and directionally the same for teriflunomide; however, the MAIC results indicated that ozanimod had significantly lower rates of SAEs than DMF, while the NMA results indicated these rates were comparable.
In terms of limitations of the MAICs included in this assessment, one important consideration is the lack of a common comparator arm for the 2-year comparisons; thus, the comparative estimates were based on unanchored MAICs (i.e. treatment comparisons without a common comparator arm). Compared with anchored MAICs (those with a common comparator arm in each trial), unanchored MAICs offer less opportunity to assess the magnitude of residual confounding bias and rely on stronger assumptions (i.e. that all effect modifiers and prognostic factors were accounted for in the population adjustments). Furthermore, while these MAICs adjust for observed differences in patient characteristics between ozanimod and each comparator, the possibility of confounding due to unobserved baseline differences cannot be ruled out. As for the limitations of the synthesis carried out herein, not all outcomes were reported in the same scale (e.g. risk differences versus odds ratios) across MAICs; the adjusted estimates, therefore, could not be visually displayed in the same graph (as was done for ARR). However, the results can still be interpreted appropriately once the scale differences are taken into account. Finally, the outcome data were qualitatively summarized across MAICs, as this synthesis did not include meta-analyses of efficacy and safety data across studies to avoid potential biases related to the pooling of comparative data from indirect treatment comparison studies with different outcome definitions and population adjustments.
The comparative evidence of ozanimod versus other oral DMTs in the treatment of RRMS generated in these previous MAICs and synthesized herein is relevant for decision makers when assessing the relative effectiveness and safety of these therapies. In its totality, this evidence suggests that ozanimod has a favorable benefit-risk profile compared with other oral DMTs and that ozanimod has a consistently favorable safety profile compared with other oral DMTs, which could lead to better medication adherence.
Conclusion
The evidence synthesized in this study indicates that, compared with the other oral DMTs evaluated in MAICs, ozanimod was associated with a consistently favorable safety profile and improved or comparable efficacy outcomes. Compared with teriflunomide and DMF, ozanimod was associated with significantly lower rates of relapse and disability progression, as well as lower rates of key safety outcomes. Compared with fingolimod and ponesimod, ozanimod had a significantly lower risk of any AEs and of AEs leading to discontinuation, while showing comparable effects on ARR. Compared with ponesimod, ozanimod was associated with significant reductions in BVL. The results from these analyses suggest that ozanimod has a favorable risk-benefit profile compared with other currently available oral DMTs in RRMS. While MAIC methodology is sufficiently robust to provide insights into the relative clinical value of ozanimod versus other oral DMTs in RRMS, the evidence synthesized in this study is subject to limitations (e.g. unobserved confounding) that only well-conducted, randomized trials can avoid.
Footnotes
Appendix
Additional differences across trials for each MAIC.

| Data elements | Fingolimod (0.5 mg) a | Teriflunomide (14 mg) b | Dimethyl fumarate (240 mg) c | Ponesimod (20 mg) d |
|---|---|---|---|---|
| Trial design | Time periods: Ozanimod trials were conducted between 2013 and 2017, while the fingolimod trials were conducted between 2006 and 2011. Geography: Ozanimod trials included higher proportions of patients from Eastern Europe (~90%) than the fingolimod trials (~2%), although they are both multinational. |
Differences were observed in patient identification, EDSS upper thresholds, and trial periods. Ozanimod trials had more stringent eligibility criteria with respect to history of relapse, variation in DMT use, which may be attributable to differing definitions of prior DMT use in the trial eligibility criteria, as well as geographic differences affecting access to other DMTs prior to trial initiation. |
The DMF trials were placebo controlled, whereas the ozanimod trials used interferon β-1a as comparator. Ozanimod trials enrolled patients with secondary progressive MS (<0.4%) and progressive relapsing MS (<1.8%) in addition to patients with RRMS (>98%), whereas DMF trials enrolled only those with RRMS. Ozanimod trials required patients to have had either at least one relapse in the 12 months before screening or at least one relapse in the 24 months before screening with at least one GdE+ lesion in the 12 months before randomization. DMF trials required patients to have had either at least one clinically documented relapse within 12 months prior to randomization or at least one GdE+ lesion within 6 weeks prior to randomization. |
RADIANCE-B trial had a higher % of patients from Eastern Europe. Comparators were different across trials (interferon β-1a for RADIANCE-B trials and teriflunomide in OPTIMUM trial). |
| Key inclusion/exclusion criteria (as noted in MAIC publication) | In the ozanimod trials, patients had a diagnosis of RMS, as defined by the 2010 revised McDonald criteria; had at least one documented relapse in the previous year before screening; and had a score between 0.0 and 5.0 on the EDSS. In the fingolimod trials, patients had a diagnosis of RMS, as defined by the 2005 revised McDonald criteria; had at least one confirmed relapse during the preceding 1 year (or at least two during the preceding 2 years); and had a score between 0.0 and 5.5 on the EDSS. |
Patients enrolled in the ozanimod trials had at least one relapse in the 12 months before screening or at least one relapse in the 24 months before screening with at least one GdE+ lesion in the 12 months before randomization. Patients enrolled in the teriflunomide trials only had at least one relapse in the preceding 12 months but one GdE+ lesion in preceding 6 weeks or 6 months. |
No major differences were noted. | OPTIMUM trial includes patients with RMS or secondary progressive MS with superimposed relapses (2.6% of patients) RADIANCE-B trial included patients with EDSS score between 0 and 5.0 at baseline versus EDSS score of up to 5.5 inclusive at baseline in OPTIMUM. There were differences in exclusion criteria related to prior treatment and the time periods over which the treatments could be administered. Additionally, there were differences in exclusion criteria in terms of MS-specific criteria, contraindications and disease history. |
| Patient characteristics | An RMS diagnosis was based on the 2010 revised McDonald criteria in the ozanimod trials and on the 2005 revised McDonald criteria in the fingolimod trials. The ozanimod trials required either one documented relapse in the prior year or one in the prior 2 years along with GdE+ lesions, whereas the fingolimod trials required either one confirmed relapse during the prior year or at least two during the prior 2 years. The upper threshold for the EDSS score for inclusion in the ozanimod trials was 5.0, whereas the upper threshold was a score of 5.5 in the fingolimod trials. Patients receiving ozanimod compared with those receiving fingolimod have shorter MS duration (6.8 versus 8.5 years, respectively) and lower likelihood of prior DMT use (29.0% versus 56.4%). |
Fewer female patients (65% versus 69%) and more Eastern European patients (90% versus 31%) were in the ozanimod group than the teriflunomide group. Patients in the teriflunomide group also had longer mean disease duration after diagnosis (teriflunomide: 4–6 years; ozanimod: 3–4 years) and after symptom onset (teriflunomide: 7–9 years; ozanimod: 6–7 years). Prior DMT use was higher in the teriflunomide group. |
Ozanimod trials required an MS diagnosis based on 2010 McDonald criteria, whereas DMF trials required an MS diagnosis based on 2005 McDonald criteria. DMF trials had higher proportions of patients who had received a prior DMT (39% versus 29%). Baseline patient characteristics also varied for sex (female: 65% ozanimod versus 72% DMF) and disease duration from diagnosis (5–6 years ozanimod versus 3–4 years DMF). |
Duration of MS since first symptom was slightly longer for OPTIMUM patients than RADIANCE-B patients .Higher percentage of patients in OPTIMUM had a prior DMT. |
| Key outcomes | Data for the 6-month CDP at 1 year was not reported in the fingolimod trial; a comparison between ozanimod and fingolimod on this outcome was not feasible. | NR | It was not possible to obtain hazard ratios for CDP at 6 months, since the CONFIRM and DEFINE studies did not report Kaplan–Meier curves at 6 months. Therefore, RRs using person-years as the denominator were used to assess CDP at 6 months. | ARR analysis: RADIANCE-B used a negative binomial regression model adjusting for region (Eastern Europe versus Rest of World), baseline age, and baseline number of GdE+ lesions. OPTIMUM used a negative binomial regression model adjusting for prior use of DMTs, EDSS category, and the number of relapses in the year prior to study entry. BVL analysis: In RADIANCE-B, change from baseline brain volume was calculated using descriptive statistics. In OPTIMUM it was calculated as least squares mean change using a mixed model with linear time effect and covariates of EDSS score strata (⩽3.5 or >3.5), DMTs within the 2 years prior to randomization strata, GdE+ T1 lesions at baseline, and baseline brain volume. In RADIANCE-B, CDP was defined as a sustained worsening in EDSS at least 1.0. In OPTIMUM, CDP was defined as an increase of at least 1.5 with baseline EDSS score of 0, at least 1.0 with baseline EDSS score of 1.0–5.0, or at least 0.5 with a baseline EDSS score of at least 5.5. RADIANCE-B reported proportion free of GdE+ T1 lesions, while OPTIMUM reported cumulative number of new GdE+ T1 lesions. Also, there were no data reported for T2 lesions in OPTIMUM trial (T2 lesions are only mentioned as part of a composite endpoint, cumulative number of combined unique active lesions). |
Swallow et al. 14
Cohan et al. 15
Cohan et al. 16
Swallow E et al. 17
ARR, annualized relapse rate; BVL, brain volume loss; CDP, confirmed disability progression; DMF, dimethyl fumarate; DMT, disease-modifying therapy; EDSS, Expanded Disability Status Scale; GdE, gadolinium enhancing; MAIC, matching-adjusted indirect comparison; MS, multiple sclerosis; RMS, relapsing multiple sclerosis; RR, rate ratio;RRMS, relapsing-remitting multiple sclerosis.
Acknowledgements
Medical writing support was provided by Loraine Georgy, PhD, of Analysis Group, Inc., and editorial support was provided by Eleanor Bush, MA, of Peloton Advantage, an OPEN Health company, funded by Bristol Myers Squibb.