Abstract
Background:
Fatigue is a prevalent non-motor symptom that often appears in the early stages of Parkinson’s disease (PD). Plasma neurofilament light chain (NfL) was elevated in PD patients and may be considered a potential biomarker for both motor and cognitive progression.
Objectives:
In this study, we explored the association between plasma NfL levels and various fatigue subtypes and the prediction of baseline plasma NfL levels for fatigue subtype conversion.
Methods:
Patients with PD were classified into four categories: persistent fatigue, never fatigue, non-persistent fatigue, and new-onset fatigue. They underwent detailed neurological evaluations at baseline and a 2-year follow-up. Plasma NfL, glial fibrillary acidic protein, phosphorylated tau181, amyloid beta 42, and Aβ40 levels in both PD patients and control subjects were measured using an ultrasensitive single molecule array.
Results:
The study enrolled 174 PD patients and 95 control subjects. Plasma NfL levels were significantly higher in the persistent fatigue group compared to the never fatigue group at the 2-year follow-up (
Conclusion:
Baseline plasma NfL levels may serve as a biomarker for predicting fatigue subtype conversion and the progression of fatigue in PD.
Introduction
Parkinson’s disease (PD) is a clinically heterogeneous disorder, characterized by a diverse array of motor and non-motor symptoms with unclear etiology.1,2 Non-motor symptoms include cognitive impairment, anxiety, depression, constipation, and autonomic dysfunction. 3 Fatigue, a prevalent non-motor symptom that often appears in the early stage of PD, 4 occurs with a frequency of 33%–58%. 5 Typically characterized by sensations of tiredness, diminished motivation, or complete physical exhaustion, fatigue significantly impacts the subjective initiative of PD patients.6,7 This, in turn, influences their daily functional capabilities and diminishes the overall quality of life. 8 Despite its high incidence, fatigue might be subjectively expressed among PD patients and has traditionally been overlooked in clinical practice.
Several studies have suggested that fatigue is related to anxiety, depression, apathy, sleep disturbances, and cognitive impairment.9,10 We have previously found that fatigue was associated with motor symptoms, sleep disturbances, and anxiety symptoms among early-stage PD patients. 11 Previous research has indicated that high levels of cerebrospinal fluid (CSF) α-syn oligomer and C-reactive protein were associated with severe fatigue in PD patients.12,13 The plasma inflammatory cytokines such as tumor necrosis factor-α and interleukin-6 were associated with fatigue in PD.14,15 A growing number of studies focused on the plasma biomarkers related to PD, such as neurofilament light chain (NfL), glial fibrillary acidic protein (GFAP), phosphorylated tau181 (p-tau181), amyloid beta 42 (Aβ42), and amyloid beta 40 (Aβ40).16–18 However, no studies have yet investigated whether these plasma biomarkers are associated with fatigue of PD.
Fatigue in PD is **non-persistent, with patients experiencing shifts between fatigue and non-fatigue.11,19,20 Currently, research on fatigue mainly focuses on the factors influencing the incidence and progression of fatigue. However, studies addressing the shift of fatigue, particularly predictive factors for fatigue conversion, are notably scarce. It remains unknown whether these biomarkers can predict the occurrence of fatigue and provide early intervention. Accordingly, we explored the difference in biomarkers among various fatigue subtypes at baseline and over time within a longitudinal cohort of Chinese PD patients. Additionally, we investigated the relationship between baseline factors, plasma biomarker levels, and fatigue conversion.
Materials and methods
Participants
Patients with PD were recruited from an ongoing perspective longitudinal cohort study conducted at the Department of Neurology, West China Hospital of Sichuan University. All participants provided written informed consent. The study was approved by the Ethics Committee of West China Hospital of Sichuan University (No. 2015-236).
A total of 174 early-stage PD patients (disease duration <3 years and Hoehn & Yahr (H&Y) stage ⩽2) and 95 healthy controls (HCs) were included in the study. All subjects were recruited from February 2015 to November 2021 and followed for up to 2 years. PD patients were diagnosed clinically according to the 2015 Movement Disorder Society clinical diagnostic criteria for PD by experienced movement disorder specialists. 21 Patients presenting with Parkinson-plus syndromes, including multiple system atrophy, progressive supranuclear palsy, cortical basal ganglia degeneration, dementia with Lewy bodies, and secondary parkinsonism (e.g., vascular, drug-induced, inflammatory, immune-mediated, normal pressure hydrocephalus, infectious, and traumatic) were excluded. We excluded patients with severe cognitive decline, significant depression or anxiety, and those who had a history of heart failure, liver disease, renal failure, and lung disease were excluded. Besides, participants undergone surgical procedures for PD were excluded, including deep brain stimulation therapy. HCs were recruited from volunteers or spouse of PD patients who have the same exclusion criteria as the participants with PD.
Clinical evaluation
Detailed clinical assessment and data collection during follow-up were previously described in our study. 3 Demographic information including age, sex, education level, age of onset, and disease duration was collected in all subjects at baseline. The clinical features of each participant were carefully evaluated at baseline, 1 year, and 2 years during the follow-up period. Assessments of motor symptoms was performed in the “OFF” state. The evaluation of non-motor symptoms was performed in the “ON” state. PD symptoms were assessed with part I, II, and III sub-scales of the Movement Disorders Society Unified Parkinson’s Disease Rating Scale (UPDRS), and H-Y staging was undertaken for all PD patients.
Montreal Cognitive Assessment (MoCA, Beijing Version) was used to assess cognitive function. 22 The 14-item Hamilton anxiety rating scale and the 24-item Hamilton depression (HAMD-24) rating scale were used to evaluate anxiety and depressive symptoms, respectively. Rapid eye movement sleep behavior disorder (RBD) was evaluated with the RBD screening questionnaire. Excessive daytime sleepiness (EDS) was assessed with the EDS scale. Parkinson’s disease sleep symptoms (PDSS) were used to assess with PD sleep scale. Levodopa equivalent daily dose (LEDD) was assessed according to the levodopa conversion formula. 23
Fatigue elevation
Fatigue was measured by the Fatigue Severity Scale (FSS), which is a validated scale for PD and recommended for both screening and severity rating. 24 The scale includes nine items, and each item is rated on a 7-point Likert scale in which 1 signifies “I strongly disagree” and 7 signifies “I strongly agree.” The total score is given as the mean score of all items and a higher total score indicates severe fatigue. A total FSS mean score of ⩾4 is used to define fatigue. 25 We classified fatigue into the following four types based on fatigue evaluation at baseline, 1-year follow-up, and 2-year follow-up 19 : persistent fatigue referred to fatigue that appeared at baseline, 1-year follow-up, and 2-year follow-up; never fatigue referred to no fatigue reported at baseline, 1-year follow-up, and 2-year follow-up; non-persistent fatigue referred to fatigue that disappeared during the 2-year follow-up; and new-onset fatigue referred to the first appearance of fatigue at the 2-year follow-up.
Measurement of plasma biomarkers
Fasting venous blood samples, comprising 4 mL of whole blood collected in EDTA anticoagulant tubes, were obtained from both PD patients and HCs. These samples were centrifuged at 4°C within 2 h (2000 rpm, 10 min), and the upper plasma was collected and stored at −80°C to avoid repeated freezing and thawing. Plasma NfL, GFAP, p-tau181, Aβ42, and Aβ40 levels were measured for all participants at baseline and re-measured for PD patients at the 1-year and 2-year follow-up. Measurements were conducted using an ultrasensitive single-molecule array (Simoa™; Quanterix, Billerica, MA, USA) implemented on the automated Simoa™ HD-X platform (GBIO, Hangzhou, China). The Neurology 4-Plex E Assay Kit (Cat No: 103670) and p-tau181 Advantage V2 Assay Kit (Cat No: 103714) were used (Quanterix). The plasma sample was randomized, blinded, and measured using a batch of reagents from the same lot.
Statistical analysis
The sample size was calculated based on a previous study 26 and performed by G*Power software. The Shapiro–Wilk test was conducted to assess the normality of data. For normally distributed variables, descriptive statistics are presented as the mean ± standard deviation (SD). Analysis of variance with Bonferroni post hoc tests was performed for multiple group comparisons. For variables that were non-normal or exhibited non-homoscedasticity, data are expressed as the median (interquartile range). Multiple comparisons were conducted using the non-parametric Kruskal–Wallis test. Categorical variables were compared using Chi-square tests or Fisher’s exact tests, as appropriate. Univariate and multivariate binary logistic regression models were applied to identify baseline factors associated with fatigue and its transformation. In the multivariate binary logistic regression model, variables were carefully selected for inclusion based on the number of available events and the strength of their univariate associations.
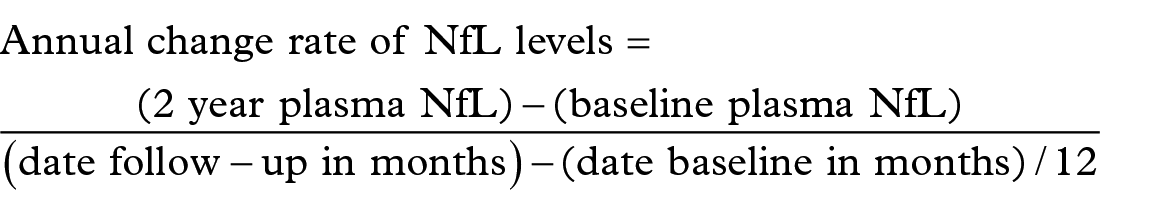
Spearman correlation analysis was conducted to evaluate the association between baseline plasma NfL levels and clinical characteristics across different groups. Receiver operating characteristic (ROC) curve analysis was performed to assess the ability of baseline plasma NfL levels to predict shifts in baseline fatigue subtypes. De Long test was used to compare the AUC value.
All analyses were carried out using SPSS version 22.0 (IBM Corporation, Armonk, NY, USA). Scatter plots were generated using GraphPad Prism version 8.0 (GraphPad Software, Inc., San Diego, CA, USA). A
Results
Comparison of clinical features between PD patients with and without fatigue in PD at baseline and 2-year follow-up
In the study, 174 patients with PD and 95 HCs were included at baseline. At the 1-year follow-up, all of the included PD patients were evaluated with clinical and plasma data. At the 2-year follow-up, seven PD patients failed to be included in the study (one patient died and six patients were lost to follow-up). All the rest were evaluated with the clinical data, but only 118 patients of them had plasma data. The age of the PD patients was 57.72 ± 11.16 years old with an age of onset of 55.75 ± 11.54 years, a disease duration of 1.88 (1.79) years, and an average H-Y stage of 2 (0). The age of HCs was 55.08 ± 7.48 years old. The comparison of the clinical features and plasma biomarkers between patients with and without fatigue at baseline, 1-year, and 2-year follow-up were shown in Table 1. Among 174 PD patients, 65 (37.36%), 64 (36.78%), and 51 (30.53%) patients had fatigue at baseline, 1-year follow-up, and 2-year follow-up, respectively. Scores of UPDRS-II, UPDRS-III, HAMA, and HAMD in PD patients with fatigue were always higher than in PD patients without fatigue at baseline and follow-up (
Comparison of clinical features between patients with and non-fatigue in PD at baseline and 2-year follow-up.
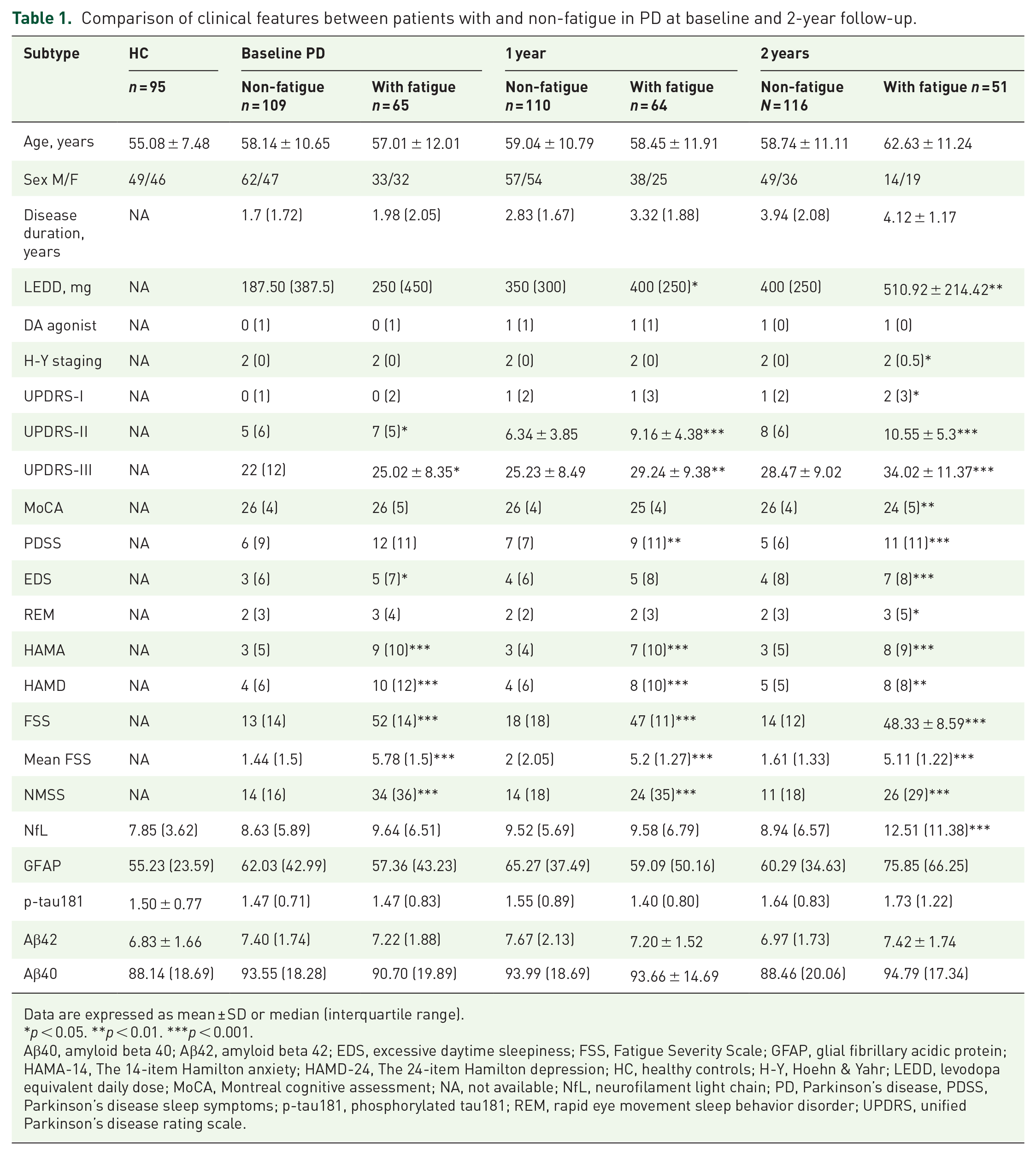
Data are expressed as mean ± SD or median (interquartile range).
*
Aβ40, amyloid beta 40; Aβ42, amyloid beta 42; EDS, excessive daytime sleepiness; FSS, Fatigue Severity Scale; GFAP, glial fibrillary acidic protein; HAMA-14, The 14-item Hamilton anxiety; HAMD-24, The 24-item Hamilton depression; HC, healthy controls; H-Y, Hoehn & Yahr; LEDD, levodopa equivalent daily dose; MoCA, Montreal cognitive assessment; NA, not available; NfL, neurofilament light chain; PD, Parkinson’s disease, PDSS, Parkinson’s disease sleep symptoms; p-tau181, phosphorylated tau181; REM, rapid eye movement sleep behavior disorder; UPDRS, unified Parkinson’s disease rating scale.

Plasma NfL levels in PD patients with fatigue. Plasma NfL levels in fatigue and non-fatigue groups were compared at baseline (a), 1-year follow-up (b), and 2-year follow-up (c). Errors bars represent mean ± SD.
The conversion of different fatigue subtypes at the 2-year follow-up
The shift of fatigue subtypes is shown in Figure 2. Longitudinally, 45.16% of patients with fatigue at baseline converted to patients without fatigue at the 2-year follow-up. Finally, 22.12% of patients without fatigue at baseline converted to patients with fatigue at the 2-year follow-up.

The shift of fatigue in patients with PD over time. Different fatigue conversion at the 1-year and 2-year follow-ups. Fatigue and non-fatigue were represented by red and blue. The number of patients with fatigue conversion was shown in the row.
In the four fatigue subgroups, 73 (41.95%) patients had never fatigue, 26 (14.94%) patients had persistent fatigue, 23 (13.22%) patients had new-onset fatigue, and 52 (29.89%) patients had non-persistent fatigue. PD patients with persistent fatigue had higher UPDRS-III, PDSS, and ESS scores than patients who had never fatigue at baseline (

Plasma NfL levels in PD patients with different fatigue subtypes. Plasma NfL levels in PD patients and HCs were compared at baseline, 1 year, and 2 years (a). Plasma NfL levels in different fatigue subtype groups were compared at baseline (b) and 2-year follow-up (c). Change in NfL levels per year in different fatigue subtypes were compared (d). Errors bars represent mean ± SD.
Correlation between fatigue subtype conversion and baseline plasma NfL levels
Univariate logistic regression analysis with persistent fatigue subtype as the dependent variable showed a significant association with baseline plasma NfL levels (odds ratio (OR) = 1.146,
Multivariate logistic regression analysis for the independent association between baseline plasma NfL levels and fatigue subtype conversions.

Results from univariable logistic regression.
Results from multivariable logistic regression: persistent group adjusted for age, LEDD, MoCA, HAMD, HAMA, PDSS, RBD, and UPDRS-III; New-onset group adjusted for age, GFAP, EDS, and UPDRS-III.
CI, confidence interval; EDS, Excessive daytime sleepiness; GFAP, glial fibrillary acidic protein; HAMA-14, The 14-item Hamilton anxiety; HAMD-24, The 24-item Hamilton depression; LEDD, levodopa equivalent daily dose; MoCA, Montreal cognitive assessment; NfL, neurofilament light chain; OR, odds ratio; PDSS, Parkinson’s disease sleep symptoms; RBD, Rapid eye movement sleep behavior disorder; UPDRS, unified Parkinson’s disease rating scale.
Prediction of fatigue subtype conversion using baseline plasma NfL levels and clinical characteristics
ROC analysis indicated that baseline plasma NfL levels predicted the fatigue persistent with an AUC of 0.64, (95% CI: 0.5113–0.7700, sensitivity = 42.31%, specificity = 89.04%). Combining the four variables most strongly associated with persistent fatigue in multivariate logistic regression analysis (plasma NfL levels, PDSS score, HAMD score, and UPDRS-III score) obtained an AUC of 0.896 (95% CI: 0.8343–0.9581, sensitivity = 80.77%, specificity = 82.19%; Figure 4(a)). The De Long test suggested that combining plasma NfL levels, PDSS scores, HAMD scores, and UPDRS-III scores significantly enhanced the AUC for predicting persistent fatigue compared to NfL alone (
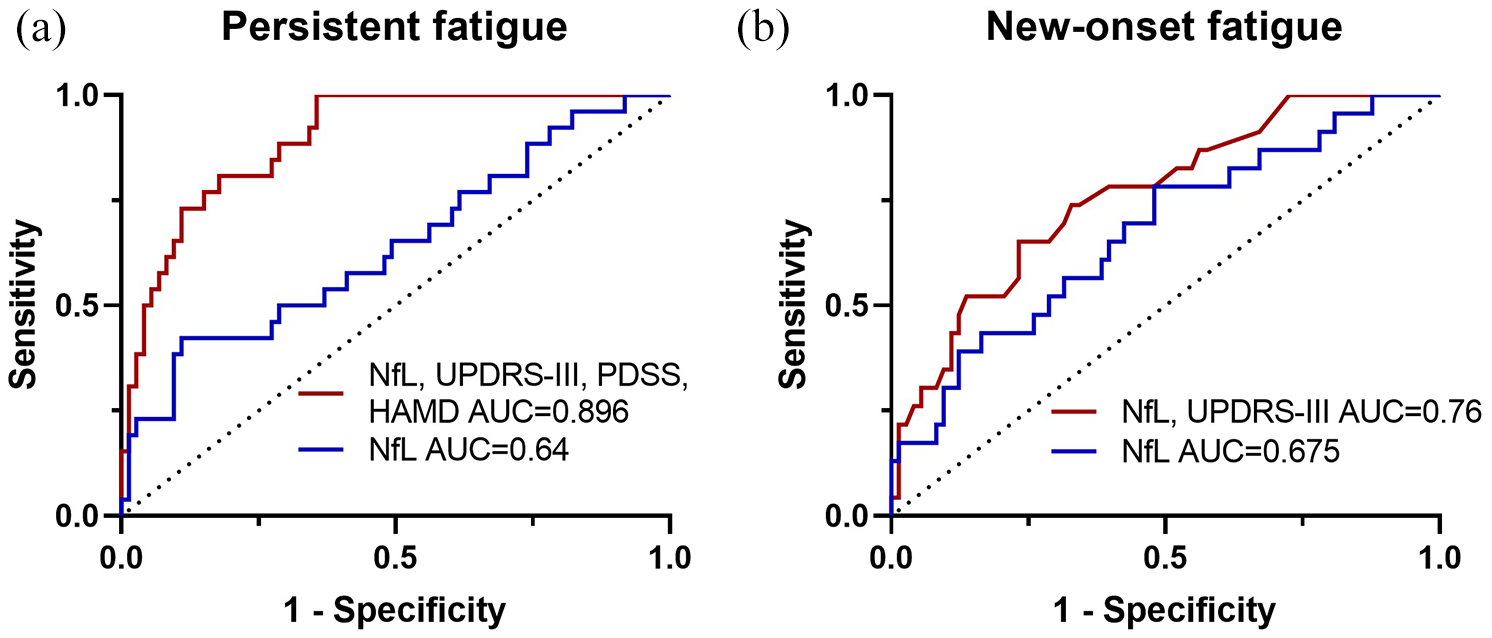
ROC analysis for prediction of fatigue conversion at the 2-year follow-up in early PD. Baseline plasma NfL levels and plasma NfL levels with clinical variables (UPDRS-III, PDSS, and HAMD scores) to predict persistent fatigue (a). Baseline plasma NfL levels and plasma NfL levels with UPDRS-III scores to predict new-onset fatigue (b).
ROC analysis indicated that baseline plasma NfL levels predicted the new-onset fatigue with an AUC of 0.675, (95% CI: 0.5480–0.8016, sensitivity = 78.26%, specificity = 52.05%). Combining the two variables most strongly associated with new-onset fatigue in multivariate logistic regression analysis (plasma NfL levels and UPDRS-III score) obtained an AUC of 0.760 (95% CI: 0.6505–0.8694, sensitivity = 65.22%, specificity = 76.71%; Figure 4(b)). Although, the De Long test revealed no statistically significant difference between the combination of NfL and UPDRS-III and NfL alone in predicting new-onset fatigue (
Associations between plasma NfL levels and FSS score
In all PD patients, plasma NfL levels were significantly associated with FSS scores at baseline (

The associations between plasma biomarkers and the FSS scores.
Discussion
In our prospective cohort study, we observed higher plasma NfL levels in patients with fatigue compared to those without fatigue at the 2-year follow-up. Our results highlighted that higher plasma NfL levels at baseline in patients without fatigue can predict the conversion to fatigue, and higher plasma NfL levels at baseline in patients with fatigue can predict persistent fatigue within a relatively short follow-up period of 2 years. However, our investigations revealed no significant associations between plasma levels of GFAP, p-tau181, Aβ42, and Aβ40 with fatigue, fatigue conversion, or progression in early-stage PD patients.
NfL mainly exists in the axons of large myelinated nerve fibers and plays a crucial role in maintaining axonal structural integrity and conduction function.27,28 When axons are damaged, NfL is released into CSF and blood. 29 Elevated plasma NfL levels have been observed in various neurodegenerative diseases, including Alzheimer’s disease, 30 amyotrophic lateral sclerosis, 30 PD,31,32 and Multiple System Atrophy. 33 Recent studies have highlighted that plasma NfL levels were associated with cognitive impairment and disease progression in PD.31,34 Consistent with prior research, 30 our results confirmed elevated plasma NfL levels in PD patients compared to HCs.
The pathophysiology of fatigue in PD remains elusive, although emerging evidence suggests abnormal functional activities in the key brain region, including frontal gyrus, insula, cingulate cortex, supplementary motor area, and postcentral gyrus are involved in its neural mechanism.35–37 Our previous study has indicated that a widespread functional deficiency within the striatal-cerebellar-cerebral cortical network is associated with the pathology of fatigue in PD. 38 A recent study revealed that elevated NfL immunoreactivity was observed in the parahippocampal and entorhinal cortex regions in Parkinson’s disease dementia/Dementia with Lewy bodies (PDD/DLB). 39 Additionally, NfL-positive neurons exhibited degenerative morphological characteristics and axonal fragmentation. 39 Furthermore, several investigations highlighted that plasma NfL levels were related to cortical neurodegeneration in PD.40,41 Therefore, we sought to explore the relationship between plasma NfL levels and fatigue in PD. Our results indicate that PD patients with fatigue had higher plasma NfL levels compared to those without fatigue at the 2-year follow-up assessment, though no differences were observed at baseline. The absence of difference in plasma NfL levels between non-fatigue and fatigue at baseline may be due to the presence of future converters within each subgroup. Another contributing factor is that the patients in our study were early-stage PD patients, with a median disease duration of 1.88 (1.79) years. Furthermore, univariate logistic analysis revealed that fatigue is associated with age, LEDD, MoCA, HAMD, HAMA, PDSS, RBD, and UPDRS-III scores, all of which may also influence the relationship between plasma NfL and fatigue. Therefore, it is essential to further explore the relationship between plasma NfL and fatigue in patients with longer disease durations and extended follow-up periods.
Fatigue in PD is often non-persistent and may shift as the disease progresses.20,42 Ahn et al. 42 demonstrated that orthostatic hypotension can serve as a predictor of fatigue in drug-naïve PD patients. Additionally, a study suggested that higher baseline levels of fatigue, EDS, and emotional apathy could forecast fatigue severity at the 1-year follow-up. 43 However, there remains a scarcity of research on biomarkers that predict the conversion of fatigue. Our study revealed that higher plasma NfL levels in PD patients without fatigue at baseline could predict fatigue convention at the 2-year follow-up. Conversely, no such predictive association was observed for GFAP, p-tau181, Aβ42, and Aβ40 levels. GFAP is considered a marker of astroglia activation. Several studies indicate that astroglia activation contributes to the development of PD, and plasma GFAP was related to cognitive and motor symptoms.16,44 Plasma Aβ42, Aβ40, and p-tau181 may be involved in the process of cognitive in PD. 45 NfL is regarded as a marker of neuroaxonal injury, and plasma NfL levels may serve as an accessible marker of neurodegeneration extent. 46 Our findings suggest that baseline plasma NfL levels could serve as a reliable biomarker for predicting fatigue conversion. However, the pathophysiological mechanism of NfL in PD fatigue still needs further investigation. We speculate that axonal damage in the cerebral cortex may be implicated in the pathophysiological of fatigue in PD.
Although fatigue and non-fatigue can transition between each other, our study revealed that 41.95% of PD patients remained never fatigue, while 14.94% were persistent fatigue over the 2-year follow-up period. Furthermore, we investigated the factors influencing the maintenance of never fatigue and the persistence of fatigue in PD. Our findings suggested that plasma NfL levels, depression, motor symptoms, and sleep disturbances at baseline were significantly associated with persistent fatigue. Additionally, the persistent fatigue group exhibited a higher rate of change in NfL levels per year compared to the never fatigue group in our study. Zhou et al. 47 suggested that the persistent fatigue group had a faster progression in total UPDRS, UPDRS-III, and PDQ-39 scores compared to the never fatigue group. Studies have shown that high plasma NfL levels were associated with faster disease progression. 27 Moreover, our results revealed that plasma NfL levels were positively associated with FSS scores in PD at baseline and 2-year follow-up. Therefore, we speculate that patients with persistent fatigue may experience more rapid motor and non-motor progression.
Our results showed the dynamic changes and relatively low levels of NfL in patients with not persistent fagtigue, compared to the persistent fatigue group, at baseline, 1-year, and 2-year follow-ups. This indicated that the levels of NfL have highly consistency with the existence and onset of fatigue. However, the univariate logistic analysis also revealed that the fatigue can be associated with age, LEDD, MoCA, HAMD, HAMA, PDSS, RBD, and UPDRS-III scores, which indicated that the conversion of fatigue in PD is complex and might need further exploration in the future.
We acknowledge the limitations of our study. First, our study was a single-center design, and the sample size was still relatively small. Therefore, larger multicenter studies are warranted in the future. Additionally, the follow-up period was only 2 years, which made it insufficient to draw the long-term progression of different fatigue subtypes in PD. Furthermore, approximately half of the PD patients in our study were receiving medication at baseline, which precluded the analysis of the progression of “pure fatigue” in PD. This study mainly focused on PD-related fatigue, as well as the conversion and progression of fatigue in PD. Therefore, the fatigue levels were not presented in the HC subjects.
Conclusion
In conclusion, PD patients with persistent fatigue and new-onset fatigue have higher plasma NfL levels at baseline than the never-fatigue subtype. Moreover, the combination of plasma NfL levels with clinical characteristics demonstrates excellent predictive utility in forecasting persistent fatigue and new-onset from the never fatigue subtype. Plasma NfL may serve as a potential biomarker for distinguishing patients with fatigue and predicting fatigue conversion in PD.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251324406 – Supplemental material for The potential use of plasma NfL as a diagnostic and prognostic biomarker of fatigue in early Parkinson’s disease
Supplemental material, sj-docx-1-tan-10.1177_17562864251324406 for The potential use of plasma NfL as a diagnostic and prognostic biomarker of fatigue in early Parkinson’s disease by Ningning Che, Jingxuan Huang, Shichan Wang, Qirui Jiang, Tianmi Yang, Yi Xiao, Junyu Lin, Jiajia Fu, Ruwei Ou, Chunyu Li, Xueping Chen and Huifang Shang in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864251324406 – Supplemental material for The potential use of plasma NfL as a diagnostic and prognostic biomarker of fatigue in early Parkinson’s disease
Supplemental material, sj-docx-2-tan-10.1177_17562864251324406 for The potential use of plasma NfL as a diagnostic and prognostic biomarker of fatigue in early Parkinson’s disease by Ningning Che, Jingxuan Huang, Shichan Wang, Qirui Jiang, Tianmi Yang, Yi Xiao, Junyu Lin, Jiajia Fu, Ruwei Ou, Chunyu Li, Xueping Chen and Huifang Shang in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
