Abstract
Background:
Efgartigimod is an approved biologic for generalized myasthenia gravis (gMG), which is an autoimmune disease and can potentially be life-threatening. However, the therapeutic response to efgartigimod among the acetylcholine receptor gMG (AChR-gMG) subtypes remains inconclusive.
Objective:
To explore the patterns and predictors for the therapeutic response to efgartigimod among AChR-gMG subtypes.
Design:
This prospective, observational study included AChR-gMG patients treated with efgartigimod at 15 centers in China with a follow-up for at least 20 weeks.
Methods:
The primary outcome was the proportion of minimal symptom expression (MSE) responders, denoted by a Myasthenia Gravis Activities of Daily Living (MG-ADL) score of 0 or 1 within 4 weeks and maintained for ⩾4 weeks. AChR antibody-positive MG (AChR-MG) subtypes were classified into early onset myasthenia gravis (EOMG), late-onset myasthenia gravis (LOMG), and thymoma-associated myasthenia gravis (TAMG). The predictive factors for MSE responders were identified by univariate and multivariate logistic regression analysis.
Results:
One hundred sixteen patients were included with a median follow-up duration of 238 days (172.5–306.3). There were 50 (43.1%) patients with EOMG, 28 (24.1%) with LOMG and 38 (32.8%) with TAMG. After efgartigimod initiation, 35 (30.2%) patients were MSE responders, and the proportion of MSE responders was highest in the LOMG group (42.9%). The MG-ADL score reduction in the LOMG group was more significant than in the EOMG group by weeks 16 and 20 (both p = 0.022). Response patterns to efgartigimod among the AChR-MG subtypes differed as measured by the proportion of improved patients and MSE. LOMG presented sustained symptom control, while EOMG and TAMG showed more fluctuations. Eight TAMG patients (21.1%) switched to another biologic (p = 0.005). Baseline MG-ADL was an independent predictor for therapeutic response to efgartigimod (p < 0.001).
Conclusion:
Our findings revealed patterns of treatment responses among AChR-gMG subtypes, with LOMG patients potentially presenting a more sustained response. These findings likely provide preliminary data for precision therapy in MG in the era of biologics.
Trial registration:
NCT04535843.
Plain language summary
Myasthenia Gravis (MG) is an autoimmune disorder that can lead to potentially life-threatening complications. Efgartigimod is an approved biologic for generalized myasthenia gravis (gMG). However, the patterns and predictors of therapeutic response to efgartigimod among the acetylcholine receptor-generalized MG (AChR-gMG) subtypes remain inconclusive. Our findings revealed patterns of treatment responses among AChR-gMG subtypes, with LOMG patients potentially presenting a more sustained response.
Introduction
Myasthenia gravis (MG) is an autoimmune disorder that affects the neuromuscular junction, leading to fatigable muscle weakness. 1 Depending on the muscle groups involved, MG can be classified as either ocular myasthenia gravis (OMG) or generalized myasthenia gravis (gMG). 2 Approximately 85% of patients with gMG patients have autoantibodies against acetylcholine receptor (AChR).3,4 Within AChR antibody-positive gMG (AChR-gMG), further clinical subtypes are defined based on the age of onset and thymoma presence, including early onset myasthenia gravis (EOMG), late-onset myasthenia gravis (LOMG), and thymoma-associated myasthenia gravis (TAMG). 2 Currently, recommended treatments for gMG include symptomatic treatment, rescue therapies with rapid action, and long-term immunotherapies. 5 However, 10%–20% of patients with MG still respond poorly to or cannot tolerate conventional treatments. 6 TAMG has a higher proportion of generalized conversion and refractory cases than non-TAMG, 7 yet it is rarely included in randomized controlled trials.
Efgartigimod, a neonatal Fc receptor (FcRn) antagonist, binds to FcRn with very high affinity, enhancing the catabolism of all immunoglobulin G subclasses and selectively reducing serum IgG levels.8,9 The ADAPT study demonstrated the efficacy and safety of efgartigimod in treating MG, as evidenced by 68% of AChR-gMG patients becoming Myasthenia Gravis Activities of Daily Living (MG-ADL) responders during the first treatment cycle; this was increased to 78% after two cycles. 10 The efficacy was further confirmed in the open-label ADAPT+ study, where a clinically meaningful improvement (CMI) in MG-ADL and Quantitative Myasthenia Gravis (QMG) scores was observed within 1 week after the first infusion. 11 Recently, efgartigimod has been approved in multiple countries and is recommended in guidelines or consensus statements.12–14
The evidence from real-world cohort studies supported the efficacy of efgartigimod in improving clinical outcomes in gMG patients.15–21 Despite these advances, the therapeutic response from different AChR-positive gMG subtypes to efgartigimod remains largely unknown. Case reports have documented both improvement and worsening of symptoms in TAMG following efgartigimod treatment.22,23 Its efficacy in TAMG remained inclusive and required more case studies. This leaves several critical questions unanswered in real-world clinical practice: (1) Is there any difference in therapeutic response to efgartigimod in AChR antibody-positive MG (AChR-MG) subtypes; (2) Which subtype of gMG would attain sustained symptom control after repeated cycles of treatment; and (3)What factors can predict the efficacy of efgartigimod in AChR-MG.
Therefore, we conducted an observational multicenter cohort study of AChR-MG patients treated with efgartigimod. We aimed to assess the patterns of therapeutic response to efgartigimod among different clinical subtypes and explore the factors that could influence drug efficacy.
Methods
Study cohort and patient selection
This multicenter, prospective, and observational cohort study included gMG patients from 15 independent neuromuscular reference centers. We recruited AChR antibody-positive gMG patients who received at least one cycle of efgartigimod from September 2023 to August 2024. The enrollment criteria were:
Symptoms and signs consistent with gMG;
Seropositive for anti-AChR antibodies by ELISA (Euroimmun, Lübeck, Germany);
Treatment with at least one cycle of efgartigimod;
Prospective follow-up for at least 20 weeks since the initiation of efgartigimod.
The following exclusion criteria were applied:
Muscle-specific kinase (MuSK) antibody-positive or double-seronegative;
Repetitive nerve stimuli suggestive of other neuromuscular junction disorders, for example, congenital MG syndrome or Lambert–Eaton syndrome;
Pregnant or planning to become pregnant during the follow-up period.
In addition, their follow-up data was not analyzed if a patient switched to another biologic such as C5 inhibitors or anti-CD20 monoclonal antibodies. However, the baseline data and data prior to switching will be maintained in the analysis.
Definitions
TAMG was defined as gMG with thymoma or a history of thymoma. For the remaining non-TAMG patients, EOMG, and LOMG were stratified by age of onset of 50 years. MG acute exacerbation was defined as worsening symptoms that require monitoring or treatment. 24 MC referred to a rapid decline in respiratory function, requiring noninvasive ventilation or intubation with mechanical ventilation. Refractory gMG was defined following German guidelines. 12 Long-term sustained intravenous immunoglobulin (IVIg) or plasma exchange (PE) was characterized by receiving repeated IVIg and/or PE over the past 3 months or for >4 times in a year. The average cycle was defined as the total number of efgartigimod treatment cycles divided by the total number of patients in the respective subgroup.
In our cohort, a reduction of ⩾2 points in the MG-ADL score from baseline was defined as CMI, while an MG-ADL score of 0 or 1 was defined as minimal symptom expression (MSE). 25 CMI or MSE responders are those who achieved CMI or MSE within 4 weeks after efgartigimod initiation and which was maintained for >4 weeks. The “Improved status” in the Myasthenia Gravis Post Intervention Status (MG-PIS) was defined as an improvement of at least three points from baseline in the MG-ADL score.
Comorbidities were assessed at baseline and during the treatment follow-up period. The definition of comorbidities followed criteria established by an Italian research team. 26 Infections at all sites were defined as infections and were not categorized as diseases of the respective sites. As this study primarily compares treatment responses across different gMG subtypes, thymoma, and thymic hyperplasia are not classified as comorbidities.
Efgartigimod administration and prospective follow-up
Efgartigimod was administered intravenously at a dose of 10 mg/kg by a 1-h infusion over a cycle of four weekly infusions. During the following flexible cyclical period, the drug was re-administered as four weekly infusions at the discretion of the treating physician if there was an acute worsening of MG-ADL.
Following efgartigimod initiation, therapeutic responses based on MG-ADL scores were prospectively collected for at least 20 weeks. Changes in prednisone dose, rescue therapies, and MG-PIS were documented every 4 weeks following treatment initiation. In our cohort, the steroid dose was generally reduced by 5–10 mg/month if patients showed stable improvement. The incidence of treatment-emergent adverse events (TEAEs) was documented throughout. The final analysis of MG-ADL, steroid dose, and MG-PIS was based on the 20-week follow-up data after treatment. We retrospectively analyzed baseline parameters, namely sex, age, age of onset, disease duration, serum antibody type, and gMG subtypes (i.e., TAMG, EOMG, and LOMG). We also considered thymectomy status; thymus condition (thymoma, thymic hyperplasia, and normal); prior treatments for MG; prednisone dose; MG-ADL score; Myasthenia Gravis Foundation of America (MGFA) classification; indications for efgartigimod as an add-on therapy; and proportion of improved patients (MG-PIS).
The reporting of this study conforms to STROBE guidelines. 27 All clinical data were collected after obtaining patients’ written consent. The study protocol was approved by the institutional review boards of the ethics committees at each participating institution (Fudan University, Huashan Hospital, 2023-1100) and adhered to the tenets of the Helsinki Declaration.
Study outcomes
The primary outcome was the proportion of MSE responders. The secondary outcomes were the incidence of TEAEs, the proportion of patients switching to other biologics, MG-PIS proportion, MSE, CMI responders, and frequency of rescue therapies. Given the variability in baseline MG-ADL scores among MG subtypes, “decrease in MG-ADL” (reduction in MG-ADL/baseline MG-ADL) was used to evaluate therapeutic responses instead of absolute values. “Patterns of therapeutic response” were defined as MG-PIS or MSE at 4, 8, 12, 16, and 20 weeks after efgartigimod treatment among the AChR-MG subtypes.
Statistical analysis
Continuous variables were presented as the mean ± SD or median (Q1–Q3), while categorical variables were expressed as n (%). Statistical analyses for different gMG subtypes involved both pairwise and multiple-group comparisons. For pairwise comparisons, continuous variables were analyzed using the Mann–Whitney test or t-test, depending on the data distribution. For multiple group comparisons, continuous variables were assessed using a one-way analysis of variance or the Kruskal–Wallis test. Categorical variables were compared using the chi-square test or Fisher’s exact test. p ⩽ 0.05 was considered to indicate statistically significant differences and confidence intervals were reported at the 95% confidence level. Predictive factors for this outcome were evaluated using univariate logistic regression models to determine odds ratios and p-values. Variables with p < 0.1 were included in a multivariate logistic regression model.
All statistical analyses were performed using R version 4.4.0. Figures were generated with R version 4.4.0 and GraphPad Prism version 9.5.1 (GraphPad Software Inc., San Diego, CA, USA).
Results
Baseline clinical features
Between September 2023 and August 2024, 4875 patients with gMG were treated across 15 research centers in China (Figure 1). Of these, 195 patients received efgartigimod. Among them, 25 patients with MuSK-MG (17.1%) and five with seronegative gMG (3.4%) were excluded. Among the remaining 165 AChR-MG patients, we included 116 AChR-MG patients who had completed at least one cycle of efgartigimod infusions. The cohort comprised 50 patients with EOMG (43.1%), 28 patients with LOMG (24.1%), and 38 patients with TAMG (32.8%). There were no significant differences among subtypes regarding the initiation status and MGFA classifications. Detailed clinical characteristics are presented in Table 1.
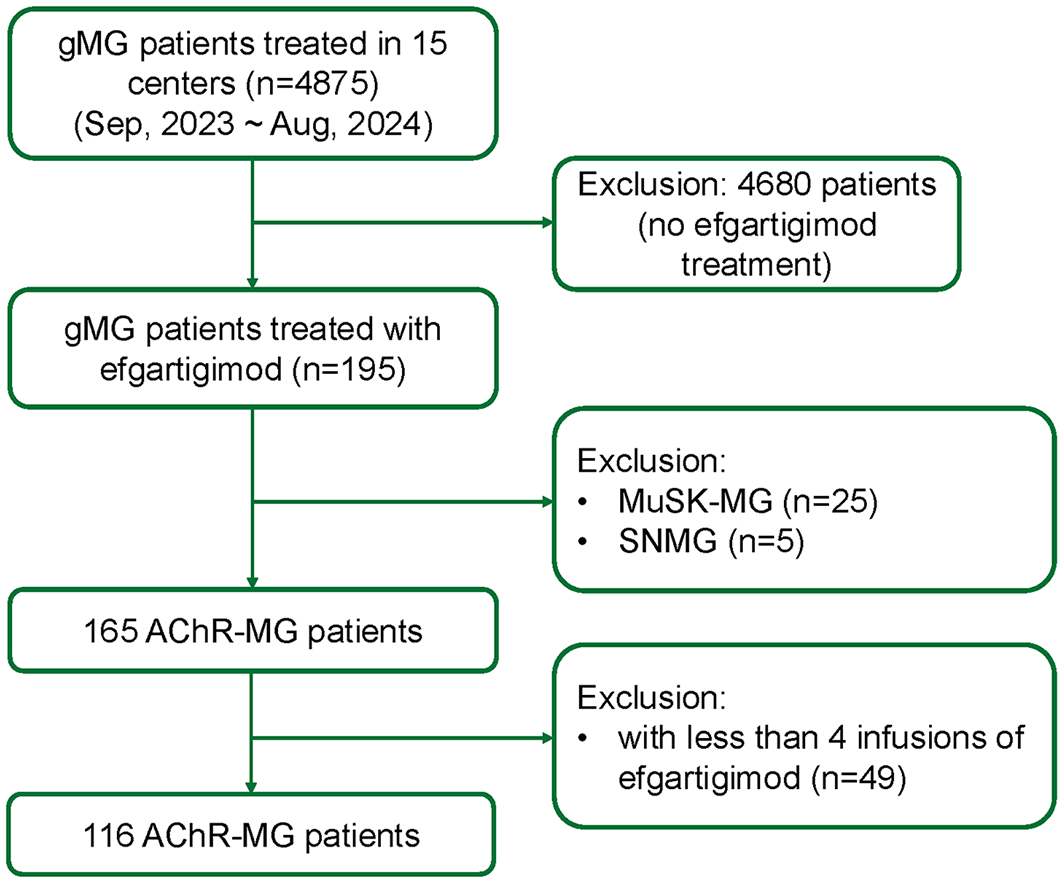
Study flowchart.
Baseline clinical features for gMG subtypes who were treated with efgartigimod (n = 116).

Baseline refers to the first infusion of efgartigimod. Disease duration was defined as the time between onset of symptom and the baseline entry. The bold values represent p-values with statistical significance.
EOMG, early-onset myasthenia gravis; gMG, generalized myasthenia gravis; LOMG, late-onset myasthenia gravis; MC, myasthenic crisis; MGAE, MG acute exacerbation; MGFA, Myasthenia Gravis Foundation of America; MGIVIg, intravenous immunoglobulin; PE, plasma exchange; TAMG, thymoma-associated myasthenia gravis.
In the cohort (n = 116), the male-to-female ratio was 28:88 with female predominance (75.8%). The median age at recruitment was 51 years (36.8–65.3), with an onset age of 41.5 years (29–60). The disease duration for the entire cohort was 4 years (1, 10). The disease duration was significantly longer in the EOMG group (6.5 years (3–10)) than in the LOMG group (2 years (0.5–5.8), p = 0.003) and the TAMG group (2.5 years (0.9–5.8), p = 0.003), respectively. Thymectomy was performed in 51 patients (44.0%), comprising 16 patients with thymic hyperplasia (31.4%), and 35 with thymoma (68.6%). Notably, one patient with LOMG had thymic hyperplasia, as indicated by the pathological studies. The WHO pathological classification of TAMG patients is shown in Table S1. The most common pathological types were B2 (9/38, 23.7%) and B3 (9/38, 23.7%).
Comorbidities were present in 66.4% of patients, with hypertension being the most prevalent (Table S2). The LOMG group had the highest proportion of comorbidities, significantly higher than that in the EOMG group (85.7% vs 54.0%, p = 0.010). The prevalence of osteoporosis was higher in the LOMG group compared to both the EOMG and TAMG groups (p = 0.041 and p = 0.004, respectively). Compared with the EOMG group, the LOMG group had a higher proportion of diabetes (p = 0.041).
Overall, 42.2% of patients were classified as refractory, with higher proportions in the EOMG (44.0%) and TAMG (52.6%) subgroups. The proportion of refractory MG was significantly higher in the TAMG group than in the LOMG group (52.6% vs 25.0%, p = 0.045). Before efgartigimod treatment, prednisone was the most used immunotherapy (87.9%), followed by tacrolimus (48.3%) and mycophenolate mofetil (22.4%). Anti-CD20 monoclonal antibodies were administered in 15 patients (12.9%), most frequently in EOMG, and were significantly more prevalent than in LOMG (p = 0.006). In addition, 12.1% of patients required long-term IVIg or PE. Upon efgartigimod initiation, all patients had an MGFA classification of II–V, with the majority classified as II/III (73.3%). The baseline dose of prednisone was significantly higher in the TAMG group than in the EOMG group (28.7 ± 18.6 vs 17.9 ± 16.6, P = 0.003).
Patterns of therapeutic response in AChR-gMG subtypes
In our cohort, 12 (10.3%) patients were lost during the follow-up and did not complete the 20-week observation period, including 10 patients with EOMG and two with LOMG. The baseline median score of MG-ADL was 8 (6–12), with no significant differences among the EOMG, LOMG, and TAMG subgroups (p = 0.232), as detailed in Table 2.
The efficacy of efgartigimod in different gMG subtypes (n = 116).

A decrease in MG-ADL was defined as MG-ADL reduction/baseline MG-ADL. CMI refers to a reduction of at least a 2-point from the baseline MG-ADL score. MSE was defined as an MG-ADL score of 0 or 1. CMI responders were those who achieved CMI within 4 weeks and maintained it for more than 4 weeks. MSE responders were denoted as achieving MSE within 4 weeks after efgartigimod initiation with sustained improvement for more than 4 weeks. The bold values represent p-values with statistical significance.
CMI, clinically meaningful improvement; EOMG, early onset myasthenia gravis; gMG, generalized myasthenia gravis; LOMG, late-onset myasthenia gravis; MG-ADL, Myasthenia Gravis Activities of Daily Living; MSE, minimal symptom expression; TAMG, thymoma-associated myasthenia gravis.
After efgartigimod initiation, participants achieved CMI with an average duration of 1.3 ± 0.9 weeks and MSE within 2.9 ± 1.7 weeks. Furthermore, 101 (87.1%) patients were CMI responders, while 35 patients (30.2%) were MSE responders. Among the gMG subtypes, the proportion of MSE responders was highest in the LOMG group (42.9%), while this was relatively low in the TAMG group (21.1%). After the first cycle of efgartigimod, the MG-ADL score decreased to 2 (1–4), representing a reduction of −71.4% (−90%, −50%) from the baseline. The MG-ADL scores for the EOMG, LOMG, and TAMG groups were reduced to 2 (1–4), 2 (0–5), and 3 (1–4), respectively. By week 16 and 20, the LOMG group showed a greater reduction in MG-ADL than the EOMG group (−72.7% [−91.7, −60.0] vs −66.7% [−87.0, −50.0], p = 0.022; −83.8% [−100, −67.3] vs −66.7% [−83.3, −50.0], p = 0.022).
During the follow-up period of 238 days (172.5–306.3), 70 (60.3%) patients received more than two cycles, and 30 (25.9%) patients were more than three cycles. In addition, 12 patients received four or more treatment cycles, of which six patients did not proceed to the fifth cycle. The average intervals from the first to the fourth cycle were 96.9 ± 52.4, 76.7 ± 52.1, and 50.1 ± 38.2 days, respectively. The majority of TAMG patients (84.2%) discontinued subsequent cycles after receiving two cycles of efgartigimod. Among patients receiving more than two cycles, LOMG patients had a lower discontinuation rate compared to EOMG patients (Figure 2).
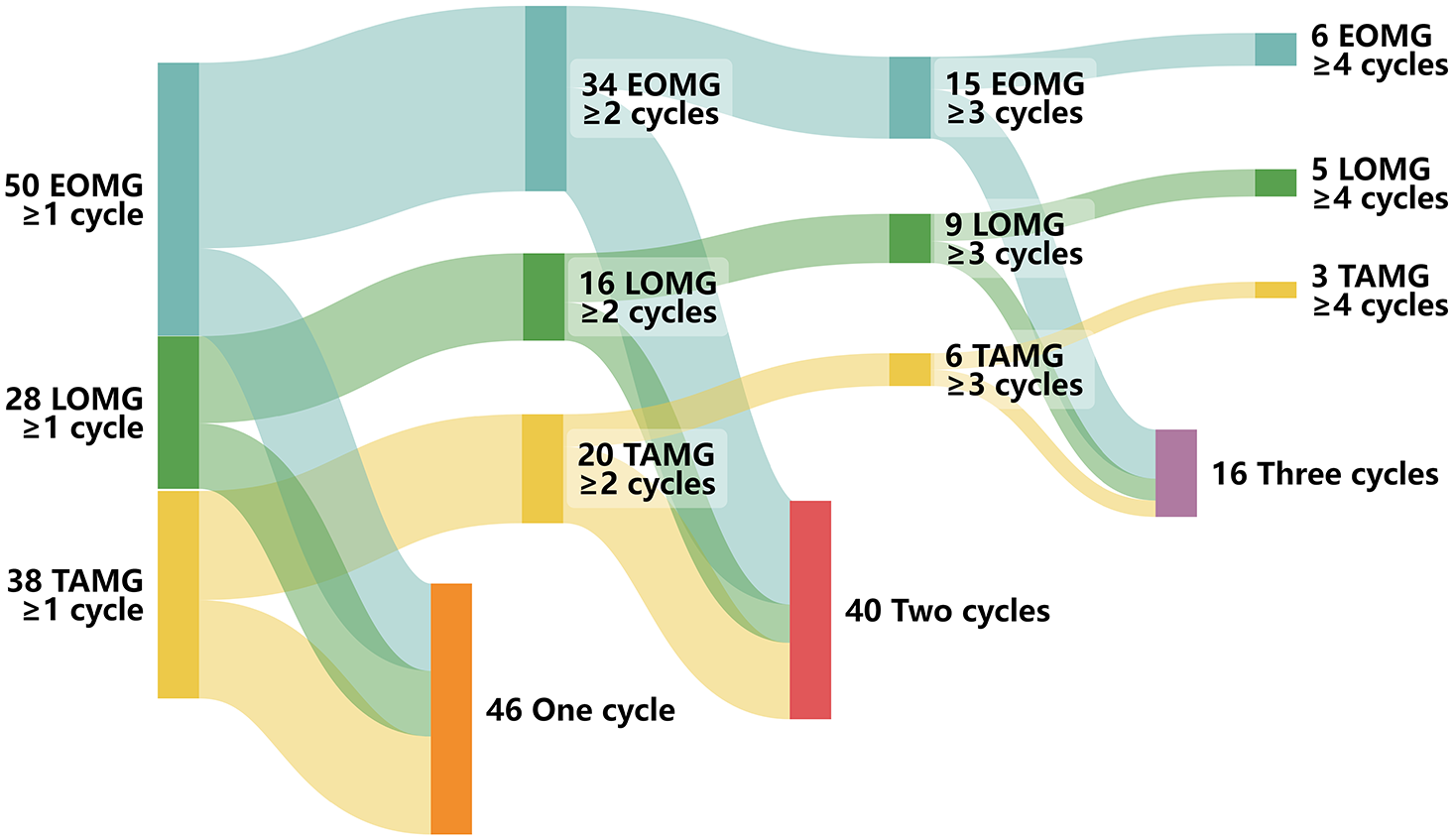
Treatment cycles in different gMG subtypes.
The patterns of therapeutic response as denoted by Improved status in MG-PIS and MSE were different among the gMG subtypes (Figure 3(a)). For the EOMG subgroup, the proportions of improved patients fluctuated from 92.0% at week 4 to 80% at week 8 and week 12. In contrast, the proportion of Improved status in LOMG increased gradually from 82.1% at week 4 to 96.0% at week 20. This further supports the sustainable therapeutic response in the LOMG group to efgartigimod. The TAMG group showed a relatively stable proportion of Improved status (80%–87%). Next, the proportion of patients achieving MSE further supported the most favorable response in the LOMG group. In comparison, the EOMG and TAMG groups showed an obvious decline at week 8 (Figure 3(b)).

The patterns of therapeutic response among AChR-MG subtypes. The trajectories of improvement status and MSE varied among the three subtypes (a, b). Time to treatment switch analysis presented significant differences (p = 0.005) (c). No statistically significant differences were observed in the aspect of rescue therapies (d). Week 4, n = 116; week 8, n = 113 (1 LOMG and 2 TAMG excluded); week 12, n = 111 (2 LOMG and 3 TAMG excluded); week 16, n = 107 (1 EOMG, 3 LOMG, and 5 TAMG excluded); week 20, n = 96 (10 EOMG, 3 LOMG, and 7 TAMG excluded); week 24, n = 85 (18 EOMG, 4 LOMG, 9 TAMG); week 26, n = 77 (21 EOMG, 8 LOMG, 10 TAMG).
Among the five TAMG patients who were perioperative, four started efgartigimod before surgery, with three of them experiencing acute exacerbations. These patients demonstrated significant reductions in their MG-ADL scores. None of them experienced TEAEs. Except for Patient 1, the others received a second cycle of treatment.
Ten patients had switched to another biological treatment (five eculizumab and five rituximab). The specific reasons are provided in Table S3. Among them, two patients switched to eculizumab after 20 weeks on efgartigimod. Next, we analyzed the time to treatment switches and rescue therapies among these subgroups. During the follow-up period, eight patients discontinued efgartigimod treatment in the TAMG subgroup, which is significantly higher than that in the EOMG subgroup (n = 1) and LOMG subgroup (n = 1; p = 0.005, Figure 3(c)). Patients who required rescue therapies were not different among subgroups including two EOMG, three LOMG, and three TAMG patients (p = 0.466, Figure 3(d)), and three TAMG of them switched to other biologic agents (two eculizumab and one rituximab; Table S4).
The average baseline steroid dose was 22.0 ± 18.9 mg. By week 20, it decreased to 12.9 ± 12.4 mg. The average steroid dose was higher in the TAMG group than in the EOMG and LOMG groups (28.7 ± 18.6 mg vs 17.9 ± 16.6 mg, p = 0.003; 28.7 ± 18.6 mg vs 20.2 ± 21.4 mg, p = 0.053; Table 3). In the follow-up period, the dose gradually decreased in three subtypes. By week 20, the steroid dose was reduced to 11.6 ± 9.2 mg in the EOMG group, 10.9 ± 16.0 mg in the LOMG group, and 16.3 ± 12.4 mg in the TAMG group.
The change of prednisone dosage in different gMG subtypes (n = 116).

EOMG, early-onset myasthenia gravis; gMG, generalized myasthenia gravis; LOMG, late-onset myasthenia gravis; TAMG, thymoma-associated myasthenia gravis.
The bold values represent p-values with statistical significance.
The changes in immunotherapies were prospectively collected. In addition to the initiation of efgartigimod, 16 patients (13.8%, including six EOMG, three LOMG, and seven TAMG) received new add-on immunosuppressants for further stabilizing MG and steroid reduction. These included tacrolimus (n = 9), mycophenolate mofetil (n = 5), and azathioprine (n = 2). Two patients (one EOMG and one TAMG) switched from tacrolimus to mycophenolate mofetil, while two other patients (one EOMG and one TAMG) switched from azathioprine to tacrolimus. In addition, one EOMG patient discontinued long-term sustained IVIg/PE and transitioned to tacrolimus.
Safety profile in gMG subtypes
None of the patients discontinued efgartigimod due to serious adverse events. Nine (7.8%) patients experienced TEAEs, primarily upper respiratory tract infections, rash, and muscle soreness (Table 4). The incidence of TEAEs did not differ significantly among the EOMG, LOMG, and TAMG groups (p = 0.578), with rates of 8%, 3.6%, and 10.5%, respectively. In addition, one patient in the EOMG group tested positive for COVID-19 following efgartigimod treatment. One TAMG patient died of multiorgan dysfunction on day 222 of follow-up due to metastasis.
Summary of TEAEs in all patients (n = 116).

The variable TEAEs refers to the incidence of TEAEs.
EOMG, early onset myasthenia gravis; LOMG, late-onset myasthenia gravis; TAMG, thymoma-associated myasthenia gravis; TEAEs, treatment-emergent adverse events.
Predictors for the therapeutic response to efgartigimod in gMG
To identify factors influencing the rapid and sustained response to efgartigimod, we divided the AChR-MG cohort into the responder group and the non-responder group based on the achievement of the primary outcome. Univariate logistic regression analysis identified sex and baseline MG-ADL as significant predictors (p ⩽ 0.05; Table 5). In addition, considering previous findings suggesting that LOMG patients may respond better to efgartigimod, we included LOMG (p = 0.097) in the multivariate analysis. Finally, only the baseline MG-ADL score was confirmed as an independent predictor in the multivariate analysis (p < 0.001).
Results of univariate and multivariate logistic regression analysis (n = 116).

Baseline refers to the first infusion of efgartigimod. Disease duration was defined as the time between onset of symptom and the baseline entry. Variables with p < 0.1 in univariate analysis were included in multivariate analysis. The bold values represent p-values with statistical significance.
EOMG, early-onset myasthenia gravis; IS, immunosuppressants; LOMG, late-onset myasthenia gravis; MC, myasthenic crisis; MG-ADL, Myasthenia Gravis Activities of Daily Living; MGAE, myasthenia gravis acute exacerbation; MGFA, Myasthenia Gravis Foundation of America; OR, odds ratio; TAMG, thymoma-associated myasthenia gravis.
Discussion
Utilizing a prospective real-world cohort of AChR-MG in this study, we explored the therapeutic response to efgartigimod among different subtypes. In the whole cohort, we observed a favorable therapeutic response, with 87.1% being CMI responders and 30.2% being MSE responders. Additionally, we identified sustained improvement in MG-ADL in the LOMG subgroup, and a higher proportion in this group to maintain multicycle infusions of efgartigimod, than the EOMG and TAMG groups. A higher proportion of treatment switching from efgartigimod was more prominent in the TAMG group than in the other groups. Moreover, we identified that patients with lower baseline MG-ADL scores were more likely to be MSE responders. These patterns of therapeutic response across AChR-MG subtypes and predictors could provide more knowledge on the maintenance of FcRn antagonists.
The ADAPT and ADAPT+ trials demonstrated parallel improvements in MG-ADL and QMG scores alongside reductions in total IgG and AChR antibodies.10,11 Our real-world data supported these findings, showing a rapid decline in MG-ADL scores across three gMG subtypes. Relatively speaking, our study identified a more favorable response with CMI responders than that in the ADAPT study (87.1% vs 77.8%). 10 This might be associated with the shorter disease duration in our cohort (median 4 years vs mean 9.7 years), which indicates that earlier initiation of efgartigimod may provide better therapeutic benefits for patients.
During the follow-up period, three gMG subtypes exhibited different response patterns to efgartigimod. The EOMG patients responded well in the first cycle but tended to experience a prolonged relapse of symptoms that corresponded to a decrease in the proportion of patients achieving both Improved status and MSE. Although TAMG patients showed rapid improvement after the first efgartigimod cycle, their responses were highly variable, with the lowest proportion of MSE responders and the highest proportion of switches to other biologic agents. The highest proportion of MSE responders and longest duration of MSE were found among LOMG patients, suggesting that LOMG may be the clinical subtype most responsive to efgartigimod. Moreover, the proportion of patients with Improved status showed an overall upward trend. LOMG appeared to respond most favorably to treatment, while EOMG and TAMG showed relatively poorer responses. This aligns with an Israeli study that identified two response patterns to efgartigimod treatment. In pattern B, marked by sustained improvement, LOMG cases were predominant, whereas in pattern A, defined by fluctuating improvement, TAMG made up two-thirds of the cases. 17 Considering both EOMG and TAMG show symptom fluctuations after efgartigimod treatment, particularly with a notable decline at week 8, could different dosing regimens help prevent this? Further clinical considerations on using tailored dosage of efgartigimod, such as infusions every 2 weeks, or in combination with immunosuppressants, are expected to improve the therapeutic outcome.
The variations in treatment response may be due to the distinct characteristics of each subtype. In our cohort, refractory cases were more prevalent in EOMG and TAMG than in LOMG. Refractory may require more frequent administration of efgartigimod, 28 which could explain the observed differences in response patterns. A hallmark of EOMG is thymic follicular hyperplasia, characterized by germinal centers and an increased number of B cells and plasma cells, which correlates with intra-thymic production of heterogeneous IgG autoantibodies targeting AChR. 29 For TAMG, the thymoma disrupts the normal structure and function of the thymus, leading to the production of self-reactive T cells that escape into the periphery and activate B cells to produce AChR antibodies when stimulated. 29 Regarding the immune response in different clinical subtypes, one study found that MG patients with thymic hyperplasia took twice as long to achieve minimal manifestation status (18 months) compared to LOMG (6 months) and TAMG (8 months), with a lower maintenance rate of MM (75.0%) than LOMG (81.7%) and TAMG (79.0%). 30 Another study found that LOMG patients required lower doses of immunosuppressants than EOMG patients to achieve a similar MG-PIS status. 31 Previous research has shown that refractory MG tends to occur at a younger age, with a higher proportion of females and a greater likelihood of thymoma or MuSK antibody positivity.32,33 For gMG with poor response to conventional immunosuppressive therapies, biologics may offer a new option. The REGAIN study and its extension strongly support the use of eculizumab in treating refractory gMG.34,35 Eculizumab has a similar effect to efgartigimod in reducing MG-ADL scores but offers a stronger effect in reducing steroid burden. 36 Rituximab, an anti-CD20 monoclonal antibody that depletes B cells, has been shown in meta-analyses to be non-inferior to eculizumab in treating refractory gMG. 37
To be noticed, LOMG group has a short disease duration from onset to the efgartigimod initiation (2 years) compared with other groups, which is potentially a contributor to the good therapeutic response as well. From a subgroup analysis stratified by the disease duration in the post-hoc analysis from the ADAPT study, response to efgartigimod based on a 3-point change in MG-ADL was relatively good (51%) in participants with short disease duration (<7 years) compared to those with long disease duration (37%; ⩾7 years; Week 0 to Week 20). 38 The therapeutic response to rituximab, a previously applied biologic targeting B cells, also manifests differently among patients with long-term disease duration versus short-term duration. A longer duration of MG at rituximab induction is associated with a less favorable response.39,40 In contrast, the achievement of minimal MG manifestations was more satisfied after low-dose rituximab among gMG with a short time since the onset, as evidenced by the RINOMAX trial, a randomized, double-blind, placebo-controlled study that took place over 48 weeks to explore the safety and efficacy of rituximab in new-onset MG. 41 In Japan, the concept of early cycles of fast-acting treatment (EFT) to achieve an early improvement and good prognosis has gradually spread in the last decade. The fact that EFT was performed more frequently in patients with short disease duration reflects such a recent change in MG treatment strategy. 42 Although benefits are more and more prominent that are brought by early intervention with fast-acting therapies, more evidence is still required for implementation since long-term safety and socio-economic factors should also be taken into account.
Patients with MG usually have coexisting autoimmune diseases (ADs) with hyperthyroidism, hypothyroidism, and vitiligo as the most prevalent comorbidities. 43 We also identified that 7.8% had concurrent ADs in this multicenter cohort. The therapeutic effect of efgartigimod on these ADs remains to be explored. In addition, efgartigimod may also be a therapeutic option for other IgG-mediated neurological immune disorders, such as stiff-person syndrome and immune-mediated myositis.44–46
The safety profile of efgartigimod is reassuring, with the most common TEAEs being headache, nasopharyngitis, and COVID-19.10,11 Over a 3-year observation period, 84.8% of patients experienced TEAEs, with five reported deaths (all related to efgartigimod). 11 However, real-world studies have shown a lower incidence of TEAEs, ranging from 11.5% to 68.4%.15–20 Serious adverse events have been rare, with only two reported cases, one each of complicated diverticular disease requiring surgery and recurrent Clostridium difficile infection.15,19 A single death was reported in an Israeli cohort, 17 likely owing to smaller sample sizes and shorter follow-up periods in these studies. In our cohort, 9 (7.8%) instances of TEAEs were recorded, including one case of COVID-19, but no serious adverse events. Notably, in an Italian cohort, 19 COVID-19 infections (10/19, 52.6%) did not result in pulmonary complications or exacerbation of myasthenia, as all patients had been vaccinated with COVID-19 mRNA vaccines before the study. Similarly, in a Japanese cohort, none of the five COVID-19-related MG exacerbations required additional treatment. 18 This may be because efgartigimod does not impair vaccine-specific IgG responses, but rather temporarily reduces the titers of all specific IgGs. 47 To be noticed, the incidence of TEAEs might be under-reported from a real-world cohort. This can be attributed to two factors: prospective collections of TEAEs by physicians might not fully represent the experience of participants, and short-term follow-up might not be able to detect long-term side effects.
To better capture the improvement and duration of patient response to efgartigimod, we introduced the MSE responder as a new primary outcome. Approximately 30% of gMG patients achieved this outcome, suggesting that efgartigimod offers a prolonged benefit to work and life quality for these patients. However, while some patients maintained MSE for about 7 weeks, the considerable internal heterogeneity highlights the need for flexible, individualized dosing. Coincidentally, the 64-week post-hoc analysis of the ADAPT study also used the time in response for MG-ADL and QMG to assess the sustained benefits of efgartigimod. 38 Although the proportions of these (59% and 44%) were higher than the MSE responder rate in our cohort, 38 this could be attributed to the longer duration of efgartigimod use in their study and the stricter endpoints we employed.
In the routine strategy for treating MG in China, if the patient has significantly improved symptoms like CMI achievement (ADL score change >2), the steroid dose is usually suggested to be reduced 5 mg each month. However, pure antibody reduction or clearance can not guarantee the therapeutic effect for treating MG if the inhibition of lymphocytes is insufficient. This is particularly important to reduce the risk of exacerbation by slow tapering of corticosteroids and IS, even if the patients are on efgartigimod and other FcRn antagonists.
The limitations of our study include: (1) Although the QMG score is a crucial component in predictive models for gMG prognosis, 48 it was not collected in our cohort. Instead, we used the MG-ADL score, which correlates well with the QMG score, as a substitute. 49 A relatively short follow-up period and lack of QMG score collection, which may have led to insufficient observation of long-term treatment outcomes and potential bias in patient prognosis; (2) The inability to entirely rule out placebo effects or the prolonged influence of prior treatment regimens; and (3) The fact that our model was found exclusively using data from Chinese gMG patients, which may limit its predictive accuracy for patients in other regions. Traditional risk factors, such as disease duration, thymoma pathology types, refractory MG, and previous acute exacerbations or MC, seem more likely to significantly influence the prognosis. Thus, it should be cautious not to use baseline MG-ADL score as the only parameter in clinical practice. Noticeably, each group is not homogeneous and the sample size for each subgroup is limited. Therefore, further validation of the safety and efficacy of efgartigimod in AChR antibody-associated gMG in larger, international multicenter cohorts is necessary.
Conclusion
This study represents a large real-world cohort to explore the efficacy of efgartigimod among different AChR-MG subtypes. After efgartigimod initiation, 101 patients were CMI responders (87.1%) and 35 patients were MSE responders (30.2%). Patients with LOMG presented sustained disease control after multicycle infusions. The baseline MG-ADL score predicts early therapeutic response to efgartigimod treatment. These findings likely provide preliminary data for precision therapy in MG in the era of biologics.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251319656 – Supplemental material for Patterns and predictors of therapeutic response to efgartigimod in acetylcholine receptor-antibody generalized myasthenia gravis subtypes
Supplemental material, sj-docx-1-tan-10.1177_17562864251319656 for Patterns and predictors of therapeutic response to efgartigimod in acetylcholine receptor-antibody generalized myasthenia gravis subtypes by Lei Jin, Zhangyu Zou, Qinzhou Wang, Wenshuang Zeng, Qilong Jiang, Jing Chen, Jianquan Shi, Yanyan Yu, Daojun Hong, Quantao Zeng, Song Tan, Yaoxian Yue, Zhouao Zhang, Yong Zhang, Xiuming Guo, Lei Du, Zhongyan Zhao, Shixiong Huang, Ying Chen, Zongtai Wu, Chong Yan, Jianying Xi, Jie Song, Sushan Luo and Chongbo Zhao in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864251319656 – Supplemental material for Patterns and predictors of therapeutic response to efgartigimod in acetylcholine receptor-antibody generalized myasthenia gravis subtypes
Supplemental material, sj-docx-2-tan-10.1177_17562864251319656 for Patterns and predictors of therapeutic response to efgartigimod in acetylcholine receptor-antibody generalized myasthenia gravis subtypes by Lei Jin, Zhangyu Zou, Qinzhou Wang, Wenshuang Zeng, Qilong Jiang, Jing Chen, Jianquan Shi, Yanyan Yu, Daojun Hong, Quantao Zeng, Song Tan, Yaoxian Yue, Zhouao Zhang, Yong Zhang, Xiuming Guo, Lei Du, Zhongyan Zhao, Shixiong Huang, Ying Chen, Zongtai Wu, Chong Yan, Jianying Xi, Jie Song, Sushan Luo and Chongbo Zhao in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-3-tan-10.1177_17562864251319656 – Supplemental material for Patterns and predictors of therapeutic response to efgartigimod in acetylcholine receptor-antibody generalized myasthenia gravis subtypes
Supplemental material, sj-docx-3-tan-10.1177_17562864251319656 for Patterns and predictors of therapeutic response to efgartigimod in acetylcholine receptor-antibody generalized myasthenia gravis subtypes by Lei Jin, Zhangyu Zou, Qinzhou Wang, Wenshuang Zeng, Qilong Jiang, Jing Chen, Jianquan Shi, Yanyan Yu, Daojun Hong, Quantao Zeng, Song Tan, Yaoxian Yue, Zhouao Zhang, Yong Zhang, Xiuming Guo, Lei Du, Zhongyan Zhao, Shixiong Huang, Ying Chen, Zongtai Wu, Chong Yan, Jianying Xi, Jie Song, Sushan Luo and Chongbo Zhao in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-4-tan-10.1177_17562864251319656 – Supplemental material for Patterns and predictors of therapeutic response to efgartigimod in acetylcholine receptor-antibody generalized myasthenia gravis subtypes
Supplemental material, sj-docx-4-tan-10.1177_17562864251319656 for Patterns and predictors of therapeutic response to efgartigimod in acetylcholine receptor-antibody generalized myasthenia gravis subtypes by Lei Jin, Zhangyu Zou, Qinzhou Wang, Wenshuang Zeng, Qilong Jiang, Jing Chen, Jianquan Shi, Yanyan Yu, Daojun Hong, Quantao Zeng, Song Tan, Yaoxian Yue, Zhouao Zhang, Yong Zhang, Xiuming Guo, Lei Du, Zhongyan Zhao, Shixiong Huang, Ying Chen, Zongtai Wu, Chong Yan, Jianying Xi, Jie Song, Sushan Luo and Chongbo Zhao in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-6-tan-10.1177_17562864251319656 – Supplemental material for Patterns and predictors of therapeutic response to efgartigimod in acetylcholine receptor-antibody generalized myasthenia gravis subtypes
Supplemental material, sj-docx-6-tan-10.1177_17562864251319656 for Patterns and predictors of therapeutic response to efgartigimod in acetylcholine receptor-antibody generalized myasthenia gravis subtypes by Lei Jin, Zhangyu Zou, Qinzhou Wang, Wenshuang Zeng, Qilong Jiang, Jing Chen, Jianquan Shi, Yanyan Yu, Daojun Hong, Quantao Zeng, Song Tan, Yaoxian Yue, Zhouao Zhang, Yong Zhang, Xiuming Guo, Lei Du, Zhongyan Zhao, Shixiong Huang, Ying Chen, Zongtai Wu, Chong Yan, Jianying Xi, Jie Song, Sushan Luo and Chongbo Zhao in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
We thank all participants recruited in this study. We are also grateful to co-investigators in the Chinese Myasthenia Gravis Collaborating Group (CMGCG): Yuzhou Guan, Peking Union Medical College Hospital; Yuwei Da, Xuanwu Hospital Capital, Medical University; Huan Yang, Xiangya Hospital, Central South University; Ting Chang, Tangdu Hospital, The Fourth Military Medical University; Yuge Wang, The Third Affiliated Hospital of Sun Yat-sen University; Huiyu Feng, The First Affiliated Hospital of Sun Yat-sen University; Bitao Bu, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology; Hongyu Zhou, West China Hospital, Sichuan University; Chunsheng Yang, Tianjin Medical University General Hospital; Hua Zhang, Beijing Hospital; Hui Deng, First Hospital of Jilin University; and Ruisheng Duan, The First Affiliated Hospital of Shandong First Medical University and Shandong Provincial Qianfoshan Hospital.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
