Abstract
Background:
Limited information exists on the healthcare resource utilization (HCRU) associated with real-world natalizumab used as a first-line (1L) versus later-line (2L+) treatment in multiple sclerosis (MS).
Objectives:
To describe natalizumab use in newly diagnosed MS patients treated as 1L or 2L+ and evaluate unadjusted annualized relapse rates (ARR) and MS-related HCRU before and after treatment initiation.
Design:
This retrospective observational study utilized Komodo Health Sentinel claims data from October 2015 to August 2022. The study included adults diagnosed with incident MS who initiated natalizumab treatment, with insurance coverage for at least 12 months before diagnosis and 24 months after. The index date was the first natalizumab claim on or after the diagnosis. Baseline was defined as the 365 days prior to the index date, truncated at the time of diagnosis. Follow-up ended at the earliest occurrence of death, insurance disenrollment, treatment discontinuation (gap ⩾45 days), switch to another disease-modifying therapy before natalizumab discontinuation, or study end.
Methods:
Relapses and HCRU were assessed using person-time methods to account for varying follow-up times. Relapses were identified using a validated claims-based algorithm, and time to first relapse was analyzed using Kaplan–Meier methods. Hazard ratios for relapse were estimated using univariate Cox models. Mean differences (MD) in HCRU between baseline and follow-up and between 1L and 2L+ treatment groups were calculated.
Results:
A total of 1174 patients in the 1L group (mean age 39.0, 72.0% female) and 394 in the 2L+ group (mean age 39.7, 79.4% female) were included. Patients in the 1L group had a significantly higher baseline ARR (1.48 vs 0.92, p < 0.001) and lower on-treatment ARR (0.28 vs 0.41 for 2L+, p < 0.001). HCRU decreased significantly in the 1L group from baseline to follow-up: hospitalizations (MD 17.01 visits/year), length of stay (LOS; MD 20.96 days/year), emergency room visits (MD 9.83 visits/year), non-natalizumab outpatient visits (MD 12.11 visits/year) and long-term care facility stays (MD 22.18 days/year, p = 0.002). The 1L group showed greater reductions in inpatient visits (MD 10.01 visits//year), LOS (MD 16.73 days/year) and non-natalizumab outpatient visits (MD 11.64 visits/year) compared to the 2L+ group.
Conclusion:
Natalizumab as a first-line treatment was associated with greater reductions in ARR and MS-related HCRU compared to later-line use.
Introduction
Multiple sclerosis (MS) is a chronic disease characterized by immune-mediated inflammatory and neurodegenerative processes within the central nervous system, predominantly in individuals ages 20–40.1,2 In patients with relapsing remitting MS, incomplete recovery after relapses, particularly when they occur early in the disease course, coupled with independent disease progression, leads to disability accumulation and diminished quality of life for MS patients. 3
Healthcare resource utilization (HCRU) and costs increase as disability progresses, with higher disability scores and disease relapses resulting in more hospitalizations and outpatient consultations. 4 Furthermore, MS poses a significant annual financial burden, with a substantial portion of costs related to managing relapses, especially in severe cases.5–7
Treating MS with disease-modifying therapies (DMTs) aims to achieve and maintain ‘no evidence of disease activity’, a state where there are no relapses, no progression in disability, and no emergence of new lesions in magnetic resonance imaging (MRI) scans. 8 Early DMT treatment reduces relapse frequency and slows disease progression.9–11 DMTs are categorized as moderate-efficacy (ME) and high-efficacy (HE), with emerging evidence suggesting that early initiation of HE-DMTs after an MS diagnosis is more effective in suppressing relapse activity.12–16 The early HE-DMT approach includes induction therapy, where patients are treated first with HE-DMTs to control disease activity early then de-escalated to an ME-DMT after a period of time of stability on HE-DMT.
Natalizumab (Tysabri®), an anti-α4 integrin monoclonal antibody, is an HE-DMT that received initial US Food and Drug Administration (FDA) approval in 2004 following the AFFIRM and SENTINEL phase-III trials. 17 Administered intravenously at a standard dose of 300 mg every 4 weeks, it effectively reduces the risk of and lowers the annualized rate of MS-related hospitalizations and disability progression in patients. 18
While natalizumab has historically been used as a second-line (2L) option following ME-DMT, limited knowledge exists regarding the real-world use of natalizumab as a first-line (1L) treatment relative to later-lines (2L+) and the subsequent treatment outcomes. This study aimed to describe the real-world use of natalizumab in both initial (i.e. 1L) and subsequent line (i.e. 2L+) treatments for newly diagnosed MS patients by analyzing unadjusted annualized relapse rates (ARR) and HCRU patterns before and during treatment.
Methods
Study design and data source
This retrospective observational study used administrative claims from the Komodo Health database during the study period from 1 October 2015 to 31 August 2022. Komodo Health is a comprehensive repository that traces the journeys of 325 million unique patients across the United States, offering a rich view of both medical and pharmacy encounters. The database contains 150+ longitudinal payer complete datasets, linking patient behaviours to individual healthcare providers and healthcare organizations thus allowing for a comprehensive understanding of complete care pathways and patient journeys. Drug utilization was identified from pharmacy claims and physician services using National Drug Code and Healthcare Common Procedure Coding System codes. Durable medical equipment (DME) and physical therapy (PT) use were identified through Current Procedural Terminology codes.
Study population eligibility criteria
The study population consisted of newly diagnosed MS patients who initiated natalizumab treatment. Patients with ⩾1 claim for MS (ICD-10 G35) between 1 October 2016 and 31 August 31 2020, were identified (see Figure 1). Initial MS diagnosis was defined as the earliest recorded MS diagnosis date. For inclusion, patients were required to have continuous health and drug plan enrolment for ⩾12 months before the initial MS diagnosis and ⩾24 months after. To ensure patients were indeed newly diagnosed with MS, those with a prior MS diagnosis or prior DMT use before the initial diagnosis date were excluded. All available data preceding the initial MS diagnosis date were used to determine the incident MS cohort. Additionally, patients were required to have ⩾1 claim for natalizumab on or after the initial diagnosis date, with the index date defined as the earliest of such claims. They also had to be ⩾18 years of age at index date and have continuous enrolment between initial diagnosis date and index date. Patients with claims for multiple DMTs on the same date any point during the study period were excluded.

Study design.
Patients were categorized into two study cohorts based on their index treatment: first-line (1L; no prior DMT between initial diagnosis and index date) or second or later-line (2L+; ⩾1 DMTs between initial diagnosis and index date). DMTs included interferons (interferon beta 1A, interferon beta 1B, peginterferon beta 1A), fumarates (dimethyl fumarate, diroximel fumarate, monomethyl fumarate), sphingosine-1-phosphate receptor modulators (fingolimod, siponimod, ozanimod, ponesimod), anti-CD20s (ocrelizumab, ofatumumab, alemtuzumab), glatiramer acetate, teriflunomide, mitoxantrone and cladribine.
The follow-up (i.e. on-treatment) period for each patient covered index date through the earliest of end of continuous enrolment of either medical or pharmacy insurance benefits, natalizumab treatment discontinuation, defined as a gap ⩾45 days between end of days of supply (assuming 28-day supply per natalizumab prescribing information) of the last natalizumab and the subsequent natalizumab administration, treatment switch, defined as initiation of any non-index DMTs prior to treatment discontinuation, death or study end. Sensitivity analysis was performed with treatment discontinuation defined as a gap ⩾90 days. Since only the month and year of death were available in the data, death date was imputed following guidance from Komodo as follows: to the last service date if the patient’s last service occurred in the death month, the 15th if the last service occurred before the death month and to the end of the month if the last service occurred within the 2 months following the death month (to account for possible claims processed after death). If last recorded service date was more than 2 months after death month, the patients were assumed not deceased, and their death date was removed.
Baseline characteristics were recorded during the baseline period (12 months prior to index date). Demographic variables measured at index date included age, sex, index year, region, insurance payer type (e.g. commercial, Medicare, Medicaid), race and ethnicity. Clinical characteristics measured in the baseline period included Charlson comorbidity index, 19 MS-related comorbidities and the previous DMT prior to 2L+ natalizumab initiation.
Outcomes
Relapses were identified using a validated claims-based algorithm.20,21 This included either hospitalization with a primary MS diagnosis or an outpatient visit with an MS diagnosis (any position) followed within 7 days by intravenous steroids, adrenocorticotropic hormone, total plasma exchange or high-dose oral corticosteroids (⩾500 mg/day of specified corticosteroids). Treatments received on days of natalizumab administration were not counted as relapse events. Multiple relapse events occurring within 30 days of each other were combined into a single relapse event. Relapses on the initial MS diagnoses date were included, and a sensitivity analysis was conducted excluding relapse events on initial diagnosis date.
Outcomes were assessed during the baseline period (truncated at initial MS diagnosis) and follow-up periods using person-time methods to accommodate varying durations of observation. Relapse was the primary outcome. The ARR was calculated by dividing the total relapses by the sum of person-years (i.e. the total follow-up time for individuals). Person-time is a clinical research measure used to calculate events rates per unit of time. Secondary outcomes included MS-related (claim with MS diagnosis in primary or secondary position) HCRU such as hospitalizations, emergency department (ED) visits and outpatient visits (differentiating between natalizumab administrations and other visits). Disability worsening HCRU was defined as the use of DME such as canes, walkers, hospital beds and wheelchairs, as well as PT and various forms of assisted living or care (e.g. nursing home, rehabilitation, assisted living or custodial living). HCRU outcomes encompassed binary assessment of HCRU utilization, counts of overall visits, counts among those with at least 1 visit and length of stay (LOS) for hospitalizations and long-term care facility stays. Visit counts and LOS variables were expressed as per-person-per-year. The calculation for each patient and period of interest involved summing up the HCRU events and dividing it by the total person-years in that period. Time before initial MS diagnosis was not included in baseline HCRU assessment.
Statistical analysis
Descriptive statistics for baseline demographic and clinical characteristics are reported as counts and percentages for categorical variables and mean and standard deviation for continuous variables. Between-group comparisons of baseline characteristics were performed using Wilcoxon rank sum test, Pearson’s Chi-squared test and Fisher’s exact test as appropriate.
The time to first relapse was analyzed by Kaplan–Meier methods, starting from the index date and censoring at end of follow-up. An exploratory analysis of the number of relapses versus follow-up time was performed to understand the distribution of relapse rates. Unadjusted ARR and 95% confidence intervals (CI) during both baseline and on-treatment periods were determined by modelling the number of relapses using separate Poisson models. A variable indicating 1L and 2L+ group membership was the sole independent variable, and person-time was incorporated as an offset. In cases of overdispersion, negative binomial regression models were used for accurate estimation. 22 Only the year prior to treatment initiation, excluding any time prior to the initial MS diagnosis, was considered for baseline ARR calculations. The hazard of relapse in 1L compared to 2L+ was estimated with a univariable Cox model. Hazard ratio (HR) and 95% CI are reported.
Unadjusted differences in baseline and follow-up HCRU were assessed within each group using the mean difference (MD) and 95% CI. Time prior to the initial MS diagnosis was excluded from baseline HCRU calculations. Statistical comparisons between baseline and follow-up HCRU within 1L and 2L+ cohorts were made with the paired t-test and McNemar’s Chi-squared test. To compare HCRU differences between the 1L and 2L+ groups, the difference in MD was examined and tested with Welch’s two-sample t-test.
Analyses were conducted with the gtsummary v1.7.0 and survival v3.5-5 packages in R v4.2.1.23–26 For all comparisons, the threshold of statistical significance was p ⩽ 0.05.
Results
Patient characteristics
In total, 1174 1L and 394 2L+ patients were identified (see Figure 2). Baseline characteristics are detailed in Table 1. Codes for identifying comorbidities are provided in Supplemental Table 1. The mean age in the 1L cohort was 39.0 years, while in the 2L+ group, it was 39.7 years. Notably, the 2L+ group had a more females (79.4% vs 72.0%, p = 0.004) and a higher prevalence of anxiety, depression, gastrointestinal disease, hypertension and arthritis (all p < 0.05) compared to the 1L group. On average, 1L patients initiated natalizumab treatment much sooner after initial MS diagnosis than 2L+ patients (mean 7.3 vs 20.1 months, p < 0.001). Among the 2L+ cohort, oral treatments (e.g. fumarates, fingolimod, siponimod and teriflunomide) were the most common 1L treatments (48.5%) followed by injectable platform therapies (45.4%), ocrelizumab (5.6%) and ofatumumab (0.5%).

Patient attrition.
Baseline patient characteristics.
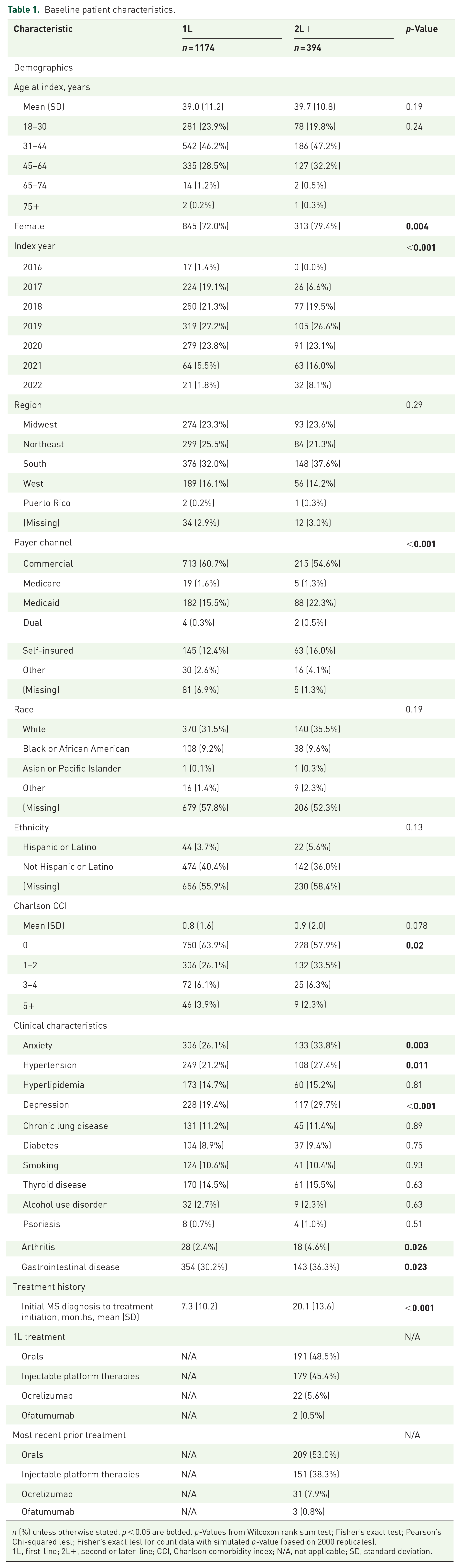
n (%) unless otherwise stated. p < 0.05 are bolded. p-Values from Wilcoxon rank sum test; Fisher’s exact test; Pearson’s Chi-squared test; Fisher’s exact test for count data with simulated p-value (based on 2000 replicates).
1L, first-line; 2L+, second or later-line; CCI, Charlson comorbidity index; N/A, not applicable; SD, standard deviation.
The 1L cohort was followed for an average of 6.2 months longer than the 2L+ cohort (mean treatment duration 30.0 vs 23.8 months, p < 0.001). The 1L group had a significantly higher proportion of individuals still receiving natalizumab treatment at the 1- and 2-year marks after starting treatment than the 2L+ group. One year after starting natalizumab, 80.3% of 1L patients and 72.6% of 2L+ patients were still receiving natalizumab treatment (p = 0.001). By the end of the second year, these percentages dropped to 64.6% and 45.7%, respectively (p < 0.001). The types of censoring events (end of follow-up) varied between the two groups. Among 1L patients, follow-up ended due to study completion (54.3%), discontinuation (23.9%) and disenrollment of healthcare plan (21.6%). In comparison, for 2L+ patients, follow-up concluded due to study completion (59.1%), discontinuation (14.0%) and disenrollment (26.6%). Rarely, follow-up ended due to death or switching to a different DMT, in both cohorts (⩽0.3%).
Notably, 94 (8%) 1L patients began natalizumab treatment on the same day as their initial MS diagnosis. These individuals were not included in the analyses that compared relapse rates and HCRU between baseline and during treatment because they lacked baseline data following their MS diagnosis.
Claims-based relapse
Within-group comparison
Compared to baseline, natalizumab treatment led to a reduction in ARR of 81% for the 1L cohort (p < 0.001) and 55% for the 2L+ cohort (p < 0.001; Figure 3). Three 2L+ patients had very high relapse activity during the follow-up period (>5 events per year) and were identified as outliers. Excluding these patients led to a decrease in the 2L+ on-treatment ARR from 0.41 (95% CI 0.34–0.49) to 0.36 (95% CI 0.30–0.44).

ARRs in baseline and on-treatment for 1L and 2L+ cohorts.
Between-group comparison
The proportion of patients experiencing relapse during natalizumab treatment was similar in both cohorts (1L: 32.2%, 2L+: 37.1%, p = 0.077). The time to relapse differed significantly between the cohorts (log-rank p < 0.001). The median time to relapse was 33.9 months for the 2L+ group, while it was not reached for the 1L group (Figure 4). Most initial relapses during treatment occurred in the outpatient setting, with 84.7% in the 1L group and 84.2% in the 2L+ group (p = 0.91).
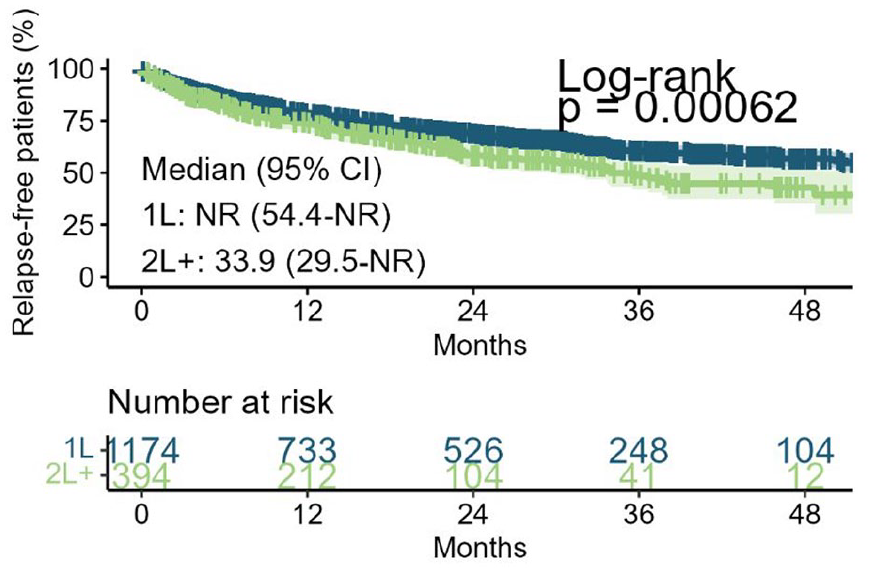
Kaplan–Meier relapse curves.
Unadjusted Cox models revealed 1L patients had a 27% lower risk of relapse compared to 2L+ patients, with a HR of 0.73 (95% CI 0.61–0.89). When comparing between the groups, the 1L cohort had significantly higher baseline ARR of 1.48 (95% CI 1.37–1.59) compared to the 2L+ group with an ARR of 0.92 (95% CI 0.82–1.02), representing a 61% higher ARR (p < 0.001). Additionally, 1L patients had a significantly lower on-treatment ARR of 0.28 (95%CI 0.25–0.31) compared to 2L+ patients with an ARR of 0.41 (95% CI 0.34–0.49), indicating a 32% lower rate (p < 0.001; Figure 3).
Results of relapse rate analysis were unchanged in sensitivity analyses that included expanding the treatment discontinuation criteria from a gap of ⩾45 to ⩾90 days and excluding relapse events on the initial MS diagnosis date. Refer to the Supplemental Table 3 for additional details.
MS-related HCRU
Within-group comparison
When considering within-group comparisons, natalizumab demonstrated a substantial reduction in MS-related inpatient HCRU compared to baseline for both the 1L and 2L+ cohorts (Supplemental Table 4). This reduction was seen in overall MS-related hospitalizations, with an average decrease of 5.43 visits per year for 1L patients (p < 0.001) and a decrease of 2.04 visits per year for 2L+ patients (p = 0.034). Among patients who had hospitalizations either at baseline or during follow-up, natalizumab treatment led to a notable reduction of 17.01 visits per year for 1L patients (p < 0.001) and 7.00 visits per year for 2L+ patients (p = 0.034). Additionally, the total LOS in hospitals decreased by 20.96 days per year for 1L patients (p < 0.001) and by 4.23 days per year for 2L+ patients (p < 0.001).
Natalizumab also contributed to a decrease in outpatient visits unrelated to natalizumab administration for the 1L cohort when compared to baseline. Overall outpatient visits were reduced by 12.01 visits per year, and visits among patients with outpatient visits at either baseline or during follow-up were reduced by 12.11 visits per year (both p < 0.001). Notably, there were no significant reductions in HCRU related to DME, PT or long-term care between baseline and follow-up for either cohort. Codes to identify DME and PT are provided in Supplemental Table 2.
Between-group comparison
In the comparison between groups, 1L patients experienced more substantial reductions in MS-related inpatient and non-administration-related outpatient HCRU between baseline and follow-up than was observed among 2L+ patients (Figure 5 and Supplemental Table 4).

Unadjusted 1L versus 2L+ difference in differences MS-related HCRU.
For 1L patients, 3.39 fewer visits per year in overall hospitalizations were observed than among 2L+ patients, and visits among patients with hospitalizations in either baseline or follow-up were further reduced by an additional 10.01 visits per year. The total inpatient LOS among those treated in 1L was 16.73 days fewer per year than the 2L+ group.
Moreover, the overall outpatient visits not related to natalizumab administration were reduced by 11.54 visits per year for 1L patients than 2L+ patients. This reduction was consistent at 11.64 visits per year among patients with outpatient visits in either baseline or follow-up.
Discussion
In this study, a large sample of newly diagnosed MS patients using natalizumab as either first (1L) or subsequent line (2L+) treatment from a nationally representative administrative claims database was examined. This analysis focused on comparing ARR and HCRU patterns within and between these groups during baseline and on-treatment periods. Our study revealed a 27% reduction in relapse risk when natalizumab was employed as 1L treatment compared to its usage in 2L+, demonstrating its enhanced efficacy in managing MS early in the disease course. Despite observing a significant 61% higher baseline ARR in the 1L group compared to the 2L+ group (1.48 vs 0.92, p < 0.001), the on-treatment ARR was notably 32% lower in the 1L patients than 2L+ (0.28 vs 0.41, p < 0.001), emphasizing the impactful reduction of relapses with early in the disease natalizumab treatment initiation. This translates to an 81% reduction in ARR while receiving treatment compared to baseline for the 1L cohort, in contrast to the 55% reduction observed in the 2L+ cohort. This clinically meaningful difference accentuates the potential benefits of early natalizumab treatment and underscores the efficacy of natalizumab as a highly effective DMT, a finding consistent with the conclusions drawn in pivotal trials like AFFIRM. 27
While the study intended to fill a key gap in the literature by reporting on the treatment outcomes of patients who received natalizumab 1L and to contextualize this information by reporting the same outcomes among those treated with natalizumab at later lines, the results should be interpreted with caution. The analyses were not designed to be a direct matched comparison between comparable groups. There are notable differences between these groups, such as treatment history – patients in the 2L+ group have failed previous therapies, while 1L patients are treatment-naïve – as well as varying comorbidities. As patients experience disease progression over the course of therapy, those treated in the 2L+ group showed a greater burden of comorbidities. This analysis presents unadjusted estimates and does not account for measured or unmeasured confounders, so caution is warranted when interpreting the between-group results.
Other literature has also reported reductions in HCRU associated with 1L initiation of natalizumab rather than later lines, as found in the present study. For example, a US study on 661 MS patients showed that natalizumab in 1L was associated with significant additional reductions of 10.01 hospitalizations/year (p = 0.009), 16.73 days/year in LOS (p < 0.001) and 11.64 visits/year in non-natalizumab-related outpatient encounters compared to initiation in 2L+. 28 These findings are in line with other research indicating a consistent pattern of decreased HCRU with natalizumab treatment. A study from the US revealed that the initiation of natalizumab was associated with a significant reduction in hospitalizations and healthcare costs. 29 Another study from Germany demonstrated a substantial decline in hospitalization rates and other healthcare resources following natalizumab treatment initiation. 30
Furthermore, the findings of this study demonstrated a prolonged time to relapse for patients treated with natalizumab 1L compared to those receiving it in later lines of treatment (median time to relapse not reached for 1L vs 33.9 months for 2L+). The real-world effectiveness of natalizumab, as exhibited through decreased risk of relapse, lower ARR and longer time to relapse for 1L patients compared to 2L+ patients, reinforces the importance of utilizing natalizumab as a 1L treatment.
When natalizumab was reintroduced into the US market in 2006, it was generally recommended as a second-line therapy in the US label due to the associated risk of progressive multifocal leukoencephalopathy (PML), a rare yet serious brain infection caused by mutated pathogenic forms of the JC virus. However, subsequent research has led to a better understanding of natalizumab-associated PML risk supporting benefit–risk discussions and patient management. Since 2013 the FDA-approved US label no longer contains language recommending second-line use but instead states that physicians should consider whether the expected benefit is sufficient to offset the risks.
Limitations
There are some limitations that should be acknowledged. First, claims data may contain incomplete or delayed information and could lead to patient selection bias. Only patients with continuous benefit coverage for ⩾24 months after MS diagnosis were included, further increasing likelihood of selection bias. Findings may not be generalizable to broader MS populations. Second, the comparisons between 1L and 2L+ did not account for differences in the study populations, including confounding variables; absence of adjusted analysis limits ability to establish causality between natalizumab treatment and observed reductions in relapse rates and HCRU. Third, claims-based relapse may have limitations compared to clinician-confirmed relapse. Relapse detection using healthcare claims data may miss mild relapse without clinical visits, potentially underestimating relapse rates and suffering from misclassification bias. To address this, several sensitivity analyses were performed including extending the treatment discontinuation gap from 45 to 90 days, excluding relapses on the initial MS diagnosis date, and for a subset of analyses, excluding three patients with unusually high relapse activity. Fourth, claims data do not contain important clinical variables such as reasons for treatment discontinuation, MRI or disability progression data. Lastly, this study focused on claims-based outcomes and did not consider other important clinical measures such as disease severity, disability progression or quality of life assessments. As such, laboratory results indicating anti-JCV antibody status were not available for analysis.
While this study offers valuable insights into the real-world use of natalizumab in treating newly diagnosed MS patients, future research could further improve upon this work by addressing several key limitations. Future studies should aim to reduce potential selection bias and include adjustments for confounding variables to allow for more robust comparisons between treatment groups. Incorporating broader clinical outcomes, such as MRI findings and disability progression, would provide a more comprehensive understanding of natalizumab’s long-term efficacy and safety. Additionally, implementing risk stratification for PML could help clarify the risk–benefit profile of using natalizumab as a first-line therapy.
Conclusion
In a real-world setting, initiating natalizumab as a 1L treatment for individuals newly diagnosed with MS resulted in a significant 81% reduction in relapse rates between baseline and follow-up. Moreover, this approach extended the time before the occurrence of the first relapse, underscoring the efficacy of early initiation of natalizumab in effectively managing the progression of the disease. Patients who began natalizumab as their initial treatment after diagnosis experienced fewer hospitalizations, ED visits and outpatient visits. This suggests the potential advantages of commencing natalizumab early in the treatment plan for MS not only in managing the condition effectively but also in reducing the overall utilization of healthcare resources.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251317949 – Supplemental material for Evaluating natalizumab first-line and later-line use in multiple sclerosis: a US claims database analysis
Supplemental material, sj-docx-1-tan-10.1177_17562864251317949 for Evaluating natalizumab first-line and later-line use in multiple sclerosis: a US claims database analysis by Robin L. Avila, Nicole S. Croteau, Fei Tang, Jason C. Simeone, Khalil Jomaa, Boyang Bian and Mattia Gianinazzi in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
This study was presented in part at the 39th Congress of the European Committee for Treatment & Research in Multiple Sclerosis, Milan, Italy, 11–13 October 2023. Writing support for this manuscript was provided by Parnian Eghbalian of Cytel, Inc.
Author’s note
Jason C. Simeone was employed by Cytel when study was conducted.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
