Abstract
Introduction:
Pregnancy is widely accepted as a period when relapses of multiple sclerosis (MS) are decreased, with an increased risk of relapse in the first months postpartum. This systematic review evaluated relapses during pregnancy and postpartum, according to disease-modifying therapy (DMT) exposure before, during, and after pregnancy, and the influence of DMT on these outcomes.
Methods:
We searched Medline and EMBASE to identify relevant publications from November 2009 to 2019 along with references lists of selected articles. Publications were filtered and assessed by two independent reviewers to ensure appropriate data extraction.
Results:
Of 469 articles identified, 28 were included for analysis including 4739 pregnancies in 5324 patients. All five studies comparing natalizumab or fingolimod (high-efficacy DMTs) use preconception versus interferon beta, glatiramer acetate, or dimethyl fumarate, or no DMT suggested that there was a greater risk of relapse during pregnancy following withdrawal of the high-efficacy DMTs. Of 10 studies evaluating relapses during pregnancy, five studies found that continuing DMTs into early pregnancy reduced relapses compared to discontinuing treatment. DMT exposure preconception generally had no effect on postpartum relapses versus no DMT; however, natalizumab or fingolimod use preconception was associated with postpartum relapse versus no high-efficacy DMT in one study. DMT exposure during pregnancy was associated with fewer postpartum relapses versus no DMT exposure in four of seven studies, while three found no difference between groups.
Conclusion:
Results of this systematic review concerning women with relapsing MS show a complex and often conflicting picture regarding DMT exposure and relapses during and after pregnancy. Although our data are limited by variability between studies, there is some evidence suggesting the use of natalizumab or fingolimod preconception is associated with increased risk of relapses during pregnancy, highlighting the need for effective disease-management strategies in these especially high-risk patients.
Introduction
Multiple sclerosis (MS) is more prevalent in women, corresponding to a female to male ratio of 3:1. 1 With a peak onset of disease averaging at 30 years of age, MS is the most common neurologic disability in women of child-bearing age and thus, treatment decisions surrounding pregnancy are of clinical importance. 2 Pregnancy is generally accepted as a period in which relapses decrease significantly, especially in the third trimester, before increasing immediately postpartum for up to a year after delivery.3,4 However, more recently published population-based data did not observe any rebound disease activity in the first 3 months postpartum. 5 In addition, a prospective, longitudinal, single-center study of 39 pregnant women with relapsing MS and 21 healthy controls measured serum neurofilament concentration during pregnancy and in puerperium. Serum neurofilament levels were found to be significantly higher in patients with relapses in the third trimester compared with controls and in patients with versus without relapses postpartum. Together, these findings suggest serum neurofilament levels reflect MS activity during and after pregnancy. 6
While some studies have indicated a number of factors, such as disease severity and relapses pre-pregnancy, may be predictive of disease activity during pregnancy and postpartum, less is known about the effects of MS treatment with disease-modifying therapy (DMT). Women have previously been advised to discontinue treatment prior to conception, considering the potential risks to the unborn fetus. 7 However, there is increasing evidence to suggest that the DMTs interferon betas, glatiramer acetate, and natalizumab do not increase the risk for adverse pregnancy and fetal outcomes, such as spontaneous abortions and congenital malformations. 8 These findings have led to the European Medicines Agency allowing for the use of interferon betas to be considered during pregnancy (if clinically needed)(Rebif)(Avonex)(Betaferon)(Extavia)9–12 and removing the contraindication of glatiramer acetate during pregnancy. 13 Safety data across all DMTs beyond the first trimester remain scarce. Conversely, cessation of DMTs with long treatment interruption periods before pregnancy – due to safety concerns – might lead to an increase in relapses and accumulation of permanent disability. 14 Furthermore, the impact of DMTs on relapses and disability progression may vary from one medication to another. As well as treatment decision making throughout pregnancy, patients must also consider their options with regard to breastfeeding. There is evidence to suggest that breastfeeding elicits a protective effect against postpartum relapses; however, uncertainty remains with regard to subgroups of patients with high disease activity who may also benefit from resuming DMTs. 15 In addition, for those who decide to forego breastfeeding for other reasons, the effectiveness of specific DMTs in reducing risk of postpartum relapse is a clinically important issue. Thus, the optimal type and timing of such treatment reintroduction has yet to be established in these patient groups. The best strategy to reduce the rate of relapse during and after pregnancy remains up for deliberation.
Against this background, we conducted a systematic review to evaluate relapse rates and disability progression during pregnancy and in the postpartum period, according to timing of exposure to DMT (before, during, or after pregnancy). The effect of exposure to any DMT versus DMT-unexposed and exposure to high- versus moderate-efficacy DMTs on these clinical status outcomes was also evaluated, along with the influence of breastfeeding. The purpose of this review was to address patient and healthcare provider needs regarding decision-making on DMT use surrounding pregnancy, with respect to clinical activity.
Methods
Search strategy
The population, exposure, comparison, and outcome (PECO) model was used to develop the inclusion criteria and search terms. 16 The population was composed of women with MS before, during, after pregnancy, or during breastfeeding. The exposure was treatment with DMTs preconception, during pregnancy or postpartum, limited to interferon beta-1a, interferon beta-1b, peginterferon beta-1a, alemtuzumab, cladribine tablets, dimethyl fumarate, fingolimod, glatiramer acetate, laquinimod, natalizumab, teriflunomide, methotrexate, cyclophosphamide, mycophenolate mofetil, azathioprine, ocrelizumab, and rituximab. The outcomes of interest were clinical status outcomes of relapse (relapse rate, proportion with ⩾ 1 relapse, number of relapses, and/or annualized relapse rate) or disability progression (changes in Expanded Disability Status Scale (EDSS) score as defined in each study) during two pregnancy periods, during pregnancy, and postpartum. Exposure definition has been grouped as follows: (1) exposed to DMT preconception versus unexposed to any DMT preconception; (2) exposed to high-efficacy DMTs preconception versus unexposed to any DMT preconception or exposed to moderate-efficacy DMT; and (3) exposed to DMTs during pregnancy, defined as ⩾ 1 prescription regardless of duration, versus unexposed to any DMT. For postpartum outcomes only, outcomes were evaluated according to timing of restarting DMT after delivery. In addition, we assessed outcomes according to breastfeeding status (e.g. exclusive, non-exclusive, or none).
The search was performed by an expert in Medline and EMBASE (on STN®) to identify relevant publications from the period November 2009 to November 2019, regardless of country of study. Conference abstracts since 2015 only were included. All aspects of the search included free text and index term searches. The search string included each DMT of interest as well as the generic concept ‘disease-modifying (drug or therapy)’ in conjunction with ‘multiple sclerosis’ and a broad profile for ‘conception, pregnancy, postpartum and breastfeeding’. The search was limited to articles mentioning ‘relapse’, ‘recurrence’, ‘flare up’, ‘exacerbation’, and/or ‘progression’. The search was restricted to publications in English. The reference lists of review articles were also manually searched to identify additional studies of interest.
This review has been registered on the PROSPERO database (CRD42020175608).
Selection criteria
Criteria for article inclusion in the analysis were as follows: (1) The study included patients with MS treated with DMTs; (2) DMT exposure could be classified according to timing of pregnancy (before, during, or postpartum); (3) Relapses or EDSS were classified according to timing of pregnancy (during or postpartum); (4) Reported comparison expressed by hazard ratio (HR), relative risk (RR), odds ratio (OR), mean/median for relapses, or EDSS; and (5) The study was a randomized control trial or observational in design. Articles with overlapping data (e.g. from the same study population and include duplicate data), general reviews, and case reports/series were excluded. Duplicate articles were removed by automated procedure.
Study selection, data extraction, and analysis
Titles and abstracts were first screened by two reviewers (SL and JD) and classified in relation to the inclusion criteria. The full texts of articles categorized as ‘unsure’ were reviewed, and then classified as excluded, included, or unsure. At this stage, those studies considered ‘unsure’ were assessed by another reviewer (MS) who was blind to their status, and discrepancies were resolved through discussion.
A standardized data collection form in Microsoft Excel was used for data extraction and data were extracted in the following categories: (1) Study characteristics such as author, year, design, number of patients and of pregnancies; (2) Exposure information such as DMTs used, timing of DMTs discontinuation before or during pregnancy, and timing of restart of DMTs among those who discontinued; (3) Outcomes definition and timing of occurrence; and (4) Breastfeeding status.
Quality assessment
The quality of each study was critically evaluated using the Newcastle–Ottawa Scale (NOS), 17 with assessment based on the following three sections: selection of the study groups, comparability, and exposure (for case–control studies) or outcome (for cohort studies). Studies were scored using a ‘star’ system. A maximum of nine stars was attainable and quality was classified as: (1) high when the majority of criteria were met, that is, each section had at least a star and the minimum total stars was eight; (2) acceptable when most of the criteria were met, that is, only one section without a star, with a minimum of six total stars; and (3) low when either few stars were assigned, or two sections lacked stars.
Results
Studies included
A total of 464 articles were identified by the search strategy and an additional 5 were identified through manual review of recent review articles (Figure 1). After automatic removal of duplicates, 189 article abstracts were screened for relevance and 82 were deemed appropriate for full assessment. Of the 82 studies assessed, 54 were excluded (31 did not meet eligibility criteria, 14 cases series/reports, 5 overlapping data, 2 non-English, 1 patient management review and 1 systematic review) and 28 studies were ultimately included for analysis. Across these studies, 4739 patients and 5324 pregnancies were included. The characteristics of the included studies are detailed in Supplementary Table s1.

PRISMA flowchart depicting the study selection and record screening process.
Pregnancy
Relapses
Among the 28 articles identified in this systematic review, 16 studies evaluated the effect of DMTs on relapses during pregnancy. The studies evaluated data from 2513 pregnancies in 2425 patients, of whom 958 were treated with DMTs before conception and 831 were exposed to DMTs during pregnancy (ranging from ~1 gestational month through to entire pregnancy, Table 1). Relapses were recorded in a number of ways including annualized relapse rate, relapse rate, percentage of patient with relapses, and number of relapses. As such, effect sizes could not be calculated and a summary of trends is provided.
Timing of DMT exposure in studies assessing relapses during pregnancy.

DMT, disease-modifying therapy; NR, not reported.
N is number of patients unless otherwise stated.
Number of pregnancies.
Five of the six articles reporting data relating to DMT exposure prior to conception compared relapses during pregnancy in patients treated with high-efficacy DMTs, predominantly natalizumab and fingolimod, with those who received moderate-efficacy DMTs (mainly interferon beta preparations or glatiramer acetate) or no DMT. Of which, all five studies suggested that there was a greater risk of relapse during pregnancy with natalizumab or fingolimod use preconception compared with both patient populations (vs untreated and vs moderate efficacy DMTs) (Tables 2 and 3).19–22,32 For instance, an Austrian cohort study found significantly higher rates of relapses during pregnancy, mainly occurring within the first or second trimester, in patients treated with predominantly natalizumab and fingolimod (29%) prior to pregnancy compared to no DMT (4.7%) and interferon beta, glatiramer acetate, or dimethyl fumarate (5.1%). This association with relapse risk was confirmed in a multivariate analysis (OR 2.91, 95% CI: 1.82–4.38). 22 In addition, the annualized relapse rate was significantly higher among natalizumab- or fingolimod-treated patients compared with interferon beta or glatiramer acetate in a cohort with 75 pregnancies included (low quality evidence); an association that remained significant after adjustment for age and disease duration (rate ratio 0.038; p = 0.003). 21
Effect of DMT exposure (preconception) on relapses during pregnancy.

ARR, annualized relapse rate; CI, confidence interval; DMT, disease-modifying therapy; NR, not reported; OR, odds ratio.
N is the number of patients unless otherwise stated.
Effect of type of DMT on relapses during pregnancy.

Adj, adjusted; ARR, annualized relapse rate; DMT, disease-modifying therapy; NR, not reported.
N is number of patients unless otherwise stated.
Time to discontinuation prior to pregnancy also emerged as a topic of clinical importance (Tables 2 and 3). A number of studies indicated an association between the length of time between stopping DMT treatment and conception, also referred to as washout period, and relapse risk during pregnancy.19,22,30,32 For patients receiving natalizumab and fingolimod, it has been recommended to respect a 2–3 month washout period before conception when planning a pregnancy. Patients with longer washout periods, for example, prior to last menstrual period, were significantly more likely to experience relapses, with one study suggesting that the risk of relapse increased by 43% per week of additional washout. 22
Ten studies evaluated relapses in relation to DMT exposure during pregnancy, mainly within the first trimester (Table 4).18,23–29,31,33 Results were diverse, with five studies conveying a potentially reduced risk of relapse in women continuing DMTs into early pregnancy18,24,27,28,33 and five proposing no effect on relapse activity,23,25,26,29,31 potentially related to the type of DMT used. Generally, studies focusing on high-efficacy DMT (natalizumab and fingolimod) exposure during pregnancy noted fewer relapses in pregnancies exposed to DMTs18,24,27,28 whereas articles assessing the use of moderate-efficacy DMTs (interferon beta formulations and glatiramer acetate) found no significant differences in relapses.23,25,26 However, one cohort study described significantly fewer relapses throughout pregnancy in patients treated with interferon beta (median 32 days exposure) than patients unexposed to DMTs (high quality evidence). 33
Effect of DMT exposure (during pregnancy) on relapses during pregnancy.
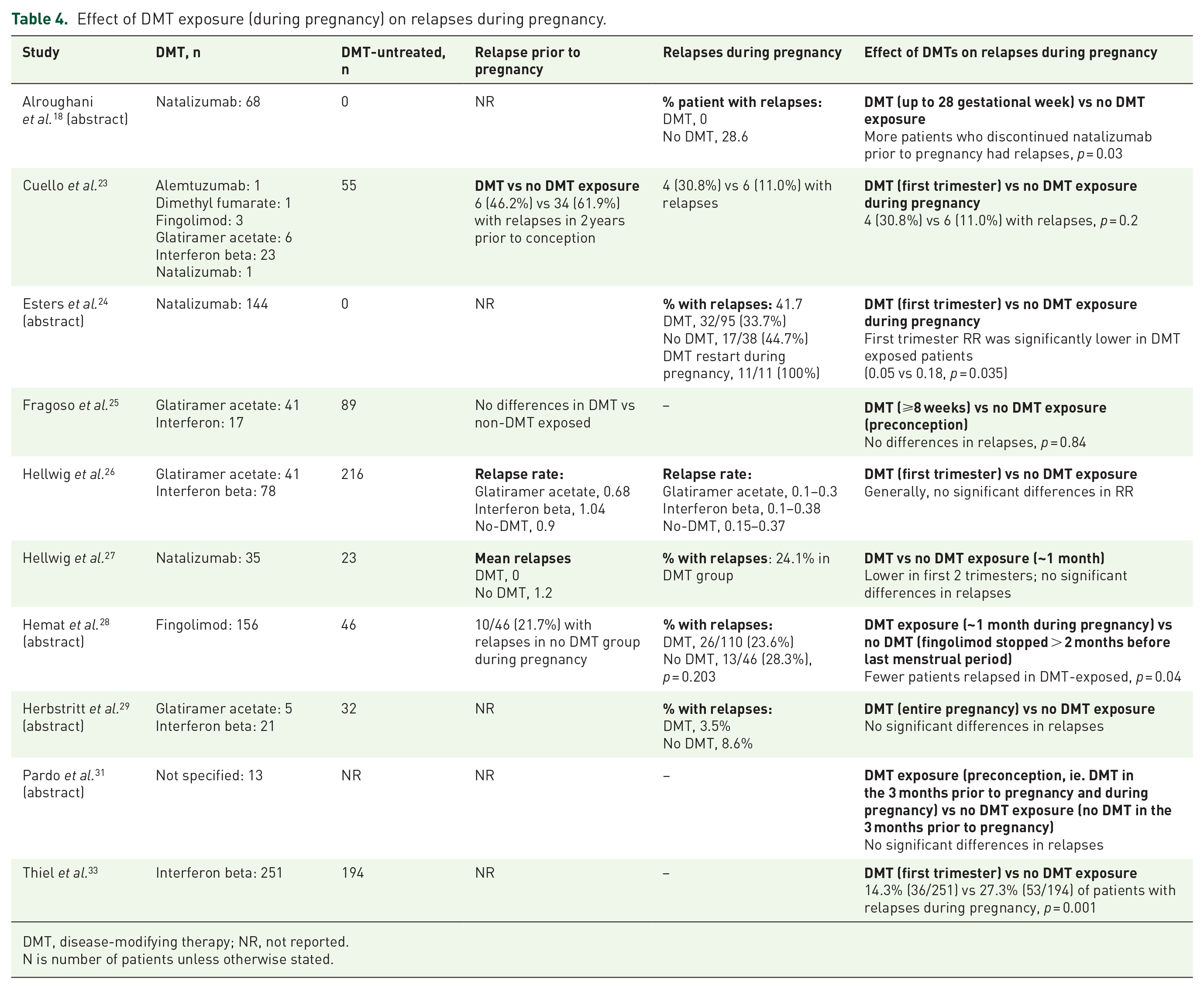
DMT, disease-modifying therapy; NR, not reported.
N is number of patients unless otherwise stated.
Disability progression (EDSS)
Few studies reported results concerning the impact of DMT exposure on disability progression during pregnancy (Table S2).22,30,34 Preconception use of natalizumab or fingolimod and longer duration of washout periods were generally associated with increased risk of disability progression.22,30
Postpartum
Relapses
Of the 28 articles included in this systematic review, 21 studies provided data on postpartum relapses in patients treated with DMTs at varying timepoints throughout pregnancy. A total of 3846 patients and 4395 pregnancies were included. Overall, 1841 pregnancies were exposed to DMTs preconception, 594 during pregnancy (range ~6 gestational weeks to entire pregnancy, Table 5) and 1004 in the postpartum period. A summary of trends is provided, given the high degree of heterogeneity in the parameters used for reporting of postpartum relapses.
Timing of DMT exposure in studies assessing postpartum relapses.

DMT, disease-modifying therapy; im, intramuscular; NR, not reported; sc, subcutaneous.N is number of patients unless otherwise stated.
number of pregnancies.
Monthly intravenous immunoglobulin.
Seven studies assessed the use of DMTs prior to and at the time of conception, and the subsequent impact on postpartum relapses (Table 6). In one of the identified studies DMT exposure within 2 years before pregnancy (median 7 months) was associated with reduced risk of relapse within 3 months postpartum (OR: 0.55, 95% CI: 0.32–0.96; p < 0.05). 38 Use of DMTs preconception did not have an effect on the risk of postpartum relapses for up to 1 year compared with those not treated with DMTs in the remaining six studies. Where the type of DMT was specified, the majority of patients were treated with injectable, moderate-efficacy DMTs.37,39 Bsteh et al. 22 found that preconception use of natalizumab and fingolimod, classified as high-efficacy DMTs, was associated with postpartum relapse with an OR of 2.11 (95% CI: 1.32–3.27) compared with those who were not treated with high-efficacy DMTs (Table 7). Other studies have reported higher rates of relapses with these high-efficacy DMTs preconception compared to injectable, moderate-efficacy DMTs or no DMT.32,40 These observations could reflect underlying disease activity of the different patient populations; the long washout periods recommended for natalizumab and fingolimod before conception may have also influenced relapses during postpartum as well as during pregnancy. For example, there was low quality evidence from Portaccio et al. investigating the effect of the length of washout period in a study of 74 pregnancies among women treated with natalizumab. Results showed that earlier discontinuation prior to conception (before last menstrual period) was associated with a more pronounced increase in the risk of relapse after delivery (p < 0.021). 32
Effect of DMT exposure (preconception) on postpartum relapses.

Adj, adjusted; ARR, annualized relapse rate; CI, confidence interval; DMT, disease-modifying therapy; HR, hazard ratio; NR, not reported; OR, odds ratio.
N is number of patients unless otherwise stated.
Monthly intravenous immunoglobulin.
Effect of type of DMT on postpartum relapses.
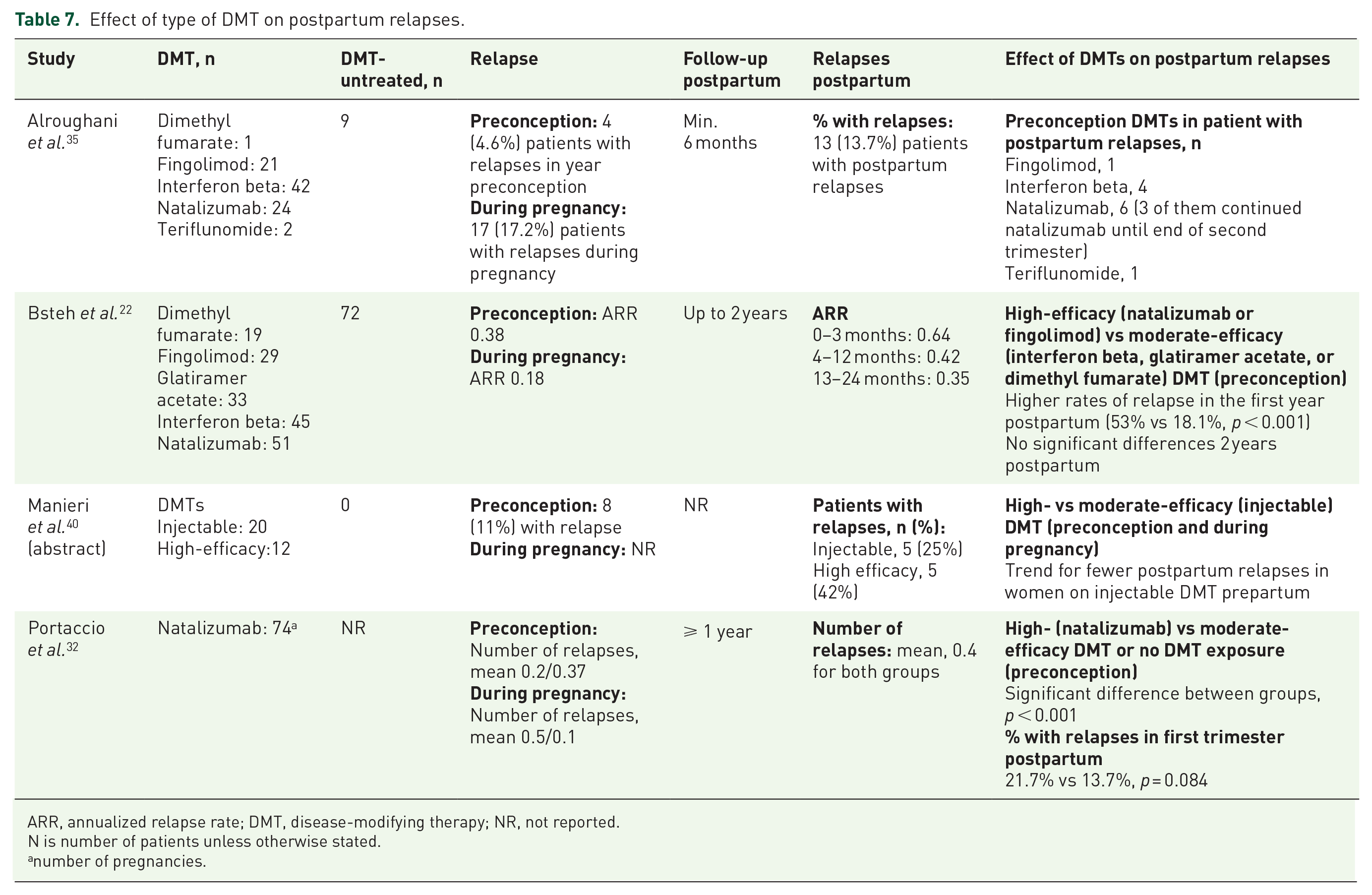
ARR, annualized relapse rate; DMT, disease-modifying therapy; NR, not reported.
N is number of patients unless otherwise stated.anumber of pregnancies.
Mixed findings were reported in regard to DMT exposure during pregnancy and postpartum relapses (Table 8). Patients exposed to DMTs during pregnancy, generally within the first trimester, had fewer postpartum relapses compared to those without DMT exposure in four of seven identified studies.18,25–27 This reduction in relapse rate was noted in both women treated with the moderate-efficacy DMTs, interferon beta formulations or glatiramer acetate,25,26 and with the high-efficacy DMT natalizumab during early pregnancy.18,27 The remaining three studies found no relevant differences in the risk of postpartum relapse between those who were and were not exposed to DMTs during pregnancy.23,29,31
Effect of DMT exposure (during pregnancy) on postpartum relapses.
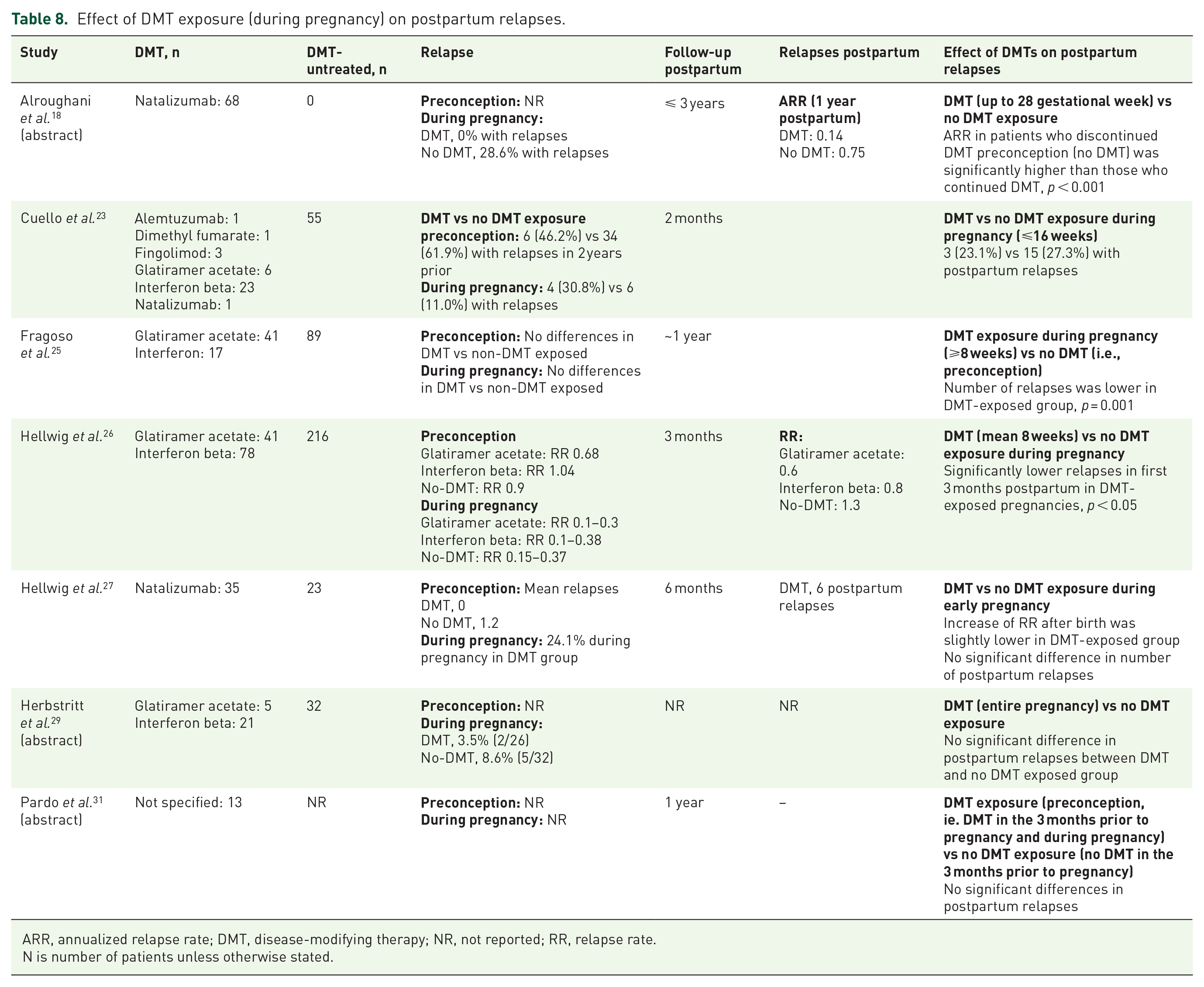
ARR, annualized relapse rate; DMT, disease-modifying therapy; NR, not reported; RR, relapse rate.
N is number of patients unless otherwise stated.
Another clinically relevant question in relation to postpartum use of DMTs is the timing of restart (Table 9). In a study of natalizumab-treated patients, reinitiating treatment more than 1 month after delivery or not at all was a significant predictor of postpartum relapses with an OR of 4.40 (95% CI: 1.28–15.16). 32 A number of other studies have reported findings to suggest that restarting DMT early (1 week to 3 months) after delivery may reduce risk of relapses in the postpartum period.28,41,43,44 Furthermore, a cohort study found that restarting DMT within the first postpartum year (OR: 0.59, 95% CI: 0.25–0.88) and shorter duration to restart (OR: 0.92, 95% CI: 0.88–0.93, per week) significantly reduced the risk of relapses. 22 In contrast, some studies reported no association between relapse risk and timing of DMT restart after birth.5,36,39 Even after adjustment for confounding factors such as exclusive breastfeeding and baseline disease activity, Beaber et al. 36 found that starting interferon beta or glatiramer acetate within 2 weeks of delivery did not reduce risk of relapse over 2 years postpartum (HR: 1.3, 95% CI: 0.5–3.4). Similarly, Langer-Gould et al. 5 reported that postpartum DMT use, as a time-dependent co-variate, had no effect on relapses in the 6 months and 1 year postpartum.
Effect of timing of DMT restart after delivery on postpartum relapses.

Adj, adjusted; ARR, annualized relapse rate; CI, confidence interval; DMT, disease-modifying therapy; HR, hazard ratio; NR, not reported; NS, non-significant; OR, odds ratio; RR, relapse rate.
N is number of patients unless otherwise stated.
Monthly intravenous immunoglobulin.
number of pregnancies.
Disability progression (EDSS)
Table S3 presents data from the 10 articles retrieved from our search that assessed the effect of DMT exposure across pregnancy on disability progression during postpartum. Preconception use of DMTs was not associated with disability progression postpartum in three studies including various DMTs.31,39,43 However, similarly to the effects on postpartum relapses, natalizumab or fingolimod exposure prior to pregnancy and long washout periods may increase the likelihood of disability progression in the first year postpartum.22,32 In a study comparing patients who discontinued fingolimod while trying to conceive with those who continued treatment until pregnancy was confirmed, few patients experienced disability progression and no significant differences between patient populations were observed. 28 Conversely, Fragoso et al. 25 reported significantly less disability progression in women exposed to DMTs for a minimum of 8 weeks during pregnancy compared with those who were not exposed, over a considerably long follow-up period averaging 45 months for both groups.
There was limited evidence on the effect of timing of DMT restart on disability progression (Table S3). Three of the four identified studies indicated that restarting of DMTs, ranging from 2 weeks to 1 year postpartum, did not reduce the risk of disability progression.22,36,43 However, the remaining study observed a significantly lower risk of disability progression in patients who restarted natalizumab within 1 month postpartum (10.7% vs 33.3%; p = 0.024). 32
Breastfeeding
Eight studies reporting the effect of breastfeeding on relapses5,22,26,37,39,42,43,45 or disability progression22,43 were identified from our search. Across these studies, 1929 patients and 2191 pregnancies were included. A total of 1097 pregnant women were exposed to DMTs during preconception (predominantly ⩽ 1 year before pregnancy), 296 during pregnancy, and 667 during postpartum (Table S4). The classification of breastfeeding varied across studies. Generally, exclusive breastfeeding was defined as breastfeeding with no formula supplementation (predominantly for ⩾ 2 months), and non-exclusive breastfeeding included complementary formula feedings. The duration of follow-up for evaluation of relapses ranged from 322,26 to 12 months postpartum,5,37,39,42,43 with one small study assessing the risk of relapses up to 36 months postpartum. 45
Table S5 summarizes findings regarding the effect of breastfeeding on postpartum relapses and disability progression. Exclusive breastfeeding significantly reduced the risk of postpartum relapse versus non-exclusive and no breastfeeding in four of six identified studies.5,26,37,45 Of these, Langer-Gould et al. 5 reported that this protective effect was also apparent in women with suboptimally controlled disease activity at conception. Hellwig et al. 37 found that the protective effect was no longer observed after introduction of supplemental feedings (adjusted HR: 1.10, 95% CI: 0.72–1.78). Two studies found that exclusive breastfeeding did not influence MS activity per multivariate analyses;42,43 however, patients with postpartum relapses were significantly less likely to have exclusively breastfed compared to those without postpartum relapses in one of the studies (27% vs 40%; p = 0.014). 42
Three studies showed non-exclusive breastfeeding (breastfeeding and supplemental formula feeding) did not significantly reduce the risk for postpartum relapse compared with those who did not breastfeed at all.5,26,37 For example, Hellwig et al. 37 found that women who breastfed non-exclusively experienced a similar risk of relapse within the first 6 months postpartum as those who did not breastfeed (adjusted HR 0.96 (95% CI: 0.45–2.03)).
Two studies included both exclusive and non-exclusive breastfeeding in their analysis and found that breastfeeding had no effect on postpartum relapse rates.22,39 It was noted that patients with postpartum relapses were significantly less likely to have breastfed compared to those without postpartum relapses in both studies (41% vs 83% and 54% vs 72%; p < 0.05). However, a high proportion of patients had restarted treatment with DMT postpartum (80–89%).22,39
Discussion
This systematic review, which aimed to assess whether the use of DMTs and timing of exposure affected relapses and disability progression during and after pregnancy, presented mixed findings. Although possible trends emerged from our analysis, such as differences between types of DMT, no clear conclusions could be drawn based on the limited, varied data available and differences in reporting methods. Overall, our findings suggest that the risk of relapse is generally low in patients treated with moderate-efficacy, injectable DMTs (interferon betas or glatiramer acetate) and those not exposed to DMTs. However, one key theme identified was a potential increased risk of relapses during pregnancy in patients who were treated with the high-efficacy DMTs natalizumab or fingolimod preconception and discontinued use while attempting to conceive, compared with the moderate-efficacy therapies interferon betas or glatiramer acetate or no DMT use. This finding may be related to disease reactivation following natalizumab or fingolimod withdrawal, as reflected in a few studies where longer times from discontinuation to conception correlated with risk of relapses and disability progression.46,47 Another potential reason for this effect is the high disease activity in patients requiring high-efficacy treatment. Thus, in patients requiring the high-efficacy DMTs natalizumab or fingolimod to manage their disease activity, these findings highlight the possible need for research into additional treatment strategies surrounding pregnancy, potentially tailored to individual patient-risk.
Similar observations were noted in some studies assessing postpartum relapses and disability progression; however, this could also be related to differences in underlying disease activity between patient populations as opposed to long-term effects of DMTs. In contrast, one registry study including pregnancies from 1967 to 2010 reported DMT use within 2 years of pregnancy independently reduces risk of postpartum relapse. 38 While newer agents have demonstrated durable treatment effects, prolonged treatment benefit (lasting over 9–12 months) of DMTs available during the study timeframe seems unlikely and no potential mechanism has been described. 44
Moreover, patients who were exposed to natalizumab or fingolimod during the first trimester generally had fewer relapses during pregnancy than those who discontinued preconception, whereas no differences in relapse risk were observed with patients taking moderate-efficacy DMTs, mainly interferon betas and glatiramer acetate. No particular trend was identified for postpartum relapses according to the type of DMT used during pregnancy. With the exception of interferon beta preparations and glatiramer acetate, the majority of DMTs currently have a precautionary warning against their use during pregnancy due to a lack of data and concerns about pregnancy and fetal outcomes. While a recent meta-analysis found no association between adverse pregnancy outcomes and DMT exposure (predominantly including studies of interferons, glatiramer acetate, and natalizumab), 8 larger studies with a wider range of DMTs are needed to guide decision-making.
The timing of DMT restart during the postpartum period may also influence the risk of relapse and disability progression. A number of studies suggested that restarting DMT after 1 week up to 3 months post-delivery may reduce the risk of relapses. However, others reported no significant differences in relapse risk; limitations of these studies included small patient numbers. The optimal treatment strategy during postpartum is yet to be established.
The influence of breastfeeding on postpartum clinical status outcomes was also examined. Given the main focus of our search was DMTs, only a limited number of studies with varied postpartum follow-up were identified. Exclusive breastfeeding, for at least 2–4 months, reduced the risk of relapse in the majority of the identified studies, likely as a result of hormonal changes including suppression of pulsatile gonadotropin-releasing hormone and luteinizing hormone that leads to lactational amenorrhea. 48 This is in line with a large, robust meta-analysis from Krysko et al. 15 that reported a conclusive 37% lower odds of postpartum relapse in mothers who exclusively breastfed compared with those who did not breastfeed or did not exclusively breastfeed postpartum. 15 Resumption of ovulation and normal menstrual cycles upon the introduction of regular formula feedings may explain the absence of benefit observed with non-exclusive breastfeeding in some studies, particularly in one study that noted no differences in relapse risk when supplemental feedings were introduced. 37 In addition, this may explain why the few studies that grouped exclusive and non-exclusive breastfeeding together observed no independent effect on clinical status compared with not breastfeeding. Thus, women should be informed of the potential protective benefits of breastfeeding as well as the general benefits to mother and infant, as recommended by the World Health Organization (https://www.who.int/health-topics/breastfeeding).
At present, given the overall risk of relapse in the postpartum period, the reintroduction of DMTs alongside breastfeeding may offer additive protection and this warrants further investigation. While most DMTs are currently contraindicated during lactation with some examples of transfer in human milk reported,49,50 patients may not need to decide between the two. For example, interferon beta preparations can be used during breastfeeding, partially based on their pharmacokinetics properties.49,51 A recent report on two lactating women receiving dimethyl fumarate also suggested there would be minimal drug exposure to an infant through human milk, proposing that dimethyl fumarate may be compatible with breastfeeding. 52 Further studies with large sample sizes are required to confirm these findings and evidence from a wider range of DMTs should be generated.
Our analysis has several limitations. First, while a comprehensive literature search strategy and stringent inclusion criteria were employed to identify appropriate articles, some of our reported observations are based on studies using markedly small sample sizes. Second, differences in statistical reporting methods were noted in the data obtained on disease outcomes, especially with regard to relapses, which precluded meta-analysis. Third, it should be noted that few studies performed adjustments for confounding factors such as age, prior disease activity and disease duration at baseline. A lack of data on alemtuzumab, cladribine tablets, dimethyl fumarate, laquinimod, teriflunomide, methotrexate, cyclophosphamide, mycophenolate mofetil, azathioprine, ocrelizumab and rituximab was also apparent within our search period. This may be in relation to label restrictions and warnings of their use during pregnancy. Therefore, any findings from our systematic review cannot be generalized to all DMTs. In addition, there were limited studies with data on disability progression, which attenuated our ability to draw conclusions on this disease outcome.
Conclusion
Results of this systematic review concerning women with relapsing MS show a complex and often conflicting picture regarding DMT exposure, relapse rates, and disease progression during and after pregnancy. Early discontinuation of natalizumab or fingolimod resulting in long washout periods may increase the risk of relapse and disease progression during pregnancy as a result of disease reactivation between drug withdrawal and conception. Thus, there is a need for effective disease management strategies for women treated with these high-efficacy DMTs prior to conception. Findings regarding postpartum relapses in women with MS exposed to DMTs were also varied. Limited evidence suggests that the use of natalizumab or fingolimod preconception with no exposure during pregnancy increases the risk of postpartum relapse and disability progression, which is likely to be explained by higher disease activity in these patients. In addition, there is evidence to suggest that exclusive breastfeeding significantly reduces the recurrence of relapses postpartum and should be considered. Thus, decision-making concerning benefit-risk of DMT use before, during, and after pregnancy for treating MS remains difficult. Treatment decisions need to be taken by the patient and healthcare professional after careful consideration of all of the potential benefits and risks.
Supplemental Material
sj-docx-1-tan-10.1177_17562864211051012 – Supplemental material for A systematic review of relapse rates during pregnancy and postpartum in patients with relapsing multiple sclerosis
Supplemental material, sj-docx-1-tan-10.1177_17562864211051012 for A systematic review of relapse rates during pregnancy and postpartum in patients with relapsing multiple sclerosis by Kerstin Hellwig, Elisabetta Verdun di Cantogno and Meritxell Sabidó in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864211051012 – Supplemental material for A systematic review of relapse rates during pregnancy and postpartum in patients with relapsing multiple sclerosis
Supplemental material, sj-docx-2-tan-10.1177_17562864211051012 for A systematic review of relapse rates during pregnancy and postpartum in patients with relapsing multiple sclerosis by Kerstin Hellwig, Elisabetta Verdun di Cantogno and Meritxell Sabidó in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-3-tan-10.1177_17562864211051012 – Supplemental material for A systematic review of relapse rates during pregnancy and postpartum in patients with relapsing multiple sclerosis
Supplemental material, sj-docx-3-tan-10.1177_17562864211051012 for A systematic review of relapse rates during pregnancy and postpartum in patients with relapsing multiple sclerosis by Kerstin Hellwig, Elisabetta Verdun di Cantogno and Meritxell Sabidó in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-pptx-4-tan-10.1177_17562864211051012 – Supplemental material for A systematic review of relapse rates during pregnancy and postpartum in patients with relapsing multiple sclerosis
Supplemental material, sj-pptx-4-tan-10.1177_17562864211051012 for A systematic review of relapse rates during pregnancy and postpartum in patients with relapsing multiple sclerosis by Kerstin Hellwig, Elisabetta Verdun di Cantogno and Meritxell Sabidó in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
Medical writing assistance was provided by Sean Littlewood and Jade Drummond of inScience Communications, Springer Healthcare Ltd, UK, and funded by Merck Healthcare KGaA, Darmstadt, Germany.
Author contributions
All authors made substantial contributions to the study conception and interpretation of the data, take responsibility for the integrity and accuracy of the analysis. All authors provided critical revision of the manuscript and approved the final version.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KH has received honoraria and research support from Bayer, Biogen, Merck Healthcare KGaA (Darmstadt, Germany), Novartis, Sanofi-Genzyme, and Teva. EVDC is an employee of EMD Serono Research & Development Institute, Inc., Billerica, MA, USA, an affiliate of Merck KGaA. MS is an employee of Merck Healthcare KGaA, Darmstadt, Germany.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This investigation was funded by Merck (CrossRef Funder ID: 10.13039/100009945).
Data availability
The copyright rules of the database producers apply. Permission to copy, electronically store, reproduce, or distribute the research results is restricted.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
