Abstract
Background:
Dementia is a serious adverse event (AE) that requires attention in clinical practice. However, information on drug-induced dementia is limited. The U.S. FDA Adverse Event Reporting System (FAERS) serves as an important resource for identifying real-world adverse drug reactions and safety signals.
Objective:
This study aimed to use FAERS data to identify drugs associated with increased dementia risk.
Design:
A secondary analysis of the FAERS database was conducted using disproportionality analysis methods.
Methods:
We reviewed dementia-related reports in the FAERS database from the first quarter of 2004 to the fourth quarter of 2023, used the Medical Dictionary for Regulatory Activity to identify dementia cases and summarized the corresponding list of potential medications, counted the dementia-causing medication classes with the highest frequency of reports, and disaggregated all medications.
Results:
The study identified 31,881 dementia-related AEs in the FAERS database, with an increasing trend over time, particularly among females and individuals over 65. Apixaban had the most reports (1631). Disproportionality analyses revealed that rivastigmine, nicergoline, aducanumab, amlodipine/atorvastatin, and dihydroergometrine had the highest risk, based on reporting odds ratio, proportional reporting ratio, and information component. Only valproate and tramadol among the top 50 drugs included a potential dementia risk in their package inserts.
Conclusion:
This study identified a list of medications associated with dementia risk, many of which lack dementia warnings on their labels. Increased monitoring is necessary for high-risk individuals, and further research is required to clarify these associations and improve patient safety.
Introduction
Dementia is a neurodegenerative syndrome characterized by a profound deterioration in cognitive abilities, significantly impacting an individual’s daily functioning and occupational performance. 1 The disease mainly affects people aged 65 and above, with a prevalence rate of 697 per 10,000, and the number of people suffering from dementia doubles approximately every 5 years. 2 Studies indicate that the economic burden of dementia is poised to escalate substantially, with estimates suggesting a total cost reaching $507.49 billion by the year 2030. 3 This financial strain places substantial pressure on both familial support structures and societal resources.
Drug-induced dementia, a subset of dementia cases, is generally considered reversible upon discontinuation of the medication. However, some researchers have reported that certain drugs, particularly those with anticholinergic drug, may be associated with an increased risk of persistent cognitive impairment.4,5 This indicates that drug-induced dementia is a complex disorder influenced by various factors, warranting focused attention and prevention efforts.
Recognizing and preventing drug-induced dementia is crucial. Currently, there is a lack of information on the adverse effects (AEs) of pharmacologic dementia. Drugs may indirectly impair cognitive performance through metabolic effects. 6 Dementia, on the other hand, is not easily diagnosed and detected due to a variety of factors, and research on the use of nonantidementia medications leading to dementia is limited. As a result, the understanding of the relationship between medications and dementia remains incomplete.
Postmarketing surveillance serves as a crucial methodology for elucidating the correlation between drugs and adverse reactions. The FDA adverse event reporting system (FAERS) database functions as a self-reporting system designed to aggregate post-marketing AEs associated with drugs and therapeutic biologics. FAERS data, being extensive and publicly accessible, are frequently employed in drug signal mining endeavors. 7 Previous studies have investigated the relationship between specific drugs and dementia using the FAERS database.6,8 To our knowledge, however, there is a lack of comprehensive studies exploring the relationship between medications and dementia risk.
The objective of this study was to conduct a thorough investigation into the risk of drug-induced dementia using the FAERS database. The primary aim was to identify pharmaceuticals that pose a risk of dementia development. By shedding light on these associations, we hope to emphasize the importance of recognizing potential dementia-related risks and maintaining vigilant monitoring within medical practice.
Methods
Data source
This study comprised three primary components: (1) data collection, (2) drug translation and collation, and (3) data analysis. It served as a retrospective pharmacovigilance investigation leveraging the FAERS database. The FAERS database encompasses post-marketing surveillance programs for all marketed drugs and therapeutic biologics. Since 2004, the database has consistently aggregated AE reports received by the FDA, incorporating submissions from healthcare professionals, consumers, and manufacturers, with quarterly updates. An extensive array of demographic information, drug profiles, indications, timelines of AEs, and associated clinical outcomes is available within the FAERS database. 9 As the FAERS is a publicly available and anonymized database, institutional review board approval and informed consent requirements were waived.
Data collection
We initiated data collection by utilizing the preferred terms (PTs) listed in the Medical Dictionary for Regulatory Activities (MedDRA) version 26.0. Our search spanned adverse reaction reports from the first quarter of 2004 to the fourth quarter of 2023. Specifically, we targeted adverse reactions related to ‘dementia’, resulting in the identification of a total of 31,881 adverse reactions. This comprehensive search allowed us to gather a robust dataset for further analysis. The PT codes utilized in this process are detailed in Supplemental Table 1.
Translation and organization of drug names
Given the multiple nomenclature that exists for drug names in the FAERS data, including brand names, generic names, chemical names, abbreviations, and even incorrect names of drugs, 10 we standardized all drug names to generic names before data mining. For this process, we utilized MedEx-UIMA 1.3.8, 11 a specialized natural language processing tool designed for extracting and normalizing medication information from clinical narratives. The standardization process involved the following steps:
First, extraction of drug names: extracting all reported drug names from the FAERS database, including both brand and generic names.
Second, normalization using MedEx-UIMA: the DRUG files provided by the FAERS database contains two fields related to the name of the drug, namely: ‘drugname’ and ‘prod_ai’. The ‘drugname’ and ‘prod_ai’ fields are mapped to RxNorm using MedEx-UIMA 1.3.8 so that each extracted drug name is mapped to the corresponding single active ingredient. MedEx-UIMA uses advanced algorithms to ensure accurate mapping and standardization of drug names.
Third, verification and quality control: cross-referencing a subset of normalized names manually to ensure the accuracy of the automated process and to confirm that all drug names were appropriately standardized.
To minimize the probability of false positives, we selected only ‘role_cod’ fields that play the role of ‘primary suspect (PS)’.
Statistical analysis
Following the FDA’s recommendations, we performed data cleaning on the DEMO table. Most importantly, duplicate records were excluded. If multiple records had the same CASEID (the identifier for FAERS cases), we retained the one with the latest FDA_DT (the date the FDA received the case). If records had the same CASEID and FDA_DT, we selected the one with the highest PRIMARYID (the unique identifier for the reports). After the de-duplication process, the total number of adverse reactions reported from 2004 to 2023 was 17,381,210, with 31,881 cases specifically related to dementia (Supplemental Table 2).
Descriptive analyses were employed to summarize the clinical profiles of patients with drug-induced dementia, encompassing factors such as age, sex, indication, regression, and reporting country. The top 50 drugs were identified based on the frequency of dementia-associated AE. In order to detect potential safety signals that may have gone undetected in pre-market clinical trials of drugs, we chose a case-number-based approach. 12 The method is effective for this purpose, as it allows us to quickly identify drugs with a higher-than-expected number of reports. These drugs underwent meticulous manual validation to ensure data accuracy and reliability. Subsequently, potential associations between drugs and dementia were hypothesized using disproportionality analysis, which calculates the ratio of observed drug-related adverse reactions to expected reactions, detecting imbalances above a predetermined threshold. 13 In this study, we deployed three signal detection methods to identify potential adverse drug reactions (ADRs) within the FAERS database: (a) Reporting Odds Ratio (ROR) 14 : This method identifies potential signals by calculating the odds of an AE being reported for a drug in question versus other drugs. A higher ROR suggests a more likely association between the drug and the event. (b) Proportional Reporting Ratio (PRR) 15 : The PRR evaluates the ratio of reports for a specific event with a drug versus all other drugs, with a significant value greater than 1 indicating a potential signal. (c) Bayesian Confidence Propagation Neural Network (BCPNN) 16 : The BCPNN uses Bayesian logic to compute the information component (IC), where a positive IC indicates a strong association between the drug and the AE. We aimed to leverage the distinct advantages of these techniques to enhance our detection capabilities, validate findings from multiple perspectives, and ensure a comprehensive and reliable identification of safety signals. The synergistic application of these diverse algorithms enables mutual validation, thereby reducing the occurrence of false positives. Furthermore, by optimizing thresholds and variances, we can identify a greater number of potential rare ADRs. An ADR is considered significant if it meets the following criteria: (1) The lower limit of the 95% confidence interval (CI) for ROR is greater than 1; (2) PRR is at least 2 with its 95% CI not dropping below 1; (3) IC025 value is positive. 14 The formulas and criteria are detailed in Table 1.
Summary of algorithms used for signal detection.

BCPNN, Bayesian confidence propagation neural network; CI, confidence interval; IC, information component; PRR, proportional reporting ratio; ROR, reporting odds ratio; χ2, chi-squared.
In this study, R version 4.3.1 was used for data acquisition, processing, and analysis.
Results
Descriptive analysis
Between the first quarter of 2004 and the fourth quarter of 2023, a total of 17,381,210 adverse events were reported in the FAERS database after de-duplication, of which 31,881 were related to dementia (Supplemental Table 2). As illustrated in Figure 1, the number of reports concerning ADRs associated with dementia exhibited a consistent upward trajectory from 2004 to 2023, reaching its zenith at 3272 cases in 2021. Thereafter, a modest decline in the number of reports was observed in both 2022 and 2023. The detailed characteristics of these cases are presented in Table 2. According to the data presented in Table 2, 16,953 cases (53.18%) were attributed to females, 12,954 cases (40.64%) to males, and 1971 cases (6.18%) to instances where sex values were unknown. Regarding age, 44.23% of the data did not provide age information, limiting our in-depth understanding of the relationship between age and drug-induced dementia. However, in the reports with clear data, the highest prevalence of dementia was observed among patients aged over 65 years (46.88%), followed by the age group of 41–64 years (7.74%). Conversely, a lower prevalence of dementia was noted among patients aged 19–40 years (0.91%), and those under 19 years of age (0.24%). Among these reports, only 3.52% involved patients with dementia, whereas a larger proportion of patients without dementia experienced dementia. The most commonly reported outcomes were other serious conditions (59.71%) and hospitalization (22.59%), followed by death (11.76%), disability (3.81%), and life-threatening (1.8%). These top five clinical outcomes all represent serious AEs that can significantly impact patients and their families and need to be taken seriously. The median time to onset of drug-induced dementia was day 142 postdose. The country with the highest number of reports of drug-induced dementia was the United States (47.55%), well ahead of Japan (5.36%), Canada (4.61%), the United Kingdom (3.8%), and Brazil (3.08%). Notably, these AEs were predominantly reported by nonhealth professionals (56.43%), with only 40.04% reported by health professionals.
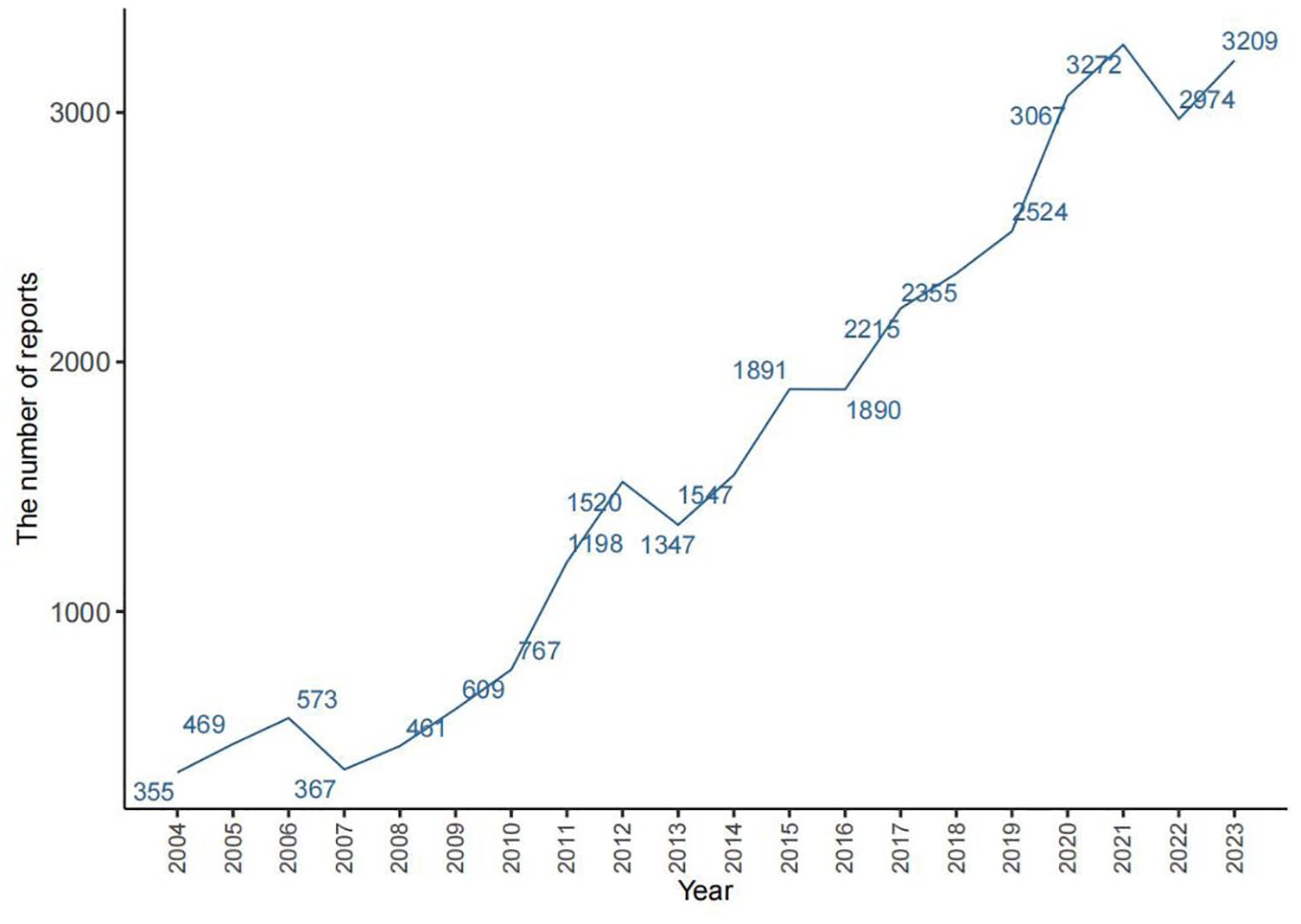
Number of reported cases of drug-induced dementia from 2004 to 2023.
Clinical characteristics of reported drug-induced dementia.

Figure 2 presents a summary of the top 50 drugs linked to dementia, ranked by the frequency of AE reports. Apixaban topped the list with 1631 reported cases, followed by rivastigmine (1094 cases), pimavanserin (863 cases), lenalidomide (834 cases), carbidopa/levodopa (776 cases), adalimumab (719 cases), insulin glargine (627 cases), quetiapine (553 cases), sacubitril/valsartan (546 cases), and etanercept (530). Of these 50 drugs, antipsychotics, antineoplastics, and antirheumatic drugs were the most prevalent, with 6 (12%) each, as well as anti-osteoporosis, hypoglycemic, and anticoagulant drugs, all at 3 (6%), and statin lipid-lowering, antihypertensive, and antimultiple sclerosis drugs at two each, each accounting for 4% of the total. Of these 50 drugs, there is only one antidementia drug: rivastigmine. Notably, of these 50 drugs associated with dementia-related adverse reactions, only five explicitly indicated dementia risks in their specifications: pregabalin, atorvastatin, enzalutamide, oxycodone, and rosuvastatin, leaving the remaining 45 without such warnings.
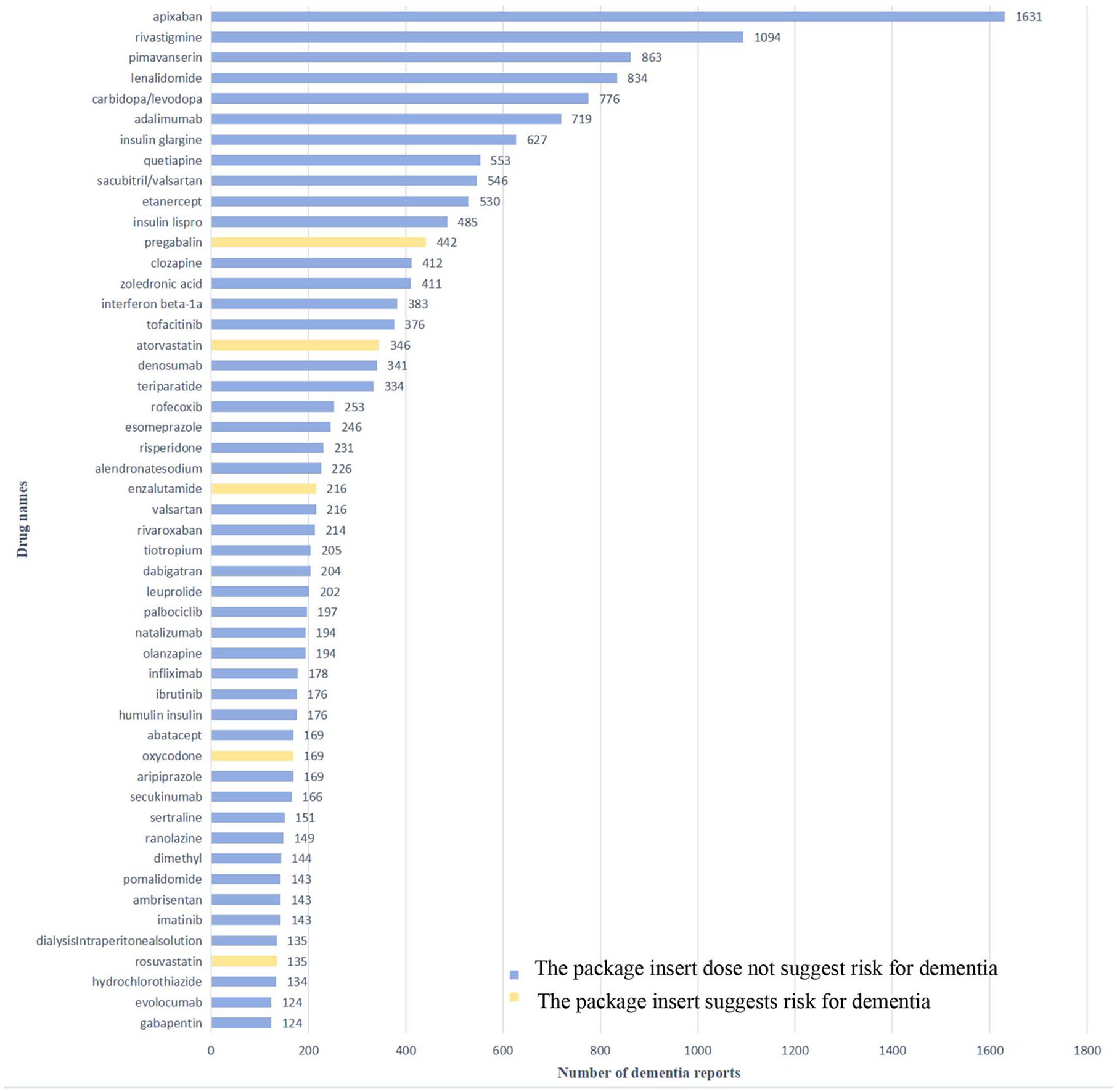
Top 50 drugs with the highest number of reported dementia.
Disproportionality analysis
Table 3 presents the top 50 drugs with the highest signal intensity according to the ROR criteria and the top 10 drugs are rivastigmine (ROR = 46.3, PRR = 43.39, χ2 = 43,813.4, IC = 5.39), nicergoline (ROR = 43.34, PRR = 40.69, χ2 = 155.08, IC = 5.35), aducanumab (ROR = 30.19, PRR = 28.89, χ2 = 592.9, IC = 4.85), amlodipine/atorvastatin (ROR = 22.68, PRR = 21.95, χ2 = 180.17, IC = 4.46), dihydroergocristine (ROR = 22.57, PRR = 21.85, χ2 = 99.6, IC = 4.45), famotidine (ROR = 22.42, PRR = 21.7, χ2 = 98.88, IC = 4.44), timolol (ROR = 20.98, PRR = 20.35, χ2 = 257.89, IC = 4.35), vildagliptin (ROR = 18.57, PRR = 18.09, χ2 = 113.14, IC = 4.18), triptorelin (ROR = 18.45, PRR = 17.97, χ2 = 1823.5, IC = 4.16) and human insulin (ROR = 17.57, PRR = 17.13, χ2 = 76.06, IC = 4.1). Among the top 50 drugs, seven were categorized as antidementia medications, while the remaining drugs fell under the classification of nonantidementia medications; however, only two drugs had a possible risk of dementia noted in the label and these were valproate and tramadol. In addition, of these 50 drugs, anti-Parkinson’s drugs were the most common of the 50 drugs with 7 (14%), followed by antihypertensive drugs with 6 (12%), hypoglycemic drugs with 3 (6%), and anticholinergic drugs with 2 (4%). In addition, to facilitate clinical identification of as many relevant drugs as possible that may cause dementia, we listed 110 major drugs according to the criteria 14 (Table 1) for signal detection (Supplemental Table 3).
Top 50 drugs for signal strength.

CI, confidence interval; IC, information component; IC025, the lower limit of 95% CI of the IC; PRR, proportional reporting ratio; ROR, reporting odds ratio; χ2, chi-squared; Y, yes; N, not.
Antidementia drug.
Discussion
To the best of our knowledge, this study is the first and largest FAERS database-based study assessing drug-induced dementia. We elucidate the drugs most closely associated with pharmacological dementia and clarify the clinical features of these AEs. The majority of these medications lack explicit warnings for dementia in their package inserts, and people are not aware of their dementia risks.
Dementia is an easily overlooked AE in medication, but it can cause serious harm and even death. Our study demonstrates that the risk of drug-induced dementia escalates with advancing age. In general, with increasing age, neurodegenerative changes associated with aging make the brain more susceptible to adverse effects of certain medications that centrally affect cognitive function that may not manifest in younger individuals. Furthermore, our findings indicate a higher risk of dementia in women compared to men, consistent with the results reported by Beydoun et al. 17 The elevated risk of dementia in women compared to men may be attributable to various factors, including lifestyle and socio-economic influences, among others. In this study, we found that among the patients reporting drug-induced dementia, except for 34.02% who did not report indications, which somewhat limited our analysis, only 3.52% of patients reporting drug-induced dementia had previous dementia, while 62.46% of patients reporting drug-induced dementia had no previous dementia. Most of the reported clinical outcomes were severe, with death in 11.76% of patients and hospitalization, disability, and other serious consequences in 86.11% of patients. Therefore, regardless of previous dementia history, healthcare professionals should be vigilant about the risk of dementia associated with medications, especially in patients with known risk factors for dementia.
The top 50 drugs with the most common reports of drug-induced dementia
Of the top 50 drugs with the most common reports of drug-induced dementia, only pregabalin, atorvastatin, enzalutamide, oxycodone, and rosuvastatin were mentioned in the inserts as potentially having a risk of dementia, with cognitive dysfunction being a common adverse reaction to pregabalin, mainly in the form of memory deficits and language deficits, while dementia was more rare with atorvastatin, enzalutamide, oxycodone, and rosuvastatin, which can be recovered after discontinuation of the drug.
Apixaban
As shown in Figure 2, apixaban, the drug with the most reported AEs (1631 cases) in dementia, is a nonvitamin K antagonist oral anticoagulant commonly used for oral anticoagulation in patients with atrial fibrillation (AF). 18 AF is an independent risk factor for dementia, 19 likely due to its contribution to microembolism and asymptomatic cerebral infarction. 20 Oral anticoagulants may prevent these conditions by inhibiting coagulation factors, 21 which may explain why multiple studies have shown that oral anticoagulants reduce the risk of dementia.20,22,23 The number of reports of drug-induced dementia in the FAERS database, in turn, is influenced by a range of factors, such as high levels of drug use and recommended levels of therapeutic dosages. Approximately one in six older adults receives oral anticoagulants, 24 with AF being the most common reason for such therapy. 19 Thus, apixaban was reported as the drug with the most dementia AEs, probably because AF is a risk factor for dementia, and apixaban is widely used in the clinical practice. Overall, apixaban is considered safe,22,25 and patients with dementia who have AF also benefit from taking it. 26 Additionally, elderly AF patients gain the greatest benefit from apixaban compared to other oral anticoagulants. 27
Anticancer, antipsychotic, and antirheumatic drugs
Anticancer, antipsychotic, and antirheumatic drugs were the drug classes with the highest number of reported cases of drug-induced dementia, with each accounting for six of the top 50 drugs reporting the highest number of cases.
Anticancer
Cancer treatment is known to have a long-term negative impact on cognitive function, often referred to as “chemo brain” or “chemo fog,” 28 although the exact mechanisms of cognitive decline remain unclear. Potential contributing factors include oxidative damage, DNA damage, apoptosis, hormonal changes, neurotoxicity, dysregulation and reduction of neurotransmitters, increased phosphorylation of tau proteins, inhibition of cerebral blood flow, and a reduction in the number of cortical spines and dendrites.29–35 Enzalutamide, a second-generation androgen receptor (AR) inhibitor, has been shown to reduce cognitive function in patients36,37 and is listed in its specification as having a risk of dementia. This may be related to its blood–brain barrier permeability and direct AR inhibitor activity. 38 Due to factors such as survival of cancer patients, objective clinical diagnostic tools for dementia, and short follow-up period, there are relatively few studies related to anticancer drugs and dementia, and no literature was retrieved on the remaining five drugs causing dementia. Neuropsychiatric symptoms of dementia (NPS) are prevalent in dementia, 39 usually manifesting as delusions and/or hallucinations, and may be associated with hyperactivation of vertebral neurons within the visual cortex and hyperactivation of limbic pathways in the midbrain.
It is clear that the number of reports of pharmacologic dementia is influenced by many factors, including social use of medications, disease interactions, progression of the disease itself, co-morbid conditions, and other factors. Therefore, the number of AE reports does not accurately reflect the relationship between medications and AE.
Disproportionality analysis
Following disproportionality analysis, we identified the 50 drugs exhibiting the strongest signals and scrutinized their package inserts. Only two drugs describe the risk of dementia in their labeling, the antiepileptic drug valproate and the analgesic tramadol, with dementia being a common AE of valproate and a rare AE of tramadol.
Dementia is the most common cause of seizures and epilepsy, and epilepsy can also be comorbid with dementia; the two disorders are a two-way association. 52 Valproate is a potent broad-spectrum antiepileptic drug, and a review 53 that included 11 papers (study populations with normal cognition at baseline and dementia after valproate treatment) showed that valproate-induced dementia is a reversible form of cognitive decline that was first reported in 1986. 54 This reversible cognitive decline can be reversed after valproate dose reduction or discontinuation, but is particularly important to diagnose because it is not easily recognized and is mistaken for neurodegenerative dementia. 53 Therefore, medical personnel should pay more attention to and recognize these patients in their clinical work, and strengthen the questioning of medication history to reduce the leakage and misdiagnosis.
The three most common drug classes associated with AEs in dementia
Among the 50 drugs with the strongest signals in the drug sensitivity analysis, seven were antidementia and anti-Parkinson’s disease drugs, and six were antihypertensive drugs. This finding prompts a critical examination of the pharmacodynamics, patient populations, and potential biases inherent in the reporting system.
Antidementia drugs
It might initially seem paradoxical that antidementia drugs, which are intended to mitigate the symptoms of dementia, are associated with the onset of the condition. However, this association could be attributed to several factors. First, protopathic bias: patients who are in the early, undiagnosed stages of dementia might be prescribed these drugs as their symptoms emerge, thus the drugs appear in the database as associated with dementia onset rather than progression. Second, disease progression: antidementia drugs might be reported because patients on these medications are inherently more likely to be diagnosed with dementia due to the progression of their underlying condition. Third, reporting bias: patients and healthcare providers are more likely to report AEs when a patient is on medications specifically for dementia, as they are already focused on this condition. However, it has been shown that acetylcholinesterase inhibitors (AChEI; rivastigmine, donepezil, and galantamine) report many adverse outcomes in dementia patients, with rivastigmine having a higher frequency of reported deaths compared to donepezil and galantamine. 57 This may be related to the fact that rivastigmine is the only AChEI available in a transdermal patch form, 58 and inappropriate use of this mode of administration can lead to high-dose exposure, severe AEs, and even death. 59 AChEI and memantine are effective treatments for dementia, 60 but we need to consider the risks and proper use of these medications to choose the safest, most effective medications for our patients.
Anti-Parkinson’s drugs
The strong association between anti-Parkinson’s drugs and dementia is also noteworthy. First, comorbidities: Parkinson’s disease (PD) and dementia share several common pathological mechanisms. Cognitive impairment is a frequent comorbidity in PD, 61 which might lead to higher reporting of dementia among patients taking these medications. Second, medication side effects: some anti-Parkinson’s medications, particularly those affecting dopamine pathways, can have cognitive side effects that might be misclassified or contribute to dementia-like symptoms. 62 Third, advanced disease stages: as PD progresses, the likelihood of dementia increases, 62 and thus the prescription of anti-Parkinson’s drugs in these stages may coincide with the emergence of dementia symptoms.
Antihypertensive drugs
First, vascular contributions: hypertension is a known risk factor for dementia.63,64 The use of antihypertensive drugs in patients at risk for vascular dementia could result in these medications being flagged in dementia reports. Second, polypharmacy: patients on anti-hypertensive medications are often older and have multiple comorbidities, increasing the risk of dementia and the likelihood of polypharmacy, which complicates attributing causation to a single drug class. Similarly, glucose-lowering drugs, antiplatelet aggregating drugs, and anticoagulants.
The remaining drugs also generated new signals, including H2 receptor antagonists (famotidine), antiarrhythmics (ranolazine), antihistamines (promethazine), and positive inotropic agents (digoxin). Despite these drugs being on the market for a long time, there are few studies examining their association with dementia. Additionally, according to the detection criteria for signaling, dementia was reported at least three times as a signal, which may lead to false-positive results. Table 3 shows that these drugs do not report a large number of dementia cases. Therefore, further research is needed to determine whether the risk of dementia should be indicated in the labeling of these drugs.
This real-world pharmacovigilance study was designed to identify drugs that may be associated with dementia. While the associations found are significant, they should be interpreted with caution. This study has several limitations that should be acknowledged. First, incomplete demographic data: a significant proportion of the FAERS reports lacked detailed demographic information. Specifically, 44.23% of reports did not include age data, and 6.18% lacked gender information. This limited our ability to analyze age and gender as covariates, which are known to influence dementia risk. Consequently, while we observed a higher prevalence of dementia in older individuals and women, these findings should be interpreted with caution. Second, there is the issue of causation versus correlation: FAERS data can highlight correlations but cannot establish causation. The associations identified require further investigation through prospective cohort studies and randomized controlled trials to elucidate the underlying mechanisms and determine whether these drugs contribute to dementia development or if the associations are due to underlying disease mechanisms. Third, reporting bias is a concern: the spontaneous reporting system of FAERS is subject to underreporting, selective reporting, and reporting bias, which can affect perceptions of the strength of associations. Fourth, confounding variables such as patients’ general health status, education level, cognitive function, comorbidities, and complications may have influenced the observed relationships. Finally, due to the lack of a known denominator (number of prescriptions for the drug), it was not possible to calculate the incidence rate using the FAERS database. Based on these limitations, further research is needed to determine whether the risk of dementia should be stated in the labeling of these drugs. Despite these limitations, the FAERS database remains an important tool for pharmacovigilance, providing valuable information for clinical practice and further research efforts.
Conclusion
In this study, we conducted a comprehensive assessment of dementia reports and their corresponding medications using data from the FAERS database. While only a few medications explicitly list dementia risk in their package inserts, our findings emphasize the importance of ongoing monitoring and assessment of drug safety, especially for medications commonly used by people at high risk for dementia. Healthcare providers should raise awareness of the potential cognitive effects of these medications and the importance of individualizing patient instructions. Additionally, it is crucial to review relevant medication histories (e.g., valproate, anticancer medications) for patients with suspected dementia to reduce misdiagnosis. Furthermore, it is imperative to explore and confirm the potential associations and pathogenesis between these drugs and dementia through further research.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251315137 – Supplemental material for Drug-induced dementia: a real-world pharmacovigilance study using the FDA Adverse Event Reporting System database
Supplemental material, sj-docx-1-tan-10.1177_17562864251315137 for Drug-induced dementia: a real-world pharmacovigilance study using the FDA Adverse Event Reporting System database by Lisi Xu, Ruonan Zhang, Xiaolin Zhang, Xiuli Shang and Daifa Huang in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
