Abstract
Background:
Despite their potential usefulness as biomarkers, no study has investigated the interactions between cerebrospinal fluid (CSF) changes of neurofilament light chain (NfL), glial fibrillary acidic protein (GFAP), growth differentiation factor 15 (GDF-15), transactive response DNA binding protein (TDP-43) and interleukin-6 (IL-6) and the core AD CSF biomarkers in the same cohort of AD patients.
Objectives:
The aim of this pilot study is to evaluate the CSF levels of these analytes in patients with AD and assess their clinical relevance in this neurological condition.
Design:
Cross-sectional study.
Methods:
We assessed the levels of NfL, GFAP, GDF-15, TDP-43 and IL-6 in the CSF samples of 52 early AD patients and evaluated their partial reciprocal correlations and those with Abeta42, p-Tau, t-Tau and Mini-Mental State Examination (MMSE), always adding age, sex and educational level as covariates.
Results:
MMSE score showed a positive correlation with the Aβ 1-42 concentrations (r = 0.485; p < 0.001), and a negative correlation with GDF-15 concentrations (r = −0.418; p = 0.002). IL-6 concentrations showed a positive correlation with NfL concentrations (r = 0.312; p = 0.026) and a negative correlation with TDP-43 concentrations (r = −0.322; p = 0.021). TDP-43 concentrations showed a positive correlation with GFAP (r = 0.33, p = 0.018). The mediation analysis suggests that the association between GDF-15 and MMSE is primarily mediated by Aβ 1-42. CSF GDF-15 concentrations were higher in AD patients with low Aβ 1-42 concentrations than those with high Aβ 1-42 concentrations (p < 0.001).
Conclusion:
Our findings highlight that CSF IL-6 levels correlate positively with markers of neuronal damage. CSF TDP-43 levels significantly correlated with GFAP, suggesting a potential link with reactive gliosis and astrocyte activation. In addition, while CSF GDF-15 levels negatively correlate with MMSE scores, mediation analysis revealed that this association is primarily indirect and mediated through Aβ 1-42 levels.
Introduction
Alzheimer’s disease (AD) is the predominant form of neurodegenerative dementia, exhibiting a progressive course that often starts with mild memory impairment, eventually significantly impacting daily functioning. 1 The pathology associated with AD is characterized by the accumulation of extracellular amyloid beta (Aβ), the formation of pathological intracellular tau protein tangles and the loss of neurons. 2
Numerous hypotheses have been put forward to elucidate the neurodegenerative mechanism underlying AD pathology, seeking to surpass the conventional ‘amyloid cascade’ 3 and ‘tau propagation’ 4 hypotheses, which prove insufficient to explain the multifaceted nature and heterogeneity of the disease. From this perspective, the hypotheses of ‘immune activation’ 5 and ‘mitochondrial dysfunction’ 6 appear highly promising, as they succeed in explaining specific aspects of the neurodegenerative process and may constitute fundamental therapeutic targets soon. 7 It is well documented that the chronic low-grade sterile inflammatory activation associated with ageing, known as ‘inflammageing’, plays a significant role in the pathological changes observed in AD. 8 In this context, the identification of molecules identified as biomarkers of inflammageing represents a highly active area of investigation. Among these, growth differentiation factor 15 (GDF-15) 9 and interleukin-6 (IL-6) 10 stand out as two well-characterized biomarkers associated with this process.
IL-6 plays a pivotal role in immune responses, inflammation and numerous physiological processes. Its synthesis is regulated by several transcription factors, including nuclear factor kappa beta (NF-κB), activator protein (AP)-1, CCAAT/enhancer binding protein (C/EBP) and cAMP response element-binding protein,11,12 and this interleukin has a critical role on different aspects of immune response and inflammation 13 becoming deregulated in several ageing-related diseases. 14
Concerning AD pathogenesis, IL-6 has been linked to Aβ aggregation and the emergence of hyperphosphorylated tau in the brain. 15 More broadly, IL-6 is involved in regulating neuronal development and adult neurogenesis, contributing to the formation of new neurons and glial cells from neural stem cells. 16 Intriguingly, IL-6 gene polymorphisms have been shown to modify the risk of late-onset AD. 17
GDF-15, a member of the TGF-β superfamily, plays a vital role in various processes such as development, cell proliferation, differentiation and tissue repair. It is abundant in both the central and peripheral nervous systems, with particularly elevated concentrations in the choroid plexus. This region secretes GDF-15 into the cerebrospinal fluid (CSF), being also damaged neurons and microglial cells responsible for its production within the central nervous system (CNS). 18 Widely studied in various neurological and non-neurological pathologies as a molecule responding to mitochondrial stress, the expression of GDF-15 increases with age and age-related pathologies and has a significant role in the immune response, notably dampening innate immunity. 19
Whether GDF-15 circulating levels differ between AD patients and age-matched healthy subjects or whether increased levels of this protein increase the risk of AD is still a matter of debate. Research on GDF-15 in the context of AD has predominantly examined its presence in the bloodstream, while studies investigating its concentration in the CSF of AD patients and its correlation with key inflammatory markers remain scarce. A previous study found no difference in CSF levels of GDF-15 in AD patients compared to healthy subjects. 20 However, the authors documented a positive correlation of GDF15 with IL-6 gene expression in the cortex of AD patients.
Additionally, aberrant protein accumulation, extending beyond Aβ and Tau, is another contributing factor to neurodegeneration. In this context, the protein transactive response DNA binding protein (TDP-43) pathology in AD is starting to become a matter of investigation. Being widely recognized for its role in amyotrophic lateral sclerosis, TDP-43 has also been shown to play a role in AD, where it tends to form aggregates in a significant percentage of patients 21 and seems to influence clinical presentation. 22 TDP-43, originally recognized for its role in RNA processing, has also emerged as a key player in immunity and neuroinflammation. 23
NfL serves as a valuable biomarker for detecting neuronal degeneration 24 and its CSF and serum concentrations are elevated in various neurological conditions. 25 Glial fibrillary acidic protein (GFAP), an intermediate filament abundantly expressed in astrocytes, has gained prominence as a reliable biomarker for astrocytic activation and injury, and CSF GFAP has emerged as a potential biomarker in AD. 26
Here, we aimed to study the CSF levels of IL-6, GDF-15, TDP-43, GFAP and NfL, their reciprocal correlations and their correlations with core AD biomarkers (i.e. amyloid, tau and p-tau levels), as well as with cognitive impairment, measured by the Mini-Mental State Examination (MMSE), in a cohort of AD patients. This analysis aims to improve our understanding of the role of these biomarkers in AD by investigating their pathophysiological and staging utility, shedding light on disease mechanisms like neuroinflammation and mitochondrial stress and exploring their connection to cognitive decline.
Methods
Study design and population
This cross-sectional pilot study was conducted between January 2022 and June 2023. We enrolled a cohort of consecutive AD patients who came to the attention of our cognitive disorders clinics (University of Siena and University of Genova) for memory complaints. All enrolled patients had a diagnosis of AD based on the 2018 National Institute on Ageing and Alzheimer’s Association guidelines, 27 and we focused on individuals with preserved Activities of Daily Living, indicating a good level of functional independence, which is typical of the very early clinical stages of the disease. 28 They underwent lumbar puncture for diagnostic purposes and the clinical workup included detailed past medical history, general and neurological examination, and a neurocognitive assessment including the MMSE that was age and educational level-corrected as per Italian population normative standards. 29 The CSF biomarkers analyzed for diagnostic purposes included Aβ 1-42, Aβ 1-40, total Tau (t-Tau) and tau phosphorylated at position 181 (P-Tau181). The cut-off value for CSF Aβ 1-42 was 450 pg/ml. However, in cases where the Aβ 1-42 concentration was above this threshold, we considered the CSF Aβ42/Aβ40 ratio, and, where available, the results from amyloid PET scans to reach the diagnosis. CSF biomarker values of the Aβ 1-42 ratio 1-42/1-40, Aβ 1-42 and p-Tau of AD patients, grouped by university centre are reported in Supplemental Materials.
The exclusion criteria were
Evidence of other potential causes of cognitive decline (e.g. subdural hematoma, malignant brain tumour);
Patients treated with immunosuppressant or immunomodulatory drugs in the previous 3 months;
Patients with a history of infectious diseases in the previous 6 months;
Past medical history suggestive of severe psychiatric comorbidities;
Pregnant or lactating patients;
Terminal neoplastic diseases with little chance of survival beyond 6 months;
Past medical history indicative of autoimmune disease;
Patients with a family history of autosomal dominant AD, those with at least three affected individuals with dementia in two or more generations and finally those with a family history of other types of genetic dementia;
Patients with a family history or any monogenic neurological disease;
History of cerebrovascular disease (i.e. stroke episodes).
During the study, the principles of Good Clinical Practice of the International Conference on Harmonization (ICH), the ‘Declaration of Helsinki’ and national and international ethical guidelines were strictly followed, the study was approved by the Local Ethics Committee of Regione Toscana Area Vasta Sud Est (protocol number 24397), and all patients signed the informed consent form.
CSF samples
CSF samples (10–12 ml) were obtained through lumbar puncture in the L3/L4 or L4/L5 interspace between 8 and 10 a.m. after an overnight fasting period, immediately collected in sterile polypropylene tubes and gently mixed to avoid possible gradient effects. All the samples were handled according to the Alzheimer’s Association unified standardized pre-analytical protocol. 30 Each CSF sample underwent measurements of Aβ 1-42, t-Tau and p-Tau181 using the validated LUMIPULSE automated immunoassay for the measurement of core AD biomarkers in CSF. The assays were performed following the manufacturer’s instructions with the LUMIPULSE® G600II instrument (Fujirebio, Ghent, Belgium) which is a cartridge-based system that uses monoclonal antibody-coated beads for capture and monoclonal antibodies for detection.
In parallel with the analysis of core biomarkers, primarily conducted for diagnostic purposes, a volume of 2 ml per patient of CSF was initially centrifuged at 2000 rpm for 10 min at 4°C, aliquoted (0.5 ml) into polypropylene tubes and then stored at −80°C awaiting further analyses. All analyses were performed at the laboratory of the Centre for Precision and Translation Medicine, University of Siena, Italy.
NfL and GFAP assay
The concentrations of NfL and GFAP were determined in patients CSF samples using commercially available immunoassay kits for NfL and GFAP – Simoa™ assay Neurology 2-Plex B (GFAP, NfL) Assay Kit (Catalog #103520; Quanterix, Billerica, MA, USA). The assays were conducted on the semi-automated ultrasensitive SR-X Biomarker Detection System (Quanterix). Samples were diluted at a ratio of 1:40 and randomly distributed on 96-well plates. Quality control (QC) samples, provided with the kit, exhibited concentrations within the predefined range and the coefficient of variance across the plates was maintained below 10%. All samples were analyzed blindly under alpha-numeric codes, and diagnostic codes were disclosed only after QC-verified NfL and GFAP concentrations were reported to the database manager. Concentrations were measured in pg/ml and documented in the database.
TDP-43 assay
The concentrations of TDP-43 were quantified in patients CSF samples using the commercially available immunoassay kit (Catalog #103293; Quanterix). The assays were conducted on the semi-automated ultrasensitive SR-X Biomarker Detection System (Quanterix). Samples were diluted at a ratio of 1:40 and randomly distributed on 96-well plates. QC samples, provided with the kit, exhibited concentrations within the predefined range, and the coefficient of variance across the plates was maintained below 10%. All samples were analyzed blindly under alpha-numeric codes, and diagnostic codes were disclosed only after QC-verified TDP-43 concentrations were reported to the database manager. The Simoa TDP-43 assay incorporates antibodies targeting amino acids 203–209 and the C-terminal region. This assay, as per the manufacturer, is designed to identify both full-length and pathologically truncated forms of the TDP-43 protein. Concentrations were measured in pg/ml and documented in the database.
GDF-15 assay
GDF-15 levels in each patient CSF sample were assessed using the GDF-15 Human ELISA kit (Bio-Techne, USA R&D Systems, Inc. Minneapolis, USA). The readings were obtained on an iMark Absorbance Microplate Reader (Bio-Rad, Milan, Italy), following the manufacturer’s instructions. Samples were diluted at a ratio of 1:2 and randomly distributed on the plates. Concentrations were measured in pg/ml and recorded in the database.
IL-6 assay
Concentrations of IL-6 in the CSF were determined using a multiplex bead-based flow cytometry assay (LEGENDplex HU Essential Immune Response Panel; BioLegend, San Diego, CA, USA). The FACSCanto II flow cytometer and LEGENDplex™ version 8.0 software (BioLegend) were utilized for sample analysis. Samples were diluted at a ratio of 1:2 and randomly distributed on the plates. Concentrations were measured in pg/ml and recorded in the database. Before analysis, the cytometer underwent calibration using set-up beads according to the manufacturer’s protocol.
Statistical analysis
Data were summarized in terms of number of patients (percentage, frequency) and median (minimum-maximum). Group differences were assessed by running two different tests: a Chi-squared test was used for testing the null hypothesis of independence between sex and centre, while a series of t-tests with Welch’s approximation was run for the other comparisons. The Shapiro–Wilk test was performed to test the null hypothesis of univariate normality. In line with published studies, NfL data were log10 transformed. 31 All other variables were kept non-transformed.
We divided our cohort of patients into two groups: a ‘low Aβ’ group (34 patients), characterized by a CSF Aβ 1-42 level below the threshold of 450 pg/ml, and a ‘high Aβ’ group (18 patients), defined by CSF Aβ 1-42 concentrations above this threshold.
The importance of age, particularly when analyzing not only NfL 32 and IL-6 33 but also GDF-15 19 concentrations has been already highlighted. Non-parametric partial correlation (Spearman’s correlation) with age as a control variable was thus performed. Partial correlation analyses between IL-6, GDF-15, TDP-43, NfL, GFAP, Abeta42, p-Tau, t-Tau, MMSE were performed, always adding age, sex and educational level as covariates.
If a variable of interest was significantly correlated with more than one variable, mediation analysis was used. Particularly, we tested two different approaches using R. First, we ran standard mediation analysis, by using the psych package, along with the mediate function, specifying 5000 bootstrap iterations for parameter estimation. This function uses Ordinary Least Squares as an estimator, thereby following the same assumptions of linear models. Considering notable deviations from normality, a more robust methodology was implemented as well. Specifically, we used the robmed package, employing the test_mediation function with 5000 bootstrap iterations for parameter estimation. This function integrates the method of moments estimator with the fast-and-robust algorithm for bootstrap analysis.
For all the aforementioned analyses a value of p < 0.05 was considered statistically significant. Analysis results and graphs were generated with Jamovi software, Version 2.3.28.0, and with R, Version 4.3.2.
Results
CSF samples of the 52 AD patients were collected, and the analytes were assessed at the laboratory of the Centre of Precision and Translation Medicine, University of Siena, Italy. Demographic and clinical features of patients are summarized in Table 1. Twenty-four patients were recruited in Genova and 28 patients in Siena. The median age of patients with AD was 74.3 years (minimum-maximum 54–84.6) and 35% were male. Considering patients from each centre separately, a statistically significant difference was observed regarding the severity of cognitive impairment assessed by the MMSE score (Table 1).
Descriptive table of demographic and neurocognitive data.

MMSE, Mini-mental State Examination.
Age and sex did not significantly differ between the patients recruited in the two Centres (Table 1).
Table 2 summarizes the median values and the 25th and 75th percentiles of CSF concentrations of all the analytes tested.
Descriptive table of analytes.

Aβ 1-42, beta-amyloid 1-42; GDF-15, growth differentiation factor-15; GFAP, glial fibrillary acidic protein; IL-6, interleukin-6; NfL, neurofilament light chain; pTAU, TAU phosphorylated; TDP-43, transactive response DNA binding protein.
We did not detect a significant correlation between baseline CSF tau levels and cognitive impairment in our cohort.
Comparing the CSF levels of the biomarkers studied, we found that GDF-15 concentrations were higher in the ‘low Aβ’ group (20F, median age 74.0 years, minimum-maximum 54–83.2) than in the ‘high Aβ’ group (13F, median age 74.8 years; minimum-maximum 65.2–84.6) (median Aβ 1-42 in the ‘low Aβ’ 500 pg/ml, minimum-maximum 85–629 pg/ml; median Aβ 1-42 in the ‘high Aβ’ 270 pg/ml, minimum-maximum 23–739 pg/ml; p < 0.001). These results are summarized in Figure 1.
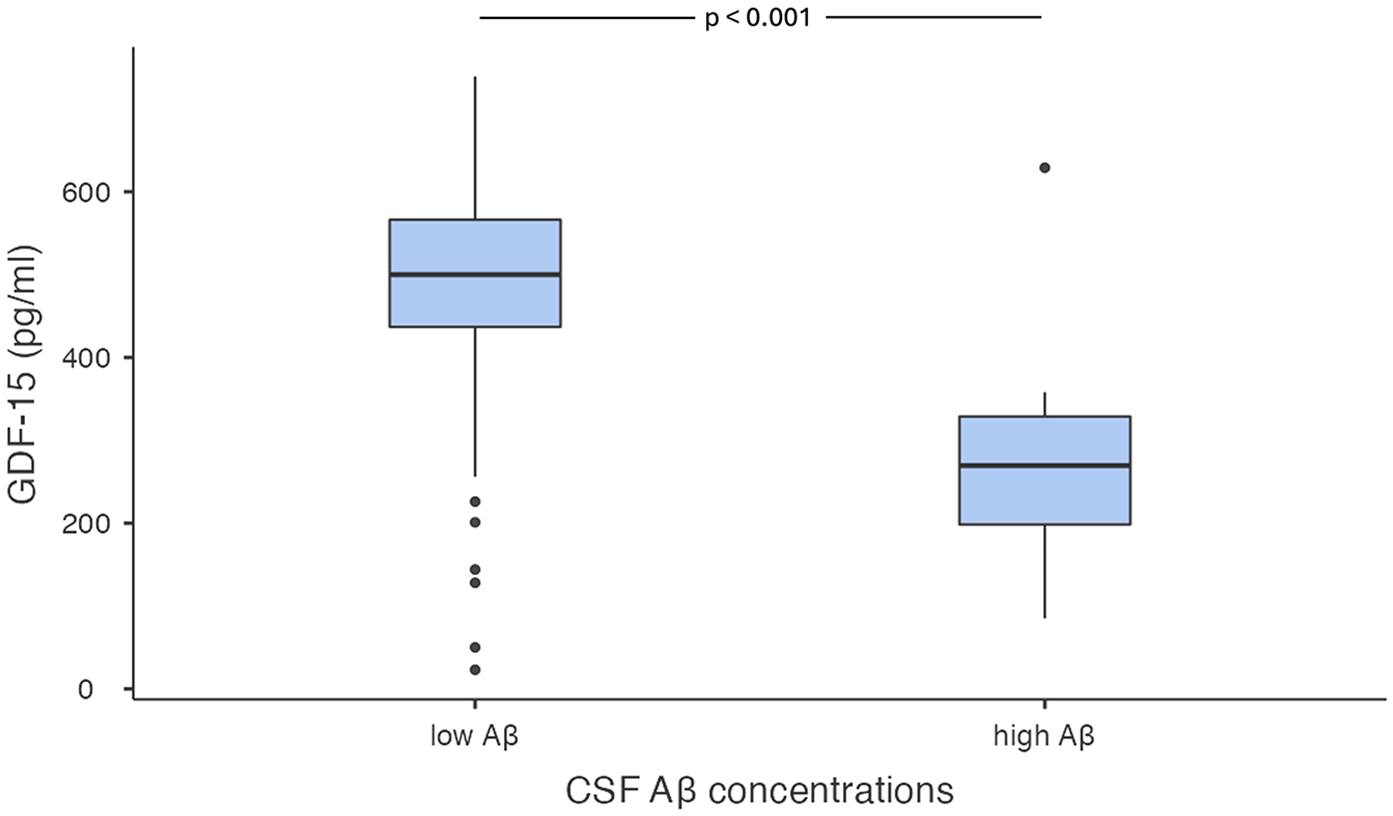
CSF levels of GDF-15 values in the ‘low Aβ’ and the ‘high Aβ’ groups of patients with Alzheimer’s disease. Box plots express the first (Q1) and third (Q3) quartiles by the upper and lower horizontal lines in a rectangular box, in which there is a horizontal line showing the median. The whiskers extend upwards and downwards to the highest or lowest observation within the upper (Q3 + 1.5 × IQR) and lower (Q1 − 1.5 × IQR) limits. p Values indicate statistical significance between the different groups.
Correlations of the concentrations of the analytes in AD patients
MMSE score showed a positive correlation with the CSF Aβ 1-42 concentrations (r = 0.48; p < 0.001), and a negative correlation with CSF GDF-15 concentrations (r = −0.41; p = 0.003). CSF IL-6 concentrations showed a positive correlation with CSF NfL concentrations (r = 0.30; p = 0.037) and a negative correlation with CSF TDP-43 concentrations (r = −0.31; p = 0.028). Additionally, TDP-43 concentrations showed a positive correlation with GFAP (r = 0.33, p = 0.018). All data correlations are included in Table 3.
Descriptive table of all results of correlation analysis.
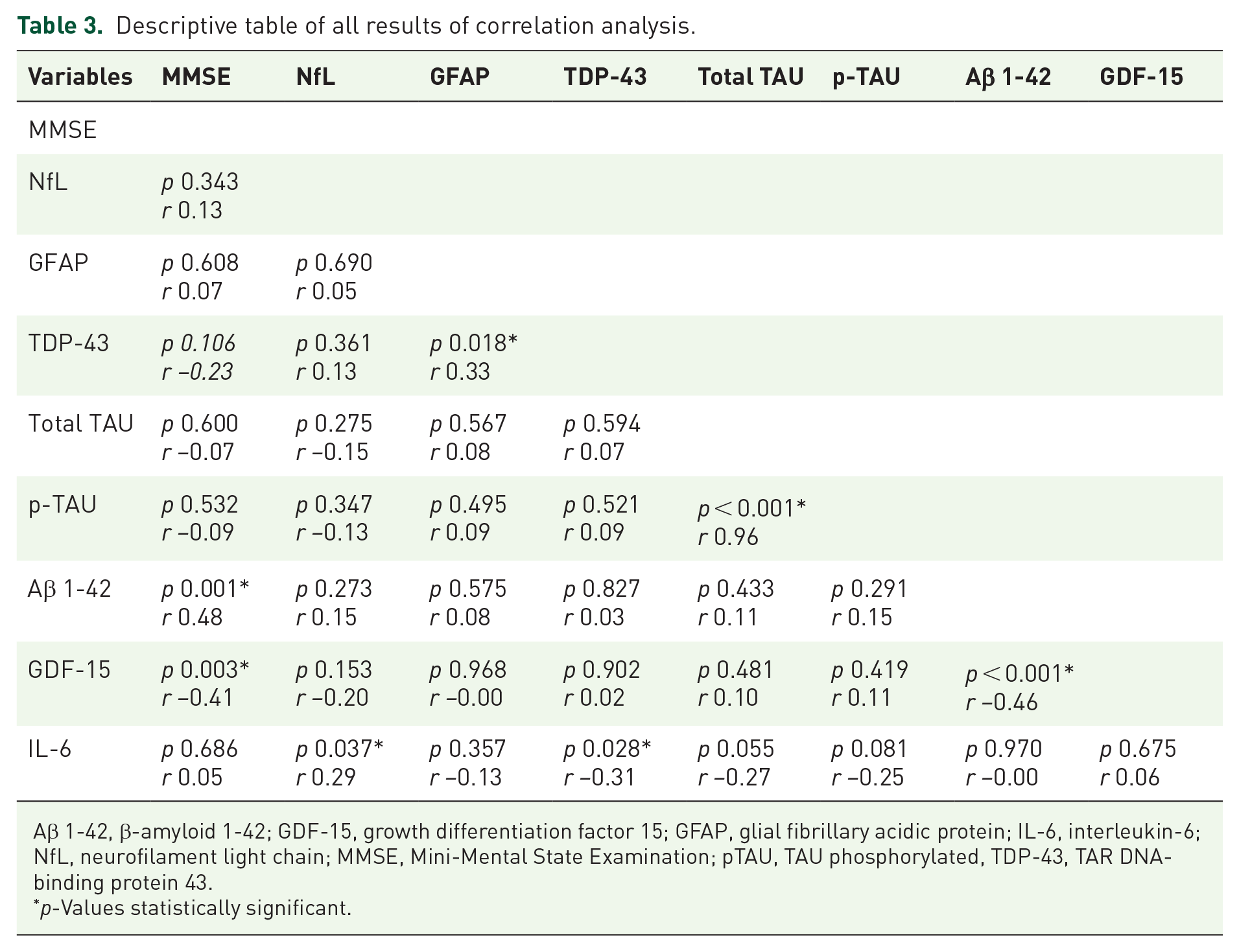
Aβ 1-42, β-amyloid 1-42; GDF-15, growth differentiation factor 15; GFAP, glial fibrillary acidic protein; IL-6, interleukin-6; NfL, neurofilament light chain; MMSE, Mini-Mental State Examination; pTAU, TAU phosphorylated, TDP-43, TAR DNA-binding protein 43.
p-Values statistically significant.
Mediation analysis
Regarding mediation analysis, we tested whether the relationship between Aβ 1-42 and MMSE was mediated by the effect of IL-6 (p = 0.525), TDP-43 (p = 0.543), GFAP (p = 0.461), none of which showed a mediation effect. Further analysis of the potential mediation of the relationship between Aβ 1-42 and MMSE via GDF-15 was also not statistically significant (p = 0.376). When examining the components of this indirect effect, the relationship between Aβ 1-42 and GDF-15 was statistically significant (p < 0.001), while the relationship between GDF-15 and MMSE was not (p = 0.359). Regarding the direct effect of Aβ 1-42 on MMSE, a significant effect was observed (p = 0.003). Lastly, the total effect (including both direct and indirect effects) of Aβ 1-42 on MMSE was highly significant (p < 0.001; Figure 2). Moreover, when we explored the relation between GDF-15 and MMSE via Aβ 1-42, mediation analysis showed that the indirect effect of GDF-15 on MMSE mediated by Aβ 1-42 was significant (p = 0.024). Examining the components of the indirect effect, a significant effect was found from GDF-15 to Aβ 1-42 (p < 0.001) and from Aβ 1-42 to MMSE (p = 0.003). The direct effect of GDF-15 on MMSE was not significant (p = 0.359). However, the total effect of GDF-15 on MMSE, including both the direct and indirect effects, was significant (p = 0.021). These findings suggest that the association between GDF-15 and MMSE is primarily mediated by Aβ 1-42 (Figure 2).
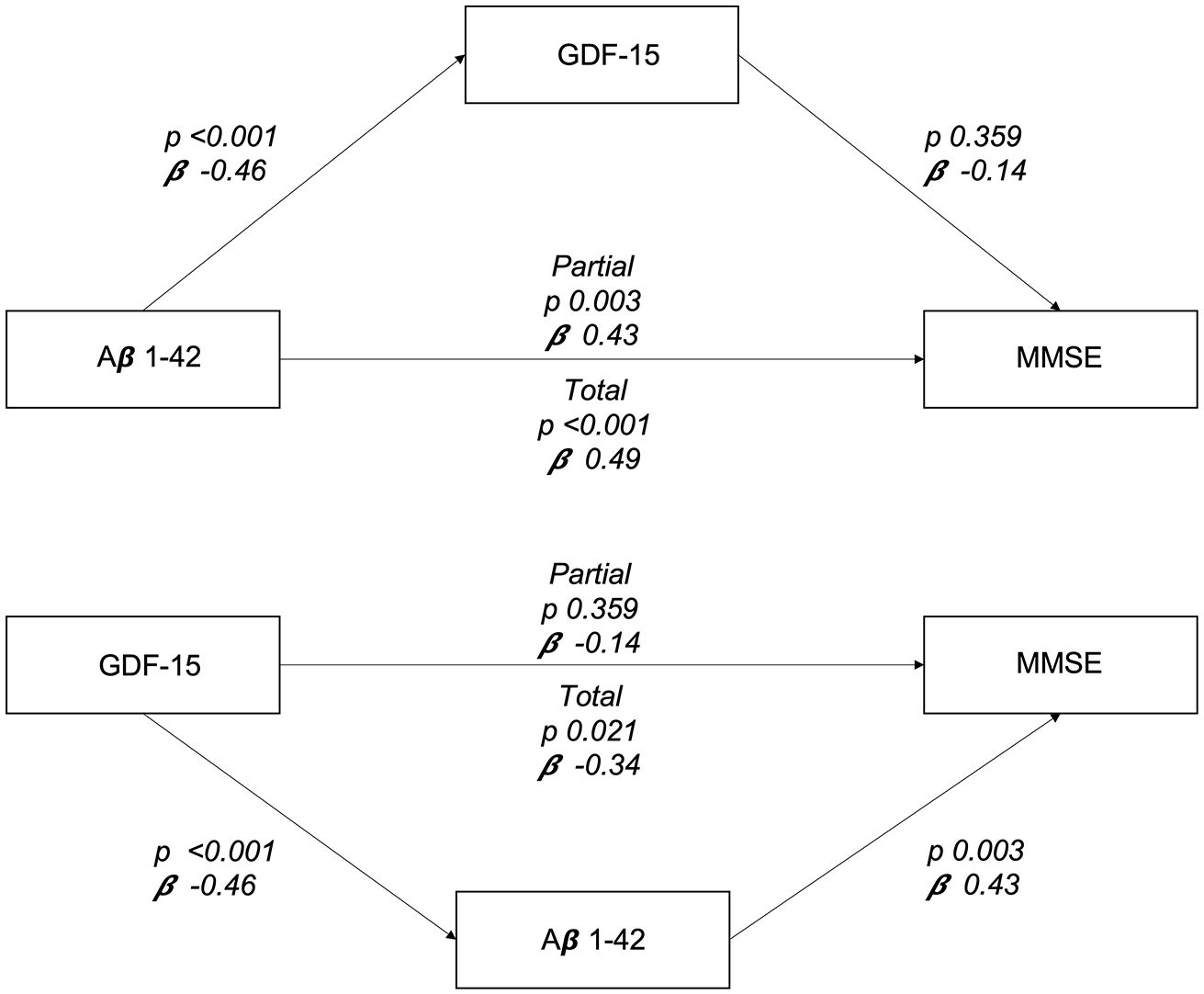
Simple mediation models representing Aβ 1-42 and GDF-15 as the mediator variable or as independent variable, MMSE score as the dependent variable. β coefficients are reported and p values are indicated.
Discussion
Here, we explored the association between two facets of inflammageing (IL-6 and mitochondrial dysfunctions) with misfolded protein levels and neural and astrocyte degenerations using CSF markers in subjects with AD. The results of this cross-sectional cohort study support the notion of a link between neuroinflammation, mitochondrial stress and cognitive impairment in AD.
The first important result is the negative correlation between CSF GDF-15 concentrations with the MMSE score. The expression of GDF-15 increases with ageing and age-related diseases, but, contrary to expectations, its CSF levels have not been shown to differ between AD patients and healthy controls. 20 Hence, GDF-15 is currently regarded more as a risk marker for the development of neurodegeneration in AD rather than a disease marker,34,35 and cognitive impairment.34,36 A recent systematic review 37 has indicated an association between elevated levels of GDF-15 and cognitive decline and proposed that the serum concentrations of GDF-15 may serve as a prospective indicator for the identification of future cognitive impairment and, indeed, an association between serum GDF-15 and cognitive performance has recently been demonstrated in a population of middle-aged adults. 38
In our population, we demonstrated that CSF GDF-15 levels were significantly associated with cognitive impairment as measured by MMSE, primarily through an indirect pathway mediated by CSF Aβ 1-42. Considering the pilot nature of this study, these findings warrant further investigation to explore the reciprocal relationships among mitochondrial stress, Aβ 1-42 deposition and cognitive impairment in AD.
It is important to notice that the direct effect of Aβ on cognition is far from resolved. Indeed, several potential indirect mechanisms linking amyloid to cognition have been proposed, starting with the impact of amyloid on tau phosphorylation, 39 the potential epileptogenic effect of amyloid 40 and the impact of amyloid on neural metabolism, the relevance of each possible pathway and their impact on specific facets of cognition 41 remain to date elusive.
Another important result of our study is the higher level of CSF GDF-15 concentrations in AD patients with low Aβ CSF concentrations compared to those with high Aβ concentrations. Evidence from in vitro studies indicates that recombinant GDF-15 may enhance Aβ clearance by cultured microglial cells 42 and studies on AD animal models demonstrate positive effects of GDF-15 on the promotion of hippocampal stem cell proliferation and migration.43,44 Therefore, a positive effect of GDF-15 on the Aβ parenchymal deposits has been hypothesized, specifically by increasing the expression of TGFβ receptor type II (TGFβRII) on microglial cells via paracrine signalling, which, in turn, promotes the activity of insulin-degrading enzyme, which facilitates Aβ breakdown. 34 Therefore, the results of our study could be interpreted as an effort by brain parenchyma to counteract Aβ aggregation and deposition.
In our study, we did not observe the positive correlation between GDF-15 and IL-6 levels that was previously reported in terms of gene expression in the cortex of AD patients. 20 This difference may be attributed to the distinct methodologies used in our research compared to that of Chiariello et al. 20 Their work focused on gene expression levels in the cortical tissue of AD patients, providing insight into local cellular responses and gene expression within brain regions directly affected by neurodegenerative processes. In contrast, our study examines CSF concentrations of GDF-15 and IL-6, which may fail to capture the same process, especially in a relatively small cohort of patients.
In our AD patients, CSF levels of TDP-43 and IL-6 are negatively correlated. This negative correlation is not surprising since TDP-43 pathology has already been associated with neuroinflammation, as evidenced by its interaction with inflammatory signalling pathways and its ability to modulate cytokine expression, including IL-6. TDP-43 has been demonstrated to curb IL-6 production in pericytes, 45 which are key players in neuroinflammation. 46 This regulation may at least partly be independent of NF-κB translocation, relying on promoting IL-6 mRNA decay, instead.47,48 Nonetheless, TDP-43 and NF-κβ have been demonstrated to interact and more precisely, TDP-43 has been found to modulate NF-κβ signalling, probably competitively binding to the nuclear translocation importin a3 (KPNA4) via its nuclear localization signal. 49
The correlation between CSF NfL and IL-6 seems to confirm previous findings 50 which highlight that both these analytes perform well as biomarkers related to neuronal damage in AD, albeit, as in our population, they are not correlated with cognitive performance. Previous studies found a negative correlation between blood levels of IL-6 and hippocampal grey matter volume,51,52 and notably, markers of chronic inflammation also exhibit a connection with elevated levels of CSF NfL. 53 The relationship between IL-6 and neurodegeneration, particularly in AD, is complex, and many aspects remain to be clarified. Some studies suggest that IL-6 may have a protective role by stimulating Aβ clearance through microglial activation; however, in contexts of chronic overexpression, IL-6 promotes neuroglial inflammation that can, conversely, exacerbate neurodegeneration. Evidence suggests that IL-6 may influence disease progression both systemically and within the brain, as this cytokine is involved in activating various signalling pathways that lead to neuronal damage. These include the Janus kinase/Signal Transducer and Activator of Transcription pathway, 54 the Mitogen-Activated Protein Kinase/Extracellular signal-Regulated Kinase pathway, 55 the Phosphoinositide 3-Kinase/Protein Kinase B/Mechanistic Target of Rapamycin pathway 56 and finally, the Nuclear Factor kappa-light-chain-enhancer of activated B cells (NF-κB) pathway. 57
The positive correlation between CSF TDP-43 and CSF GFAP levels observed in our study may suggest a potential role for TDP-43 in promoting reactive gliosis, a phenomenon previously described in neurodegenerative conditions involving TDP-43. 58 Specifically, phosphorylated TDP-43 deposition in the absence of hippocampal sclerosis has been associated with neurodegeneration and gliosis.59,60 However it remains unclear whether astrogliosis is a nonspecific response to neuronal loss or a more targeted reaction to TDP-43 pathology.
The lack of correlation between baseline CSF tau levels and cognitive impairment in our cohort is consistent with studies suggesting that longitudinal changes in tau levels may better reflect cognitive decline in AD than single time-point measurements.61–63
Our study has several limitations that should be acknowledged. First, the sample size was relatively small, which may have reduced the statistical power of our analyses and limited our ability to detect certain correlations. Second, the cross-sectional design prevented us from tracking longitudinal changes in biomarkers and cognitive decline over time, which could have provided deeper insights into the significance of these biomarkers for the progression of the disease. Furthermore, we analyzed a broad range of biomarkers, but additional markers (e.g. more specific biomarkers of neuroinflammation and mitochondrial dysfunction) could have provided a more detailed characterization of AD pathology. In addition to GDF-15, which is considered a marker of mitochondrial stress response, other more specific biomarkers of mitochondrial damage, such as mitochondrial DNA copy number, could be measured in future studies to fully characterize mitochondrial dysfunction in AD.
Conclusion
Our findings highlight that CSF IL-6 levels correlate positively with markers of neuronal damage (such as NfL). Importantly, CSF TDP-43 levels showed a significant correlation with GFAP, suggesting a potential link with reactive gliosis and astrocyte activation, which warrants further investigation into its role in astrocytic pathology in AD.
In addition, our study emphasizes the importance of GDF-15 in AD. While CSF GDF-15 levels negatively correlate with MMSE scores, mediation analysis revealed that this association is primarily indirect and mediated through Aβ 1-42 levels. This suggests that GDF-15 may influence cognitive impairment by modulating amyloid dynamics, rather than having a direct impact on cognitive performance. Moreover, higher CSF levels of GDF-15 in AD patients with low Aβ concentrations may reflect an endogenous attempt to counteract Aβ aggregation and deposition, potentially mediated by microglial activation.
Overall, these results support the hypothesis that mitochondrial stress and neuroinflammatory markers are linked to neurodegeneration and cognitive impairment in AD, albeit further longitudinal studies are needed to explore their precise roles and potential utility as biomarkers for tracking disease progression.
Supplemental Material
sj-docx-1-tan-10.1177_17562864251314773 – Supplemental material for CSF IL-6, GDF-15, GFAP and NfL levels in early Alzheimer disease: a pilot study
Supplemental material, sj-docx-1-tan-10.1177_17562864251314773 for CSF IL-6, GDF-15, GFAP and NfL levels in early Alzheimer disease: a pilot study by Domenico Plantone, Matteo Pardini, Carlo Manco, Delia Righi, Paolo Alessandro Alì, Dario Arnaldi, Virginia Pelagotti, Federico Massa, Miriana d’Alessandro, Elena Bargagli and Nicola De Stefano in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864251314773 – Supplemental material for CSF IL-6, GDF-15, GFAP and NfL levels in early Alzheimer disease: a pilot study
Supplemental material, sj-docx-2-tan-10.1177_17562864251314773 for CSF IL-6, GDF-15, GFAP and NfL levels in early Alzheimer disease: a pilot study by Domenico Plantone, Matteo Pardini, Carlo Manco, Delia Righi, Paolo Alessandro Alì, Dario Arnaldi, Virginia Pelagotti, Federico Massa, Miriana d’Alessandro, Elena Bargagli and Nicola De Stefano in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
