Abstract
Background
Acute ischemic stroke (AIS) is a leading cause of death and disability. Glucagon-like peptide-1 receptor agonists (GLP-1RA) reduce atherosclerotic events in type 2 diabetes and obesity, and meta-analyses of cardiovascular outcome trials (CVOTs) suggest a modest reduction in incident stroke. Their safety and efficacy within acute AIS pathways remain uncertain.
Methods
We conducted a narrative review by searching MEDLINE (PubMed), Cochrane CENTRAL, and ClinicalTrials.gov for studies through January 2026. We included in vitro and animal stroke models, observational studies, randomized controlled trials, and meta-analyses reporting AIS-related outcomes or stroke prevention endpoints with GLP-1RA.
Results
Experimental models commonly show reduced infarct volume and improved neurological outcomes, with proposed mechanisms including attenuation of excitotoxicity, apoptosis, oxidative stress, neuroinflammation, and blood-brain barrier disruption, alongside signals of angiogenesis and neurogenesis. Translation is limited by heterogeneity of agents, timing, dosing, and routes, and by uncertainty over direct central nervous system versus systemic mediation. CVOTs and meta-analyses support long-term stroke risk reduction, whereas observational studies and small AIS trials mainly inform feasibility, metabolic control, and safety, with efficacy unproven. Ongoing stroke-dedicated trials should define patient selection, exposure–response relationships, and interactions with thrombolysis or thrombectomy, while prospectively incorporating imaging and biomarker endpoints to test mechanisms.
Conclusions
Current evidence does not support routine GLP-1RA use as an acute neuroprotective therapy in AIS in humans. At present, GLP-1RA should be considered primarily for secondary prevention in patients with established indications, pending dedicated stroke trials clarifying acute safety, optimal timing/dosing, interactions with reperfusion therapies, and functional endpoints.
Keywords
1. Introduction
Stroke is the second leading cause of death worldwide and the third leading cause of combined death and disability, with the heaviest burden observed in low- and middle-income countries.1,2 According to the Global Burden of Disease (GBD) Stroke Project, the impact of stroke is expected to nearly double between 2020 and 2050 in terms of disability-adjusted life years (DALYs), mortality, and healthcare costs. 3 At the same time, type 2 diabetes mellitus (T2DM), one of the greatest global health threats of the 21st century, represents a major risk factor for ischemic stroke. 4 Patients with T2DM not only face a twofold higher risk of stroke compared to those without diabetes but also have a worse prognosis following a cerebrovascular event. 5
Glucagon-like peptide-1 receptor agonists (GLP-1RA), initially developed as glucose-lowering agents and later approved for obesity management, have emerged as drugs with substantial cardiometabolic benefits. Beyond their glucose- and weight-lowering effect, large cardiovascular outcome trials (CVOTs) have demonstrated their efficacy in reducing atherosclerotic cardiovascular disease (ASCVD) events, as well as provide benefits in heart failure (HF) and chronic kidney disease (CKD).6,7 Current guidelines recommend GLP-1RA or sodium–glucose cotransporter 2 (SGLT2) inhibitors with proven cardiovascular benefit as first-line therapy in patients with T2DM and established ASCVD or CKD, independent of baseline glycated hemoglobin (HbA1c) levels. 8 In a recent meta-analysis of randomized controlled trials (RCTs) including cerebrovascular events in endpoints, treatment with GLP-1RA reduced the risk of stroke by 16%. 9 Yet, evidence directly addressing their role in acute ischemic stroke (AIS) and stroke prevention remains limited.
The hypothesis of GLP-1RA–mediated neuroprotection in AIS is supported by experimental data showing that these agents counteract detrimental metabolic and vascular processes in the brain.10,11 GLP-1 crosses the blood-brain barrier (BBB) and has already demonstrated pre-clinical neuroprotective efficacy in Alzheimer’s Disease and neurodegenerative conditions, but also stroke.12-14
The present narrative review summarizes preclinical and clinical evidence regarding GLP-1RA in AIS and secondary stroke prevention, focusing on mechanistic plausibility, agent-level heterogeneity, clinical safety considerations, and key gaps that inform future stroke-dedicated trial design.
2. Methodology
A literature search was conducted in MEDLINE (PubMed), the Cochrane Central Register of Controlled Trials (CENTRAL), and ClinicalTrials.gov to identify relevant publications available up to January 2026. The search algorithm used included terms: (“GLP-1” OR “glucagon-like peptide-1 receptor agonist” OR “GLP1” OR albiglutide OR dulaglutide OR semaglutide OR exenatide OR lixisenatide OR liraglutide OR efpeglenatide) AND (“stroke” OR “ischemic stroke” OR “brain infarct” OR “brain ischemia”).
Titles and abstracts were screened for relevance by the authors, and full texts were reviewed when appropriate. Studies were selected based on the scope of the narrative review and included preclinical studies, observational studies, clinical trials, meta-analysis and relevant review articles addressing GLP-1-mediated neuroprotection and stroke-related outcomes. Articles not written in English or not directly related to ischemic stroke or lacking clinical or mechanistic relevance were excluded. Reference lists of key articles were also screened to identify additional relevant publications.
Given the narrative design, no formal risk-of-bias assessment was performed and no quantitative meta-analysis was undertaken within this review. The selected literature was synthesized qualitatively and organized thematically, with emphasis on (i) mechanistic insights derived from in vitro and in vivo stroke models, (ii) clinical evidence from observational studies, cardiovascular outcome trials and post-hoc analyses investigating stroke endpoints, and (iii) emerging studies specifically evaluating the role of GLP-1RA in the setting of AIS.
Preclinical Studies Evaluating GLP-1RA Administration After Stroke Onset

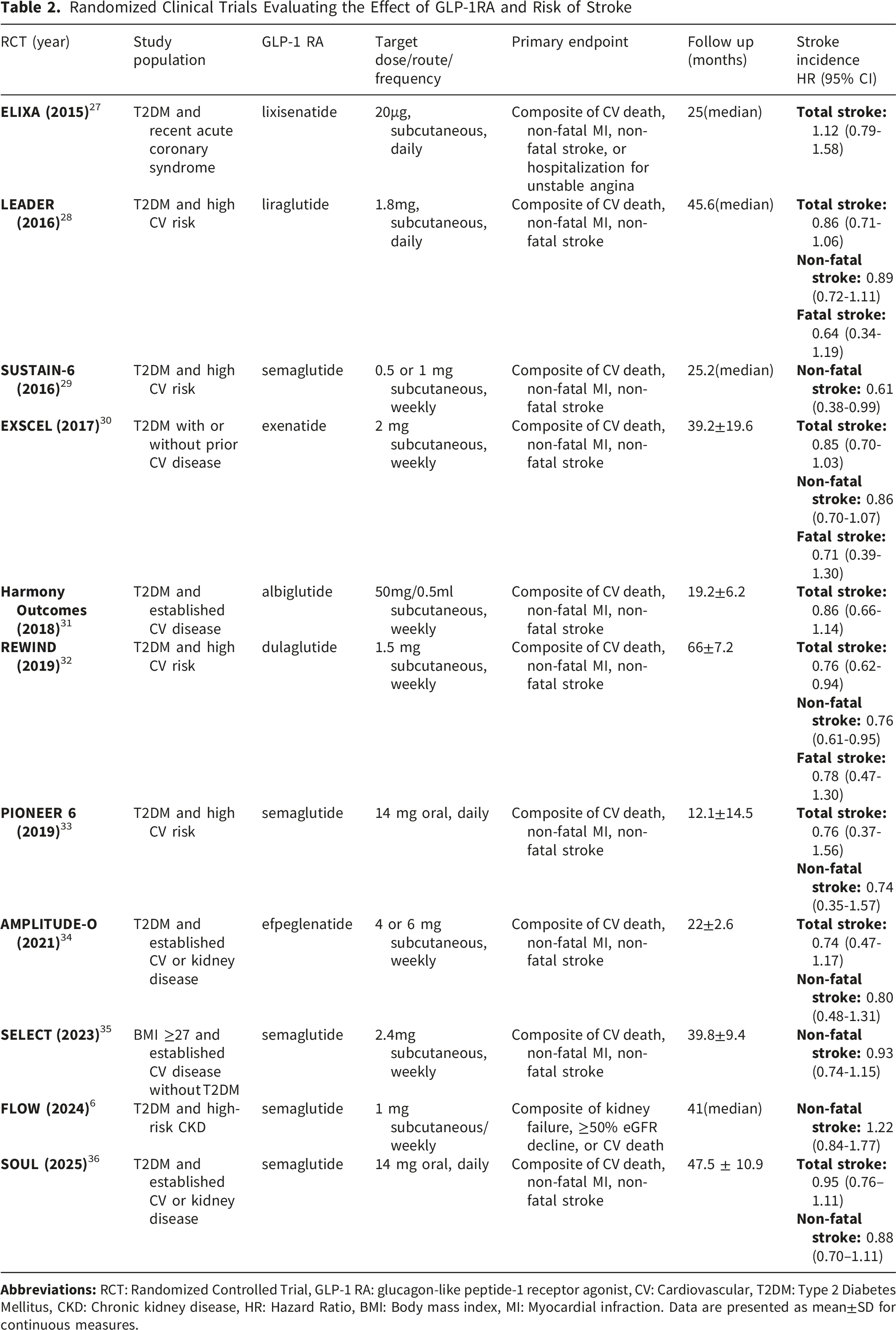
Randomized Clinical Trials Evaluating the Effect of GLP-1RA and Risk of Stroke
3. Preclinical Studies
3.1. In Vitro Studies Investigating the Neuroprotective Effects of GLP-1RA
In vitro studies have demonstrated that stimulation of the GLP-1 receptor (GLP-1R) protects hippocampal neurons against toxicity induced by amyloid-β peptide, Fe2+, and glutamate.37,38 Furthermore, activation of the GLP-1R by GLP-1 and exendin-4 (Ex-4) has been shown to induce neurite outgrowth in the PC12 cell line and to protect neurons via a cascade involving the second messenger cyclic AMP (cAMP).38-40
Cell proliferation can be assessed by the abundance of Ki-67, a nuclear protein expressed exclusively in actively dividing cells. Importantly, reduced Ki-67 staining conventionally indicates decreased cellular proliferation. In vitro studies have shown that administration of the Ex-4 increases Ki-67 staining, suggesting that GLP-1R activation plays a role in promoting cell division and proliferation. 41 Furthermore, incorporation of the thymidine/uracil analog bromodeoxyuridine and assessment of adenosine triphosphate (ATP) levels in primary rodent cultures demonstrated a significant increase in the number of brain stem cells in vitro following GLP-1RA treatment. Similar proliferative effects were also observed in ST14A cells, a rat striatal embryonic cell line. 42 GLP-1 administration to cultured neurons has been shown to protect against glutamate-induced alterations in calcium currents, thereby reducing the risk of excitotoxic cellular damage. 43
3.2. Animal Studies Evaluating the Efficacy of GLP-1RA in Ischemic Stroke Models
The first preclinical animal study to evaluate the efficacy of GLP-1RA was conducted by Li et al in a mouse model of stroke, using intracerebroventricular infusion of the peptide exendin-4 (Ex-4). The study demonstrated that the neuroprotective effects were mediated through GLP-1R activation, as Ex-4 was ineffective in GLP-1R knockout mice. 44
Subsequent studies using systemic routes reported neuroprotection with intravenous administration of Ex-4 in transient focal ischemia, with effects observed when administered at onset or within one hour, and diminished efficacy when delayed beyond a few hours in some models. These benefits were associated with increased cAMP and reduced oxidative stress and inflammation, and appeared partly independent of glycemic control. 15
Pretreatment with intraperitoneal Ex-4 in T2DM rats reduced brain injury, with dose-dependent effects that were already noticed at a clinically relevant dose (0.1 μg/kg). 45 In hyperglycemic mouse models, Ex-4 reduced infarct growth and improved functional outcomes even when glucose lowering was comparable with insulin, suggesting mechanisms beyond glycemia alone.46,47 Liraglutide has also shown beneficial signals in several models, including delayed administration paradigms in selected studies.16,20
Across preclinical studies, neuroprotective effects have been linked to reduced infarct volume, apoptosis, oxidative stress, and inflammation, and to improvements in cerebral blood flow, neurogenesis, and angiogenesis, with functional benefits in cognitive and motor domains; these studies have used exendin-4,15,17 liraglutide,16,20,23,24 rhGLP-1 (recombinant human GLP-1), 18 lixisenatide, 21 semaglutide, 22 PEx-4 (exendin-4 loaded poly-microspheres), 48 proGLP-1 (long acting GLP-1RA), 49 DMB (GLP-1R agonist/modulator), 50 dual GLP-1/Glucose-dependent Insulinotropic Peptide (GIP) agonist (GLP-1/GIPDA), 19 and oxyntomodulin (a co-activator of GLP-1R and glucagon receptor) 51 (Table 1). Two studies investigating multiple doses of GLP-1RA concluded that neuroprotection was dose-dependent. Notably, a key translational finding was that GLP-1RA retained neuroprotective effects when administered up to 24 hours and even 3 days after stroke onset.25,52 Nevertheless, the heterogeneity of agents, dosing regimens, and administration routes is a major confounder for translation, particularly given differences in molecular structure, half-life, and potential CNS exposure/BBB penetrance across GLP-1RA. Accordingly, neuroprotection observed with one agent or regimen should be considered agent- and context-specific rather than class-uniform, until confirmed by head-to-head translational work or stroke-dedicated clinical trials.
3.3. Mechanisms of GLP-1RA-Mediated Neuroprotection in Ischemic Stroke
Although multiple convergent pathways have been proposed, mechanistic attribution remains challenging because many studies use different agents, doses, time windows, and outcomes, and some effects may reflect indirect systemic changes rather than direct CNS actions.
3.3.1. Cellular and Molecular Mechanisms
Antiapoptotic effects of GLP-1RA were demonstrated by increased expression of Bcl-2, an antiapoptotic protein, and reduced levels of Bax and caspase-3, key proapoptotic proteins. This reduction in apoptosis was consistently observed in both normoglycemic and diabetic stroke models.22,53,54 The anti-inflammatory effects of GLP-1RA were associated with reduced levels of inflammatory markers, including myeloperoxidase, interleukin (IL)-1β, IL-6, IL-18, tumor necrosis factor-α, matrix metalloproteinase-9 (MMP-9), and cyclooxygenase-2, alongside increased levels of the anti-inflammatory cytokine transforming growth factor-β.24,26,55 Several animal studies in both diabetic and non-diabetic rodent models of experimental stroke have reported improved redox status following GLP-1RA administration, characterized by reduced malondialdehyde levels and increased concentrations of glutathione and superoxide dismutase, indicating attenuated oxidative stress.15,16,21,56,57
3.3.2. Neurovascular Mechanisms
GLP-1RA have been associated with pro-angiogenic responses in experimental stroke models, including increased VEGF expression within ischemic brain regions following stroke induction. 58 The phosphatidylinositol-3 kinase (PI3K)/protein kinase B (Akt), PKA, and Src signaling pathways have also been implicated in angiogenesis-related mechanisms. 59 Compared with non-GLP-1RA-treated controls, GLP-1RA treatment has been shown to improve cerebral blood flow after AIS, 48 and also enhance BBB integrity by reducing MMP-9 expression and limiting vascular immunoglobulin G extravasation. 57
3.3.3. Neurogenesis, Neuroplasticity and Neurotrophic Factors Secretion
Excessive activation of glutamate receptors is a major contributor to neuronal damage after stroke. GLP-1RA may reduce neuronal injury in the brain, an effect that is likely mediated by attenuation of glutamate-induced excitotoxicity. 60 This effect has also been correlated with increased production of brain-derived neurotrophic factor and enhanced cholinergic marker activity.61,62 Preclinical studies of GLP-1RA have shown activation of several intracellular signaling pathways implicated in neuroprotection, including cAMP/PKA/CREB, PI3K/Akt, MAPK/ERK, Wnt/β-catenin, and Nrf2/heme oxygenase-1.52,62,63 Semaglutide treatment enhanced neurogenesis in neural progenitor cells, as evidenced by significantly increased levels of neurogenesis-specific biomarkers, including doublecortin (a microtubule-associated protein), stromal cell-derived factor-1a (SDF-1a) and its receptor CXCR4, as well as nestin. 22 Furthermore, neuroplasticity is believed to improve functional recovery and outcomes after stroke.64,65 Evidence suggests that GLP-1RA in the hippocampus, particularly through signaling involving neurotrophic tyrosine kinase receptor type 2 (NTRK2) and mammalian target of rapamycin (mTOR), contribute to the enhanced neuroplasticity observed with GLP-1RA treatment. 65
These mechanistic signals are largely derived from experimental models and may not translate linearly to humans with heterogeneous stroke subtypes, comorbidities, and concurrent reperfusion and acute care interventions. Clarifying whether putative benefits require meaningful CNS exposure versus systemic mechanisms remains a key translational priority.
3.3.4. Integrated Mechanistic Relevance to Stroke Recovery
Taken together, preclinical findings suggest that GLP-1RA may exert multimodal effects that extend beyond acute neuronal survival and could plausibly support post-stroke recovery. In experimental models, GLP-1RA have been associated with attenuation of excitotoxicity, apoptosis, oxidative stress, neuroinflammation, and BBB dysfunction, alongside signals consistent with neurovascular repair and plasticity (e.g., angiogenesis, neurogenesis, neurotrophic pathways, and improved perfusion).15,16,21,22,24,26,48,52-60,62-65 These effects have been accompanied by improved performance on selected cognitive and motor assessments evaluating functional outcome in a few models.15,16,19,21,23 Functional outcomes were evaluated using heterogeneous neurological scoring systems, with reported benefits observed from as early as 24 hours up to 28 days after stroke in some studies.20,23 Because long-term recovery depends on vascular repair and adaptive remodeling in peri-infarct tissue as well as infarct limitation, such pathways are biologically relevant. However, their clinical applicability remains uncertain.
4. Clinical Evidence
4.1. Overview of Observational Studies Evaluating Outcomes in AIS Patients With Prior GLP-1RA Exposure or Early Post-Stroke Initiation
Observational evidence specifically linking pre-stroke GLP-1RA exposure to AIS outcomes is limited, and available studies frequently mix prevalent GLP-1RA use at the time of the index event (a proxy for prior exposure) with GLP-1RA initiation after AIS (post-event treatment). Accordingly, these data should be interpreted cautiously due to residual confounding, including confounding by indication, heterogeneity in exposure definitions and timing, and potential time-related biases.
In a small retrospective cohort of 123 patients with diabetes hospitalized for AIS, GLP-1RA use documented at admission and/or prescribed at discharge, therefore partially reflecting prior exposure, was associated with numerically lower 1-year stroke recurrence (Log Odds Ratio [LOR] 1.13, 95% CI 3.21–0.95, P=0.28) and higher odds of functional independence at 90 days (LOR 2.38, 95% CI 0.12–4.87, P=0.06), but these associations did not reach statistical significance. 66
In a larger retrospective study of 69,005 patients with ischemic stroke treated with intravenous thrombolysis, GLP-1RA initiation within 6 months after the index event was associated with lower all-cause mortality (HR 0.61, 95% CI 0.39–0.93), fewer emergency department visits (HR 0.78, 95% CI 0.63–0.95), and fewer inpatient hospitalizations during follow-up (HR 0.69, 95% CI 0.55–0.85). 67 Notably, this analysis evaluated early post-stroke initiation rather than pre-event exposure and cannot establish neuroprotection in the acute phase.
Finally, in a real-world cohort of 73,750 patients with diabetes and AIS, GLP-1RA users within 12 months after the index event were propensity-score matched to non-users; GLP-1RA use was associated with lower risks of major adverse cardiovascular events and cardiovascular (HR 0.71, 95% CI 0.54–0.94, P =0.02) and all-cause mortality (HR 0.59, 95% CI 0.46–0.76, P<0.001). 68 While supportive of secondary prevention benefits, these findings primarily inform outcomes during the chronic post-stroke period and remain hypothesis-generating.
4.2. Overview of Clinical Trials Investigating GLP-1RA in Stroke Prevention
Multiple CVOTs have evaluated licensed GLP-1 receptor agonists in high-risk cardiometabolic populations, with stroke captured predominantly as a secondary endpoint (Table 2).6,27-36 Overall, the signal for stroke prevention appears favorable but heterogeneous across agents and trials. The most consistent reductions have been observed with dulaglutide in total stroke (REWIND; HR 0.76, 95% CI 0.61–0.95, P = 0.01) and non-fatal stroke (REWIND; HR 0.76, 95% CI 0.62–0.94, P=0.01) 32 and subcutaneous semaglutide in non-fatal stroke (SUSTAIN-6; HR 0.61, 95% CI 0.38–0.99, P=0.04), 29 whereas several other CVOTs reported neutral effects on stroke outcomes. In newer semaglutide programs, stroke endpoints have generally remained directionally favorable but not uniformly significant.6,33,35,36 Importantly, these CVOT data primarily inform long-term cerebrovascular prevention, given their design, follow-up duration, and population characteristics, rather than acute stroke biology or early neurological recovery.
Notably, RCTs on dual GLP/GIP receptor agonist tirzepatide versus placebo, reported no stroke events as an endpoint. Results are awaited from SURMOUNT-MMO RCT with tirzepatide in patients with obesity, without diabetes and high or established CV risk. The primary endpoint is a 5-component composite outcome including nonfatal stroke. 69
Beyond primary trial reports, exploratory analyses provide additional nuance. In REWIND, secondary analyses suggested a reduction in ischemic stroke (HR 0.75, 95% CI 0.59–0.94) and disabling stroke outcomes, defined as any stroke with modified Ranking Scale (mRS) of 3 or more, among those experiencing stroke during follow-up (HR 0.74, 95% CI 0.56–0.99, P=0.04). These effects appeared broadly consistent irrespective of prior stroke history. 70 For semaglutide, pooled post-hoc analyses across SUSTAIN-6 and PIONEER-6 suggested a reduction in total stroke (HR 0.68, 95% CI 0.46–1.00; P = 0.048), with signals driven mainly by a reduction in small-vessel occlusion (HR 0.51, 95% CI 0.29–0.89; P = 0.017) and potentially modified by age and atrial fibrillation status. 71
Meta-analyses of CVOTs consistently support a modest relative reduction in stroke risk with GLP-1RA compared with placebo and suggest this benefit is broadly robust across dosing frequency, administration route, and diabetes status. More specifically, a meta-analysis published in 2018 was among the first to evaluate the effect of GLP-1RA on stroke prevention and reported a statistically significant 13% reduction in the risk of total stroke with GLP-1RA compared with placebo (relative risk 0.87, 95% CI 0.78–0.98, P = 0.021). 72 This finding is consistent with a more recent meta-analysis of 11 CVOTs published up to 2023, including 82,140 participants, in which GLP-1RA were associated with a 16% relative reduction in stroke risk compared with placebo. The benefit was consistent regardless of treatment frequency, administration route (oral vs subcutaneous), or diabetes status. 9 Another meta-analysis of 11 RCTs included 85,373 patients comparing GLP-1 RAs with placebo, reported that prior stroke history had greater stroke protection (HR 0.73, 95% CI 0.62–0.86) than MI protection (HR 0.88, 95% CI 0.77–1.01). 73 However, these summary estimates should still be interpreted within the context of chronic prevention trials, where stroke is not typically the primary outcome and mechanistic attribution to direct neuroprotection remains uncertain.
4.3. Evidence From Clinical Trials Investigating the Effect of GLP1-RA in the Setting of AIS
Early interventional studies provide preliminary insights into the feasibility and short-term safety of GLP-1 receptor agonist administration in the AIS setting. However, evidence regarding efficacy remains limited. In the first pilot study, exenatide was administered to 11 patients with acute stroke during hospitalization for post-stroke hyperglycemia management. Treatment was well tolerated, safe, and associated with a low rate of hyperglycemia (4.9%), without evidence of symptomatic hypoglycemia. 74 A randomized prehospital exenatide trial demonstrated feasibility but was terminated early after enrolling 19 patients, precluding meaningful efficacy inference. 75 The TEXAIS phase II trial reported that exenatide within 9 hours did not improve neurological impairment at 7 days versus placebo, although it reduced hyperglycemic events without hypoglycemia. 76
The recently published LAMP trial evaluated liraglutide in 636 patients with acute minor ischemic stroke (NIHSS ≤3) or high-risk transient ischemic attack (ABCD2 ≥4) and type 2 diabetes in China, reporting reduced stroke recurrence (HR 0.56, 95% CI 0.34–0.91, P=0.02) and improved 90-day outcomes (OR 1.95, 95% CI 1.28–3.00, P=0.002). 77 While encouraging, these data apply to a specific, selected population and do not establish class-wide neuroprotection in AIS.
Taken together, these studies suggest that selected GLP-1RA can be administered early after ischemic events under trial conditions and may improve metabolic control, but they do not establish a clinically meaningful neuroprotective effect in AIS, nor do they define optimal timing, dosing, or patient selection. Evidence from ongoing stroke-dedicated trials, such as ASSET enrolling AIS patients without diabetes 78 and GALLOP enrolling patients with large-vessel occlusion eligible for thrombectomy, 79 will be important to clarify feasibility, safety, and outcomes in broader AIS pathways.
4.4. Potential Mechanisms Underlying Clinical Effects
Several pathways have been proposed to explain how GLP-1RA reduce stroke risk. However, in clinical settings these effects are more likely to be mediated predominantly through indirect, systemic pathways rather than direct neuroprotection. Established extra-cerebral effects of GLP-1RA, including reductions in HbA1c, systolic blood pressure, body weight, atrial fibrillation, and dyslipidemia, may collectively contribute to stroke prevention by improving modifiable cardiovascular risk factors. In addition, GLP-1 receptor agonists have been shown to influence insulin resistance and visceral adiposity, processes that may further reduce cerebrovascular risk. Experimental and clinical studies also suggest potential anti-atherosclerotic effects, including reductions in plaque burden, enhanced plaque stability, and modulation of macrophage phenotype toward an anti-inflammatory profile (Figure 1).56,80,81 While these mechanisms provide a plausible biological framework for long-term cerebrovascular benefit, direct neuroprotective mechanisms in AIS remain largely extrapolated from preclinical models and have not yet been conclusively demonstrated in humans. Direct and indirect mechanisms for neuroprotection with GLP-1 RA. Abbreviations: GLP-1 RA: glucagon-like peptide-1 receptor agonist, NTRK2: neurotrophic tyrosine kinase receptor type 2, mTOR: mammalian target of rapamycin, MAPD: microtubule-associated protein doublecortin, SDF-1a: stromal cell-derived factor-1α CREB: response element binding protein, P13K: phosphatidylinositol-3 kinase, MAPK: mitogen-activated protein kinase, BDNF: brain-derived neurotrophic factor, VEGF: vascular endothelia growing factor. Created in https://BioRender.com
5. Challenges and Limitations
5.1. Translation of Preclinical Evidence Into Clinical Practice
Our review highlights experimental signals for the AIS setting and consistent evidence for long-term stroke risk reduction in high-risk cardiometabolic populations. Human stroke is heterogeneous in etiology, territory, infarct dynamics, comorbidities, and concurrent acute care interventions, whereas preclinical studies often use homogeneous rodent models with limited comorbidity burden, restricting generalizability. Some experimental paradigms, namely bilateral carotid occlusion, selected lesion models, incompletely recapitulate human AIS pathophysiology and clinical time courses. Moreover, studies employing intracerebral or intracerebroventricular administration are not directly comparable to clinical practice, as they bypass BBB and systemic pharmacokinetics. 44 Uncertainty also remains whether CNS benefits require direct central exposure or can be achieved through systemic mechanisms, including glycemic variability, inflammation and endothelial function, which has major implications for translation and agent selection. Additional limitations include small sample sizes, variable methodological rigor, heterogeneity of agents, and differences between experimental and clinically approved dosing.
5.2. Optimal Dose and Timing of Administration
The doses employed in preclinical studies are far higher than the clinical doses used to treat T2DM. In a study with Ex-4 administered intraperitoneally, a dose-dependent effect already significant at the clinical dose of 0.1 μg/kg was noted, but the protective effect was stronger at 1 μg and 5 μg/kg. 45 Several preclinical studies administered GLP-1RA after stroke onset - a strategy most closely aligned with the potential clinical use of these agents in GLP-1RA–naïve patients presenting with AIS - and reported neuroprotective benefits.25,52
In contrast, a recent meta-analysis demonstrated that the stroke risk reduction associated with GLP-1RA was independent of dosing frequency, route of administration (oral vs subcutaneous), and diabetes status. 9 However, these chronic prevention data do not define the acute therapeutic time window, the dose–response relationship in AIS pathways, or whether any putative benefit would require direct CNS exposure versus systemic effects. Accordingly, extrapolation from experimental paradigms to human AIS should be cautious, and definitive conclusions about optimal timing, dosing, and acute neuroprotection cannot yet be drawn. These uncertainties support the need for stroke-dedicated trials with prespecified exposure targets, timing windows, and mechanistic endpoints.
5.3. Agent-Level Heterogeneity
GLP-1RA are not pharmacologically interchangeable. They differ in molecular structure, duration of action, and potential CNS access, including BBB penetrance and CNS distribution. 82 This heterogeneity may contribute to variability in neuroprotection signals and limit direct comparison across studies using different compounds, routes, and dosing schedules. Accordingly, any potential acute stroke benefit should be interpreted as agent- and regimen-specific until confirmed by dedicated trials, and future research should explicitly account for these pharmacologic differences through agent selection, exposure assessment, and prespecified subgroup analyses.
5.4. Optimizing Study Populations
The careful selection of study populations will be essential for future trials evaluating the benefits of GLP-1RA in acute stroke. Stroke patients are highly heterogeneous, and outcomes vary according to stroke subtype, vascular territory, acute management pathways and the presence of comorbidities. A key design consideration is whether to enroll patients treated with intravenous thrombolysis and/or mechanical thrombectomy—currently the only proven disease-modifying interventions in AIS—because these subgroups allow assessment of feasibility and safety within contemporary reperfusion workflows, potential interactions with recanalization therapies, and effects on infarct evolution and early neurological outcomes. Another important consideration will be whether to include both patients with or without diabetes, as preclinical studies suggest that the neuroprotective effects of GLP-1RA may occur independently of glycemic control. Another key question relates to whether patients with prior ASCVD, CKD, or obesity should be prioritized, since these populations may derive additional cardiovascular benefits from GLP-1RA. Age and frailty should also be considered, as older patients often have distinct risk profiles and treatment responses compared with younger cohorts. Careful stratification of these populations in future trials will be crucial to identify the subgroups most likely to benefit from GLP-1RA therapy in the setting of AIS.
5.5. Potential Adverse Effects
The most common reported adverse events of GLP-1RA are gastrointestinal, namely mild nausea, vomiting and delayed gastric emptying. While generally tolerable in the limited AIS trial data available,74-76 these effects warrant careful consideration in AIS because dysphagia, impaired consciousness, sedation/anesthesia for procedures, and baseline aspiration risk are common in acute stroke pathways. Emerging perioperative guidance has highlighted aspiration concerns during anesthesia/deep sedation in patients receiving GLP-1RA, which may be relevant to thrombectomy workflows and acute imaging and procedural needs.83,84 Volume status and hemodynamic management are also central in AIS, particularly in older adults and those with HF/CKD, and should be systematically assessed in trials. Additionally, a retinopathy signal was observed in SUSTAIN-6, reinforcing the need for caution in patients susceptible to rapid glycemic changes and for prespecified ocular safety monitoring in relevant populations.29,85
6. Anticipated Trials and Future Directions
Although multiple CVOTs have evaluated GLP-1RA, stroke has largely been captured as a secondary endpoint. Future studies should prioritize stroke-specific outcomes, including incident stroke, stroke subtype, and recurrence, as well as extend evaluation to populations with and without diabetes, including those with prediabetes or elevated ASCVD risk. While RCTs remain the gold standard, increasing background GLP-1RA use may complicate recruitment and interpretation in some settings. Nevertheless, guideline recommendations remain indication-specific and evidence-driven (e.g., GLP-1RA for obesity/ASCVD and SGLT2 inhibitors for CKD/heart failure), 8 and widespread uptake has not precluded dedicated trials in new indications and outcomes.
Importantly, although GLP-1RA are established as chronic preventive therapies, acute-phase testing can still be clinically relevant when mechanisms extend beyond long-term risk-factor modification. In AIS, future trials should incorporate design features that improve feasibility and mechanistic resolution, including imaging-based enrichment providing information on infarct core/penumbra, perfusion profiles, and BBB markers, standardized short-term outcomes, such as early neurological worsening, infarct growth, and pragmatic or registry-based randomization to reduce recruitment barriers. Adaptive approaches and integration of validated prognostic tools may further increase efficiency, particularly given evolving background therapy.
7. Conclusions
The quest for effective neuroprotective therapies in AIS remains a major unmet need. GLP-1RA have strong biological plausibility and well-established cardiometabolic benefits, with consistent evidence supporting ASCVD risk reduction and a potential reduction in stroke risk in high-risk populations. However, evidence supporting direct neuroprotection in AIS in humans is currently insufficient, and available AIS trials primarily inform feasibility, metabolic control, and selected early outcome signals in restricted populations rather than definitive efficacy. Emerging data from RCTs conducted in the setting of AIS are hypothesis-generating and warrant confirmation in larger, stroke-dedicated studies that address agent selection, timing and dosing, acute safety, and interactions with reperfusion therapies. Therefore, at present GLP-1RA should be considered primarily for long-term secondary prevention in patients with T2DM or obesity, while their role in AIS remains to be determined.
Footnotes
Ethical Considerations
This article is a narrative review and does not involve any studies with human participants, human data, or human tissue.
Author Contributions
V.S., F.B, H.M contributed to the conception and design of the work. V.S contributed to drafting the work and F.B, H.M reviewing it critically for important intellectual content. K.T., A.C., T.K., E.C.R., M.R. and D.P. provided critical review for important intellectual content, interpretation and analysis of data for the work. All authors contributed to the final version of the manuscript, approved its submission and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TK has received honoraria for lectures from AstraZeneca, Boehringer Ingelheim, Menarini, ELPEN, Pharmaserve Lilly and Novo Nordisk, for advisory boards from Novo Nordisk and Boehringer Ingelheim, and has participated in sponsored studies by Eli-Lilly, AstraZeneca and Novo Nordisk. ECR has received speaker honoraria and consulting fees for clinical trials unrelated to this work (Novartis, Sanofi, NovoNordisk, Pfizer, AstraZeneca/BMS, Eli Lilly, MSD/Vianex, Boehringer Ing, Amgen, GSK, Plus Pharmaceuticals, WinMedica, Servier). HM participated in educational, research and consulting activities supported by healthcare companies, including AstraZeneca, Boerhinger Ingelheim, Menarini, Novartis, Pfizer, Sanofi. FB has received research grants from Amgen, Boehringer Ingelheim, Eli Lilly, Novartis, Novo Nordisk; honoraria and consulting fees from Menarini Hellas, Novartis, Novo Nordisk, Sanofi and Viatris. DP, AC, MR, VS and KT declare no conflict of interest.
