Abstract
Introduction:
Mounting evidence suggests that glucagon-like-peptide-1 receptor-agonists (GLP-1 RAs) attenuate cardiovascular-risk in type-2 diabetes (T2DM). Tirzepatide is the first-in-class, dual glucose-dependent-insulinotropic-polypeptide GIP/GLP-1 RA approved for T2DM.
Patients and methods:
A systematic review and meta-analysis of randomized-controlled clinical trials (RCTs) was performed to estimate: (i) the incidence of major adverse cardiovascular events (MACE); and (ii) incidence of stroke, fatal, and nonfatal stroke in T2DM-patients treated with GLP-1 or GIP/GLP-1 RAs (vs placebo).
Results:
Thirteen RCTs (9 and 4 on GLP-1 RAs and tirzepatide, respectively) comprising 65,878 T2DM patients were included. Compared to placebo, GLP-1RAs or GIP/GLP-1 RAs reduced MACE (OR: 0.87; 95% CI: 0.81–0.94; p < 0.01; I2 = 37%), all-cause mortality (OR: 0.88; 95% CI: 0.82–0.96; p < 0.01; I2 = 21%) and cardiovascular-mortality (OR: 0.88; 95% CI: 0.80–0.96; p < 0.01; I2 = 14%), without differences between GLP-1 versus GIP/GLP-1 RAs. Additionally, GLP-1 RAs reduced the odds of stroke (OR: 0.84; 95% CI: 0.76–0.93; p < 0.01; I2 = 0%) and nonfatal stroke (OR: 0.85; 95% CI: 0.76–0.94; p < 0.01; I2 = 0%), whereas no association between fatal stroke and GLP-1RAs was uncovered (OR: 0.80; 95% CI: 0.61–1.05; p = 0.105; I2 = 0%). In secondary analyses, GLP-1 RAs prevented ischemic stroke (OR: 0.74; 95% CI: 0.61–0.91; p < 0.01; I2 = 0%) and MACE-recurrence, but not hemorrhagic stroke (OR: 0.92; 95% CI: 0.51–1.66; p = 0.792; I2 = 0%). There was no association between GLP-1RAs or GIP/GLP-1 RAs and fatal or nonfatal myocardial infarction.
Discussion and conclusion:
GLP-1 and GIP/GLP-1 RAs reduce cardiovascular-risk and mortality in T2DM. While there is solid evidence that GLP-1 RAs significantly attenuate the risk of ischemic stroke in T2DM, dedicated RCTs are needed to evaluate the efficacy of novel GIP/GLP-1 RAs for primary and secondary stroke prevention.

Introduction
Glucagon-like peptide-1 receptor agonists (GLP-1RAs) have attracted widespread attention during the past few years, as a novel class of type 2 diabetes (T2DM) medications with improved efficacy on glycemic control and a safety profile aligned with other classes of antidiabetics. 1 GLP-1RAs exert their antihyperglycemic effects by activating the GLP-1, an endogenous incretin that regulates glucose-dependent insulin and glucagon secretion, delays gastric emptying and increases satiety postprandially.2,3 Among the currently available, FDA-approved GLP-1RAs for T2DM, exenatide and lixisenatide, are based on the exendin-4 molecule, a naturally occurring peptide isolated from the venom of the Gila monster (Heloderma suspectum), with a 53% homology to the human GLP-1. 4 Conversely, liraglutide, albiglutide, dulaglutide and semaglutide are classified as human GLP-1RA analogs, that bear a >90% homology to the native GLP-1 and are synthesized by conjugation of GLP-1 with molecules that reduce its renal excretion and prolong its plasma half-life. 4 Besides glycose regulation, recent evidence suggests that GLP-1RAs exert pleiotropic effects in the cardiovascular and cerebrovascular system, with evidence from cardiovascular outcome trials (CVOTs) linking their clinical use to significant reduction of vascular risk and mortality in T2DM patients.1,5
The effective integration of GLP-1RAs into the arsenal of antidiabetic treatments along with their recent inclusion in clinical practice guidelines both for primary prevention of cardiovascular disease and secondary stroke prevention,6,7 has accelerated the pace for the development of newer, enforced molecules that act as dual glucose-dependent insulinotropic polypeptide (GIP) and GLP-1 receptor agonists. Tirzepatide comprises the first-in-class, FDA-approved GIP/GLP-1-RA agent, with preliminary evidence from phase 2 and 3 randomized clinical trials (RCTs) supporting its strong cardioprotective potential. Compared to GLP-1RAs, the so-far available evidence indicates that tirzepatide’s effects on glycemic control and weight loss supersede those of GLP-1RAs. 8 Besides the shared GLP-1R-mediated pathways, the GIP component of dual GIP/GLP-1 receptor agonists is hypothesized to elicit additional anorexigenic and lipid-lowering effects: (i) by acting centrally on anorexigenic neurons in the brain; and (ii) by promoting lipid storage in adipose tissue in the periphery.9,10 Moreover, tirzepatide appears to have additional cardiovascular and cerebrovascular benefits, displaying antihypertensive effects and attenuating endothelial dysfunction and circulation of inflammatory molecules in vivo. 11 While the cardiovascular safety of tirzepatide in the T2DM population remains under investigation in the context of the ongoing SURPASS CVOT trial, 12 so-far published data point toward a low risk of major adverse cardiovascular events (MACE), including stroke, in tirzepatide-treated T2DM patients.
The aim of the present systematic review and meta-analysis was to evaluate the risk of MACE and stroke in T2DM patients treated with GLP-1RAs or GIP/GLP-1 RAs. In particular, we sought to update the results of previous meta-analyses by our group 13 and others (summarized in Supplemental Table-S1),14 –20 given the availability of new trial data from a GLP-1 RA RCT 21 and GIP/GLP-1 RA RCTs and provide comparative estimates for cardiovascular outcomes in GLP-1 RA or GIP/GLP-1 RA-treated T2DM patients. Results from all so-far published placebo-controlled RCTs with prospectively collated and centrally adjudicated MACE in T2DM patients under GLP-1RAs and GIP/GLP-1 RAs were identified and meta-analyzed.
Methods
Standard protocol approvals and registrations
The present systematic review and meta-analysis is reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta- Analyses (PRISMA) statement. 22 Only publicly available published studies were used for meta-analysis. Ethical Committee approval was waived due to study design (systematic review and meta-analysis). The study protocol, comprising pre-determined PICOS (Population, Intervention, Comparison, Outcome and Study) framework, was a priori designed and registered at the PROSPERO database (CRD42023481699). The authors declare that all supporting data are available within the article and its Supplemental Files.
Data sources and searches
In this systematic review and meta-analysis, two independent reviewers (MIS, AT) searched for published randomized placebo-controlled trials testing GLP-1RAs or GIP/GLP-1 RA in T2DM patients. Eligible RCTs were identified by systematic search in MEDLINE (via PubMed) and Scopus databases. The combination of search strings for all database queries included the following search terms: “glucagon-like peptide-1 receptor agonist,” “dual GLP-1/GIP receptor agonists,” “exenatide,” “lixisenatide,” “liraglutide,” “albiglutide,” “dulaglutide,” “semaglutide,” “tirzepatide,” “randomized controlled trial,” “placebo,” “stroke,” “MACE,” or “major adverse cardiovascular events.” The full search algorithms used in MEDLINE and SCOPUS databases are provided in the Supplement. Our search was restricted to RCTs, while no language restrictions were applied. The search spanned from each electronic database’s inception to January 17th, 2023. An additional manual search of bibliographies of articles meeting study inclusion criteria was conducted to ensure the comprehensiveness of the literature.
Placebo-controlled RCTs that reported on MACE 23 or stroke in T2DM patients treated with GLP-1RAs or GIP/GLP-1 RAs were eligible for inclusion. Exclusion criteria comprised: (1) RCTs that were not placebo-controlled; (2) RCTs investigating compounds of GLP1-RAs combined with other drugs, including combined regimens of insulin degludec and liraglutide (IDegLira); (3) study population of <300 patients; (4) reported outcomes not aligned with our inclusion criteria; (5) observational studies, narrative and systematic reviews, case-series or case-reports, commentaries, pre-prints or non-peer reviewed studies, and conference abstracts. In case that studies had overlapping data, we retained the study with the largest dataset. All retrieved studies were assessed by two reviewers (MIS, AT) independently and any disagreements between reviewers were resolved by consensus after discussion with a third tie-breaking evaluator (GT).
Quality control, bias assessment and data extraction
For relevant domains of each included study, the risk of bias was assessed using the Cochrane Collaboration risk of bias tool. 24 Two independent reviewers (MIS, AT) performed quality control and bias assessment, and consensus after discussion with the corresponding author (GT) was reached in case of disagreement. For further analyses, data including the name of the first author, year of publication, study design, follow-up duration, sample size, patient population, and event type (i.e. MACE, all-cause stroke, fatal stroke, nonfatal stroke, all-cause mortality, cardiovascular mortality, fatal myocardial infarction [MI], nonfatal MI) were extracted from individual studies in structured reports.
Publication bias across individual studies was evaluated graphically using funnel plots, 25 whereas funnel plot asymmetry was assessed using Egger et al.’s linear regression test, 26 and the threshold of the statistical significance was set at p < 0.10.
Outcomes
An aggregate data meta-analysis was performed including all identified placebo-controlled RCTs that reported the incidence of MACE or stroke in T2DM patients treated with GLP-1RAs or GIP/GLP-1 RAs. The predefined primary outcomes of interest were twofold: (i) the incidence of MACE, all-cause and cardiovascular mortality; (ii) the incidence of all-cause stroke, fatal, and nonfatal stroke. Secondary outcomes included the incidence of ischemic and hemorrhagic stroke, fatal and nonfatal MI. In addition, recurrence rates of MACE were evaluated among patients with prior history of established cardiovascular disease, and history of MI or nonfatal stroke. Subgroup analysis was performed based on antidiabetic treatments either with GLP-1 RAs or GIP/GLP-1 RAs. In addition, sensitivity analysis was performed to evaluate potential differences in GLP-1 RAs effects at different time-points (i.e. at 12 and 24 months of treatment).
Statistical analysis
Meta-analysis was performed using R–software version 3.5.0 (packages: meta and metafor). All intended outcomes of interest were handled as dichotomous variables, and all the associations evaluating the effect of GLP-1RAs or GIP/GLP-1 RAs with different outcomes are reported as odds ratios (OR) with their corresponding 95% confidence intervals (CI). Risk differences (RD) between GLP-1 RAs and placebo were calculated based on probability point estimates acquired from Kaplan-Meier plots using WebPlotDigitizer. 27 The random-effects model of meta-analysis (DerSimonian and Laird) was utilized for estimation of the pooled estimates. We used the Q test to assess subgroup differences. The I 2 and Cochran Q statistics were employed for heterogeneity assessment. With respect to the qualitative heterogeneity interpretation, I 2 values>50% and values>75% were considered to represent either substantial or considerable heterogeneity, respectively. The significance level was set at 0.1 for the Q statistic, 28 while the equivalent z test with a two-tailed p value < 0.05 was considered statistically significant for each pooled estimate.
Results
Literature search and included studies
The systematic database search yielded 228 records from MEDLINE and 362 records from SCOPUS databases. After exclusion of duplicates and articles that were out-of-scope, 90 records were considered eligible for inclusion and were assessed in full. After reading the full-text articles, 75 more were further excluded (Supplemental Table-S2). Finally, we identified 15 eligible studies for inclusion (9 RCTs on GLP-1 RAs in T2DM,21,29 –36 2 post-hoc analyses of RCTs on GLP-1 RAs,37,38 4 RCTs on tirzepatide in T2DM39 –42), comprising a total of 65,878 T2DM patients. All original studies were placebo-controlled RCTs, and Supplemental Table-S3 summarizes their main characteristics. In Figure 1, the PRISMA flowchart of the meta-analysis is presented.
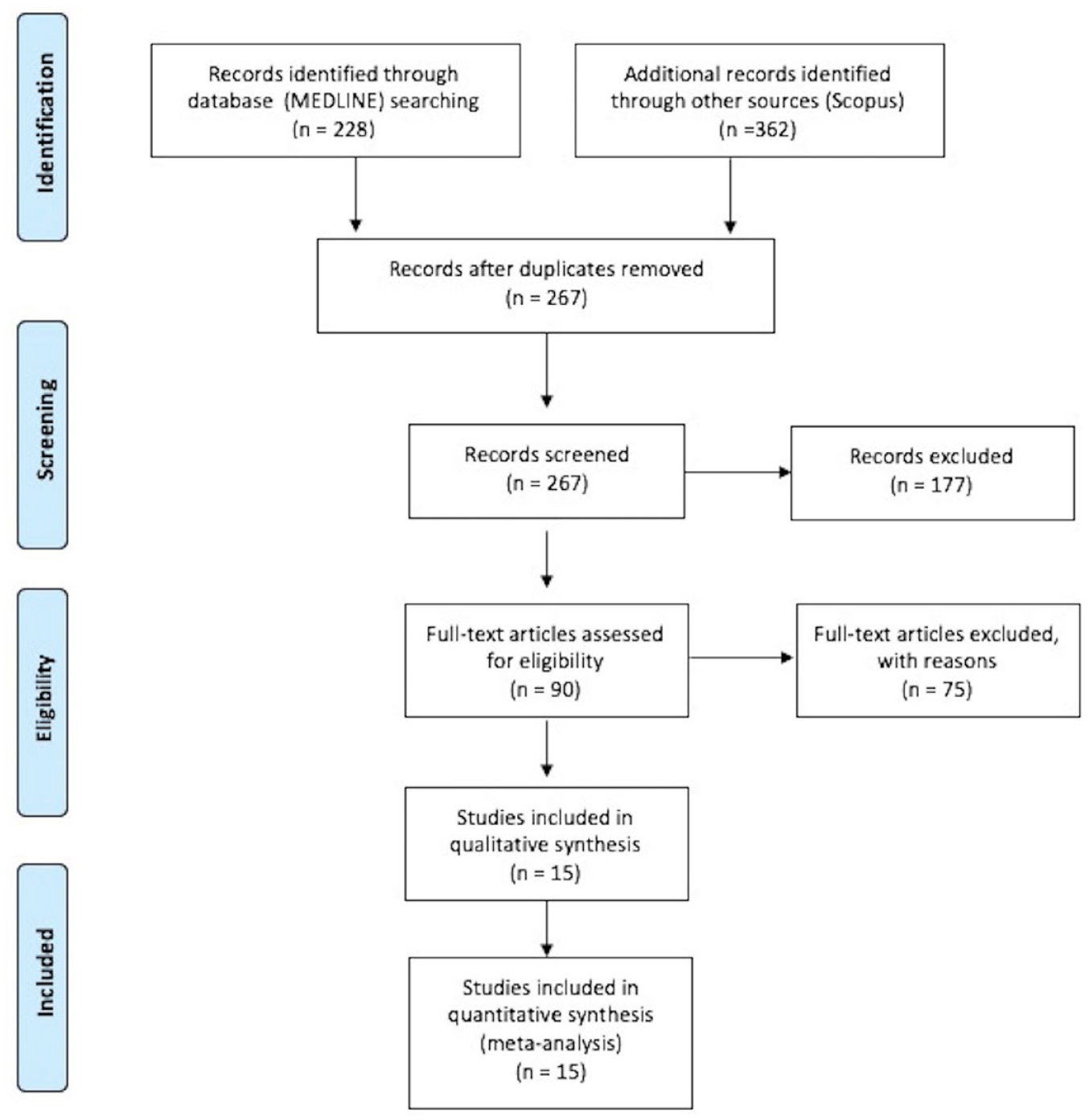
PRISMA flowchart diagram presenting the selection of eligible studies.
Quality control and publication bias of included studies
The risk of bias of studies included in the present meta-analysis is presented in Supplemental Figure S1. The risk of bias was considered low in all the included RCTs.
Funnel plot symmetry inspection and Egger statistical testing were performed for outcomes involving ⩾10 studies. 25 Accordingly, no asymmetry was revealed for assessment of publication bias among trials reporting MACE (p = 0.3015; Figure S2), cardiovascular mortality (p = 0.2606; Figure S3) and all-cause mortality (p = 0.3570; Figure S4) between treatment with GLP-1RAs or GIP/GLP-1 RAs and placebo.
Primary outcomes
When compared to placebo, treatment with GLP-1RAs or GIP/GLP-1 RAs was associated with significant reduction of MACE (13 RCTs; OR: 0.87; 95% CI: 0.81–0.94; p for Cochran Q < 0.01; I 2 = 37%; Table 1, Figure 2), all-cause mortality (12 RCTs; OR: 0.88; 95% CI: 0.82–0.96; p for Cochran Q < 0.01; I 2 = 21%; Figure S5) and cardiovascular mortality (10 RCTs; OR: 0.88; 95% CI: 0.80–0.96; p for Cochran Q < 0.01; I 2 = 14%; Figure S6). Additionally, GLP-1RAs reduced the odds of all-cause stroke (8 RCTs; OR: 0.84; 95% CI: 0.76–0.93; p for Cochran Q < 0.01; I 2 = 0%; Figure 3) and nonfatal stroke (9 RCTs; OR: 0.85; 95% CI: 0.76–0.94; p for Cochran Q < 0.01; I 2 = 0%; Figure 4(a)), but no significant association was uncovered for fatal stroke (8 RCTs; OR: 0.80; 95% CI: 0.61–1.05; p for Cochran Q = 0.105; I 2 = 0%; Figure 4(b)). Notably, as the 4 included RCTs on tirzepatide in T2DM reported no stroke events (i.e. zero events in both treatment and placebo groups), these RCTs contributed no data to the respective meta-analyses of all-cause stroke, fatal and nonfatal stroke.
Overview of vascular outcomes among patients with type 2 diabetes treated with GLP-1RAs or GIP/GLP-1 RAs versus placebo.
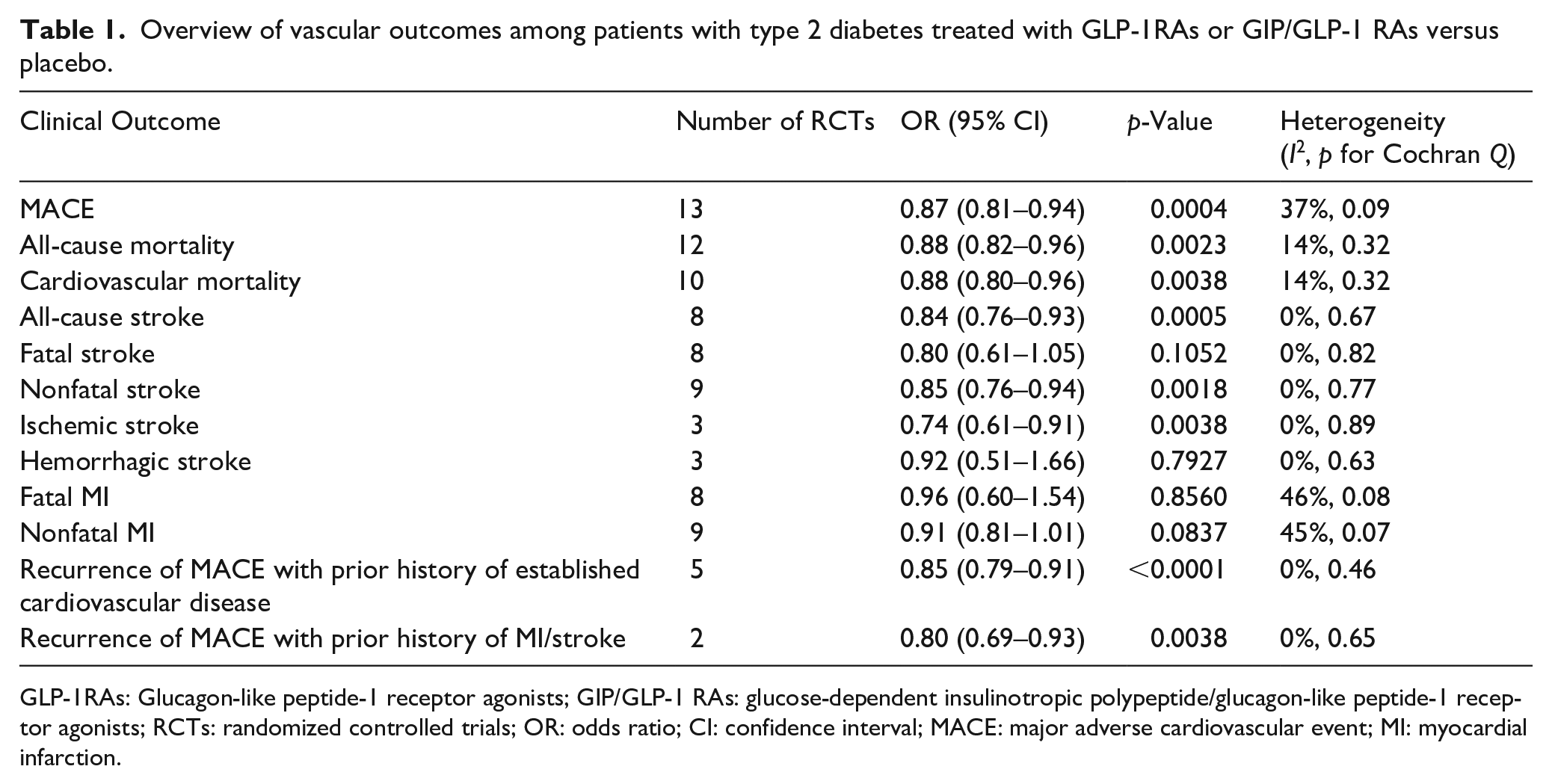
GLP-1RAs: Glucagon-like peptide-1 receptor agonists; GIP/GLP-1 RAs: glucose-dependent insulinotropic polypeptide/glucagon-like peptide-1 receptor agonists; RCTs: randomized controlled trials; OR: odds ratio; CI: confidence interval; MACE: major adverse cardiovascular event; MI: myocardial infarction.
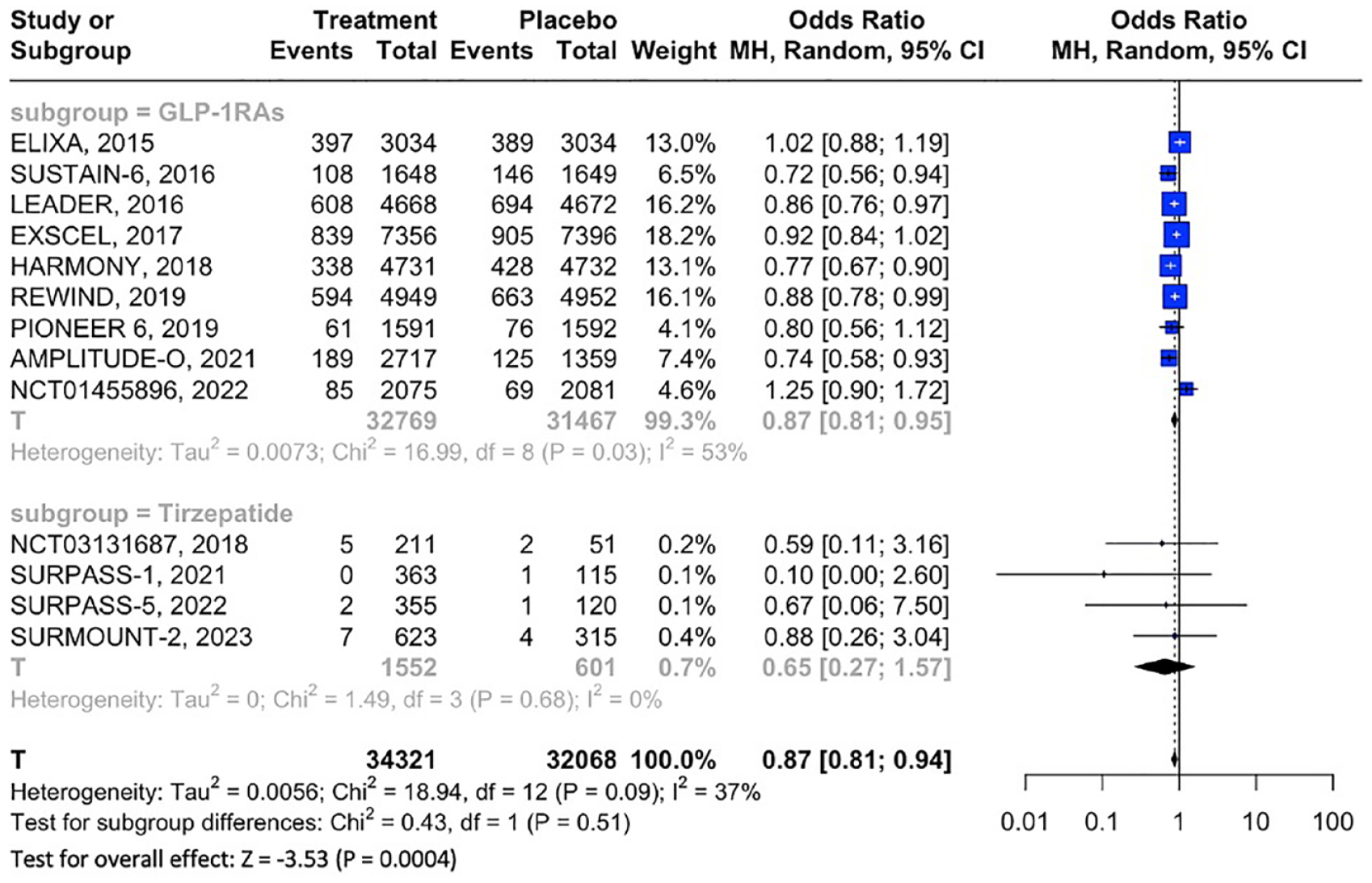
Forest plot comparing the risk of MACE in T2DM patients treated with GLP-1RAs or GIP/GLP-1 RAs versus placebo.
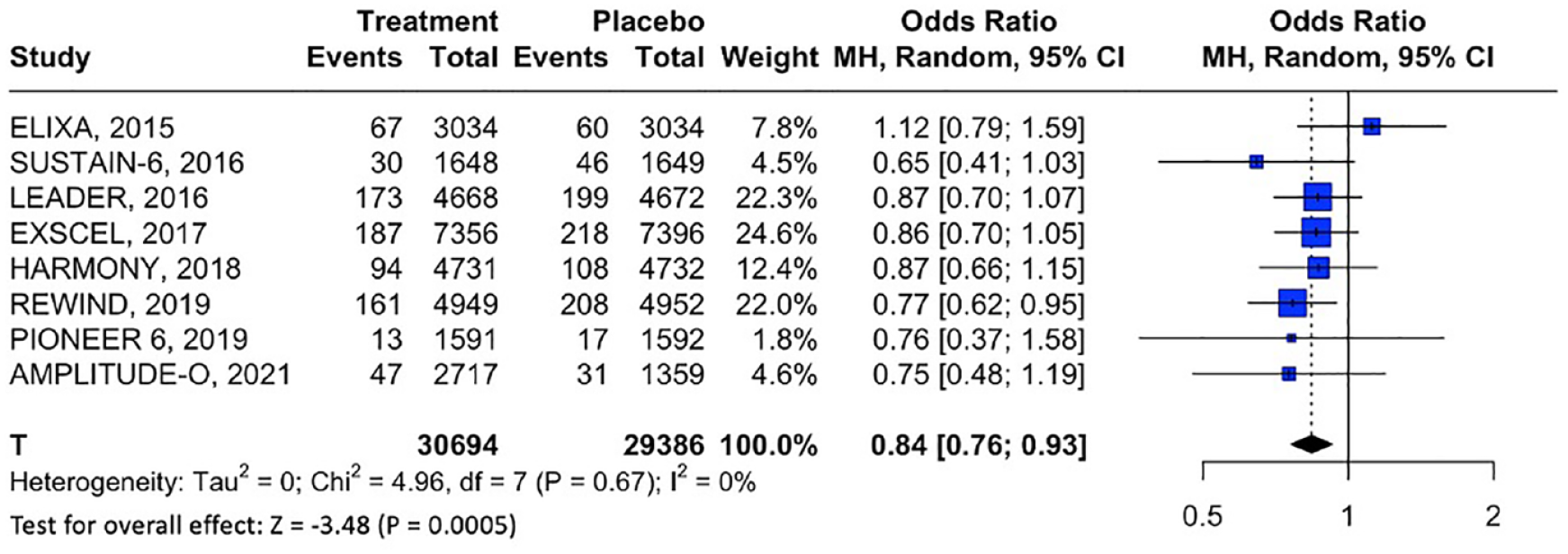
Forest plot comparing the risk of all-cause stroke in T2DM patients treated with GLP-1RAs versus placebo.
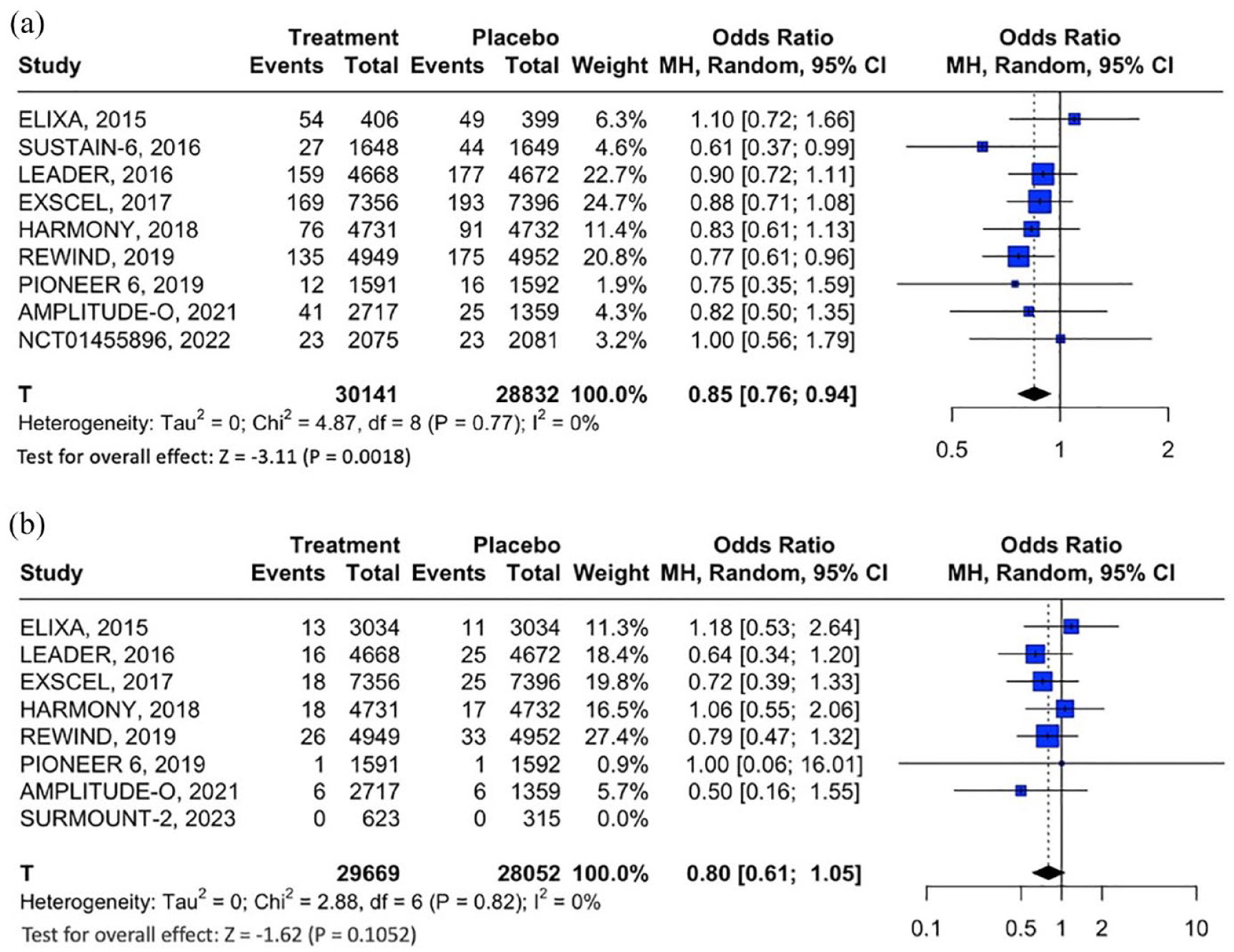
(a) Forest plot comparing the risk of nonfatal stroke in T2DM patients treated with GLP-1RAs versus placebo and (b) Forest plot comparing the risk of fatal stroke in T2DM patients treated with GLP-1RAs versus placebo.
Secondary outcomes
Concerning stroke subtypes, treatment with GLP-1RAs was associated with significant reduction in the odds of ischemic stroke (3 RCTs; OR: 0.74; 95% CI: 0.61–0.91; p for Cochran Q < 0.01; I 2 = 0%; Figure 5), but no association was noted for hemorrhagic stroke (3 RCTs; OR: 0.92; 95% CI: 0.51–1.66; p for Cochran Q = 0.792; I 2 = 0%; Figure S7). Conversely, no clear association of GLP-1RAs or GIP/GLP-1 RAs was disclosed for either fatal MI (8 RCTs; OR: 0.96; 95% CI: 0.60–1.54; p for Cochran Q = 0.856; I 2 = 46%; Figure S8) or nonfatal MI (9 RCTs; OR: 0.91; 95% CI: 0.81–1.01; p for Cochran Q = 0.084; I 2 = 45%; Figure S9).
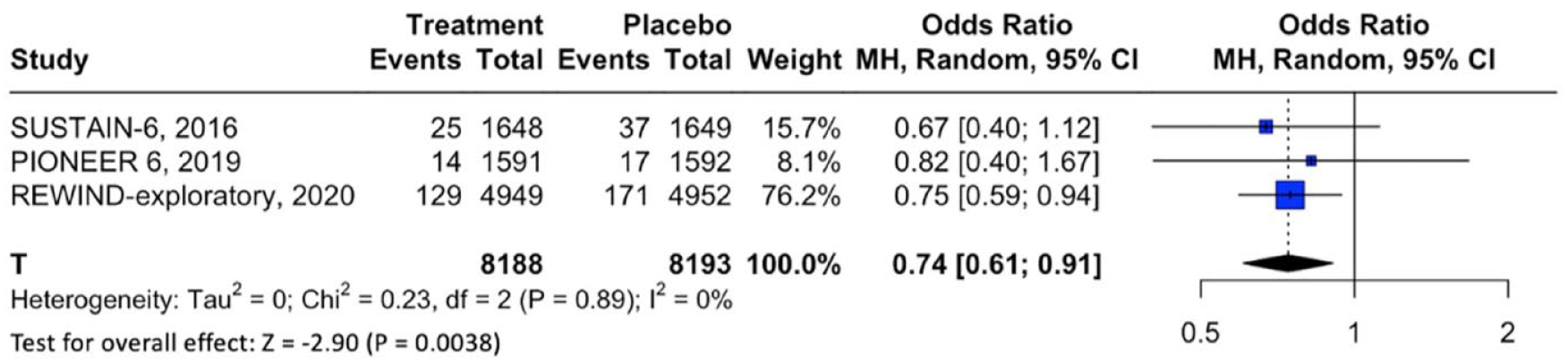
Forest plot comparing the risk of ischemic stroke in treatment with GLP-1RAs versus placebo.
Regarding secondary prevention, GLP-1RA treatment was associated with reduced incidence of recurrent MACE among patients with prior established cardiovascular disease (5 RCTs; OR: 0.85; 95% CI: 0.79–0.91; p for Cochran Q < 0.01; I 2 = 0%; Figure S10) and among patients with prior history of MI or stroke (2 RCTs; OR: 0.80; 95% CI: 0.69–0.93; p for Cochran Q < 0.01; I 2 = 0%; Figure S11).
Subgroup analyses on GLP-1RA versus GIP/GLP-1 RA treatment, revealed no significant subgroup effects on MACE (p for subgroup differences = 0.51; Figure 2), all-cause mortality (p for subgroup differences = 0.33; Figure S5), cardiovascular mortality (p for subgroup differences = 0.19; Figure S6), or fatal MI (p for subgroup differences = 0.17; Figure S8). Sensitivity analyses revealed a trend toward greater MACE reduction with increasing duration of GLP-1 RA treatment from 12 months (RD: −0.006; 95% CI: −0.024 to 0.012; p = 0.498; Figure S12
Discussion
The findings of the present systematic review and meta-analysis demonstrate that treatment with GLP-1RAs or GIP/GLP-1 RAs is associated with significant reduction of MACE, all-cause mortality and cardiovascular mortality in the T2DM patient population. In addition, treatment with GLP-1 RAs leads to significant attenuation of all-cause stroke and nonfatal stroke in patients with T2DM. Notably, no stroke events were reported in the 4 included RCTs on the GIP/GLP-1 RA tirzepatide in T2DM (i.e. zero events in both the treatment and placebo group in all trials). Consequently, no inferences can be drawn regarding potential associations between GIP/GLP-1 RAs and stroke based on the currently available evidence.
The present findings on GLP-1RA use in T2DM patients are concordant with results of prior meta-analyses from our group 13 and others (summarized in Supplemental Table S1),14 –20 as well as data from real-world observational and pharmacovigilance studies,43,44 that have established the robust cardiovascular and cerebrovascular benefits from GLP-1RAs in T2DM and laid the ground for their broader incorporation in clinical practice guidelines.6,7 Compared to previous works, the present updated meta-analysis has included data from the recently published placebo-controlled NCT01455896, a pre-approval CVOT that investigated cardiovascular outcomes in T2DM patients treated with the ITCA 650 device, that delivers subcutaneous infusion of exenatide via an osmotic mini-pump, and showed significant reduction in MACE in ITCA 650-treated patients. 21 Importantly, subgroup analyses revealed consistent effects of GLP-1 RAs both in primary and secondary prevention (i.e. in the latter case in patients with prior history of MI or stroke, or established cardiovascular disease). In this context, it is worth noting that the line of evidence on GLP-1 RAs’ cardiovascular potency exceedingly grows, including the very recent publication of the SELECT trial in patients with pre-existing cardiovascular disease and overweight or obesity, 45 with GLP-1 RAs steadily claiming their place in cardiovascular disease prevention also beyond T2DM.
Besides GLP-1RAs, the current meta-analysis included data from four placebo-controlled RCTs assessing the cardiovascular and cerebrovascular safety of the novel GIP/GLP-1 RA tirzepatide in T2DM patients. Data from GLP-1RAs and GIP/GLP-1 RAs trials were pooled for the primary outcome meta-analysis; nevertheless, the pooled effect on MACE was clearly driven by the significant effects of GLP-1RAs, as no association between MACE and tirzepatide treatment was uncovered. Similarly, no associations between all-cause and cardiovascular mortality and tirzepatide treatment were disclosed. Accordingly, the test for subgroup differences (i.e. GLP-1 RAs vs GIP/GLP-1 RAs) indicated no significant subgroup effect; however, the smaller number of RCTs and T2DM patients in the tirzepatide subgroup may have hindered detection of subgroup differences. Some additional methodological nuances should be clarified. First, although all four tirzepatide RCTs were either phase 2 or 3 trials, they were not designed as CVOTs and were evidently underpowered to evaluate cardiovascular efficacy as indicated by the small sample sizes and short follow-up periods. With respect to the latter, it is intriguing that CVOTs on GLP-1 RAs use in T2DM have documented divergence in cardiovascular outcomes between treatment and control groups after the first year of GLP-1 RA treatment.30,37 Notably, in the current meta-analysis, sensitivity analysis also revealed time-dependent effects of GLP-1 RAs in stroke prevention. Second, contrary to CVOTs on GLP-1 RAs that included T2DM patients with established cardiovascular disease or at high cardiovascular risk, tirzepatide trials randomized T2DM patients irrespective of presence of prior cardiovascular disease or cardiovascular risk factors, a fact that may account for the very low number of recorded MACE, and possibly for type II errors.
In accordance with our findings, a previous meta-analysis that assessed tirzepatide’s cardiovascular safety in T2DM, 46 including RCTs with at least one placebo or active comparator arm, found no significant differences in MACE, all-cause and cardiovascular mortality between tirzepatide-treated patients and controls. Compared to the aforementioned study, we restricted our analysis to placebo-controlled RCTs and additionally included the recently published data of the SURMOUNT-2 trial. 42 Still, given the methodological limitations mentioned, the cardioprotective benefits from tirzepatide in T2DM likely remain underestimated. Notably, the so-far available evidence on GIP/GLP-1 RAs efficacy in T2DM is striking, with tirzepatide (15 mg once weekly) appearing superior both compared to placebo and GLP-1RAs for glycemic control and body weight reduction, and also superior to basal insulin for glycemic control, without increasing the risk for hypoglycemia. 47 Given the complementary and synergistic effects of GIP and GLP-1 receptor agonism, 48 the results of the SURPASS CVOT trial that will comprehensively assess tirzepatide’s cardiovascular safety and efficacy against the GLP-1 RA dulaglutide in patients with T2DM and established cardiovascular disease, are eagerly awaited. 12
In addition, we performed subgroup analyses, assessing the association between GLP-1 RA treatment and different stroke subtypes. Our findings demonstrate that GLP-1 RA use is associated with significant reduction in the risk of ischemic stroke, while no similar effect was detected for haemorrhagic stroke. In line with these findings, a post-hoc analysis of the SUSTAIN 6 and PIONEER 6 trials on the GLP-1 RA semaglutide, 49 showed significant reduction in the incidence of all-cause stroke in treated patients versus controls; an effect that was mainly driven by reduction of the odds of ischemic stroke and in particular, small-vessel occlusion (i.e. lacunar stroke). Interestingly, subgroup analyses also revealed that the net clinical benefit from GLP-1 RA use on cerebrovascular outcomes (i.e. stroke) was greater compared to cardiovascular outcomes (i.e. with nonsignificant effect on fatal and nonfatal MI), a finding that has been replicated to date in a number of RCTs and meta-analyses.13,18 Moreover, it is compelling that the heterogeneity in reported stroke outcomes of the present meta-analysis was exceptionally low, with a significant number of trials contributing data to the pooled analyses. Whether these findings imply a stronger neuroprotective potential of GLP-1 RAs as suggested in prior studies remains to be established.50 –52
Some limitations of the present meta-analysis should be acknowledged. First, as this was an aggregate data meta-analysis, potential associations between clinical parameters, comprising concomitant treatments, comorbidities, T2DM duration, and clinical outcomes could not be evaluated. Second, as MACE was the primary outcome of all included RCTs, stroke meta-analyses were based on secondary outcome assessment. In light of the robust evidence of GLP-1 RAs’ effects for stroke prevention, it should be emphasized that dedicated RCTs designed to evaluate GLP-1 RAs and GIP/GLP-1 RAs efficacy and safety specifically in stroke are direly needed. Third, the generalizability of the present findings may be limited, since RCTs on GLP-1 RAs or GIP/GLP-1 RAs included T2DM patients at high cardiovascular risk or without cardiovascular risk factors, respectively. In addition, we could not assess the potential role of GIP/GLP-1 RAs in stroke due to unavailability of data for meta-analysis from the so-far published RCTs. Thus, well-designed CVOTs with adequate sample sizes and follow-up periods are needed to comprehensively evaluate the safety and efficacy of tirzepatide in stroke prevention in T2DM. Fourth, despite differences in plasma half-life and structural homology of GLP-1 RAs to GLP-1 receptor, no subgroup analyses were pursued, since previous meta-analyses from our group and others have already established that MACE and cardiovascular benefits are independent of the structural basis of the GLP-1 receptor analogs.13,18 Fifth, as no data were available for ischemic stroke subtypes, potential beneficial effects of GLP-1 RAs for prevention of ischemic stroke due to large-artery atherosclerosis, cardioembolism or small-vessel occlusion could not be evaluated and remain to be explored in future studies.
In conclusion, our systematic review and meta-analysis provides evidence of strong potential from GLP-1 RA or GIP/GLP-1 RA treatment in reducing MACE, all-cause and cardiovascular mortality in the T2DM population. Most importantly, GLP-1 RAs significantly attenuate the risk of ischemic stroke. Given the current dearth of evidence on tirzepatide, the jury is still out on whether this novel agent may parallel the effects of GLP-1 RAs in stroke prevention. Consequently, well-designed RCTs are needed to cast more light onto the potential cardiovascular and cerebrovascular effects of GIP/GLP-1 RAs in T2DM.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241234238 – Supplemental material for Risk of major adverse cardiovascular events and stroke associated with treatment with GLP-1 or the dual GIP/GLP-1 receptor agonist tirzepatide for type 2 diabetes: A systematic review and meta-analysis
Supplemental material, sj-docx-1-eso-10.1177_23969873241234238 for Risk of major adverse cardiovascular events and stroke associated with treatment with GLP-1 or the dual GIP/GLP-1 receptor agonist tirzepatide for type 2 diabetes: A systematic review and meta-analysis by Maria-Ioanna Stefanou, Aikaterini Theodorou, Konark Malhotra, Diana Aguiar de Sousa, Mira Katan, Lina Palaiodimou, Aristeidis H Katsanos, Ioanna Koutroulou, Vaia Lambadiari, Robin Lemmens, Sotirios Giannopoulos, Andrei V Alexandrov, Gerasimos Siasos and Georgios Tsivgoulis in European Stroke Journal
Footnotes
Acknowledgements
None
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent - ethical approval
This study did not require an ethical board approval or written informed consent by the patients according to the study design (systematic review and meta-analysis).
Guarantor
GT
Contributorship
Dr. Stefanou participated in study concept and design, acquisition of data, analysis and interpretation, and prepared the first draft of the manuscript. Dr. Theodorou participated in acquisition of data, analysis and interpretation, and prepared the first draft of the manuscript. Dr. Mahlotra participated in acquisition of data and contributed to critical revision of the article for important intellectual content. Dr. Aguiar de Sousa participated in acquisition of data, critical revision of the article for important intellectual content. Dr. Katan contributed to analysis, critical revision of the article for important intellectual content. Drs. Palaiodimou, Katsanos, Koutroulou, Lambadiari, and Lemmens participated in critical revision of the article for important intellectual content. Drs. Giannopoulos, Alexandrov, and Siasos contributed to critical revision of the article for important intellectual content. Dr. Tsivgoulis was responsible for study design, analysis and interpretation, critical revision of the article for important intellectual content.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
