Abstract
Background:
Disease progression is observed across the spectrum of people with multiple sclerosis (MS) and identification of effective treatment strategies to halt progression remains one of the greatest unmet clinical needs.
Objectives:
The Canadian Prospective Cohort Study to Understand Progression in MS (CanProCo) was designed to evaluate a wide range of factors associated with the onset and rate of clinical disease progression in MS and to describe the interplay between these factors.
Design:
A prospective cohort study.
Methods:
CanProCo is a national, prospective, observational cohort study that has recruited 944 individuals from 5 large academic MS centers in Canada. Participants include people with radiologically isolated syndrome (RIS), early relapsing-remitting and primary progressive MS (RRMS, PPMS), and healthy controls (HCs). Annually, participants complete self-reported questionnaires, undergo clinical evaluation and, if clinically indicated, magnetic resonance images (MRIs) of the brain and cervical spinal cord; in a subset of participants (n = 399), blood, and research MRIs of the brain and cervical spinal cord are collected. Linkages to health administrative databases are available at three sites.
Results:
Overall, 944 participants were recruited (53 HCs, 63 RIS, 751 RRMS, 77 PPMS). RIS and MS participants had a mean age of 39.0 years and 70.5% female. The mean time since diagnosis was 2.7 years. There were differences observed in the Expanded Disability Status Scale score and components of the MS performance test (walking speed test, manual dexterity test, processing speed test, and low-contrast visual acuity) between RIS and MS subtypes. Questionnaires revealed more symptoms of depression and anxiety and impaired physical and mental quality of life in people with RIS/MS versus HCs and differences across RIS/MS subtypes.
Conclusion:
Physical and mental neurological disability is prevalent even in the earliest stages of MS. Transdisciplinary approaches such as those used in CanProCo are needed to better characterize clinical progression in MS. Additional CanProCo results, including MRI, biological, and pharmaco-economic data will be forthcoming. Going forward, CanProCo’s data sharing and collaborative vision will facilitate numerous global collaborations, which will inform the development and implementation of effective interventions for people with MS around the world.
Introduction
One of the greatest unmet needs in the field of multiple sclerosis (MS) is treatments that definitively modify the course of neurological disease worsening, or progression in MS. This recognition has led to numerous international efforts aiming to address this unmet need—one of the largest being the International Progressive MS Alliance. 1 The Canadian Prospective Cohort (CanProCo) Study to Understand Progression in MS was designed with the overarching aim to better understand the multitude of factors, and interactions between these factors that lead to onset, heterogeneity, and magnitude of progression in MS.
CanProCo is a large, national prospective cohort study of healthy control (HC) participants and individuals with radiologically isolated syndrome (RIS), and in early stages of different subtypes of MS (clinically isolated syndrome (CIS) with dissemination in space per 2017 McDonald Criteria, 2 relapsing-remitting MS (RRMS), and primary progressive MS (PPMS)) followed over 5 years at one of five participating academic MS centers in Canada. CanProCo participants contribute a wide range of clinical, self-reported, biological, and imaging data during yearly study visits. Participants in British Columbia (one site) and Alberta (two sites) agree to link study data to provincial health administrative data. The considerable breadth and depth of data collected in CanProCo will allow for a thorough evaluation of biological factors (e.g. immunophenotyping) extending to social determinants of health (e.g. health system factors) that are relevant to disease worsening across the spectrum of MS. Knowledge gained from CanProCo may enable a better understanding of the mechanisms underlying MS disease onset and progression that will in turn inform future research, clinical trials, MS practice, and health policy, ultimately improving the lives of people living with MS in Canada and beyond.
This manuscript describes the baseline demographic and clinical characteristics of CanProCo participants, which will serve as a reference for current and future studies utilizing CanProCo data.
Methods
A comprehensive description of the CanProCo study rationale, inclusion and exclusion requirements, and study design has been previously published 3 and the protocol adheres to Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. Briefly, study participants were enrolled at five academic MS research centers in Canada between April 2019 and March 2022. Data collected from all participants constitute the “foundation cohort” (core data); and a subcohort of participants contribute additional data, described further below (Figure 1).
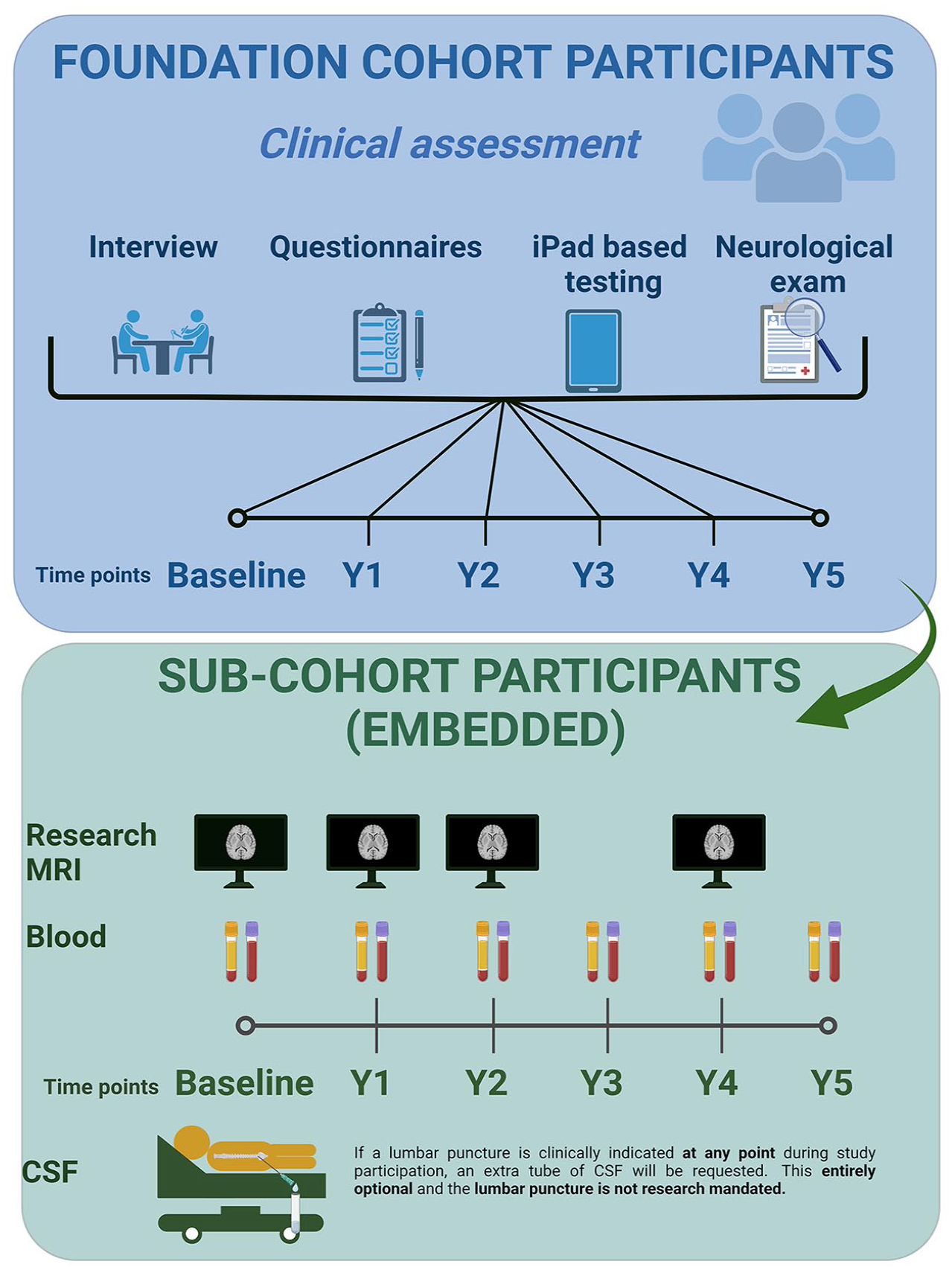
CanProCo study schematic.
Inclusion criteria for the foundation cohort and specific subcohorts in participants with RIS (per 2009 Okuda criteria), CIS with dissemination in space, RRMS, and PPMS as well as healthy control (HC) participants are summarized in Table 1. All CanProCo participants form the “foundation” cohort and contribute clinical data and clinical MRIs. A subset of individuals from the CanProCo foundation cohort with RIS, CIS/RRMS, and PPMS who meet more restrictive criteria (as outlined in Table 1) and who are willing to give blood and participate in research MRIs at each study visit constitute the RIS, CIS/RRMS, and PPMS subcohorts. All RIS participants participated in the RIS subcohort. For the remainder of this manuscript, those with CIS and dissemination in space will be grouped together with RRMS and referred to collectively as the “RRMS subgroup.”
CanProCo inclusion and exclusion criteria in foundation cohort and subcohorts.

CIS, clinically isolated syndrome; EDSS, expanded disability status scale; PPMS, primary progressive multiple sclerosis; RIS, radiologically isolated syndrome; RRMS, relapsing-remitting multiple sclerosis.
Participants were recruited via referral through a member in their circle of care, or self-referral from advertisement (e.g. posters and website). Each CanProCo participant signed an informed consent form approved by the study site’s research ethics board.
All research staff received training regarding study protocols and standard operating procedures. Ongoing training and data audits ensure continued data quality. Additional details of data standardization and quality assurance are detailed in the methods manuscript. 3
Data collection
A comprehensive description of all clinical, imaging, and biological questionnaires collected in CanProCo, and linkage to health administrative data have been described previously. 3 Briefly, CanProCo data collection includes demographic and clinical information, a clinician-assessed outcome: expanded disability status scaled (EDSS) score administered by an experienced neurologist, performance-based outcomes (iPad-based multiple sclerosis performance test (MSPT)), patient-reported outcomes collected by various questionnaires (Table 2), and clinical MRIs of the brain conforming to accepted guidelines, 6 and research-grade MRIs of the brain and cervical spinal cord which include advanced sequences in subcohort participants (Figure 1). The MSPT7,8 is an iPad-administered set of tests and questionnaires that includes three tests similar to those that constitute the multiple sclerosis functional composite 9 including the manual dexterity test (similar to the 9-hole peg test), the walking speed test (similar to the 25-foot timed walk), the information processing test (similar to the symbol digit modalities test). The MSPT also includes visual tests, including high-contrast visual acuity and low-contrast letter acuity.
CanProCo questionnaires administered.
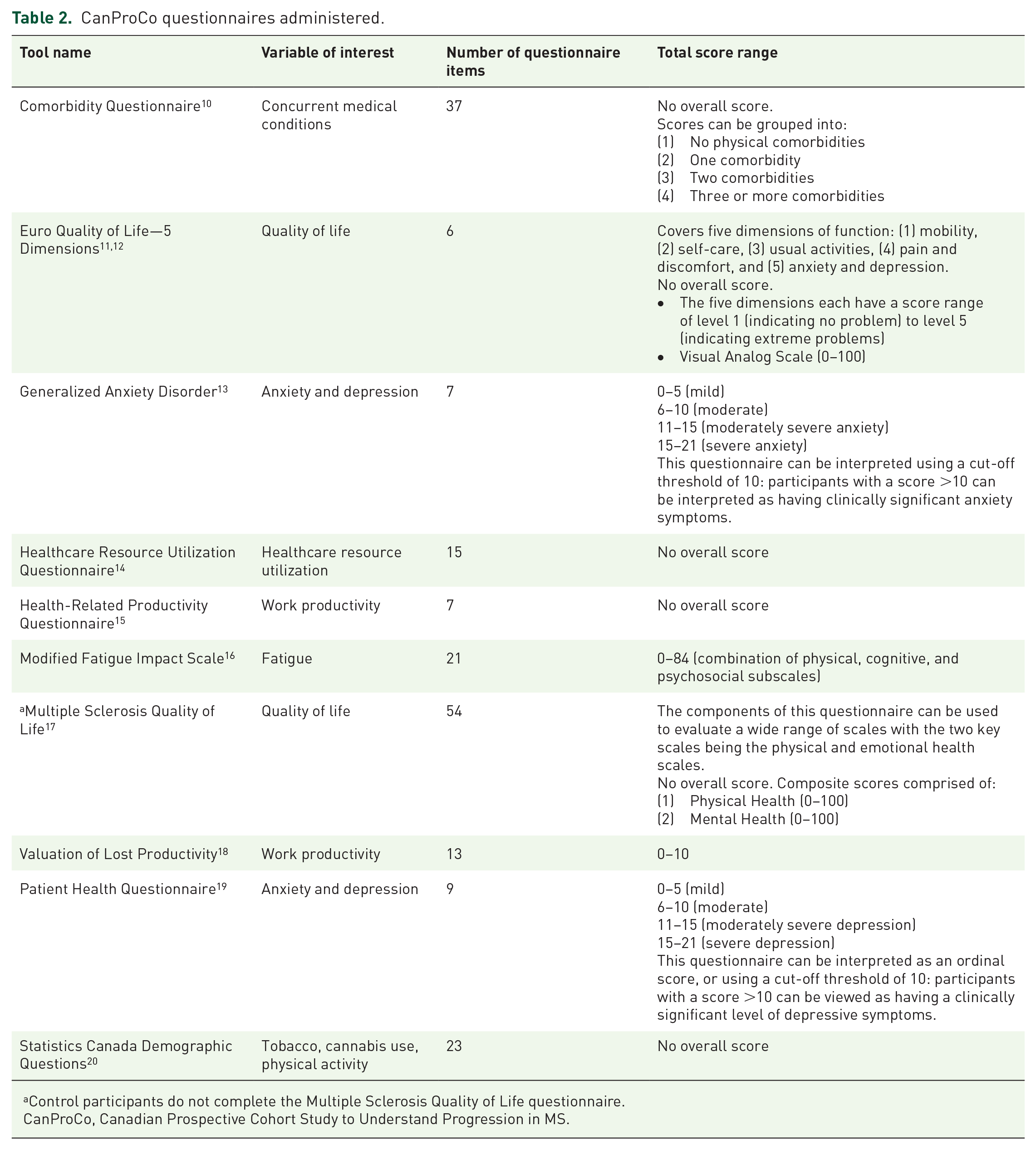
Control participants do not complete the Multiple Sclerosis Quality of Life questionnaire.
CanProCo, Canadian Prospective Cohort Study to Understand Progression in MS.
For questionnaires, the Patient Health Questionnaire (PHQ-9) 19 can be interpreted as an ordinal score, or using a cut-off threshold of 10: participants with a score >10 can be viewed as having a clinically significant level of depressive symptoms. Similar to the PHQ-9, the Generalized Anxiety Disorder questionnaire 13 (GAD-7) can be interpreted using a cut-off threshold of 10: participants with a score >10 can be interpreted as having clinically significant anxiety symptoms.
The (Euro quality of life, 5 dimensions11,12 (EQ-5D)) items cover five dimensions of function: (1) mobility, (2) self-care, (3) usual activities, (4) pain and discomfort, and (5) anxiety and depression. The components of the MS quality of life (QOL) can be used to evaluate a wide range of scales with the two key scales being the physical and emotional health scales.
Standardized case report forms (CRFs) were used to collect participant baseline data (Supplemental Documents 1a–d). Demographic details were self-reported during the baseline study visit. Information related to RIS/MS disease onset, diagnosis, relapse, and treatment history was abstracted from the medical chart and confirmed by the participant during the study visit.
A total of 70 mL of blood was collected from HCs and subcohort participants at each study visit: 60 mL was collected in ethylenediaminetetraacetic acid-treated tubes for immediate isolation of peripheral blood mononuclear cells (PBMC) (50 mL) and plasma (10 mL), and 10 mL was collected in a tube with gel for optimal serum collection. PBMC, plasma, and serum samples were frozen at −80°C, while PBMCs were subsequently stored in liquid nitrogen. In consenting participants undergoing spinal taps for clinical reasons, a single additional 5 mL tube of cerebrospinal fluid (CSF) was taken for research purposes and spun down. Cell pellet and supernatants were frozen at −80°C. CSF samples were obtained at any point during study participation, and repeat samples were collected if the participant provided consent and underwent another spinal tap for clinical reasons.
Data storage
Clinical data were recorded on CRFs at each study site and uploaded to the central study database hosted by the Praxis Spinal Cord Institute’s Global Research Platform. All biological specimens were shipped to the biological sample repository sites, the Centre Hospitalier de l’Université de Montréal, and the University of Calgary. De-identified MRI data collected at each site (clinical and research MRI scans) were sent electronically via secure file transfer protocol, where they were stored in the UBC MSMRI Imaging Repository, a secure server in the UBC EduCloud infrastructure.
Statistical analyses
Descriptive statistics were used to describe the data in all participants, and in subgroups. All data available from each participant were included. Chi-squared tests, Fisher’s exact tests, Wilcoxon rank-sum tests, and Kruskal–Wallis tests were used for group comparisons. Hypotheses were tested at the p < 0.05 level of confidence. In view of the descriptive nature of this analysis, adjustments for multiple tests were not used. MSPT outcomes were converted into Z-scores which were provided by the device, and calculated from regression-based equations derived from a healthy volunteer study. 17
Ethics approval
Each CanProCo participant provided a written informed consent form approved by the study site’s research ethics board. For full details on consent and ethical approval, please see the ethics statement at the end of this article.
Results
A total of 944 adults (n = 891 patients (n = 63 RIS; n = 751 RRMS; n = 77 PPMS); and n = 53 HC participants) attended baseline study visits between April 2, 2019 and March 2, 2022. All study participants contributed data to the analyses presented below, but not all participants had complete baseline datasets.
Geographic distribution
Participants were distributed throughout five large academic MS centers with similar numbers from each site: Toronto, 161; Montreal, 206; Edmonton, 177; Calgary, 206; Vancouver, 194.
Demographic and clinical characteristics
At baseline, the mean age of the 891 people with RIS/MS was 39.0 years (SD = 10.3) and 70.5% were female. HCs (n = 53) had a mean age of 37.0 years (SD = 11.3) and 67.9% were female (Tables 3–6). The mean time since diagnosis of all MS/RIS participants was 2.7 years (SD = 2.7). When demographics were assessed by disease subtype: RIS (n = 63) participants were a mean age of 42.3 years (SD = 10.3) and 77.8% female with a mean time since diagnosis of 2.2 (SD = 2.7) years, CIS/RRMS (n = 751) participants were a mean age of 37.5 years (SD = 9.6), 72.7% female, and a mean time since diagnosis of 2.8 (SD = 2.7) years, and PPMS (n = 77) participants were a mean age of 51.0 years (SD = 7.7), 42.9% female, with a mean time since diagnosis of 2.6 (SD = 2.6) years. Notably, CIS/RRMS subcohort patients (who had to be treatment-naïve at the first study visit) had a mean time since diagnosis of 0.9 (SD = 1.4) years. There were no differences observed between RIS/MS and HC participants with respect to age and sex. PPMS participants were older, had a lower proportion of females, and had a longer time since symptom onset than RRMS participants.
Demographics, social determinants, and health behavior.

The p-value <0.05 for RIS versus RRMS group comparisons.
The p-value <0.05 for RRMS versus PPMS group comparisons.
HC, healthy controls; MS, multiple sclerosis; PPMS, primary progressive multiple sclerosis; RIS, radiologically isolated syndrome; RRMS, relapsing-remitting multiple sclerosis; SD, standard deviation.
Clinical characteristics.

Unless otherwise specified.
The p-value <0.05 for RIS versus RRMS group comparisons.
The p-value <0.05 for RRMS versus PPMS group comparisons.
In these PPMS participants, the possibility of new/worsening symptoms due to a relapse was raised during their disease course, but the treating neurologist ultimately deemed these symptoms to be consistent with a primary progressive MS disease course.
DMT, disease-modifying therapy; EDSS, expanded disability status scale; HC, healthy controls; IQR, interquartile range; MS, multiple sclerosis; PDDS, patient-determined disease steps disability score; PPMS, primary progressive multiple sclerosis; RIS, radiologically isolated syndrome; RRMS, relapsing-remitting multiple sclerosis; SD, standard deviation.
Demographics, social determinants, and health behaviors of subcohort participants.

The p-value <0.05 for RIS versus RRMS group comparisons.
The p-value <0.05 for RRMS versus PPMS group comparisons.
MS, multiple sclerosis; PPMS, primary progressive multiple sclerosis; RIS, radiologically isolated syndrome; RRMS, relapsing-remitting multiple sclerosis; SD, standard deviation.
Clinical characteristics of subcohort participants.

Unless otherwise specified.
The p-value <0.05 for RIS versus RRMS group comparisons.
The p-value <0.05 for RRMS versus PPMS group comparisons.
In these PPMS participants, the possibility of new/worsening symptoms due to a relapse was raised during their disease course, but the treating neurologist ultimately deemed these symptoms to be consistent with a primary progressive MS disease course.
DMT, disease-modifying therapy; IQR, interquartile range; MS, multiple sclerosis; PDDS, patient-determined disease steps; PPMS, primary progressive multiple sclerosis; RIS, radiologically isolated syndrome; RRMS, relapsing-remitting multiple sclerosis; SD, standard deviation.
CanProCo participants had a wide range of self-reported ethnicities. The most common ethnicities of MS/RIS participants included: 78.8% white, 4.5% South Asian, 4.4% Arab/West Asian, 4.3% mixed race, and 2.5% Black and Caribbean. In HCs, the most common ethnicities were: 69.8% white, 5.7% South Asian, 5.7% Latin American, 5.7% Korean, 5.7% mixed race, 3.8% Arab/West Asian, and 3.8% Black and Caribbean. Most MS/RIS participants (78.9%) had some post-secondary education. Most MS/RIS participants were employed full-time or part-time (72.5%) with 12.9% unemployed due to disability. A lower proportion of participants with PPMS were employed full-time or part-time in comparison to those with RRMS (55.8% vs 73.9%, respectively, p = 0.001). Nearly two-thirds of MS/RIS participants were partnered (62%), 29.3% were single, and 8.3% reported being divorced or separated. A higher proportion of PPMS participants reported being divorced or separated compared to RRMS participants (18.2% vs 7.5%, respectively, p = 0.004). There was a range of reported alcohol consumption in the past year: 15.6% of MS/RIS participants reported no alcohol consumption, and 22.6% reported alcohol consumption less than once monthly. When the frequency of having had more than five drinks of alcohol on one occasion in the past year was evaluated, most MS/RIS participants reported a frequency of “never” (44.3%) or that such occurrences took place less than once per month (36.2%) in the past year. A greater proportion of PPMS participants reported never consuming more than five drinks of alcohol on one occasion in the past year in comparison to RRMS (64.6% vs 45.3.0%, p < 0.05). Mean body mass index was 27.4 (SD = 7.4) among MS/RIS participants. Three-quarters (74.6%) of participants with MS reported no known family history of MS.
The most common presenting clinical symptom in MS/RIS participants was sensory (32.6%), followed by multifocal (22.5%) and visual (17.6%). In RRMS, the most common presenting symptoms were sensory (37.4%), multifocal (27.6%), and visual (20.9%); while in PPMS the most common presenting symptoms were pyramidal (46.8%), multifocal (29.9%), and sensory (11.7%). With regards to disease-modifying treatment (DMT) use: 51.9% of all RIS/MS participants were on DMT at baseline. Of note, one RIS participant (1.6%), 56.6% of RRMS, and 53.2% of PPMS participants were on DMT at baseline. When evaluating DMT use at baseline in subcohorts where there were specific inclusion/exclusion criteria, including DMT use prior to enrollment (Table 6): only 3.7% of RRMS subcohort participants were on DMT (n = 9 on minocycline due to standard clinical practice at one site, and n = 1 on glatiramer acetate, started on a patient before the study visit, but after the baseline bloodwork was collected), and 48.5% of PPMS. Most CanProCo MS/RIS participants reported using Vitamin D supplementation (92.3%), at doses ranging from 1000 to 10,000 IU daily.
Clinical disability measures
Baseline median EDSS in all MS and RIS participants was 1.5 (interquartile range, IQR = 1.5) (Tables 4 and 6). The median EDSS was lowest among those with RIS (0 (IQR = 1.5)), followed by those with RRMS (1.5 (IQR = 2.0)), and PPMS (4.0 (IQR = 2.0), p < 0.001). Based on the patient-determined disease steps score, the majority of MS/RIS participants reported having no (60.7%) or only mild (20.8%) disability at baseline: in RIS 87.3% and 7.9%; RRMS 64.3% and 21.7%; and PPMS 3.9% and 22.1% reported having no or mild disability, respectively, with significant differences across subgroups (p < 0.001).
The MSPT was completed by n = 641 participants, and both raw scores and Z-score are summarized in Table 7. Based on the mean Z-scores, participants with PPMS were generally impaired, but RIS and RRMS did not demonstrate clear evidence of impairment (mean Z-scores in the range of 0). When comparing subgroups, PPMS demonstrated greater deficits in each test in comparison to RRMS (p < 0.001). On the other hand, there were significant differences between RRMS and RIS in PST and visual acuity at 100% (p < 0.05), but these results were not adjusted for multiple comparisons.
Multiple sclerosis performance test raw and Z-scores.
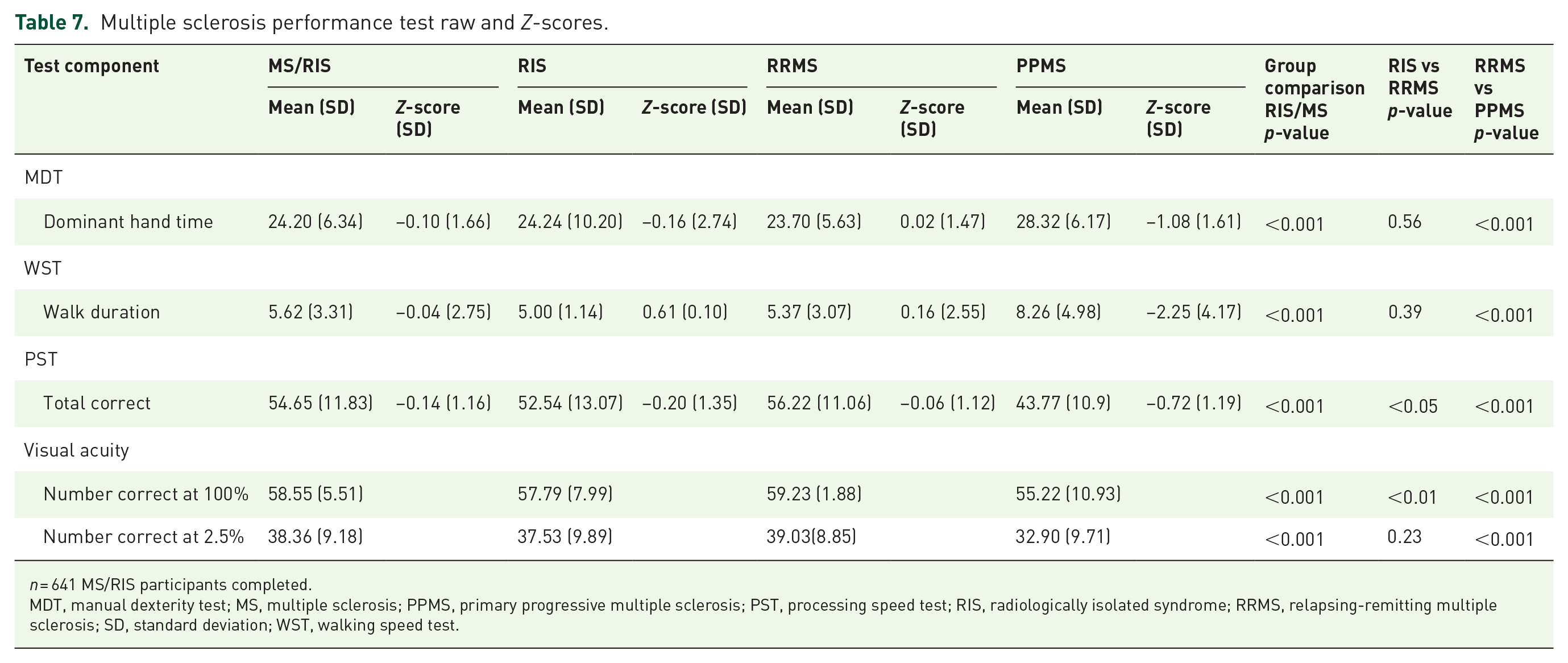
n = 641 MS/RIS participants completed.
MDT, manual dexterity test; MS, multiple sclerosis; PPMS, primary progressive multiple sclerosis; PST, processing speed test; RIS, radiologically isolated syndrome; RRMS, relapsing-remitting multiple sclerosis; SD, standard deviation; WST, walking speed test.
There were differences observed across CanProCo study sites in the distribution of participants, and many key demographic and clinical characteristics, summarized in Supplemental Table 1. As such, CanProCo site is a covariate that will need to be taken into account for relevant future analyses.
Questionnaires
Results of select questionnaires evaluating depressive symptoms (PHQ-9), 19 anxiety symptoms (GAD-7), 13 and QOL (EQ-5D11,12 and MSQOL 18 ) are summarized below (Table 8). Results of additional questionnaires, including workplace productivity, 10 comorbidities, 16 and other QOL scales21,22 have been or will be reported in subsequent publications. 23
Results of select questionnaires.

p-values for group comparisons generated using Wilcoxon rank-sum tests due to the skewed distributions. PHQ-9 (n = 880 MS/RIS and n = 44 HC completed); GAD-7 (n = 837 MS/RIS and n = 34 HC completed); EQ5D (n = 880 MS/RIS, n = 44 HC completed); MSQOL-54 (n = 878 MS/RIS completed and not administered to HC).
EQ-5D, Euro quality of life, 5 dimensions; GAD-7, Generalized Anxiety Disorder; HC, healthy controls; MS, multiple sclerosis; MSQOL, MS quality of life; PHQ-9, Patient Health Questionnaire; PPMS, primary progressive multiple sclerosis; PST, processing speed test; RIS, radiologically isolated syndrome; RRMS, relapsing-remitting multiple sclerosis.
The median PHQ-9 score was higher in people with RIS/MS vs HCs (median score of 6 vs 2, p < 0.0001), reflecting more symptoms of depression in people with RIS/MS. Based on the PHQ-9, the prevalence of clinically meaningful elevations in depressive symptoms in the MS/RIS was 29.5% while it was significantly lower at 6.8% (95% CI: 2.1%–19.6%) in HCs (unadjusted OR = 5.7 (95% CI: 1.8–29.2), p = 0.0005). When comparing subgroups, there was no difference in PHQ-9 scores across RIS and MS subtypes (p = 0.25).
The median GAD-7 score was also higher in people with RIS/MS compared to HCs (4 vs 2, p < 0.01), reflecting more symptoms of anxiety in those with RIS/MS. The prevalence of clinically significant anxiety was 16.6% (95% CI: 14.2%–19.3%) in RIS/MS and 6.8% (95% CI: 2.1%–19.6%) in HCs (OR = 2.72 (95% CI: 0.85–13.9, p = 0.09), which was numerically lower, but not statistically significant.
The median EQ-5D health status score was higher in those with RIS/MS versus HCs (90 vs 80, p < 0.001). Specifically, people with RIS/MS reported significantly higher (worse) scores in all domains but self-care versus HCs. When comparing across RIS/MS subgroups, participants with PPMS reported worse scores in mobility, self-care, usual activities, and pain/discomfort domains (all p < 0.001); however, they did not differ significantly with respect to the anxiety and depression domain from those with RIS/RRMS (p = 0.64). EQ-5D subscale scores for mood and anxiety were concordant with results observed with the GAD and PHQ-9 (p < 0.01).
In people with RIS/MS, the median physical health score of the MSQOL was 90 (IQR = 35 (65–100)) and the median emotional health score was 72 (IQR = 28 (84–56)). The MSQOL was not administered to HCs. When evaluating across RIS/MS subgroups, there were group differences in physical health (p < 0.001) but not mental health (p = 0.29). When pairwise comparisons were performed there was greater physical disability in PPMS versus RRMS, but not mental disability. On the other hand, there were no differences observed in any of the measures between RRMS and RIS, suggesting that the level of physical and mental disability was similar in these two groups.
Additional CanProCo results, including MRI, biological, and pharmaco-economic data will be forthcoming.
Discussion
CanProCo is a national prospective cohort study designed to shed insight into the many relevant factors and how they interact to result in disease worsening in MS. An understanding of the determinants of progression sets the stage for the development of targeted therapies, including experimental drugs and treatment strategies that can prevent and meaningfully alter the trajectory of progression in MS, which is one of the field’s major unmet clinical needs.
In this manuscript, we reported the baseline demographic and clinical characteristics of CanProCo participants: HCs and a relatively early group of people with RIS/MS, who are also reflective of the “spectrum” of MS—spanning from those without overt clinical symptoms (RIS) to those with early RRMS, to those with early PPMS. There were expected abnormalities in various measures of neurological disability in those with established MS (RRMS and PPMS) and RIS in comparison to HCs having similar distributions of age and sex. Moreover, there were expected differences observed in clinical characteristics between disease subtypes, including more composite disability as measured by the EDSS and the MSPT and more physical disability and other domains based on patient-reported outcomes in those with PPMS versus RRMS. One important observation is that in individuals with RIS, there were findings in the questionnaires evaluating mood symptoms and QOL, which is in keeping with recent literature demonstrating that when sensitive clinical tools are utilized, people with RIS have evidence of subtle neurological deficits. 24 In addition, we observed lower mood (including nearly 30% in the clinically meaningful range), higher anxiety, as well as lower QOL across the spectrum of MS in comparison to HCs, even in early RRMS and RIS. Interestingly, when evaluating MS disease subtypes, PPMS consistently had more physical disability, but mental health was comparable to those with RRMS. Moreover, there was no difference observed between RRMS and RIS in the questionnaires evaluating mood and QOL, suggesting that those without overt clinical symptoms of MS (RIS) are, in many ways, indistinguishable from those with established MS. These findings make it evident that, across the spectrum of MS, neurological disability as measured by a variety of tools (physical health, mental health, pain, and general QOL) are negatively affected, even in the very earliest stages of the disease. It will be of high interest to monitor these measures longitudinally to evaluate how they change over time in CanProCo participants and whether there are differences in trajectories of change across disease subtypes and according to various demographic/clinical characteristics.
There are additional national cohorts of people with MS that have been established around the world in recent years. Similar to CanProCo, some of these cohorts are collecting a wide range of data on participants, including clinical, biological, and imaging data. Specific examples include the Ausimmune/Auslong study that recruited patients between 2003 and 2006 and collected blood samples, a range of clinical information, and basic MRI data, 25 and the Queensland cohort. 26 In recent years, both Australian cohorts have reported on a wide range of findings, including findings of interest on diet and its relationship to MS disease activity, neuropsychiatric features of MS, proteomics related to MS risk, as well as employment.27–31 The German National MS Cohort (German Competence Network Multiple Sclerosis (KKNMS) networks) also recruited a group of CIS and RRMS patients nearly a decade ago and has reported on a wide range of topics32–35 including genetics, environmental factors, pain, fatigue, as well as fluid biomarkers in MS. Finally, a recently established national cohort is the Scottish Future MS Cohort which has recruited a group of newly diagnosed people with RRMS (n = 440) and was established with the goal of developing predictive tools to aid MS clinical care. This cohort recently reported baseline and early longitudinal clinical results. 36 While there are similarities between these studies and CanProCo, a few distinct features of CanProCo are worth highlighting. CanProCo was specifically designed with the aim of understanding mechanisms of disease progression in MS and includes people with MS of distinct subtypes as well as RIS that will allow an evaluation of progression across the spectrum of MS. Moreover, when evaluating differences in data collection, CanProCo includes a wide range of biological factors, both clinical and research MRIs (including spinal cord MRIs), a range of clinical and performance-based measures, including those collected using digital devices, and also includes the ability to link to health administrative data at three of five sites. Thus, the depth and breadth 3 of data collection in our cohort is unique and will be invaluable to understand the wide range of factors that relate to disease progression in MS. This is a particular strength of CanProCo since MS is a complex, chronic, slowly progressive disease that will inevitably require multimodal tools to facilitate a comprehensive understanding of disease mechanisms. Finally, another noteworthy feature of CanProCo is that there was more diverse racial/ethnic representation compared to other contemporary cohorts: 21.2% of CanProCo participants were non-white racially, which is reflective of the ethnic diversity in large, academic MS centers in Canada. The ethnic diversity observed in CanProCo is a much higher proportion than other cohorts including the Queensland cohort 26 (Direct Communication with Dr Simon Broadley, January 8, 2024), the Ausimmune/Auslong cohort 25 (Direct Communication with Dr Bruce Taylor, January 18, 2024), the Scottish cohort, 36 and recent phase III clinical trials, 37 all which reported >97% white participants. The KKNMS38,39 (direct communication and data on file with Drs Anke Salmen, Ralf Gold, and Heinz Wiendl, January 8, 2024) also observed a substantially higher proportion of white participants (95%). Following disease trajectories of non-white participants in CanProCo will be informative to understand how race/ethnicity may influence disease progression in MS.
One of the exploratory aims of CanProCo is to utilize novel technologies to improve clinical disease monitoring in MS, which is a great unmet clinical need. Specifically, CanProCo incorporated digital devices, including the iPad-based MSPT, and FLOODLIGHT, a smartphone app designed for MS40,41 in the early stages of the study. Though FLOODLIGHT was unfortunately retired due to decisions made by the application sponsor, there remains great interest in validating novel digital measures that may assist with more accurate disease monitoring in MS, which is another great unmet clinical need. With the upcoming extension of CanProCo, the hope is to incorporate novel technologies, including digital tools, smartphone apps, novel biological analysis tools, as well as artificial intelligence to analyze the “big data” emerging from this study.
Another unique aspect of CanProCo is its funding structure, which is a hybrid of non-profit organizations, government funders, and industry partners. Despite the inherent complexities related to multiple funding sources, CanProCo’s success demonstrates that such hybrid funding schemes are feasible and should be considered for large, national, resource-intensive studies that necessitate budgets that would be prohibitive for single funding agencies. The hope is that CanProCo’s unique funding structure will set a precedent for the field so that future large-scale studies can be funded through such hybrid mechanisms. With the planned extension of CanProCo to 10 years of follow-up, multiple funding sources are being considered, including non-profit organizations, philanthropy, industry partners, and government funding agencies.
Another component of CanProCo’s design worthy of discussion is that this is a study that was designed to facilitate collaborations with existing and ongoing studies around the world, with the overarching goal of improving the efficiency of scientific discovery. It is increasingly recognized that collaborative efforts are key to minimizing redundancy and accelerating scientific progress in a field. 42 In the initial design stages of CanProCo, efforts were made to utilize similar inclusion/exclusion criteria 43 and data collection strategies (utilizing standardized MRI protocol, 6 use of MSPT device) with existing collaborative studies to facilitate eventual data sharing, including international pragmatic studies such as TREAT-MS, DELIVER-MS,43,44 as well as previously mentioned international cohort studies such as KKNMS, 36 as well as MS-PATHS. 45 Moreover, CanProCo is seeking funding to establish an open science platform that will allow for the release of data (if aligned with the CanProCo data management scheme) to qualified investigators around the world. Furthermore, for any investigators utilizing CanProCo data, analyzed data will be required to be returned to the repository and made available to qualified investigators, further fueling collaborations and scientific discovery efficiency. Although CanProCo is still in its early stages of collaboration and data sharing, we expect that these efforts will substantially increase in the years to come. Looking ahead, it would be beneficial for the field if most studies could be designed to facilitate such collaborations, and data sharing was encouraged or even mandated with the aim of accelerating scientific progress in the field.
There are a number of limitations in CanProCo that will need to be taken into account for subsequent analysis. First, the COVID-19 pandemic led to a halt in study activities at all CanProCo sites for a minimum of 6 months. Despite this, CanProCo was able to complete study recruitment within 1 year of the expected recruitment period. To adjust to the COVID-19 pandemic, several modifications were made to the study protocol including enabling study visits to be delayed for up to 6 months, and allowing for partial virtual data collection when in-person visits were not anticipated to be possible within the 6-month grace period. These factors will need to be taken into account when data are analyzed. The success of CanProCo recruitment despite the restrictions of a global pandemic is reflective of the motivation and willingness of people living with MS to continue to participate in a research study in tenuous circumstances and also illustrates how research studies can pivot and adjust to unexpected circumstances to ensure success. While CanProCo includes people with MS (pwMS) from wide geographic area catchment areas across Canada, selection bias or limitations in generalizability are still an important concern as study participants were recruited from the largest academic urban MS centers in Canada, making it likely that our study population is not entirely representative of all people in early stages of MS in Canada. Moreover, CanProCo was specifically designed with the purpose of trying to understand factors related to progression in MS, thus including people with relatively early MS of different subtypes, making it reflective of an early MS population, rather than MS across the entire disease span. Finally, as with many longitudinal cohort studies, participant retention will be an ongoing concern, and our study team is making every effort to increase participant retention. These efforts include hosting annual educational events developed to engage and inform study participants, increasing patient and public engagement with the development of patient expert panels (who regularly provide feedback on study design, analysis priorities, and information dissemination), the publication of periodic newsletters for participants in both official languages (English and French), regular study result dissemination, as well as distributing CanProCo-branded merchandize to increase participant morale.
An important lesson learned from the launch of CanProCo is that the most time-consuming and rate-limiting steps for study initiation were the legal and privacy requirements to set up data-sharing agreements across multiple institutions. Each CanProCo site institution, related to differences in provincial legislature and institutional requirements, had unique requirements to protect participant privacy at the time of study launch. Moreover, there were specific legal requirements that differed across institutions that ultimately resulted in substantial delays with study initiation. Incorporating novel smartphone-based technologies and the data storage related to such new devices was a major challenge for many sites, as many ethics boards and contracts offices were unfamiliar with the use of novel technologies in research studies at the time of CanProCo launch. Given that we are living in an era where multisite, multinational data-sharing studies are common, and that there are continuously novel technological devices being integrated into clinical trials, together with ever-changing privacy legislation, our experience highlights the need for streamlining study approval processes, particularly those involving privacy legislation, to prevent similar delays. Moving forward, in the MS field and beyond, it is evident that multiple stakeholders (clinicians, scientists, patient representatives, legal entities, hospital administrative representatives, and global ethics and regulatory agencies) will need to collectively evaluate these requirements and find a more efficient way to facilitate multisite, multinational research so that such collaborative studies are not hampered to such an extent in the future.
In conclusion, we summarized baseline demographic and clinical characteristics of CanProCo participants, which reflects an early cohort of people living with RIS, MS, and HCs. We expect that this manuscript will serve as a starting point for numerous future studies and collaborations involving our dataset in the years to come. CanProCo’s unique study design, overarching goal, integration of scientific disciplines, as well as potential to collaborate with existing databases and mandate to share data widely make it a resource that will benefit the field in the years to come. Over time, we expect that findings stemming from CanProCo and other collaborative international efforts will bring us one step closer to developing more effective treatment strategies that can definitively modify the course of disease worsening in MS, ultimately improving patient outcomes for people living with MS in Canada and around the world.
Supplemental Material
sj-docx-1-tan-10.1177_17562864241273045 – Supplemental material for The Canadian Prospective Cohort Study to understand progression in multiple sclerosis: baseline characteristics
Supplemental material, sj-docx-1-tan-10.1177_17562864241273045 for The Canadian Prospective Cohort Study to understand progression in multiple sclerosis: baseline characteristics by Jiwon Oh, Nathalie Arbour, Fabrizio Giuliani, Melanie Guenette, Shannon Kolind, Larry Lynd, Ruth Ann Marrie, Luanne M. Metz, Alexandre Prat, Alice Schabas, Penelope Smyth, Roger Tam, Anthony Traboulsee, Voon Wee Yong and Scott B. Patten in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864241273045 – Supplemental material for The Canadian Prospective Cohort Study to understand progression in multiple sclerosis: baseline characteristics
Supplemental material, sj-docx-2-tan-10.1177_17562864241273045 for The Canadian Prospective Cohort Study to understand progression in multiple sclerosis: baseline characteristics by Jiwon Oh, Nathalie Arbour, Fabrizio Giuliani, Melanie Guenette, Shannon Kolind, Larry Lynd, Ruth Ann Marrie, Luanne M. Metz, Alexandre Prat, Alice Schabas, Penelope Smyth, Roger Tam, Anthony Traboulsee, Voon Wee Yong and Scott B. Patten in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-3-tan-10.1177_17562864241273045 – Supplemental material for The Canadian Prospective Cohort Study to understand progression in multiple sclerosis: baseline characteristics
Supplemental material, sj-docx-3-tan-10.1177_17562864241273045 for The Canadian Prospective Cohort Study to understand progression in multiple sclerosis: baseline characteristics by Jiwon Oh, Nathalie Arbour, Fabrizio Giuliani, Melanie Guenette, Shannon Kolind, Larry Lynd, Ruth Ann Marrie, Luanne M. Metz, Alexandre Prat, Alice Schabas, Penelope Smyth, Roger Tam, Anthony Traboulsee, Voon Wee Yong and Scott B. Patten in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-4-tan-10.1177_17562864241273045 – Supplemental material for The Canadian Prospective Cohort Study to understand progression in multiple sclerosis: baseline characteristics
Supplemental material, sj-docx-4-tan-10.1177_17562864241273045 for The Canadian Prospective Cohort Study to understand progression in multiple sclerosis: baseline characteristics by Jiwon Oh, Nathalie Arbour, Fabrizio Giuliani, Melanie Guenette, Shannon Kolind, Larry Lynd, Ruth Ann Marrie, Luanne M. Metz, Alexandre Prat, Alice Schabas, Penelope Smyth, Roger Tam, Anthony Traboulsee, Voon Wee Yong and Scott B. Patten in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-5-tan-10.1177_17562864241273045 – Supplemental material for The Canadian Prospective Cohort Study to understand progression in multiple sclerosis: baseline characteristics
Supplemental material, sj-docx-5-tan-10.1177_17562864241273045 for The Canadian Prospective Cohort Study to understand progression in multiple sclerosis: baseline characteristics by Jiwon Oh, Nathalie Arbour, Fabrizio Giuliani, Melanie Guenette, Shannon Kolind, Larry Lynd, Ruth Ann Marrie, Luanne M. Metz, Alexandre Prat, Alice Schabas, Penelope Smyth, Roger Tam, Anthony Traboulsee, Voon Wee Yong and Scott B. Patten in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The authors would like to acknowledge Ms Fasna Raufdeen for her assistance with data analysis for this manuscript, Ms Perrine Coquelet for creating ![]() , the NationMS cohort study, Germany: Anke Salmen, Ralf Gold, Heinz Wiendl, and KKNMS collaborators for sharing data related to discussion points, and CanProCo’s Scientific Advisory Board: Drs Amit Bar-Or, Helmut Butzkueven, Daniel S. Reich, Alan Thompson, Heinz Wiendl, and Christina Wolfson.
, the NationMS cohort study, Germany: Anke Salmen, Ralf Gold, Heinz Wiendl, and KKNMS collaborators for sharing data related to discussion points, and CanProCo’s Scientific Advisory Board: Drs Amit Bar-Or, Helmut Butzkueven, Daniel S. Reich, Alan Thompson, Heinz Wiendl, and Christina Wolfson.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
