Abstract
Background:
Post-traumatic acute brain swelling (ABS) is a major cause of elevated intracranial pressure and thus mortality. The current definition of post-traumatic ABS has certain limitations, and there is limited information available regarding ABS associated with traumatic acute subdural hematoma (ASDH).
Objectives:
To investigate the incidence, risk factors, and clinical outcomes of ABS associated with traumatic ASDH.
Design:
Retrospective study.
Methods:
Data for 161 patients diagnosed with traumatic ASDH were retrospectively collected. Novel computed tomography-based criteria were proposed for diagnosing ABS in patients with ASDH and determining its incidence. Univariate and multivariate logistic regression analyses were performed to explore the risk factors of post-traumatic ABS. The Glasgow Outcome Scale (GOS) score, mortality, and functional prognosis of all patients at discharge and the proportion of intraoperative malignant brain bulge in surgical patients were taken as clinical outcome measures.
Results:
A total of 45 (28%) patients experienced post-traumatic ABS, exhibiting significantly lower Glasgow Coma Scale scores on admission (p < 0.001). The incidence of hemispheric and whole-brain swelling was 8.1% and 19.9%, respectively. Risk factors independently associated with post-traumatic ABS were: (1) age [odds ratio (OR) = 0.917, p < 0.001]; (2) platelet to white blood cell ratio (PWR) (OR = 0.887, p = 0.012); and (3) traumatic subarachnoid hemorrhage (SAH) (OR = 4.346, p = 0.005). The ABS cohort had a lower GOS score [2 (1–3) versus 4 (3–5); p < 0.001], higher mortality (46.7% versus 6.9%; p < 0.001), and higher proportion of unfavorable functional prognosis (75.6% versus 34.5%; p < 0.001) upon discharge compared to the no ABS cohort, along with higher proportion of intraoperative malignant brain bulge (43.8% versus 0%; p < 0.001).
Conclusion:
The incidence of ABS associated with ASDH is significantly high overall. Patients with ASDH who have young age, low PWR, and traumatic SAH are at an increased risk of developing post-traumatic ABS, and therefore of poor clinical outcomes.
Keywords
Introduction
Traumatic acute subdural hematoma (ASDH) is the most severe subtype of traumatic brain injury (TBI) due to its high disability and mortality rates. Acute brain swelling (ABS) following TBI is a common clinical phenomenon, which often leads to refractory elevated intracranial pressure (ICP) and rapid neurological deterioration. 1 The lethality of ASDH primarily arises from its frequent association with primary and/or secondary brain damage, including contusion, laceration, edema, or swelling. 2 The mortality of ASDH associated with ABS has been reported to exceed 75%, even when treated with early surgery. 3
Decompressive craniectomy (DC) following hematoma evacuation is a routine procedure used for the treatment of patients with ASDH, particularly those with concomitant ABS prior to surgery. 4 The main mechanisms of malignant brain bulge, an extremely dangerous and urgent situation during DC, include late-onset intracranial hematoma and ABS. Among them, ABS plays a dominant role in the development of intraoperative malignant brain bulge and has the highest mortality.5,6 Thus, it is crucial for neurosurgeons to understand potential risk factors for the development of ABS associated with ASDH. This helps identify individuals at higher risk of developing post-traumatic ABS and enables early diagnosis as well as prompt management of elevated ICP.
On computed tomography (CT), post-traumatic ABS typically presents with two distinct patterns: hemispheric swelling accompanied by an ipsilateral ASDH and whole-brain swelling without significant intracranial lesions (the so-called diffuse brain swelling).1,3,7–9 Currently, there is a lack of standardized criteria for diagnosing post-traumatic ABS; consequently, the actual incidence of ABS associated with ASDH is unknown. Furthermore, few studies have specifically focused on identifying risk factors of ABS associated with ASDH and evaluating its impact on patients’ clinical outcomes.
A previous multicenter retrospective study has demonstrated that ICP monitoring is associated with reduced mortality and improved functional outcomes in patients with severe diffuse TBI. 10 However, the availability of ICP monitoring equipment remains restricted in most centers, leading to a heavy reliance on imaging examinations for the management of post-traumatic ABS. Here, we proposed novel CT-based diagnostic criteria for ABS associated with ASDH. The criteria incorporate an evaluation of sulcal effacement at the vertex, which has been proven to be a reliable indicator for predicting elevated ICP after TBI. 11 The objectives of this study were threefold: (1) to determine the incidence of ABS associated with ASDH, including hemispheric and whole-brain swelling; (2) to identify potential risk factors for ABS associated with ASDH by analyzing clinical, laboratory, and radiological variables; and (3) to evaluate the clinical outcomes of ASDH associated with ABS.
Methods
Patient selection
Patients diagnosed with traumatic ASDH who were treated in the First Hospital of Putian City from September 2018 to September 2023 were identified in this study. The inclusion criteria comprised consecutive patients with non-penetrating head injury, age ⩾18 years, and with an ASDH located in the convexity of the hemisphere. The exclusion criteria were as follows: (1) time interval from trauma to diagnosis exceeding 24 h; (2) history of head trauma, brain tumor, stroke, or craniotomy; (3) long-term anticoagulant or antiplatelet therapy; (4) presence of concomitant epidural hematoma or massive intracerebral hematoma; and (5) bilateral ASDHs. The flow chart (Figure 1) illustrates the process of selecting patients enrolled in the study.
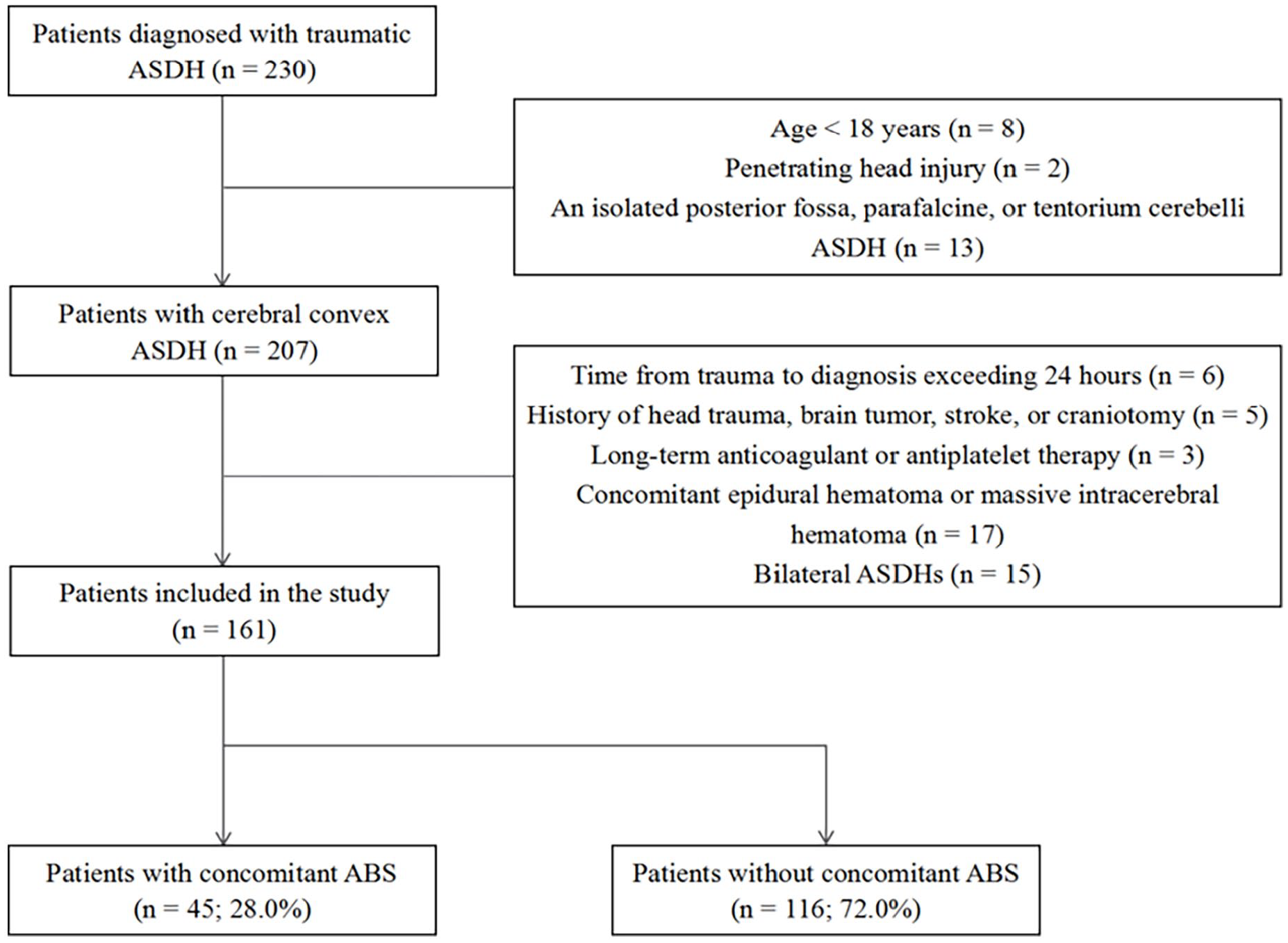
Flowchart of patient screening.
Definitions
All patients were initially admitted to the emergency department after trauma and then underwent an initial CT scan of the head. The diagnosis of post-traumatic ABS was made on the basis of CT findings. In addition to examining the basal cisterns, an additional evaluation of sulcal effacement at the vertex 11 was also conducted using axial sequences of CT with a thickness of 5 mm. If necessary, both axial and coronal sequences of thinner CT were utilized to determine the status of the sulci and cisterns. The CT images were independently reviewed by two experienced neurosurgeons (HL and SW) who were blinded to other clinical data of patients. A diagnosis of hemispheric swelling was established based on any of the following criteria: (1) the midline shift exceeded the maximum thickness of hematoma [Figure 2(a)] 3 ; (2) a thin ASDH (defined as the maximum thickness of hematoma <10 mm), and sulcal effacement at the vertex that occurs only on the ipsilateral hemisphere [Figure 2(b) and (c)]. The diagnostic criteria for whole-brain swelling were as follows: (1) compression or absence of the basal cisterns; (2) sulcal effacement at the vertex that occurs on the bilateral hemispheres [Figure 2(d) and (e)]. The criteria for identifying intraoperative malignant brain bulge was: the brain tissue gradually protruded outside the edge of the bone window shortly after opening of the dura mater, and then it was significantly compressed by the edge of the bone window and unable to be restored even with remedies such as hyperventilation and/or dehydrating agents. 6 A routine CT scan was performed immediately on patients who had largely stable vital signs after surgery. If no new intracranial hematoma was found on the contralateral side, malignant brain bulge was considered to be the primary phenomenon caused by ABS; otherwise, it was deemed secondary.

Three cases of brain swelling associated with ASDH. (a) A 35-year-old female patient with an isolated ASDH (black arrow) on the left presented with hemispheric swelling due to a significant midline shift of 10.5 mm (yellow line) that exceeds the maximum thickness of the hematoma (7.0 mm). (b and c) A 51-year-old male patient with a thin ASDH (black arrow) on the left and concomitant other injuries (including multiple cerebral contusions and lacerations and scattered SAH) presented with hemispheric swelling due to sulcal effacement at the vertex of the ipsilateral hemisphere. Note the insignificant midline shift. (d and e) A 48-year-old male patient with a thin ASDH (black arrow) on the right and concomitant other injuries (including focal contusion and laceration over the right temporal lobe, and diffuse SAH) presented with whole-brain swelling due to the absence of the basal cisterns (yellow arrow) and sulcal effacement at the vertex of the bilateral hemispheres.
Data collection
The inpatient records of enrolled patients were extracted from the electronic medical record system. The proportion of patients who exhibited hemispheric and whole-brain swelling was calculated. Demographic information and clinical data on admission included sex, age, chronic diseases (hypertension or diabetes), mechanism of injury, Glasgow Coma Scale (GCS) score, mean arterial pressure, hypotension (defined as systolic blood pressure <100 mmHg), and symptomatic epilepsy. To evaluate the status of post-traumatic neuroinflammation, venous blood samples on admission were collected for routine tests to determine the neutrophil to lymphocyte ratio (NLR), platelet to white blood cell ratio (PWR), and platelet to lymphocyte ratio (PLR). Radiographic findings, including occipital skull fracture, multiple contusions and lacerations, intraventricular hemorrhage, primary brain-stem injury, and traumatic subarachnoid hemorrhage (SAH), were evaluated on initial CT scan.
Outcome measures
The primary clinical outcome measures included the Glasgow Outcome Scale (GOS) score, mortality, and functional prognosis at discharge. Under the GOS rating system, a score of 1 signifies death, 2 signifies persistent vegetative state, 3 signifies severe disability, 4 signifies moderate disability, and 5 signifies good recovery. 12 In terms of functional prognosis, a GOS score of 1–3 indicates an unfavorable prognosis, while a score of 4–5 indicates a favorable prognosis. The secondary outcome measure was the proportion of surgical patients who experienced intraoperative malignant brain bulge.
Statistical analysis
Statistical analyses were conducted using SPSS version 22.0 for Windows (SPSS Inc., Chicago, IL, USA). Continuous variables were presented as mean ± standard deviation or median (interquartile range, IQR); categorical variables were presented as count (percentage). All patients were divided into ABS group and no ABS group. For group comparisons, the Student’s t test, Mann-Whitney U test, and Pearson’s chi-square or Fisher’s exact test were used for normally distributed continuous variables, non-normally distributed continuous variables, and binary categorical variables, respectively. The possible risk factors for ABS were initially selected through univariate analysis, followed by identification of independent risk factors using binary multivariate logistic regression analysis. A two-tailed p-value <0.05 was considered statistically significant. The multivariate model was described by odds ratio (OR) and its 95% confidence interval (CI).
Results
Incidence of ABS
A total of 230 patients diagnosed with ASDH were reviewed in this study. According to the predefined inclusion and exclusion criteria, 161 patients were ultimately enrolled in the analysis. Of these, 45 patients experienced post-traumatic ABS, resulting in an overall incidence rate of approximately 28.0%. Furthermore, the incidence of whole-brain swelling was higher than that of hemispheric swelling. Specifically, hemispheric swelling was observed in 13 (8.1%) patients and whole-brain swelling in 32 (19.9%) patients (Table 1).
Clinical characteristics of the 161 enrolled patients diagnosed with ASDH.

Upon admission.
ABS, acute brain swelling; ASDH, acute subdural hematoma; DC, decompressive craniectomy; GCS, Glasgow Coma Scale; IQR, interquartile range; SAH, subarachnoid hemorrhage.
Clinical characteristics
Of the 161 patients, 90 (55.9%) were male. The median age (IQR) was 60 years (48–69 years). The median GCS (IQR) was 14 (8–14). Thirty-one (19.3%) patients had chronic diseases. Traffic accident was the most frequent mechanism of injury (73 patients; 45.3%), followed by fall (63 patients; 39.1%), fall from a height (21 patients; 13.0%), and others (four patients; 2.5%). Hypotension was observed in five (3.1%) patients and symptomatic epilepsy in three (1.9%) patients. The presence of occipital skull fracture was found in 21 (13.0%) cases, multiple contusions and lacerations in 60 (37.3%) cases, intraventricular hemorrhage in 8 (5.0%) cases, primary brain-stem injury in 4 (2.5%) cases, and traumatic SAH in 80 (49.7%) cases on initial CT. There were 55 (34.2%) patients who underwent surgical hematoma evacuation after admission, of whom 48 (29.8%) underwent additional DC. The clinical characteristics of the 161 patients are summarized in Table 1. In addition, the ABS group had significantly lower GCS scores on admission compared to the no ABS group (p < 0.001) (Table 2).
Comparison of characteristics between the ABS and no ABS groups.

p Values <0.05 are highlighted in bold.
Upon admission.
ABS, acute brain swelling; GCS, Glasgow Coma Scale; IQR, interquartile range; NLR, neutrophil to lymphocyte ratio; PLR, platelet to lymphocyte ratio; PWR, platelet to white blood cell ratio; SAH, subarachnoid hemorrhage; SD, standard deviation.
Risk factors for post-traumatic ABS
The comparison of different characteristics between the ABS group (n = 45) and the no ABS group (n = 116) is presented in Table 2. There was a significant difference in age between the groups (p < 0.001), with the ABS group having a comparatively smaller age distribution. The ABS group had more frequent traumatic SAH (p < 0.001), along with significantly lower mean arterial pressure (p = 0.026), lower PLR (p = 0.015), and lower PWR (p < 0.001). No significant differences were observed between the groups in terms of sex, mechanism of injury, and NLR, as well as the presence of chronic diseases, hypotension, symptomatic epilepsy, occipital skull fracture, multiple contusions and lacerations, intraventricular hemorrhage, and primary brain-stem injury (all p > 0.05).
A multivariate logistic regression analysis was conducted to determine the variables, including age, mean arterial pressure, PLR, PWR, and traumatic SAH, that could help predict the development of ABS in patients with ASDH (Table 3). The results showed that younger age (OR = 0.917, 95% CI = 0.885–0.950, p < 0.001), lower PWR (OR = 0.887, 95% CI = 0.809–0.974, p = 0.012), and presence of traumatic SAH (OR = 4.346, 95% CI = 1.567–12.054, p = 0.005) were independent risk factors for post-traumatic ABS, while mean arterial pressure and PLR had no significant associations with the development of ABS. The OR associated with age was 0.917, indicating that for every 1-year decrease in age, patients with ASDH have an 8.3% increased risk of developing post-traumatic ABS.
Multivariate logistic regression analysis of risk factors for patients who developed ABS.
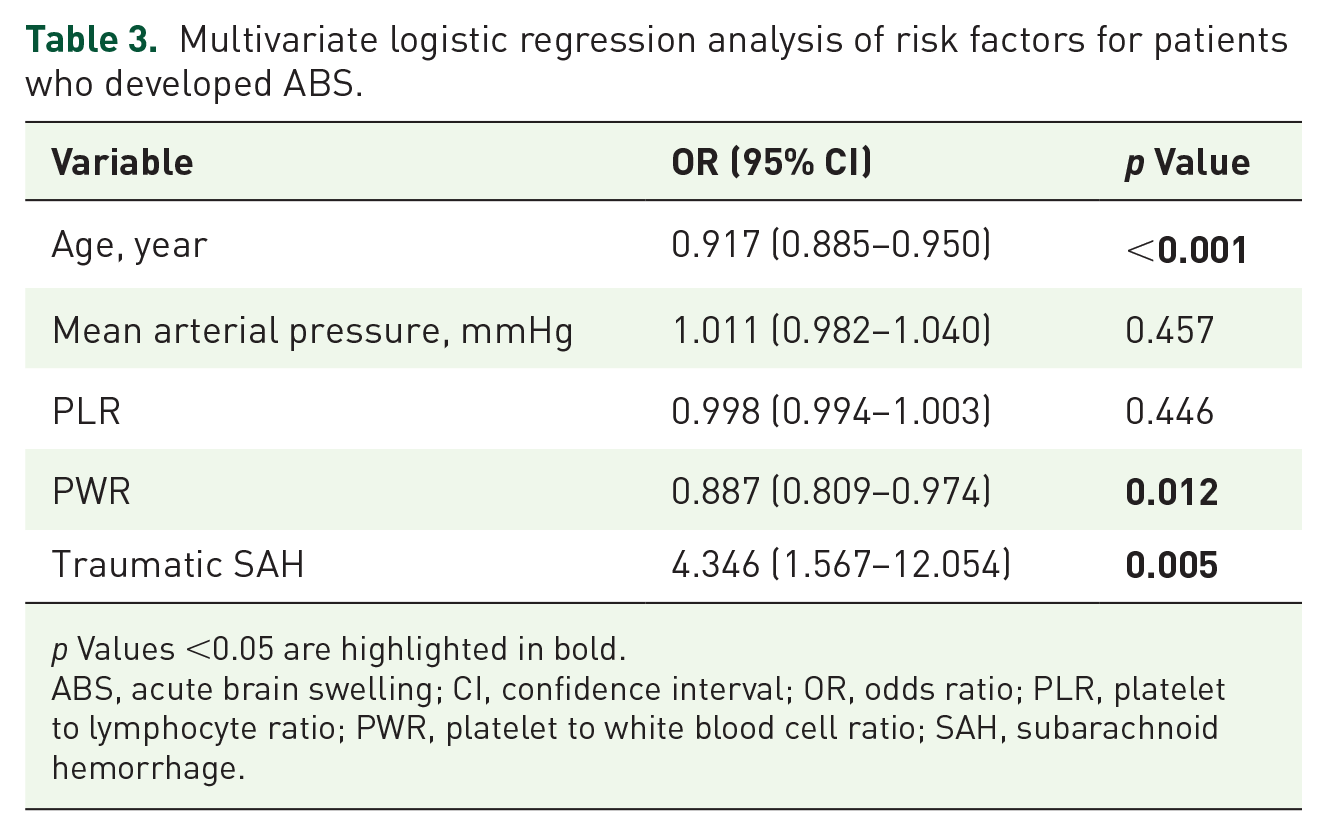
p Values <0.05 are highlighted in bold.
ABS, acute brain swelling; CI, confidence interval; OR, odds ratio; PLR, platelet to lymphocyte ratio; PWR, platelet to white blood cell ratio; SAH, subarachnoid hemorrhage.
Clinical outcomes
Regarding clinical outcomes at discharge, the median GOS score (IQR) for the ABS group was 2 (1–3) versus 4 (3–5) in the no ABS group (p < 0.001). It is worth noting that those dying patients who were voluntarily discharged from the hospital had a GOS score of 1 due to their anticipated imminent demise. In addition, there were significant differences in mortality and functional prognosis between the groups (both p < 0.001), indicating that the ABS group had a poorer functional prognosis and higher mortality compared to the no ABS group (Table 4). Figure 3 shows the proportion of patients in the different GOS score groups between the groups. In the ABS group, 32 (71.1%) patients underwent surgical treatment, of whom 10 (31.3%) experienced primary brain bulge during surgery and 4 (12.5%) experienced secondary intraoperative brain bulge. Twenty-three (19.8%) patients underwent surgical treatment in the no ABS group, and none of them experienced intraoperative brain bulge. Among 55 surgical patients, the proportion of patients who experienced intraoperative malignant brain bulge was significantly different between the groups (43.8% versus 0%; p < 0.001) (Figure 4).
Comparison of discharge outcomes between the ABS and no ABS groups.

p Values <0.05 are highlighted in bold.
ABS, acute brain swelling; GOS, Glasgow Outcome Scale; IQR, interquartile range.

Comparison of the proportion of patients in different prognostic scores between the groups.

Comparison of the proportion of surgical patients with intraoperative malignant brain bulge between the groups.
Discussion
Post-traumatic ABS typically occurs within a few hours after TBI. When it exacerbates during a craniotomy and subsequently causes brain tissue to rapidly bulge out of the bone window, this phenomenon is also known as primary intraoperative malignant brain bulge. This study focused on ABS occurring shortly after trauma in patients with traumatic ASDH. According to the definition employed, approximately 28% of patients experienced post-traumatic ABS. Patients with concomitant ABS had worse clinical outcomes than those without concomitant ABS. Traumatic SAH, age, and PWR were closely associated with the development of post-traumatic ABS.
Currently, there is a lack of consensus on the definition of post-traumatic ABS, particularly regarding whole-brain swelling. In previous studies,1,3,8 whole-brain swelling was diagnosed on the basis of compression or absence of the cerebrospinal fluid space, which mainly involves the basal cisterns, mesencephalic cisterns, and/or third ventricle. Additionally, a diagnosis of hemispheric swelling was made by enlargement of the affected hemisphere in volume.3,13 Although neurosurgeons can easily identify the absence of the cerebrospinal fluid space, determining its compression is more challenging and subjective. To avoid observer variability, Lang et al. 1 defined whole-brain swelling according to the absence of the basal cisterns or third ventricle. Nonetheless, the diagnosis based on the status of the basal cisterns or third ventricle still has undeniable limitations. In clinical practice, for certain patients without concomitant whole-brain swelling, it is possible that the morphological abnormalities of these anatomical structures can be caused by the space-occupying effect of focal injuries adjacent to the skull base. Furthermore, although no significant midline shift is observed in certain patients with a thin or small ASDH, the disappearance of the ipsilateral sulci and cisterns actually indicates the presence of hemispheric swelling. A recent study has demonstrated that sulcal effacement at the vertex is a reliable predictor of elevated ICP following TBI. 11 One of the purposes of this study was to ascertain the incidence of ABS associated with ASDH, encompassing hemispheric and whole-brain swelling. Therefore, we proposed novel CT-based criteria to clearly define this easily overlooked subtype of TBI. The criteria include an additional evaluation of sulcal effacement at the vertex, aiming to enhance the diagnostic accuracy and comprehensiveness of post-traumatic ABS. Our study found that the incidence of hemispheric and whole-brain swelling in patients with ASDH was 8.1% and 19.9%, respectively. The overall incidence is similar to that reported previously. 3
In the past, post-traumatic ABS has been widely believed to be primarily driven by two potential mechanisms: acute cerebrovascular engorgement resulting from vasomotor paralysis and brain edema dominated by cellular edema.14,15 However, these previously proposed hypotheses were insufficient to clearly elucidate the pathophysiological mechanism of ABS associated with ASDH. On the one hand, damage to the vasomotor centers located in the hypothalamus and brainstem should theoretically result in whole-brain swelling rather than just hemispheric swelling. On the other hand, brain edema typically occurs at a later stage after TBI, but this cannot fully explain the rapid onset of ABS. Previous clinical studies have found no correlation between the thickness of ASDH and post-traumatic ABS.3,13 However, the impact of hematoma on the underlying hemisphere cannot be completely disregarded. A recent study in an ASDH model in rats has revealed that cerebral circulation disorders, particularly dysregulated venous circulation caused by compression of the hematoma, may have a significant impact on the development of post-traumatic ABS. 16 It is evident that the pathophysiology of ABS associated with ASDH is intricate and shows an inconsistency between hemispheric and whole-brain swelling. Therefore, further studies, including comprehensive clinical analyses of risk factors, are needed to enhance the understanding of the underlying pathophysiology.
The GCS score can reflect the severity of TBI. The ABS group had lower GCS scores on admission. This finding might be explained by the fact that elevated ICP caused by ASDH with concomitant ABS is more pronounced than that caused solely by a hematoma, thereby resulting in a lower GCS score. Our study found that traumatic SAH was an independent risk factor for post-traumatic ABS. This finding is consistent with some previous reports.3,5 The present results showed that traumatic SAH occurred in 77.8% of patients with concomitant ABS. SAH is characterized by bleeding in the subarachnoid space that can result in the release of neuroinflammatory substances and subsequent cerebral vasospasm. 17 Previous studies have suggested that the mechanisms of vasospasm following traumatic SAH may be similar to those identified in aneurysmal SAH. 18 In non-traumatic SAH, the occurrence of vasospasm typically presents a significant delay and is associated with delayed cerebral ischemia and subsequent elevated ICP. The available data on the time course of vasospasm following traumatic SAH remains limited. Consequently, the precise mechanism by which traumatic SAH affects the development of post-traumatic ABS remains unclear. Age was found to be independently associated with the development of post-traumatic ABS. The present finding suggests that younger patients with TBI have a higher risk of developing post-traumatic ABS. Younger patients have higher brain volume and, therefore, lower cerebrospinal fluid space, which could potentially contribute significantly to the development of ABS. In addition, the compliance of cerebral blood vessels naturally decreases with age, which may reduce the responsiveness to acute cerebral vasodilation or vasospasm. Systemic inflammatory markers such as NLR, PWR, and PLR are new prognostic indicators in neurological disorders.19,20 To our knowledge, this study is the first to explore the impact of these inflammation markers on the development of ABS associated with ASDH. The multivariate analysis revealed that low PWR on admission was an independent risk factor for post-traumatic ABS. It might be explained by the hyperactivity of neuroinflammation after diffuse TBI. PWR can be reduced with a decrease in platelet count or with an increase in white blood cell count. White blood cell levels are elevated in the early stage of TBI, and this elevation is somewhat dependent on the severity of brain injury. 21 It has been found that catecholamine released following diffuse TBI results in leukocytosis.14,22 In addition, the mechanism underlying the reduction of platelets in TBI may be associated with the activation of the coagulation cascade and subsequent platelet consumption. 19
The clinical outcomes of ASDH are not solely attributed to the space-occupying effect of the hematoma itself but also to the additional presence of ABS. Our study revealed that 75.6% of patients with concomitant ABS had an unfavorable functional prognosis upon discharge. Additionally, although most patients in the ABS group underwent surgical treatment, the mortality rate was as high as 46.7%. DC is a life-saving surgical procedure for patients with refractory elevated ICP following TBI who have not responded to pharmacologic therapy. However, the impact of this operation on the prognosis of patients with TBI remains controversial. 23 An additional DC was performed in this study for patients who presented with ABS or dilated pupils prior to surgery, as well as those deemed at high risk of developing severe brain edema after hematoma evacuation. Previous studies have demonstrated that ICP monitoring or combined external ventricular drainage can effectively manage elevated ICP in patients with diffuse TBI, thereby potentially obviating the necessity for DC in certain cases.7,10 In this study, half of the patients who experienced secondary brain bulge survived due to a timely second craniotomy performed on the contralateral side. Several stepwise intracranial decompression techniques, such as pre-craniotomy hole drilling, slow cutting of the dura mater, and gradual decompression via external ventricular drainage or ICP monitoring, have been reported to effectively decrease the occurrence of secondary brain bulge to a certain extent. 12 However, these measures may not sufficiently prevent primary brain bulge in clinical practice. In this study, nearly all surgical patients who experienced primary brain bulge succumbed to refractory elevated ICP shortly after surgery. In a retrospective study, Wu et al. 5 identified several risk factors for intraoperative primary malignant brain bulge in patients with ASDH, including bilaterally dilated pupils, SAH, and a shorter injury-to-surgery interval. The pathophysiology of primary malignant brain bulge remains unclear, and dysregulated venous circulation may be the underlying mechanism that warrants more attention in future studies. 16
This study has the following limitations. First, it was a single-center retrospective study with a small sample size, and a power analysis was not performed for sample size estimation. Second, given that the mechanism of post-traumatic ABS is complex and not entirely clear, a separate analysis of risk factors for hemispheric or whole-brain swelling associated with ASDH was not further conducted. Third, hypoxemia may be an important risk factor for the development of post-traumatic ABS 24 ; however, this variable was not considered in this study due to incomplete arterial blood gas data. Finally, although only a very small proportion of patients had a small concomitant parafalcine ASDH, it remains uncertain whether such hematomas affect the presence of sulcal effacement at the vertex.
Conclusion
The present study’s findings provide neurosurgeons with additional valuable insight into ABS associated with ASDH, thereby serving as a significant supplement to the existing literature. Post-traumatic ABS occurs frequently in patients with ASDH and is significantly associated with worse clinical outcomes compared to those without concomitant ABS. Patients who have young age, low PWR, and traumatic SAH are at greater risk for developing post-traumatic ABS. Therefore, neurosurgeons should pay particular attention to such patients for early detection and prompt management of elevated ICP.
