Abstract
Background:
Several studies have demonstrated that early childhood and adolescent obesity are risk factors for multiple sclerosis (MS) susceptibility. Obesity is thought to share inflammatory components with MS through overproduction of pro-inflammatory adipokines (e.g., leptin) and reduction of anti-inflammatory adipokines (e.g, adiponectin). Recently, drug repurposing (i.e. identifying new indications for existing drugs) has garnered significant attention. The US Food and Drug Administration Adverse Event Reporting System (FAERS) database serves not only as a resource for mining adverse drug reactions and safety signals but also for identifying inverse associations and potential medication repurposing opportunities.
Objective:
We aimed to explore the association between weight-loss-inducing drugs and MS using real-world reports from the FAERS database.
Design:
Secondary analysis of existing data from the FAERS database.
Methods:
We conducted a disproportionality analysis using the FAERS database between the fourth quarter of 2003 and the second quarter of 2023 to explore associations between MS and weight loss-inducing drugs. Disproportionality was quantified using the reporting odds ratio (ROR). An inverse association was defined when the upper limit of the 95% confidence interval for ROR was <1.
Results:
We found an inverse association between MS and anti-diabetic weight loss-inducing drugs including semaglutide (ROR: 0.238; 95% CI: 0.132–0.429), dulaglutide (ROR: 0.165; 95% CI: 0.109–0.248), liraglutide (ROR: 0.161; 95% CI: 0.091–0.284), empagliflozin (ROR: 0.234; 95% CI: 0.146–0.377), and metformin (ROR: 0.387; 95% CI: 0.340–0.440). No inverse associations were found for other weight loss-inducing drugs such as phentermine, bupropion, topiramate, zonisamide, and amphetamine. An exception was naltrexone (ROR: 0.556; 95% CI: 0.384–0.806).
Conclusion:
Our findings suggest a potential consideration for repurposing anti-diabetic weight loss-inducing drugs including semaglutide, dulaglutide, and liraglutide (glucagon-like peptide-1 receptor agonists), empagliflozin (sodium-glucose cotransporter-2 inhibitor), and metformin (biguanide), for MS. This warrants validation through rigorous methodologies and prospective studies.
Keywords
Introduction
The relationship between multiple sclerosis (MS) and obesity is multi-faceted. Obesity is a driver of systemic and local inflammation. Several studies have demonstrated that early childhood or adolescent obesity increases the risk of MS. Obesity is also a disease modifier in MS. Newly diagnosed patients with MS who are obese tend to experience more severe disease and poorer outcomes. 1 Moreover, obesity has been reported to be associated with a less favorable response to disease-modifying therapies. 2
Recently, drug repurposing, defined as researching new indications for already approved drugs, is gaining attention as a rapid and cost-efficient strategy for developing new treatments. Pharmacovigilance data which are primarily used to identify adverse drug reactions by screening for disproportionate reporting can also serve as a data mining source for identifying inverse associations and potential medication repurposing opportunities. In this study, we aimed to explore the association between weight loss-inducing drugs and MS using reports from the US Food and Drug Administration Adverse Event Reporting System (FAERS).
Methods
FAERS is a publicly available database containing information on spontaneous reporting of adverse events (AEs) and medication errors. We performed a disproportionality analysis of FAERS between the fourth quarter of 2003 and the second quarter of 2023 using the OpenVigil2.1-MedDRA-v24 software3,4 to identify associations between MS and weight loss-inducing drugs. Disproportionality analysis is a validated method for detecting significant associations between drugs and AEs. We investigated both anti-diabetic medications with weight loss-inducing effects, including semaglutide, dulaglutide, liraglutide, tirzepatide, empagliflozin, canagliflozin, dapagliflozin, and metformin, as well as other weight loss-inducing drugs, including orlistat, phentermine, bupropion, topiramate, zonisamide, amphetamine, and naltrexone. To include reports where these medications were listed as concomitant medications, rather than exclusively as primary or secondary suspects for the AE of interest, no filter was applied based on the role of the drug. If a drug was associated with ⩽5 drug-event pair (i.e. drug-MS) combinations, it was excluded from the analysis. Disproportionality was quantified using the reporting odds ratio (ROR) and its 95% confidence interval (CI). ROR is the odds of a certain event (in this case, ‘multiple sclerosis’) occurring with the drug of interest, compared to the odds of the same event occurring with all other drugs in the database. An inverse association was defined when the upper limit of the 95% CI for ROR was <1.
Results
Table 1 shows the number of reports for MS as an event versus reports of other events for each weight loss-inducing drug. The following medications were not included in the analysis due to an inadequate number of reports (i.e. ⩽5): tirzepatide, orlistat, canagliflozin, and dapagliflozin. The results of the disproportionality analysis are presented in Figure 1. Inverse associations were found between MS and the following anti-diabetic weight loss-inducing drugs: semaglutide (ROR: 0.238; 95% CI: 0.132–0.429), dulaglutide (ROR: 0.165; 95% CI: 0.109–0.248), liraglutide (ROR: 0.161; 95% CI: 0.091–0.284), empagliflozin (ROR: 0.234; 95% CI: 0.146–0.377), and metformin (ROR: 0.387; 95% CI: 0.340–0.440). Except for naltrexone (ROR: 0.556; 95% CI: 0.384–0.806), no inverse associations were found for other weight loss-inducing drugs such as phentermine (ROR: 0.856; 95% CI: 0.46–1.593), bupropion (ROR: 1.198; 95% CI: 1.052–1.365), topiramate (ROR: 1.422; 95% CI: 1.218–1.659), zonisamide (ROR: 1.054; 95% CI: 0.679–1.634), and amphetamine (ROR: 1.494; 95% CI: 1.224–1.825).
Number of reports of MS as an adverse event versus reports of adverse events other than MS for each drug of interest with weight loss-inducing effect based on the data from the FDA Adverse Event Reporting System database.

GIP, glucose-dependent insulinotropic polypeptide; GLP-1, glucagon-like peptide-1; MS, multiple sclerosis; SGLT-2, sodium-glucose cotransporter-2.
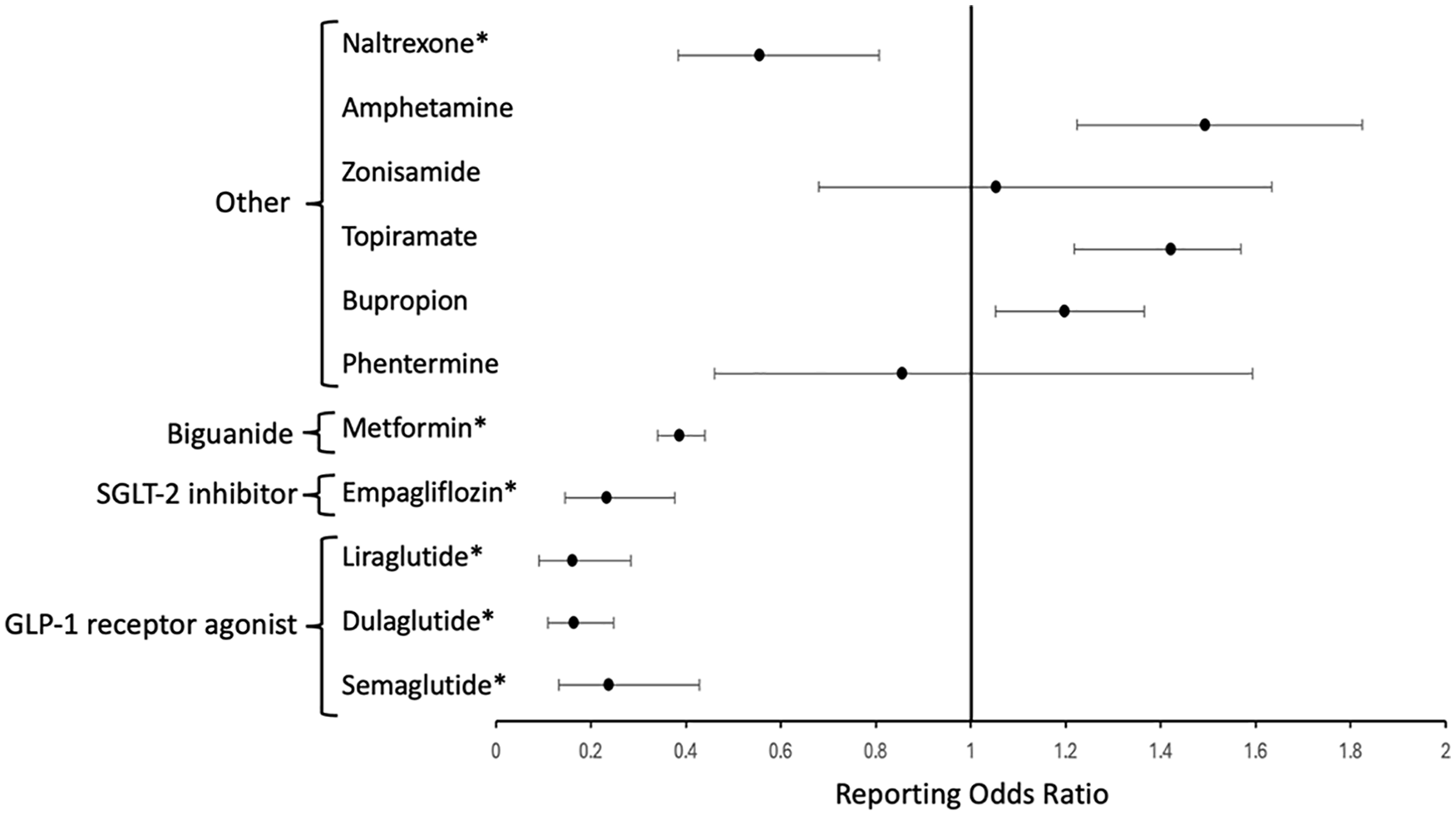
Disproportionality analysis illustrating the association between multiple sclerosis and weight loss-inducing drugs based on the data from the FDA Adverse Event Reporting System database. The reporting odds ratio represents the odds of a certain adverse event (in this case, ‘multiple sclerosis’) occurring with the drug of interest, compared to the odds of the same adverse event occurring with all other drugs in the database. An asterisk (*) denotes the presence of an inverse association, defined when the upper limit of the 95% CI for reporting odds ratio is less than 1.
Discussion
We found an inverse association between MS and anti-diabetic weight loss-inducing drugs including semaglutide, dulaglutide, and liraglutide [glucagon-like peptide-1 (GLP-1) receptor agonists], empagliflozin [sodium-glucose cotransporter-2 (SGLT-2) inhibitor], and metformin (biguanide). These findings suggest a potential for repurposing these medications for MS.
The precise biological mechanisms linking MS and obesity remain elusive. Obesity is believed to share inflammatory components with MS. Excess macronutrients in adipose tissues trigger the release of inflammatory mediators, including leptin, tumor necrosis factor-α, and interleukin-6, while decreasing the production of adiponectin (an anti-inflammatory adipokine), predisposing to a pro-inflammatory state and oxidative stress. 5 Obesity is thus conceptualized as a state of low-grade chronic inflammation.
While initially designed as insulinotropic and anti-hyperglycemic agents for type 2 diabetes treatment, GLP-1 receptor agonists are now receiving growing attention for their neurotrophic and neuroprotective properties. Of note, brain pericytes, a critical cellular component of the blood–brain barrier (BBB), express the GLP-1 receptor. Activation of GLP-1 receptor is thought to improve brain pericyte function resulting in restoration of vascular integrity and BBB functions. 6 The potential efficacy of GLP-1 receptor agonists specifically dulaglutide and liraglutide toward MS has been shown in studies of experimental autoimmune encephalomyelitis (EAE). Dulaglutide caused a reduction in clinical manifestations and histopathological outcomes, and the incidence of encephalitogenic Th1/Th17 cells in EAE mice. 7 Liraglutide treatment delayed the onset, reduced the severity of EAE, and increased mitochondrial manganese superoxide dismutase in the brain of EAE rats, suggesting the capacity of GLP-1 receptor agonists against oxidative stress. 8 In addition, liraglutide improved the remyelination process by stimulating oligodendrocyte progenitor cell differentiation in a cuprizone-induced mouse model. 9 While recognizing the speculative nature, these neuroprotective properties of GLP-1 receptor agonists may provide a potential perspective for interpreting our study’s findings.
SGLT-2 inhibitors, another novel class of anti-diabetic drugs, although not yet specifically studied in EAE or MS, have also been shown to have anti-inflammatory effects by reducing reactive oxygen species, lessening BBB leakage, and decreasing microglia burden. SGLT-2 is expressed in pericytes, and the inhibition of SGLT-2 has been shown to ameliorate ischemic brain injury in mice through pericyte protection and to increase the level of brain-derived neurotrophic factor. 10
Metformin, a common oral treatment for type 2 diabetes, attenuates EAE by modulating key immunopathological mechanisms, including Th17 and T-reg cell balance. Metformin has also been demonstrated to enhance the regenerative capacity of aged oligodendrocyte progenitor cells, improving remyelination in aged rodents after focal demyelination. 11
The only non-anti-diabetic medication in our study that was found to have an inverse association with MS was naltrexone. Endogenous stimulation of opioid growth factor by low-dose naltrexone has been demonstrated to reverse the course of progressive EAE and limit relapse frequency in relapsing-remitting EAE. 12 Nevertheless, rigorous clinical studies of naltrexone for MS are limited and yield mixed findings.
Recognizing FAERS database limitations is important. It does not provide definitive proof of a causal (or inverse) relationship between product exposure and reported events. The absence of a control group makes it difficult to differentiate between associations and coincidences. FAERS relies on voluntary reporting, likely missing some AEs. Adjustment for clinical characteristics and reliable evaluation of synergistic effects are not possible due to the nature of the database. Moreover, the absence of a known denominator (the number of prescriptions for the product) prevents incidence calculation using FAERS data.
Overall, this study hints at the possibility of considering anti-diabetic drugs with weight loss-inducing effects, especially GLP-1 receptor agonists, for potential repurposing opportunities in MS. These findings should be validated through complementary methodologies and prospective studies.
