Abstract
Multiple sclerosis (MS) was defined as a rare disease in China due to its low prevalence. For a long time, interferon β was the only approved disease-modifying therapy (DMT). Since the first oral DMT was approved in 2018, DMT approval accelerated, and seven DMTs were approved within 5 years. With an increasing number of DMTs being prescribed in clinical practice, it is necessary to discuss the standardized MS treatment algorithms depending on the disease activity and DMT availability. In this review paper, more than 20 Chinese experts in MS have reviewed the therapeutic progress of MS in China and worldwide and discussed algorithms for treating relapsing MS (RMS) based on the available DMTs in China, providing insights for establishing the standardized RMS treatment algorithms in this country.
Plain language summary
In this review paper, more than 20 Chinese experts in MS have reviewed the therapeutic progress of MS in China and worldwide and discussed algorithms for treating relapsing MS (RMS) based on the available DMTs in China, providing insights for establishing the standardized RMS treatment algorithms in this country: 1) CIS and RRMS account for more than 90% of the MS patients and most of them are mild to moderate; 2) MS patients should initiate DMT treatments as soon as the disease has been diagnosed in order to reduce the risk of disease progression; 3) Patients who have been diagnosed with MS should start treatment with fundamental DMTs unless the disease course has been highly active; 4) MAGNIMS score may be a suitable and simplified assessment tool for measuring treatment response to DMTs; 5) Patients treated with corticosteroids and NSIS should be switched to the standardized DMT treatment during remission in accordance with disease activity.
Infographic
Introduction
In 2018, multiple sclerosis (MS) was included on the first list of rare diseases in China, which was published by the National Health Council of China. Since then, the approval of disease-modifying therapies (DMTs) and inclusion of the national reimbursement drug list (NRDL) have been accelerated. 1 Before 2018, interferon β (IFN-β) was the only DMT in China, but it is now rarely used in the country. Within 5 years, seven DMTs [glatiramer acetate (GA), teriflunomide, dimethyl fumarate (DMF), fingolimod, siponimod, ozanimod, and ofatumumab] and one symptom management drug (fampridine) were approved by China National Medical Products Administration (NMPA) and marketed in China, as of 31 December 2023. All of these MS drugs, with the exception of GA, were included in the NRDL, 2 which has greatly improved the prognosis and treatment affordability for patients with MS.
Several categories of DMT have been in clinical use worldwide: IFN-βs, teriflunomide, fumarates, sphingosine-1-phosphate receptor modulators (S1PRMs), anti-CD20 monoclonal antibodies (anti-CD20s), alemtuzumab (anti-CD52 monoclonal antibody), cladribine, and natalizumab (α4-integrin antagonist).3,4 As a new class of DMTs, Bruton tyrosine kinase inhibitors (BTKi) are under development. 5 Nowadays, the DMTs used in China have covered most of the different efficacy classes compared with those used in Europe and the US, despite including only eight DMTs. Since these DMTs were approved through an accelerated process, almost all of them, with the exception of teriflunomide, were approved with clinical trial waivers. 6 With regard to clinical decision-making, it is necessary to discuss to treat MS patients with the appropriate DMTs.
The aims of this review paper are as follows: (1) to summarize the demographic characteristics of MS patients in China; (2) to describe the efficacy of DMTs available in China; and (3) to explore the treatment algorithms of MS based on the DMTs available in China. This manuscript was developed by a group of Chinese experts on MS and have reviewed the literature about the progress of MS treatment both domestically and internationally.
Characteristics of MS patients in China
Demographics
The female:male ratio of MS patients in China is approximately 2:1,7–14 which is consistent with the global sex ratio, according to the Atlas of MS and MSBase.15,16 The standardized diagnosis and treatment of MS in China was initiated later than the developed countries. 17 The age at MS diagnosis varies among the different Chinese regions and the different hierarchical levels of hospitals, as there is health inequity. According to a nationwide hospital-based study, the mean age at MS diagnosis was found to be 45.3 years old. 7 However, in leading neurological centers where there are MS specialists and professional multidisciplinary teams, the mean age at diagnosis is ~30 years old,8,18,19 which is consistent with data from the Atlas of MS. 15 Therefore, the early diagnosis should be promoted to ensure the early initiation of treatment.
Based on the disease activity and severity of MS in China, mild to moderate MS is more common. More than 90% of patients with MS in China were diagnosed with clinically isolated syndrome (CIS) and relapsing-remitting MS (RRMS),8–12,18,20 which is a higher prevalence than in the global data. 21 In a multicenter retrospective study, 84.2% of MS patients experienced no or one relapse within 1 year, and patients with Expanded Disability Status Scale (EDSS) ⩽3.0 accounted for 59.1% of MS patients. 12 In a cross-sectional survey conducted in different regions of China, 84.0% of the patients had experienced ⩽1 relapse in the previous year. 13 Moreover, in a prospective, single-center study, EDSS <4.0 accounted for 92.2% of MS patients, 51.2% of the patients completely recovered, and 37.8% partially recovered from the first relapse. 19 Furthermore, in the data from the MSNMOBase registry, 82.5% of patients had ⩽1 relapse in the first 2 years after disease onset. 20
DMT usage
There are variations in DMT usage among the different regions of China and among the different hierarchical levels of hospitals. The rate of DMT usage was reported to be more than70% in the leading neurological centers,8,22 which was higher than the global results (56%), and the findings in Europe (52%) and the Americas (50%), according to the Atlas of MS. 23 However, in some nationwide, multicenter studies, the rate of DMT usage was approximately 30–40%,9,10,13 which was higher before oral DMT was approved (10%) in China, 14 but still lower than the rates in Australia (64%) and Germany (57%).24,25 Improving the overall DMT usage in China is key to improve the prognosis of MS patients.
Initiation of RMS treatment in China
MS is a chronic inflammatory and immune-mediated demyelinating disease of the central nervous system (CNS). 26 Several hypotheses have been proposed regarding its pathogenesis, including the involvement of Epstein–Barr virus infection, low vitamin D, high latitude, risk genes, smoking, and obesity. 27 However, the exact triggers of MS remain unclear, and no treatments have been able to reverse disease progression. The therapeutic strategy involves prevention of relapses and delaying disability progression, and the treatment drugs were named DMT. 28 The major mechanisms of action (MoA) of DMTs involve anti-inflammatory and neuroprotective effects through various signaling pathways (Figure 1). 29

Pathology of multiple sclerosis and mechanism of available disease-modifying therapies in China. IFN-β could affect multiple levels of cellular functions to increase the levels of anti-inflammatory agents and reduce the levels of pro-inflammatory cytokines. 30 GA is a noninterferon immunomodulator, which has a similar effect as that of IFN-β on multiple levels of cellular functions. 31 Teriflunomide is a selective, non-competitive, reversible inhibitor of dihydro-orotate dehydrogenase (DHODH), which prevents the proliferation of T- and B-lymphocytes in the periphery. 32 DMF is defined as the Nrf2 activator, 33 which could further activate Keap1/Nrf2/ARE signaling pathway to extert its anti-inflammatory and neuroprotective effects. 34 S1PRMs have a dual action on S1PRs as the pharmacological antagonists or agonists to prevent the lymphocyte exit into circulation from secondary lymphoid organs. 35 Anti-CD20s could directly target CD20+ B cells to deplete B cells. 36
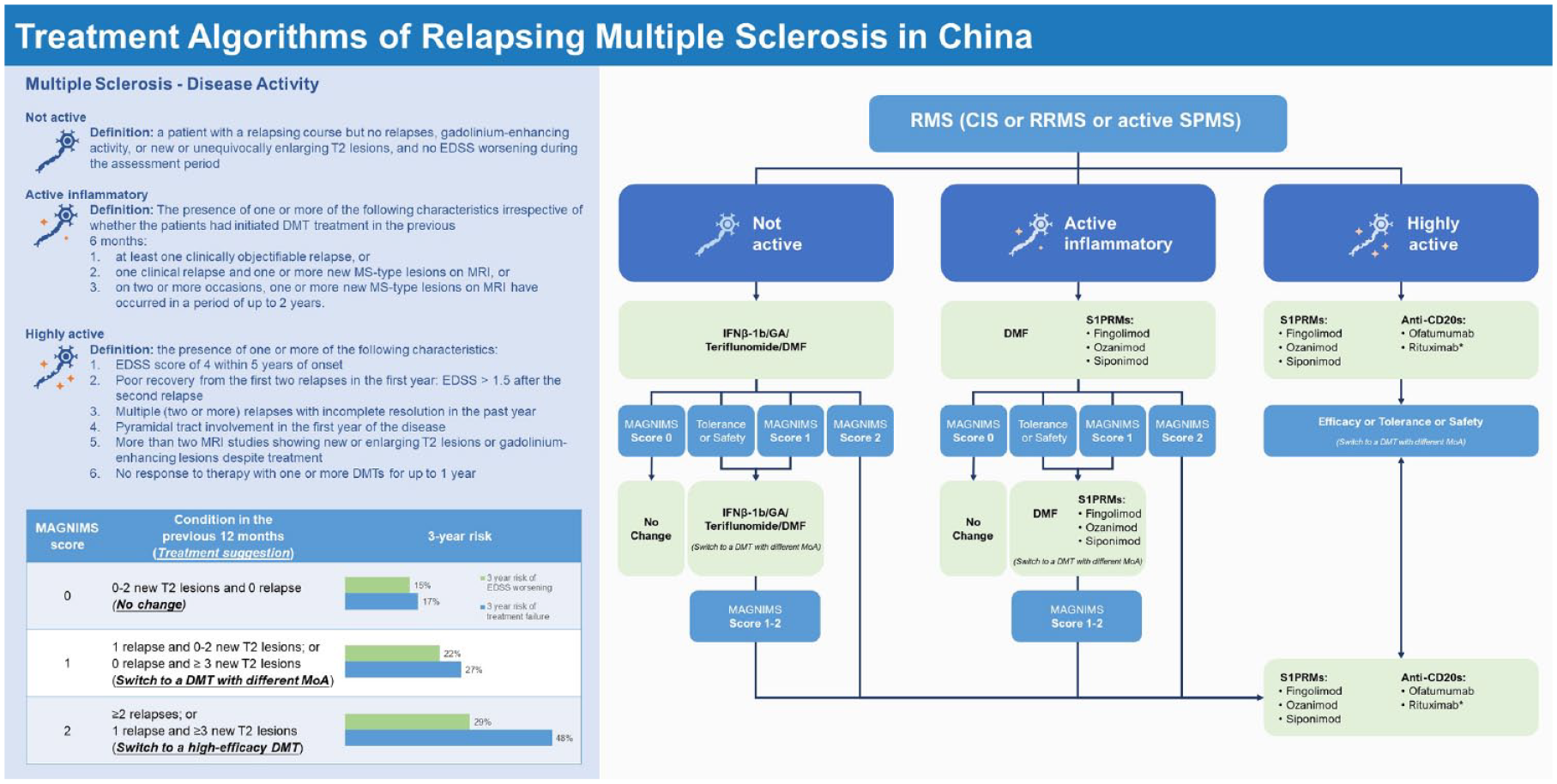
Before 2018, low-dose steroids, nonspecific immunosuppressant (NSIS), and rituximab were used to treat MS in China during the remitting phase. 37 It was impractical to treat MS patients according to disease activity and prognostic factors or to develop personalized treatment strategies. After teriflunomide was approved at the end of 2018, oral DMTs were gradually promoted into clinical practice, and an increasing number of MS patients initiated DMT treatment with more and more approved DMTs. 37 To date, eight DMTs have been approved by NMPA (Table 1), 1 and they are all indicated for the treatment of RMS, including CIS, relapsing-remitting multiple sclerosis (RRMS), and active secondary-progressive MS (SPMS), in adults. Fingolimod was also approved to treat pediatric patients (⩾10 years old) with MS. With the different classes of DMTs and the status of NRDL inclusion, the treatment strategy may differ from that in other countries. Against this background, it is now time to explore the treatment algorithm to ensure that neurologists treat MS patients appropriately in China (Figure 2), 38 especially physicians who have limited experience in MS diagnosis and treatment.
Pivotal data of disease-modifying therapies approved in China.
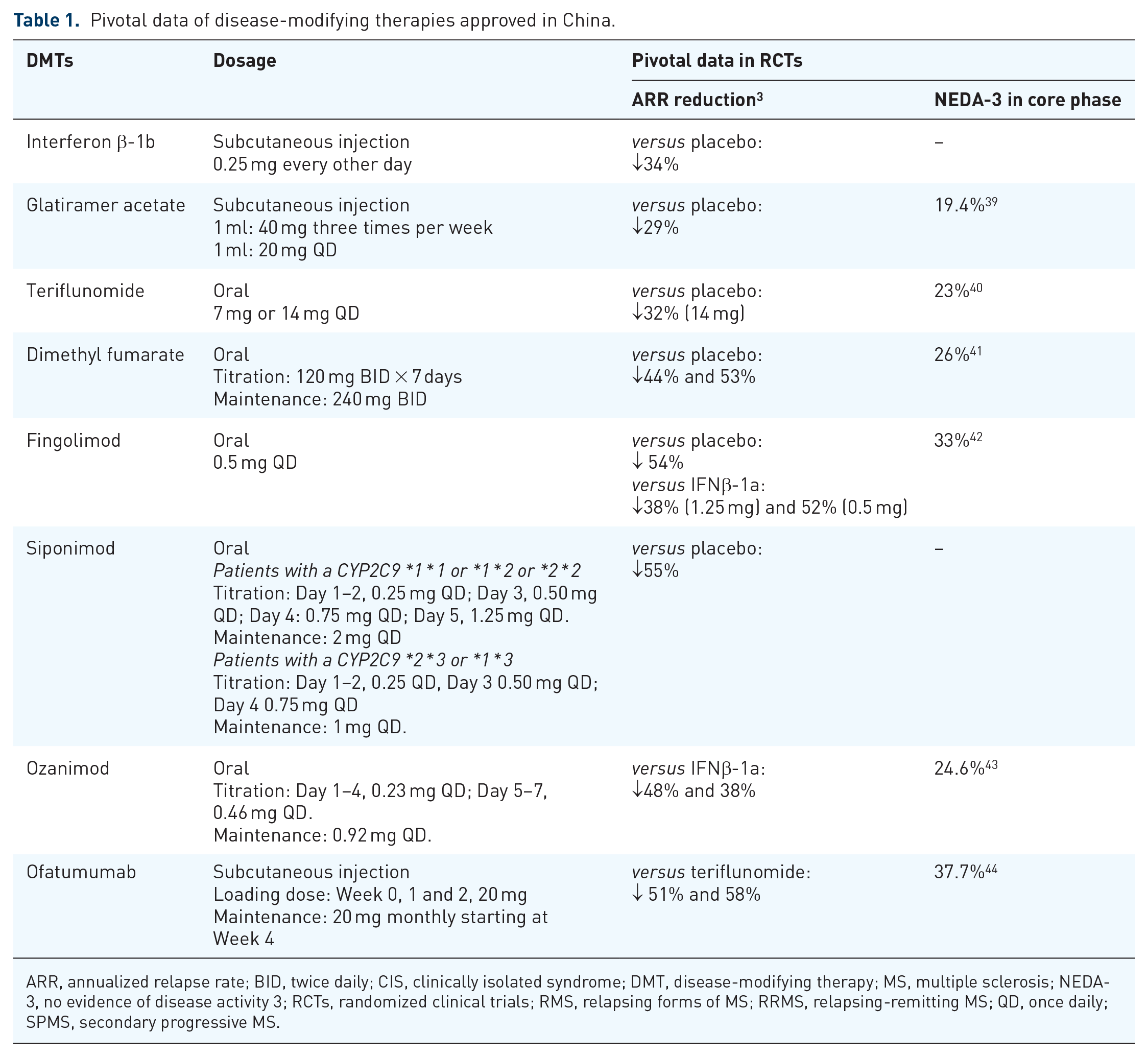
ARR, annualized relapse rate; BID, twice daily; CIS, clinically isolated syndrome; DMT, disease-modifying therapy; MS, multiple sclerosis; NEDA-3, no evidence of disease activity 3; RCTs, randomized clinical trials; RMS, relapsing forms of MS; RRMS, relapsing-remitting MS; QD, once daily; SPMS, secondary progressive MS.

Treatment algorithms of relapsing–remitting MS based on the available disease-modifying therapy in China. MS patients should start treatment with fundamental DMTs unless the disease course becomes highly active. MS patients with highly active MS must be treated with high-efficacy therapies. DMF and S1PRMs are recommended to treat patients with MS with an active inflammatory MS. If patients cannot tolerate current DMTs or have some safety concerns, a switch to a DMT with a different mechanism of action but similar efficacy is highly recommended to maximize the clinical benefits and avoid overtreatment. When the patients show a suboptimal response to the current treatment, it should be considered to switch to another DMT with a different MoA or with different efficacy profile depending on the disease activity, which could be assessed using MAGNIMS score: No change of DMT in patients with Score 0; Switching to a DMT with a different MoA and with similar efficacy in patients with Score 1; and switching to a DMT with higher efficacy in patients with Score 2.
Disease activity
In 2013, the US National Multiple Sclerosis Society (NMSS) further modified the MS subtype as ‘not active’ and ‘active’ on the basis of RRMS, SPMS, and primary progressive MS (PPMS) on the basis of 1996 version.45,46 In clinical practice, MS was often categorized as mild to moderate and (highly) active based on relapse frequency, new magnetic resonance imaging (MRI) findings (mainly T2), EDSS score, and other clinical features.47–49 There was no clear definition of mild to moderate cases, and the definition of (highly) active disease varied. In 2021, German MS guidelines further categorized MS into mild, active inflammatory, and highly active. 50 Recently, the Chinese MS guideline (2023 version) was published and clearly defined the highly active disease. 51 In this review paper, to help Chinese physicians better stratify their MS patients, MS is categorized into not active, active inflammatory, and highly active (Figure 2) by using the combination of the NMSS classification, the 2021 German MS guidelines and Chinese MS guideline (2023 version). Patients with mild to moderate disease are divided into those with ‘not active’ and ‘active inflammatory’ MS. Some minor modifications of the definitions are also made based on the clinical situations of MS in China.
Highly active MS is defined as the presence of one or more of the following characteristics51,52:
(1) EDSS score of 4 within 5 years of MS onset
(2) Poor recovery from the first two relapses in the first year, such as EDSS >1.5 after the second relapse 53
(3) Multiple (two or more) relapses with incomplete recovery in the past year
(4) Pyramidal tract involvement (EDSS functional pyramidal score ⩾2) in the first year of the disease 6
(5) More than two MRI studies showing new or enlarging T2 lesions or gadolinium-enhancing lesions despite treatment
(6) No response to therapy with one or more DMTs for up to 1 year
Active inflammatory MS is defined as the presence of one or more of the following characteristics, irrespective of whether the patients had initiated DMT treatment in the previous 6 months 50 :
(1) at least one clinically objectifiable relapse
(2) one clinical relapse and one or more new MS-type lesions on MRI
(3) on two or more occasions, one or more new MS-type lesions on MRI have occurred in a period of up to 2 years.
‘Not active’ MS is defined as a patient with a relapsing course but no relapses, gadolinium-enhancing activity, or new or unequivocally enlarging T2 lesions, and no EDSS worsening during the assessment period. 46
Early initiation
The overall DMT usage is still much lower than in the leading neurological centers in China. MS patients should initiate DMT treatments as soon as the disease has been diagnosed in order to reduce the risk of disease progression.49,50,54–56 In the BENEFIT trial, initiation of IFNβ-1b treatment in patients with CIS greatly delayed the time to clinically definite MS and ‘McDonald MS’, with reductions of risk by 50% and 44%, respectively. 57 Additionally, similar results were observed upon treatment with IFNβ-1a (in the CHAMPS, ETOMS, and REFLEX trials) and with teriflunomide (in the TOPIC trial).58–61 An international observational cohort study has also shown that the early initiation of DMT treatment significantly reduced the risk of RRMS converting to SPMS.62,63 Adequate evidence supporting early treatment of MS has thus been accumulated.
Treatment options
There have been debates about whether to treat RRMS patients with early high-efficacy therapies (HET) or with the traditional escalation strategy.64–67 Two large clinical trials, DELIVER-MS (NCT03535298) and TREAT-MS (NCT03500328) that are evaluating the long-term outcomes of early HET versus the escalation approach are currently ongoing. At present, an escalation strategy (Figure 2) is still recommended in many countries.49,50,54,68 Patients who have been diagnosed with MS should start treatment with fundamental DMT unless the disease course is highly active, as recommended in many countries.49,50,54,68 The fundamental DMTs are also named platform DMTs,54,69 which refer to IFNβ-1b, GA, teriflunomide, and DMF. MS patients with highly active MS must be treated with HETs,47,49,54,70 which refer to anti-CD20s (ofatumumab and rituximab) and S1PRMs (fingolimod, siponimod, and ozanimod) in China. Rituximab has not been indicated to treat patients with MS in China, although a phase III randomized clinical trial (RCT) (RIFUND-MS) proved its efficacy in CIS and RRMS patients. 71 Moreover, in an observational cohort study, rituximab was noninferior to ocrelizumab in terms of disability-related outcomes, but rituximab treatment demonstrated a high risk of relapse. 72 Furthermore, a retrospective case series (n = 9) revealed that low-dose rituximab (100 mg every 6 months) showed cost-effective results and a good safety profile in Chinese patients with RRMS. 73
DMF and S1PRMs can be prescribed for treatment of patients with active inflammatory MS. There were no head-to-head clinical trials comparing the efficacy of teriflunomide, DMF, and S1PRMs. However, real-world studies have provided sufficient evidence for comparison. A nationwide cohort study from Denmark provided Class II evidence that DMF was more effective and had a lower incidence of discontinuation owing to disease breakthrough than teriflunomide in patients with RRMS. 74 Additionally, similar results were also reported in other real-world studies and meta-analysis. Nevertheless, several other real-world studies demonstrated that the efficacy of teriflunomide was comparable to that of DMF. Moreover, the comparison of DMF and fingolimod was controversial because some studies reported that the efficacy of DMF was comparable to that of fingolimod, whereas only two studies reported that fingolimod was superior to DMF as a second-line treatment. Studies comparing the three DMTs have shown that DMFs exhibited superior efficacy to that of teriflunomide and comparable to that of fingolimod.75,76 Therefore, DMF and S1PRMs are suitable for treating patients with active inflammatory MS, especially when taking the treatment cost into consideration.
Treatment switching
Treatment response
No treatment can be guaranteed to cure MS. The original goal of treating MS was to delay the disability progression, 55 which is usually evaluated using the EDSS. 77 The development of Rio score and modified Rio score aimed to evaluate the treatment response to DMTs (Table 2).78,79 Rio score was developed with the initial aim of measuring the response to IFN-β treatment (platform treatment), which was then modified in 2013.78,79 It was found that the modified Rio scores 0, 1, and 2–3 could predict the 3-year probability of disease progression at the rates of 24%, 33%, and 65% (p < 0.001), respectively. 79 It was also proven that the modified Rio score could predict the response to fingolimod and natalizumab treatment in patients with highly active RRMS. 80
Treatment response evaluation using MAGNIMS Score and modified Rio Score.
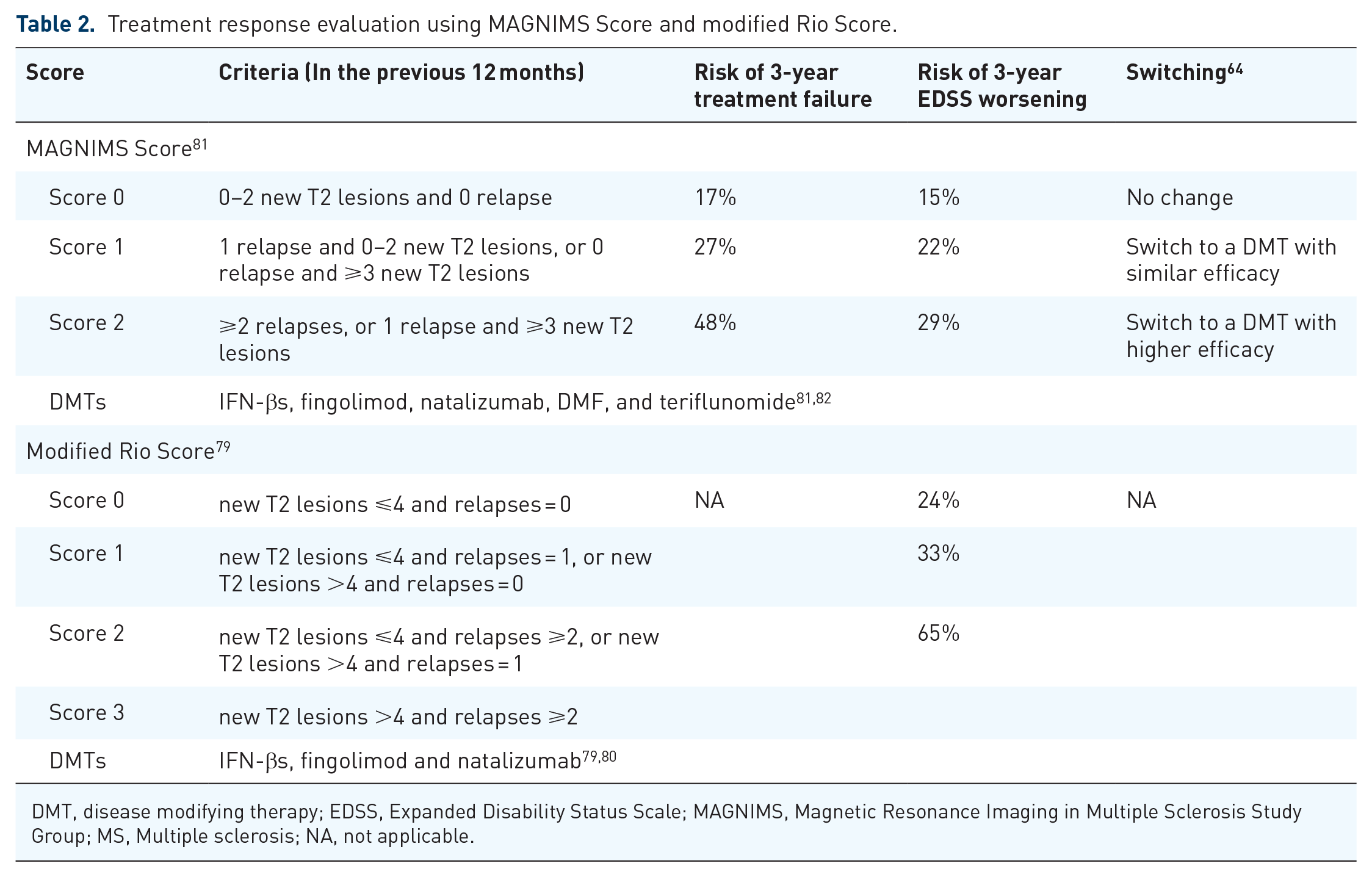
DMT, disease modifying therapy; EDSS, Expanded Disability Status Scale; MAGNIMS, Magnetic Resonance Imaging in Multiple Sclerosis Study Group; MS, Multiple sclerosis; NA, not applicable.
No evidence of disease activity (NEDA) has become the favored treatment goal with the increasing number of DMTs approved in recent years.83,84 In the CLIMB study, NEDA-3 (no relapses, no sustained progression, and no MRI activity) at 2 years positively predicted 78.3% of patients with a progression-free status (EDSS score change ⩽ 0.5) at 7 years. 85 In addition, in the EPIC study, NEDA-3 in the first 2 years was not associated with time to EDSS 6 or time to SPMS after a 10-year follow-up. 86 Meanwhile, it has been very difficult to achieve a good NEDA-3 during RCTs (Table 1), although a high NEDA-3 proportion per year was observed in a real-world setting.19,41,87 Thus, there is a need to further validate NEDA-3 as a long-term predictor of prognosis.
In 2016, the Magnetic Resonance Imaging in MS (MAGNIMS) score was proposed to assess the response to IFN-β after 1 year of treatment (Table 2). 81 Scores of 0, 1, and 2 could predict the 3-year risk of treatment failure at the rates of 17%, 27%, and 48% (p < 0.001), respectively. 81 Scores of 0, 1, and 2 could also predict the 3-year risk of EDSS worsening at the rates of 15%, 22%, and 29% (p < 0.001), respectively. 81 This scoring system could predict IFN-β treatment failure and EDSS worsening for up to 15 years. 88 In addition, MAGNIMS score could also predict the response to treatment with other DMTs, such as fingolimod, natalizumab, DMF, and teriflunomide. 81 This scoring system has been cited as a tool for evaluating treatment switching if a suboptimal response is observed.49,54,64 Therefore, MAGNIMS score may be a suitable and simplified assessment tool for measuring treatment response to DMTs in China. Notably, ⩾2 spinal cord/brainstem lesions indicated a high risk of disease progression, which should be carefully monitored, as mentioned in the 2023 Chinese MS guideline. 51
MS standardized assessment
The regular assessment of MS in China has been published by the Pan-Yangtze River Delta Collaborative Group for Diagnosis and Treatment in Multiple Sclerosis. 17 A comprehensive assessment should be performed, including MRI, neurological functions, such as EDSS, Timed 25-Foot Walk, 9-Hole Peg Test, Symbol Digit Modalities Test, and ophthalmic assessment, including vision, field of vision, optical coherence tomography, and visual-evoked potential. The standardized, comprehensive assessment should be performed at diagnosis and before treatment initiation. Re-baseline and re-assessment should be done every 3–6 months after treatment initiation, and regular assessment should be conducted at least once a year.
DMT switching
None of the available DMTs could completely prevent clinical relapses, MRI activity, and disability progression. When patients show a suboptimal response to the current DMT, switching to another DMT with a different MoA or different efficacy profile should be considered depending on the disease activity, which could be assessed using MAGNIMS score (Table 2).47,64,89 If patients cannot tolerate the current DMT or have some safety concerns, a switch to a DMT with a different MoA but similar efficacy is highly recommended to maximize the clinical benefits and avoid overtreatment (Figure 2).
The off-label use of DMTs is a global medical issue, existing in at least 89 countries because of the availability and affordability of on-label DMTs. 90 As mentioned above, corticosteroids, NSIS, were mainly prescribed to patients with MS who received treatment during remission before the approval of oral DMT in China. 91 However, no evidence has shown the long-term benefits of corticosteroids in patients with MS during remission,92,93 and there are many limitations for treating patients with MS using NSIS regarding the safety concerns, such as cardiotoxicity, severe infections, malignancy, and chromosomal aberrations. 47 NSIS are usually prescribed for treating patients with highly active MS or aggressive MS,47,52 which is also recommended in the 2023 Chinese MS guideline. 51 The feasibility of NSIS switching to fundamental DMTs has been proven by clinical trials and real-world studies.17,94,95 Therefore, patients with not active and active inflammatory MS who are using corticosteroids and NSIS should be switched to the standardized DMT treatment during remission in accordance with disease activity, especially with an increasing number of DMTs approved in China.
De-escalation and discontinuation
There have been very limited data discussing de-escalation and discontinuation, and expert recommendations/guidelines varied in different countries.47,49,50,54 In 2020, the Canadian MS Working Group mentioned that de-escalation could be taken into consideration in patients on long-term (>5 years) immunosuppressant therapy. 54 For younger patients (<60 years old) who have been clinically stable for >5 years, de-escalation or discontinuation may cause breakthrough disease activity, so a maintenance therapy should be given in this group. 54 For older patients (>60 years old), a poorer response to DMTs was observed, whereas there was an increasing risk of side effects, such as infection, due to immunosenescence and comorbidities.96,97 Clinical trials of DMTs mainly included patients aged 18–55 years old,98–101 so there is a lack of evidence on efficacy and safety in elderly patients. It is still controversial whether DMTs should be de-escalated or discontinued in elderly MS patients.96,97,102–104
Other factors influencing clinical decision-making
In addition to the clinical benefits, the risk of DMTs should be considered when making clinical decisions. MS can be assessed regularly following the expert recommendation from the Pan-Yangtze River Delta Collaborative Group for Diagnosis and Treatment in Multiple Sclerosis. 105 On the basis of the standardized assessment, a comprehensive evaluation should be performed to provide optimized treatments to MS patients in China.
Pregnancy
Most patients with newly diagnosed MS in China are females aged 30–50 years. 7 Family planning should be considered when initiating a DMT treatment. So far, only GA can be prescribed to treat patients with RMS during pregnancy in China. DMF exposure (n = 379) during pregnancy did not increase the incidence of birth defects or the rate of spontaneous abortion compared with those in general population. 106 The lack of DMF/MMF accumulation makes it suitable for use until confirmed pregnancy; however, it should still be discontinued during pregnancy itself. 107 Teriflunomide has been associated with teratogenicity from studies on male sperm, which may last up to 2 years after discontinuation. 108 Teriflunomide (pregnancy category X) is contraindicated in pregnant women, and women of childbearing age should use reliable contraception if they are being administered with it. 108 Teriflunomide elimination can be accelerated using cholestyramine or charcoal if required. 109 Data from the Novartis Safety Database reported no congenital anomalies, reports of B-cell depletion, immunoglobulin/hematological abnormalities, or serious infections in MS patients (n = 30) or live births (n = 17) exposed to ofatumumab during pregnancy or 6 months prior to the last menstrual period. 110 However, the sample size in that study was small, so caution should be taken when prescribing ofatumumab in female patients with family planning. Moreover, fetal malformation was reported in MS patients treated with fingolimod in the first trimester. 111
Progressive multifocal leukoencephalopathy
Progressive multifocal leukoencephalopathy (PML) is a rare but severe demyelinating disease of the CNS caused by reactivation of John Cunningham virus. 112 It is a serious safety concern for patients who are immunocompromised, as well as for individuals receiving immunosuppressive/modulatory therapy. 112 No DMTs in China can be guaranteed to avoid the risk of PML, with the exception of GA. As of 21 July 2021, 12 confirmed PML cases of patients treated with DMF have been recorded, and the PML risk of DMF was reported to be 1.07 per 100,000 patient-years of DMF exposure. 113 As of April 2020, the PML risk of fingolimod was 0.131 per 1000 patient-years. 114 Additionally, the PML risk of fingolimod in Japan is higher (estimated 0.652 per 1000 patients) than the worldwide level (0.083 per 1000 patients). 115 A suspected PML case caused by teriflunomide has also been reported. 116 Leflunomide, whose active metabolite is teriflunomide, has been reported to be associated with PML. 117 Moreover, PML cases have been reported in patients taking anti-CD20s. 118 Although no PML cases have been found in MS patients taking ofatumumab, cases were reported in patients with chronic lymphocytic leukemia. 118 Therefore, irrespective of which DMTs are used, the risk of PML should be carefully monitored and managed.
Rebound
For cell-trafficking DMTs, referred to S1PRMs and natalizumab, treatment cessation may result in the rebound of disease activity.70,119,120 Rebound activity after the cessation of fingolimod was most commonly mentioned.119–121 A case report also revealed substantial disease exacerbation after the cessation of siponimod. 122 The risk of rebound activity should be evaluated when starting the treatment. Management to mitigate this possibility should be planned when switching to treatment with other DMTs. 123 A shorter washout period (as short as ⩽7 days) might be a favorable strategy for managing the rebound activity, according to the Japanese post-marketing surveillance of DMF, 124 The potential risk of PML should be evaluated depending on the subsequent DMTs. 125
Hepatitis B virus and tuberculosis infections
Hepatitis B virus (HBV) and tuberculosis (TB) infections are more common in China than those in Europe and the US.126,127 The incidences of acute and chronic HBV infection were reported to be 4.6 and 54.5 per 100,000 person-years in China. 128 Screening for HBV is required when starting DMT treatment, but only anti-CD20s are contraindicated in HBV infection129,130 because anti-CD20s increase the risk of mortality caused by HBV reactivation.131–133 In patients with active HBV, fingolimod cannot be used until resolution of the active phase. 129 The risk of HBV reactivation of teriflunomide and DMF has not been assessed but is likely to be low. 129 No publications have reported the risk of HBV reactivation upon the prescription of siponimod and ozanimod due to their short approval time. According to experiences from Italy, patients with MS are suggested to receive HBV vaccination. 134 Meanwhile, in patients with HBsAb titer negativity or <100 mIU/mL, antiviral prophylaxis is a good way of preventing HBV reactivation, 134 but drug-drug interaction should also be taken into consideration when prescribing antiviral prophylaxis and DMT simultaneously.
The incidence of TB infection was estimated to be 59 per 100,000 individuals (2020) in China, which is the second highest rate among countries globally. 127 In this context, two concerns should be raised: (1) The risk of TB reactivation may increase when prescribing DMTs and (2) TB infection may trigger MS. 135 TB screening should be done when initiating DMTs in MS patients who are at high risk of TB infection.135–137 DMTs affecting lymphocytes were reported to have a higher risk of indeterminate IFNγ release assay results, a measure for screening TB. 138 For patients with MS having negative TB results, TB screening should be repeated annually. 135 Meanwhile, for MS patients with latent TB, DMT initiation should not be delayed until anti-TB treatment has been completed, but clinical and laboratory monitoring should regularly be performed. 135 Because some anti-TB treatments and DMTs increase the potential risk of drug-induced liver injury,139,140 drug-drug interaction and TB reactivation should be taken into consideration when making clinical decisions.
Traditional Chinese medicine
Traditional Chinese medicine (TCM) has played an important role in the treatment of various diseases for thousands of years in China. TCMs have been used in Chinese patients with MS (~10%) who received treatment during the remission phase. 91 So far, TCM, acupuncture, moxibustion, tuina, and traditional sports have shown certain effects in the management of MS. 141 Yishen Jiedu decoction was proven to reduce the frequency of clinical relapse and prevent EDSS worsening through regulation of T lymphocyte subtypes and IgG. 142 In addition, Yishen Busui Tongluo decoction with methylprednisolone was shown to further improve the symptoms of neurological deficits, Multiple Sclerosis Impact Scale (MSIS-29) scores, and fatigue status compared with methylprednisolone alone during the acute phase. 143 The combination of acupuncture and herbal decoction also demonstrated favorable effects in MS patients. 144 The integration of Chinese and Western medicine has been explored across several observational studies.145,146 Therefore, TCM could be considered for the management of both the disease and the symptoms, as well as adverse event management, but drug-drug interaction should also be taken into consideration when planning to integrate Chinese and Western medicine.
Pediatric onset MS
The incidence of pediatric-onset MS (POMS) is 0.055 per 100,000, which is much lower than adults (0.288 per 100,000) in China. 7 The relapses were more frequent147,148 and the mental disorders were more common in pediatric patients than in adult patients.147–149 Early intervention is necessary to prevent disease progression; however, RCTs and regulatory approval of DMT for POMS are lacking. To date, only three DMTs have been approved for POMS: fingolimod by the European Medicines Agency (EMA), U.S. Food and Drug Administration and NMPA, DMF, and teriflunomide by EMA because of the promising efficacy and safety from three RCTs: PARADIGMS, CONNECT, and TERIKIDS.150–152 In China, fingolimod is the only DMT approved for POMS, which makes the management of POMS difficult. Nevertheless, patients with POMS should still be treated with DMTs as early as possible as per the approved indications and evidence.
Patient preference
MS is a chronic disease that requires long-term management. Patient preference is important for maintaining treatment adherence. Many factors impact treatment adherence, including the route and frequency of administration, intensity of monitoring, cost, and status of reimbursement under health insurance schemes. 70 There should be an open dialogue between patients and physicians in order to maximize the clinical benefits and patient adherence.47,55,70
Consideration for future MS treatments in China
The standardized DMT treatment of MS started late in China due to limited DMT availability before 2018. Because MS is defined as a rare disease in China, clinical neurologists in China have limited experience of using DMTs in clinical practice. An algorithm for stratifying patients in terms of disease activity is executable to help clinical neurologists choose a suitable DMT for MS patients. For the first time, we developed a simplified treatment algorithm based on the DMTs available in China, providing reasonable suggestions for treating MS patients in China. This algorithm will help physicians in China treat MS patients appropriately while also helping physicians in other regions who have limited experience of MS management.
Although the eight DMTs cover the full spectrum of efficacy classes from low, medium to high, there was only one high-efficacy monoclonal antibody, ofatumumab, which could not meet the medical needs of MS patients, especially for those with highly active MS. More HETs, such as natalizumab, cladribine, and alemtuzumab, are needed to provide more treatment options to MS patients with highly active MS or poor prognosis.
With the accumulation of clinical experience, personalized treatments will be considered depending on the prognostic factors in future. Recently, a new concept, high-definition medicine, has been proposed, 153 which involves human beings being treated as a comprehensive system by integrating big personal data at baseline regarding health, genetic risk and genomics, epigenomics, along with cellular, mosaicism, immunome, food, and nutrition tracking, imaging, pharmacogenetics and pharmacogenomics, and artificial intelligence technology, among others. This sophisticated analysis will achieve high-definition prevention and high-precision treatment for MS patients in the future.
