Abstract
Glatiramer acetate (GA) represents one of the most common disease-modifying therapies for multiple sclerosis. GA is currently approved for patients at high risk of developing clinically definite multiple sclerosis (CDMS) after having experienced a well-defined first clinical episode (clinically isolated syndrome or CIS) and for patients with relapsing-remitting multiple sclerosis (RRMS). GA's efficacy and effectiveness to reduce relapse frequency have been proved in placebo-controlled and observational studies. Comparative trials have also confirmed the lack of significant differences over other choices of treatment in the management of relapse frequency, and long-term studies have supported its effect at extended periods of time. Additionally, RRMS patients with suboptimal response to interferon β may benefit from reduced relapse rate after switching to GA, and those with clinically isolated syndrome may benefit from delayed conversion to CDMS. All these results, together with its proven long-term safety and positive effect on patients’ daily living, support the favorable risk-benefit of GA for multiple sclerosis treatment.
Introduction
Multiple sclerosis (MS) is a demyelinating and neurodegenerative disease affecting young adults, being the most frequent chronic neurological disease in this segment of population. Sometimes it can be detected before any clinical symptoms when a magnetic resonance imaging (MRI) study is done for other reasons (eg, radiological isolated syndrome or RIS); some persons with RIS will convert to MS after some years delay. In the most common cases, there is a first clinical manifestation or relapse typical of a demyelinating disease called clinically isolated syndrome (CIS). After some time, other relapses will ensue, making it possible to establish the diagnosis of clinically definite multiple sclerosis (CDMS). Nowadays, the diagnosis can be done as soon as MRI shows enhancing and not enhancing lesions after the CIS. Later on, patients enter a period of attacks and remissions, so-called relapsing-remitting multiple sclerosis (RRMS). Some 10 to 20 years later, the majority of patients enter a phase of disability progression with or without superimposed relapses called secondary progressive multiple sclerosis (SPMS). This is the typical course for 90% of MS patients; another 10% will have a progressive disease since onset, with or without exacerbations, called primary progressive multiple sclerosis (PPMS).
The approved treatments for MS at present include glatiramer acetate (GA-Copaxone®) and interferon beta (IFNβ), which has several different molecules and routes of administration: IFNβ-1a by intramuscular route at a dose of 30 μg per week (Avonex®); IFNβ-1a by subcutaneous route at a dose of 22 μg or 44 μg three times per week (Rebif®); and IFNβ-1b by subcutaneous route at a dose of 250 μg every other day (Betaseron® or Betaferon® and Extavia®). Other available drugs are Mitoxantrone (Novantrone®), Natalizumab (Tysabri®), and Fingolimod (Gilenya®). All these drugs have differences in efficacy and in their safety profile that are not within the scope of this review.
Glatiramer acetate (GA—Copaxone®; Teva Pharmaceuticals Ltd.), formerly known as copolymer 1, is a peptide mixture of four amino acids initially designed as an analogue of myelin basic protein. 1 It was synthesized in the 1960s and proved to be effective in preventing and suppressing experimental autoimmune encephalomyelitis, the primary animal model of multiple sclerosis, in a variety of animals including guinea pigs, rabbits, mice, and monkeys. 2 5 Early exploratory studies addressed the potential benefit of GA for MS treatment, 6 9 which was confirmed in aUS phase III trial that proved its efficacy in reducing the relapse rate in patients with relapsing-remitting MS (RRMS) without inducing significant side effects. 10 The benefit shown by GA administration to patients with RRMS in these studies finally led GA to being first approved in 1996 in the United States.
Even though the mechanism of action of GA is not completely understood, it has been shown to be an immunomodulatory agent with neuroprotective properties. 11 It also appears to induce GA-reactive Th2 immunoregulatory cells in experimental models of experimental autoimmune encephalomyelitis, 12 which may cross the blood-brain barrier and stimulate the secretion of anti-inflammatory cytokines and growth factors. 13 Additionally, GA has been shown to stimulate secretion of neurotrophins that might protect axons and promote injured neurons to be repaired. 12
All these properties may contribute to the beneficial effect of GA reported by clinical trials carried out in patients with RRMS either initiating immunomodulatory therapy10,14–17 or switching from IFNβ. 18 20 Long-term studies confirmed the efficacy and safety of GA during exposure periods extended up to 15 or 22 years,21,22 observational studies verified its benefit under clinical practice conditions, 23 27 and patient-oriented research supported its favorable effect on daily living. 28 33 In addition, patients with clinically isolated syndrome (CIS) have also been shown to benefit from GA treatment, delaying conversion to clinically definite MS (CDMS).34,35
Currently, GA is approved in many countries worldwide for the treatment of patients who have experienced a well-defined first clinical episode and are determined to be at high risk of developing CDMS and for the reduction in frequency of relapses in ambulatory patients with RRMS. This article aims to review the use of GA in patients with RRMS or CIS, mainly in terms of the management of relapse frequency.
Pharmacokinetic Profile, Metabolism, and Mechanism of Action
GA is the acetate salt of a mixture of synthetic polypeptide composed of L-glutamic acid, L-alanine, L-tyrosine, and L-lysine, with a molar residue ratio 0.14:0.34:0.43:0.09, an average length of 45 to 100 amino acids and a molecular mass of 4.7–11.0 kDa. The pharmacokinetic profile of GA has not been assessed in patients with MS. However, the data available from in vitro studies and those performed in healthy volunteers showed that the active substance was rapidly absorbed, with maximum plasma concentration of 69 to 605 ng/mL, an area under the plasma concentration-time curve from time zero to six hours of 1,644 to 67 532 ng · min/mL, and time to the maximum plasma concentration between 15 and 30 minutes after 60 mg of GA was subcutaneously administered to healthy volunteers. 36 A large part of the dose is rapidly degraded to smaller fragments in subcutaneous tissues, and studies carried out with radiolabeled doses of GA in animal models showed that the highest radioactive levels were in the stomach and thyroid, while the lowest radioactive levels were in the brain, possibly because of the high polarity and hydrophilic nature of GA prevent it from crossing the blood-brain barrier. 36
Even though the mechanism of action has not been completely elucidated, GA is thought to act by immunomodulating the pathways involved in the pathogenesis of MS and inducing both neuroprotective and remyelinating effects (Fig. 1). 11 GA has been shown to bind with high affinity to major histocompatibility complex class II molecules present on the surface of myelin basic protein-recognizing antigen presenting cells,37,38 acting either as a blocking peptide or as an antagonist and resulting in suppression of autoimmune T cell response, anergy, or both. 38 The binding of GA and major histocompatibility complexes to receptors on T-reactive cells in the bloodstream also triggers T cells to proliferate and secrete cytokines. 12 In fact, GA has been shown to induce some populations of GA-specific regulatory CD4+ and CD8+ T cells, as well as to induce a shift towards a Th2 pattern of anti-inflammatory cytokines. 39 42 Even though GA does not cross the blood-brain barrier, GA-reactive Th2 cells can do so and can also be reactivated by central nervous system (CNS) antigens such as myelin brain protein, which would induce the expression of anti-inflammatory cytokines that cause bystander suppression.42,43 In addition, GA has been shown to alter B cell function, inducing antibodies against GA. 44 46 However, these antibodies have been shown not to compromise GA activity,45,46 but, on the contrary, they have been suggested to enhance remyelination in chronic lesions. 47 Furthermore, GA has been demonstrated to act on antigen presenting cells, inhibiting monocyte reactivity, 48 exerting an antiproliferative effect of monocyte-derived dendritic cells on lymphocytes, 49 modulating monocyte and dendritic function that contribute to Th2 deviation,48,50,51 and triggering the production anti-inflammatory cytokines by monocytes and dendritic cells.49,52
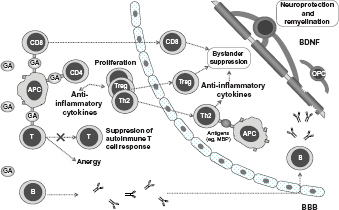
Overview of glatiramer acetate mechanism of action.
Furthermore, GA has been shown to stimulate secretion of neurotrophins such as brain-derived neurotrophic factor (BDNF) in both animal models13,53 and patients with MS,54,55 which might contribute to protect axons and promote injured neurons to be repaired. 12 Additionally, GA has also been associated with remyelination through increasing proliferation, differentiation, and survival of oligodendrocyte precursor cells in injury sites. 56 The increase in oligodendrocyte precursor cells has been suggested to be associated with elevation of insulin-like growth factor-1 and BDNF, which might be involved in the mechanism of proliferation and maturation of oligodendrocyte precursor cells into oligodendrocytes. 57 However, it is still difficult to differentiate whether the reduction in neuronal and myelin injury results from the release of BDNF or from the blockade of inflammatory pathways. 58
Clinical Studies
Efficacy
Utility of glatiramer acetate versus placebo
The efficacy of GA has been widely evaluated during the last three decades. In 1995, the first data on a Phase III clinical trial evaluating GA's effect in comparison with placebo was published.
10
This was the US pivotal trial of GA in RRMS, a Phase III, randomized, double-blind, placebo-controlled study in which a total of 251 patients diagnosed with RRMS according to Poser's criteria were randomly assigned (1:1) to receive placebo or GA. The objective of the study was to compare the therapeutic impact and tolerance of daily subcutaneous injections of 20 mg of GA in comparison with placebo over two years, using the number of relapses as the primary endpoint (Table 1). The results obtained showed that the 2-year relapse rate was reduced by 29%, from 1.68 in patients receiving placebo to 1.19 in those receiving GA (
Summary of glatiramer acetate efficacy versus placebo and interferon β in relapsing-remitting multiple sclerosis.

Another double-blind, randomized, placebo-controlled trial subsequently addressed the effect of GA in comparison with placebo in a European/Canadian population of patients diagnosed with RRMS.
15
A total of 239 patients were randomized (1:1) to receive placebo or 20 mg of GA administered as daily subcutaneous injections (Table 1). The analysis of the primary endpoint showed a statistically significant fewer number of enhancing lesions in the GA group (36.8 vs. 26.0;
Utility of glatiramer acetate versus interferon
Several clinical trials have directly evaluated the efficacy of GA in comparison with IFNβ in patients with RRMS, reporting the lack of significant differences in primary endpoints assessing reductions in relapse rates (Table 1).14,16,17
The REGARD Study was an open-label, randomized, phase IV trial carried out in 764 treatment-naïve patients diagnosed with RRMS according to McDonald criteria who had experienced at least one relapse during the year before being enrolled in the study.
16
Patients were randomly assigned (1:1) to receive 44 μg of IFNβ-1a administered subcutaneously three times per week or 20 mg of GA administered subcutaneously once daily. The analysis of the primary endpoint showed that the number of relapses over the 96 weeks of the study was lower than expected in both IFNβ-1a and GA groups (126 vs. 132) and there were no significant differences between treatment groups in the time to the first relapse (hazard ratio [HR] 0.94, 95% CI 0.74, 1.21;
The BEYOND Study, a randomized, parallel-group, multicenter, phase III trial, randomized (2:2:1) a total of 2,244 treatment-naïve patients with RRMS meeting McDonald and International Panel diagnostic criteria to receive either 250 μg or 500 μg of IFNβ-1b subcutaneously every other day or 20 mg of GA subcutaneously once-daily for at least two years.
17
The primary endpoint analysis showed that the relapse risk did not significantly differ among treatment groups (500 μg IFNβ-1b vs. 250 μg IFNβ-1b: HR 0.93, 95% CI 0.81–1.07,
The BECOME Study was a comparative, randomized, phase IV trial performed to assess the efficacy of IFNβ-1b and GA by using once-monthly MRI.
14
A total of 75 patients with RRMS or CIS were randomly assigned (1:1) to receive either 250 μg of IFNβ-1b subcutaneously every other day or 20 mg of GA subcutaneously once a day. MRIs were performed for a maximum of two years using a specific protocol optimized to detect enhancement, and the primary endpoint was the number of combined active lesions per scan in the first year, defined as the sum of the contrast-enhancing lesions in T1 plus the new T2 lesions on the MRIs that had appeared since the most recent examination. The results obtained showed that patients receiving either IFNβ-1b or GA showed a similar median number of combined active lesions per patient and MRI (0.63 vs. 0.58;
In light of the above, the results achieved in the primary endpoints of these three comparative studies were similar between INFβ-1b and GA treatments, including the time to the first relapse, risk of relapse, and number of combined active lesions. Likewise, similar results between treatments were observed in other variables such as ARR, disease progression, and many MRI assessments (eg, T2-active lesions, volume of gadolinium-enhancing lesions, T1-hypointense lesion volume, and lesion-free patients). In addition, the decreased relapse rate observed in these comparative trials was much greater than in the US pivotal trial. 10 The most likely explanation is the existence of differences in patient populations derived from the diagnosis of RRMS according to different criteria, that is, Poser's criteria in the US pivotal trial and McDonald criteria in comparative trials. 59 In fact, the currently used McDonald criteria based on imaging diagnosis techniques rather than in clinical criteria enable MS to be diagnosed and treated earlier. Therefore, the results of the REGARD, BEYOND, and BECOME studies more likely represent the efficacy that might be expected after starting GA treatment in patients newly diagnosed with RRMS. 59
Long-term utility of glatiramer acetate
Despite the increasing trend to recommend the initiation of disease-modifying therapies as early as possible, a reduced number of studies have approached the assessment of patients after long-term GA treatment (Table 2).
Summary of long-term efficacy of glatiramer acetate in patients with relapsing remitting multiple sclerosis.

One of these studies is the open-label extension phase of the US GA pivotal trial, which has reported the effect of GA treatment for 6 years,60,61 8 years, 62 10 years, 63 and up to 15 years. 21 The last data cut-off was performed in February 2008, at which 100 out of the 232 (43.1%) patients who had received at least one dose of GA from the beginning of the trial still remained in the extension phase and the average length of GA treatment was of 13.6 years. 21 The results obtained at this data cutoff showed a reduction of 77.7% in the ARR, from 1.12 before starting GA to 0.25 at the 15-year analysis.
Another study assessing long-term effects of GA was performed in 46 patients who received GA as compassionate use for RRMS up to 22 years. 22 After a mean duration of GA treatment of 16.3 years, mean ARR also declined significantly from 3.06 prior to study entry to 0.09 at the last observation. Additionally, approximately 72% of patients were still relapse-free throughout the course of GA treatment.
Utility of glatiramer acetate in terms of reducing brain atrophy
Brain atrophy is a well-known phenomenon in patients with MS, 64 which may be related to focal tissue damage at earlier points in time and represents the most significant MRI predictor of disability at follow-up. 65 Brain volume loss is a dynamic process that mildly affects RRMS patients and usually occurs despite receiving disease-modifying treatments. 66 Even though several studies have approached the effect of GA on brain atrophy, the variable results obtained did not enable us to reach clear conclusions.
The short-term assessment of brain loss in patients included in the European/Canadian placebo-controlled trial did not find a significant effect of GA on reducing brain loss in patients with RRMS over an 18-month follow-up period, in which brain volume decreased by 1.4% and 1.2% in patients originally randomized to placebo and GA, respectively. 67 Another study carried out in patients with RRMS also reported a decline of 2.0% in the percentage of central brain volume change over a 14-month follow-up, which was correlated with higher scores on the Expanded Disability Status Scale (EDSS) and T2 lesions at baseline. 68 Additionally, a recently published study in patients with RRMS or CIS showed a steady decrease in brain volume not explained by changes in inflammation detected in monthly MRI irrespective of IFNβ or GA treatment. 69 In fact, an average monthly decrease of 1.3 cm3 was observed in spite of an average transient increase of 1.2 cm3 per lesion resulting from focal inflammatory activity.
In contrast, the US pivotal trial reported a nearly three-fold lower annual decline in brain volume over a 2-year GA treatment in comparison with placebo. 70 Similarly, two large randomized comparative trials of IFNβ and GA that included the assessment of changes in brain volume showed lower decreases in whole brain volume during the first year of treatment with GA. 17 However, one of them did not detect such significant differences among treatments during the second and third years of treatment,16,17 while the other still detected lower brain volume loss during the second year of GA treatment. 16 In both studies, the magnitude of brain volume loss was greater at the beginning of the treatment, which might partly be associated with a reduction in inflammatory edema (pseudoatrophy) that was more prominent in IFNβ than in GA.16,17
Even though little data is currently available on the long-term effect of GA in brain atrophy, the extended open-label follow-up of the US pivotal trial appears to show that greater long-term treatment with GA might partially prevent loss of brain parenchyma in patients with RRMS.
71
In fact, brain loss was greater in patients originally on placebo, showing that a delay in initiating GA treatment might result in a long-term progression of MRI-measured pathology that could be prevented. In addition, a recently published comparison of RRMS treatments such as the administration of 30 μg of IFNβ intramuscularly every week, 250 μg of IFNβ subcutaneously every other week or 20 mg of GA subcutaneously once a day over a 5-year period showed that GA enabled the least brain volume loss in comparison with high-dose IFNβ (−2.62% vs. −2.27%;
The variable results obtained in the above-mentioned studies might be partly derived from differences in study designs and durations, which may not be adequate to reveal beneficial effects in a chronic disease like MS. 72 Thus, further research on brain volume variation is still needed to clarify the effect of GA treatment.
Utility of switching from interferon to glatiramer acetate
Several studies have been performed to address the effect of switching the treatment from IFNβ to GA in patients with suboptimal response to IFNβ, which showed clear improvements in relapse rates (Table 3).
Summary of studies reporting switching relapsing-remitting multiple sclerosis treatment from interferon β to glatiramer acetate.
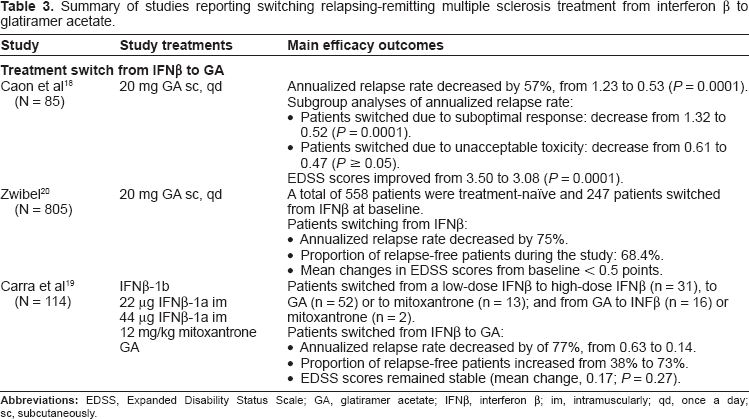
The first was a prospective observational study carried out in 85 patients with RRMS who had received weekly intramuscular doses of IFNβ-1a for at least 18 months.
18
These patients switched to GA because of persisting suboptimal response (n = 62) or unacceptable toxicity (n = 23) to IFNβ. The analysis of the primary endpoint showed that GA enabled ARR to be reduced from 1.23 to 0.53 (
A subsequent prospective open-label study was conducted in a heterogeneous population of patients with RRMS who received GA on a compassionate-use basis, which included a cohort of patients who had discontinued IFNβ-1b as a result of inadequate response or toxicity. 20 A total of 805 patients were enrolled into the study: 558 treatment-naïve patients and 247 patients previously treated with IFNβ-1b. Patients who had previously received IFNβ-1b were older, showed more advanced disease, and reported greater disability according to EDSS scores at baseline. ARR decreased by 75% in both treatment-naïve patients and those previously receiving IFNβ-1b after an average duration of GA treatment of 20.3 months and 14.8 months, respectively. In addition, 69.5% of treatment-naïve patients and 68.4% of patients previously treated with IFNβ-1b remained relapse-free throughout the 3.5 years of the study. The median time to the first relapse was 1391 days for treatment-naïve patients and could not be estimated for prior IFNβ-1b patients because 50% of these patients had not relapsed by the study end.
Another prospective observational study carried out in 114 patients with RRMS assessed the impact on clinical outcomes of switching immunotherapy in patients who did not respond adequately to initial treatment or experienced unacceptable side effects. 19 Patients were switched from a low-dose IFNβ to high-dose IFNβ (n = 31), to GA (n = 52), or to mitoxantrone (n = 13) and from GA to INFβ (n = 16) or mitoxantrone (n = 2). All groups obtained benefit from switching the treatment over the 3-year post-switch treatment period. However, the best results were obtained after switching to GA or mitoxantrone in terms of improving ARR, increasing the proportion of relapse-free patients, and the stabilization of EDSS scores. In fact, patients whose treatment was switched from IFNβ to GA even reached a reduction in the ARR of 77%, from 0.63 to 0.14, and the proportion of relapse-free patients increased from 38% to 73%.
Utility of glatiramer acetate in clinical practice
Even though clinical trials provide strong evidence, their strictly controlled conditions and homogeneous patient populations may entail differences in the effectiveness and safety observed in clinical practice. This fact has reinforced the need to perform observational studies worldwide in order to assess the effect of GA in routine clinical practice.
Two database analyses of large populations of patients with MS under treatment with intramuscular IFNβ or GA have addressed the use of these drugs in US institutions.23,26 The results obtained showed that the use of GA was associated with a significantly lower estimated 2-year risk of relapse in comparison with IFNβ-1a, which reached 5.18% and 10.01% (
A German study also compared immunomodulatory treatments for RRMS such as intramuscular IFNβ-1a, subcutaneous IFNβ-1a, IFNβ-1b, and GA administered under clinical practice conditions using a structured clinical database.
25
The relapse rate was reduced to a comparable extent with all regimens after six months of treatment. However, the reduction achieved after 24 months was greater in patients receiving GA (−0.81;
The results obtained in a Spanish cohort of patients with RRMS treated with GA for two years showed a 65.2% reduction in the relapse rate, which was 58.3% in the subgroup of patients that switched from IFNβ because of suboptimal response.
27
At the second year of treatment, the proportion of relapse-free patients was 46.6%. Similarly, another observational study carried out in a Spanish population of 104 patients with CDMS or RRMS was recently published showing the beneficial effect of GA administration under clinical practice conditions in terms of both relapse rate and disease progression.
24
In fact, the relapse rate was reduced by 60% (1.0 vs. 0.4;
Utility of glatiramer acetate in the clinically isolated syndrome
The PreCISe trial was a randomized, double-blind, placebo-controlled, phase III trial that aimed to assess the effect of GA on conversion to CDMS in patients with CIS.
34
A total of 481 patients were enrolled in the study and randomly assigned (1:1) to receive daily subcutaneous injections of 20 mg of GA or placebo. The analysis of the primary endpoint showed that patients receiving GA attained a 45% reduction in the risk of conversion to CDMS during the placebo-controlled phase of the study (HR 0.5, 95% CI 0.40–0.77;
Safety
Long-term studies have confirmed the safety profile of GA administration in patients with RRMS.21,22,60–63 In fact, results from the follow-up of RRMS patients included in the US pivotal trial up to 15 years showed that the adverse events thought to be related to GA were as expected according to previously published information. 21 These adverse events included local reactions at the injection site such as erythema, pain or edema, and symptoms of self-limiting immediate post-injection reactions such as vasodilatation, chest pain, palpitation, tachycardia, or dyspnea. No immune diseases, immunosuppression, or malignancies were reported to be associated with GA. Neither was there evidence of hematologic, hepatic or renal dysfunction, nor emergence of time-dependent adverse events. Additionally, the analysis of reasons for patient discontinuation showed that only 23 (9.9%) out of the 232 patients who had received at least one dose of GA withdrew because of adverse events. The remaining reasons for discontinuation comprised lost to follow-up (n = 14) and patient decision/other (n = 95), which included patient perception of disease worsening (n = 29), desire to switch or combine therapies (n = 26), difficulty/inability/unwillingness to adhere to study protocol (n = 32), and pregnancy (n = 8). Similarly, the results obtained from the long-term, compassionate-use study of GA up to 22 years in RRMS patients showed similar adverse events to those previously reported, mainly including injection site reactions such as soreness, redness, swelling, or itching. 22 Six patients who had taken GA up to 22 years also reported the occurrence of lipoatrophy, but no one reported skin necrosis over the course of the study. Only two (4.4%) patients discontinued because of adverse events (immediate post-injection reactions), while other reasons for discontinuation included patient withdrawal of consent (n = 15), protocol violations (n = 5), patient desire to try another therapy (n = 3), pregnancy (n = 1), and lost to follow-up (n = 1). Furthermore, the adverse events reported in patients with CIS were consistent with the known safety profile of GA, including injection-site reactions in 135 (56%) patients and immediate post-injection reactions in 47 (19%) patients as the most frequently reported adverse events. 34 Other adverse events reported irrespective of being related to GA, such as lymphadenopathy, urticaria, influenza-like illness, constipation, pruritus, erythema, vomiting, rash, and blurred vision, were described in up to 5.3% of patients. A low number of withdrawals was also reported in patients with CIS, reaching only 14 (5.8%) patients.
Some anecdotal reports have informed that during treatment with GA, some other side effects may seldom occur, including nausea, joint pain, severe muscle tension or spasticity, impotence, or decreased interest in sex.
Economic advantages
Cost variations among countries have been described as a result of differences in the cost of patients’ care, drug usage and clinical practice, as well as because of variations in the estimates of cost-effectiveness and the lack of homogeneity in the studies’ design. 75 However, overall favorable results have been obtained in the budget impact and cost-effectiveness analyses performed comparing GA with other immunomodulatory agents. These favorable results might be helpful to optimize health resources and improve budget control of MS treatment.
Assessment of immunomodulatory therapies for RRMS through a model constructed from the perspective of US health care showed that GA was the best strategy of the four immunomodulatory therapies used for RRMS, with incremental costs per quality-adjusted life-years (QALY) of $416,301, $303,968, $310,691, $258,465 for subcutaneous IFNβ-1a, intramuscular INFβ-1a, IFNβ-1b, and GA, respectively.
76
Additionally, another model for US health care showed that subcutaneous IFNβ-1a, IFNβ-1b and GA had the most favorable cost per relapse avoided ($80,589, $87,061 and $88,310, respectively) in contrast to intramuscular INFβ-1a, whose cost-effectiveness ratio was the least favorable ($141,721).
77
The total 2-year costs of these treatments were estimated to be $52,010, $50,389, $48,473 and $49,068 for IFNβ-1b, subcutaneous IFNβ-1a, intramuscular INFβ-1a, and GA, respectively. Two additional database analyses also approached the costs of these treatments in the US, reporting lower costs during GA treatment.23,26 Indeed, the use of GA represented a 7% reduction in total 2-year direct medical costs in comparison with intramuscular IFNβ-1a ($44,201 vs. $41,121;
Further analyses in several countries have recently addressed the cost-effectiveness of immunomodulatory therapies for MS. A budget impact analysis of first-line treatments of RRMS considering a Spanish cohort of 22,255 patients was recently published addressing the costs of both IFNβ and GA. 79 The results obtained showed a mean global budget impact per year of €260,775,470, with an average cost per patient of €11,540 per year. The exclusion of GA would increase the budget impact by 3.2% (€8,419,214; €372 per patient per year). The analysis of the hypothetical scenario in which one treatment would only be administered, the lower budget impact would be obtained with GA (mean annual budget impact per patient, €9,715). A later study assessing the first-line treatments of RRMS in Spain showed that GA (€322,510) was the least costly strategy during the 10-year time-horizon of the study, followed by intramuscular IFNβ-1a (€329,595), IFNβ-1b (€333,925), and subcutaneous IFNβ-1a (€348,208). 80 The average cost per patient for these disease-modifying therapies were €42,454, €47,532, €48,752, and €65,475, respectively. The health benefits provided by these treatments ranged from 4,117 QALY per patient with GA to 4,177 QALY per patient with intramuscular IFNβ-1a. This last treatment was a dominant strategy compared with subcutaneous IFNβ-1a and IFNβ-1b, but it was not considered cost-effective in comparison with GA since the incremental costs per QALY gained with intramuscular IFNβ-1a exceeded the €30,000 per QALY threshold commonly used in Spain. The comparison of costs between the treatment with GA and fingolimod has been recently addressed in a Spanish model (unpublished data). The cost for the first year of treatment with GA was €9,439 in contrast to €19,602 with fingolimod, reaching a difference of €10,163, which represented a 2.1 times higher cost of fingolimod than GA. Additionally, a recently published Italian survey of MS costs which also included natalizumab reported direct costs per patient per year of €9,501 for intramuscular INFβ-1a, €8,553 for IFNβ-1b, €11,255 for 44 mg of subcutaneous IFNβ-1a, €9,883 for 22 mg of subcutaneous IFNβ-1a, €8,174 for GA, and €21,817 for natalizumab, representing GA the least expensive biological disease-modifying agent for MS treatment. 81
Place in Therapy and Patient Preference
MS treatment has considerably evolved in the last decades. However, GA and INFβ still remain the main treatment choices for patients with RRMS. New drugs have been approved recently, but, despite having proved their short-term efficacy, their long-term safety profile has not been completely clarified.
Natalizumab is a recombinant humanized anti-α4-integrin antibody recently indicated as single disease-modifying therapy in patients with highly active RRMS despite treatment with INFβ. The approval of this monoclonal antibody by the EMA in 2006 generated considerable optimism especially for patients with active disease in spite of being under first-line treatment in whom advancing therapy to natalizumab represents a logical approach for disease management. 82 However, the initial excitement was tempered by the unexpected development of progressive multifocal leukoencephalopathy. 83 Additionally, a primary central nervous system lymphoma (PCNSL) has also been reported in a 40-year-old patient after 21 infusions of natalizumab administered as montotherapy. 84 Since the prevalence of PCNSL in patients with MS is low and the patient was otherwise not immunocompromised, the association between natalizumab treatment and PCNSL cannot be ruled out. The occurrence of opportunistic infections and malignancies also represents a conspicuous concern either on natalizumab or other new emerging therapies for MS treatment. 83
Cladribine is an antineoplastic and immunosuppressive substance currently indicated for the treatment of hairy cell leukemia. However, the EMA declined approval for it to be used in patients with MS in October 2010 because it considered that the risks in patients under this treatment might not compensate the benefits.
Fingolimod is a sphingosine 1-phosphate receptor modulator approved by the EMA in 2011 as single disease-modifying therapy in patients with highly active RRMS despite treatment with IFNβ and for those with rapidly evolving severe RRMS. This approval enabled the first oral treatment for MS to be available for patients with RRMS. However, its introduction in the market has been gradual and slower than expected because of its higher annual cost and practical inconveniences derived from heart monitoring before and after the first dose.
The main handicap for these therapies is the occurrence of adverse events. However, other factors such as the appearance of neutralizing antibodies may also represent an important drawback. In fact, therapy-induced neutralizing antibodies are a major problem for INFβ treatment, since they might seriously compromise its biologic activity. 85 These neutralizing antibodies may be induced in up to 35% of patients under treatment with INFβ and reduce its efficacy on relapses and MRI lesions, particularly at high titers. 82 Additionally, persistent antibodies against natalizumab were detected in about 6% of natalizumab-treated patients, which might be associated with infusion-related adverse events and efficacy loss.86,87
The overall safety of GA21,22 and the absence of neutralizing antibodies that compromise its activity45,46 represent important advantages for MS treatment. GA efficacy10,14–17 and effectiveness 23 27 under routine clinical practice conditions have also been widely proved in patients with RRMS. In these patients its efficacy has been shown to be comparable to IFNβ14,16,17 and its long-term effect has also been confirmed.21,22 RRMS patients with suboptimal response to IFNβ may also benefit from switching to GA, 18 20 and patients with CIS under treatment with GA may delay their conversion to CDMS. 34 Additionally, administration of GA may favorably affect disabling symptoms of MS such as depression, fatigue, and spasticity that might interfere with patients’ work performance and other activities of daily living. In fact, GA does not negatively affect depression, which is why GA is the treatment choice for patients with history of depression 88 and might even exert an antidepressant effect through its neuroprotective and anti-inflammatory activity. 32 GA has also been shown to decrease fatigue and work absenteeism31,33,89 as well as to improve spasticity in patients with RRMS previously treated with IFNβ.29,30 These effects might impact on patients’ perception of their quality of life. Even though data on health-related quality of life are still scant, the results obtained from a recently published study approaching its assessment in RRMS patients receiving GA in daily clinical practice confirmed early gain in health-related quality of life in treatment-naïve patients after 6 months of treatment that was sustained at 12 months. 28 Early discontinuation of treatment precluded patients from improving health-related quality of life, which might be explained by the prevention of the full effect of GA, as relapse reduction usually occurs after six months of treatment. 28 Therefore, optimal adherence to therapy must be ensured to improve both treatment outcomes and quality of life. The major reasons for treatment discontinuation are patient decision, lost to follow-up, and the occurrence of adverse events. 90 Additionally, several factors such as high levels of self-efficacy,91,92 hope, perceived health care provider support, and no previous use of other immunomodulators have been associated with enhanced adherence to GA, 91 while greater level of disability, spasticity, and problems with literacy may negatively influence adherence. 92 Predictors of adherence might differ between treatment-naïve patients and those switching from IFNβ to GA, so previous experience should also be considered when tailoring interventions to increase adherence to treatment and further research is encouraged to overcome potential barriers. 93
Taken together, the proven safety, efficacy, effectiveness, and effect on daily living make GA a preferable choice for patients diagnosed with RRMS and those with CIS at high risk of developing CDMS. Furthermore, the cost-effectiveness studies confirmed that GA represents an efficient treatment alternative for MS in comparison with other therapeutic alternatives.23,26,76–81 These facts reinforce the important role of GA in the treatment of MS.
The emergence of new treatments might conspicuously change the therapeutic approach to MS in the near future. Even though greater efficacy might be achieved with these treatments, their risk-benefit balance considering their long-term efficacy and safety must be carefully analyzed. In fact, data from clinical trials and previous experience in therapeutic areas different from MS have proved that the use of some emerging therapies might entail the occurrence of opportunistic and community-acquired infections, malignancies, and autoimmune diseases. 83 The effect and length of these complications might be unpredictable, which might considerably compromise the attempts to make safe and reasonable decisions about MS treatments. Therefore, the use of GA in the future would not only depend on its risk-benefit analysis but also in the risk-benefit analysis of emerging treatments such as drugs currently approved for the treatment of other diseases, new biological agents, and other drugs currently under development.
Conclusions
The current data available support the continuous use of GA from the initial first-line treatment after the diagnosis of CIS 34 to its prolonged administration during the course of RRMS to reduce the occurrence of relapses.10,14–17,21–27 The studies addressing the direct comparison of GA and IFNβ have shown a similar effect in the management of RRMS relapse frequency14,16,17 In addition, the reduction in relapse rate during GA administration has been shown to be greater in more recently published trials14,16,17 than that observed in the US GA pivotal trial. 10 The magnitude of reduction in relapse rates shown in more recent trials could be considered more representative of the effect that might be obtained after starting GA for the early treatment of RRMS at present. Furthermore, patients with suboptimal response to IFNβ may also benefit from reducing relapse rates after switching to glatiramer actetate. 18 20
GA has been shown to have an overall safety profile both in patients with CIS 34 and during long-term administration to patients with RRMS.21,22,60–63 No opportunistic infections, malignancies, or immune-mediated disorders were reported. However, the occurrence of immediate post-injection reactions and local injection-site reactions associated with GA still need to be followed up.
In conclusion, the proven efficacy of GA for CIS and RRMS, its comparable effects to other treatment choices, and its favorable safety profile over long-term periods support the positive risk-benefit of GA for the treatment of MS. However, the future of GA will not only depend on its own risk-benefit balance but also on the risk-benefit analysis of other emerging therapies.
Author Contributions
Conceived and designed the manuscript: OFF. Analyzed the data: OFF. Wrote the first draft of the manuscript: OFF. Contributed to the writing of the manuscript: OFF. Agree with manuscript results and conclusions: OFF. Jointly developed the structure and arguments for the paper: OFF. Made critical revisions and approved final version: OFF.
Funding
Medical writing support provided by Dynamic S.L. was funded by TEVA Pharma S.L.U. No other external funding was provided for the preparation of this manuscript.
Competing Interests
The corresponding author (Oscar FernÁndez, MD, PhD) has received honoraria as consultant in advisory boards, and as chairmen or lecturer in meetings, and has also participated in clinical trials and other research projects promoted by Biogen- Idec, Bayer-Schering; Merck-Serono, Teva, Almirall, Allergan and Novartis.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest. Provenance: the authors were invited to submit this paper.
Footnotes
Acknowledgements
Medical writing support was provided by Esther Álvarez-García at Dynamic S.L. during the preparation of this paper, funded by TEVA Pharma S.L.U. Responsibility for opinions, conclusions, and interpretation of data lies with the author.
