Abstract
Background:
There are limited data on the real-world healthcare resource use (HCRU) and management costs of myasthenia gravis (MG) in England.
Objective:
This study aims to assess the burden of disease for patients with MG in England.
Design:
A retrospective, observational cohort study of adult patients diagnosed with MG, using data from the Hospital Episode Statistics data warehouse.
Methods:
Patients with a first-ever recorded diagnosis of MG between 30 June 2015 and 30 June 2020 were followed up until 30 June 2021 or death, whichever occurred first. Post-diagnosis patient characteristics, treatment patterns, HCRU, and costs were described. Costs were evaluated using National Health Service reference costs.
Results:
A total of 9087 patients with a median follow-up time of 2.9 years (range, 1.7–4.3 years) were included. The mean age at diagnosis was 66.5 years and 53% of the patients were male. A large proportion of patients (72.8%) were admitted as inpatients during follow-up with a mean number of 1.3 admissions. Patients hospitalized for MG-related complications spent a mean of 9.7 days per patient-year in the hospital. During follow-up, 599 (6.6% of the total cohort) and 163 (1.8%) patients had a record of rescue therapy with intravenous immunoglobulin (IVIg) and plasma exchange (PLEX), respectively. Rituximab was administered to 81 (0.9%) patients and 268 (2.9%) patients underwent thymectomy. In those patients receiving rescue therapy or rituximab, >10% received at least three cycles of the same treatment. The average annual cost of hospital admissions across all patients treated with IVIg, PLEX, and rituximab were £907,072, £689,979, and £146,726, respectively.
Conclusion:
A majority of MG patients required hospitalization or accident and emergency attendance, resulting in high HCRU and costs. A subset of patients required rescue therapy (including IVIg and PLEX), rituximab administration, ventilation, or thymectomy.
Introduction
Myasthenia gravis (MG) is a rare, chronic autoimmune disease that affects the neuromuscular junction (NMJ). 1 MG is characterized by the production of pathogenic autoantibodies that bind to components of the NMJ, with the most common being the acetylcholine receptor, muscle-specific tyrosine kinase (MuSK), or lipoprotein receptor-related protein 4. 2 The prevalence and incidence of MG in the UK have been estimated at 33.7 [confidence interval (CI): 32.7–34.7] cases per 100,000 population and 2.46 (CI: 2.34–2.59) cases per 100,000 person-years, respectively, and both are on the rise since 2008. 3
Acetylcholinesterase inhibitors (AChEIs) are the preferred first-line symptomatic treatment, followed by corticosteroids, non-steroidal immunosuppressants, and thymectomy. 4 In cases of myasthenic exacerbation or myasthenic crisis (a life-threatening condition due to respiratory muscle weakness and swallowing difficulties), immediate hospitalization and rescue therapy with plasma exchange (PLEX), intravenous immunoglobulin (IVIg), ventilation, or immunoadsorption may be required.5–7 Patients are considered to have refractory MG if they fail to respond adequately to conventional therapies, are unable to reduce immunosuppressive therapies without relapses, or have frequent myasthenic crises even while on therapy. 6 Care of these patients places a significant burden on the healthcare system.
A recent systematic review including 16 studies found that the current body of literature on healthcare resource use (HCRU) and cost of MG management is sparse and limited to a few geographical settings and resource categories. 8 In England, one study found that patients with refractory MG had significantly higher secondary HCRU than patients with non-refractory MG; however, the study did not include an estimation of associated costs of care. 9 The current study aimed to evaluate the burden of MG on secondary care settings across England, and to gain a better understanding of the baseline characteristics, treatment, HCRU, and associated costs in patients with newly diagnosed MG.
Methods
Data source and study population
Hospital Episode Statistics (HES) is a secondary care data warehouse that contains pseudonymized records of all patients admitted to the NHS hospitals in England, with data stored on hospital diagnoses, procedures, treatment, HCRU [including inpatient admissions, attendance at outpatient departments, and accident and emergency (A&E)], and associated costs for the past 10 years. 10 In this retrospective, longitudinal, observational cohort study, adult patients (age ⩾18 years) were included if they had a first-ever recorded diagnosis of MG (index date) between 30 June 2015 and 30 June 2020 (eligibility period) in the HES. Patients were described for the 12-month period prior to the index date (baseline) and followed up from the index date until 30 June 2021 or death, whichever occurred first. Access to HES was provided under license via Harvey Walsh Ltd from National Health Service (NHS) Digital (Data Sharing Agreement: DARS-NIC-05934-M7V9K).
The International Statistical Classification of Diseases and Related Health Problems 10th Revision codes were used to define diagnoses including MG (G700) recorded either in inpatient or outpatient settings. Of note, as the recording of diagnosis does not form any part of the UK NHS costing system in an outpatient setting, a significant under-recording of patients receiving diagnosis in that setting is therefore likely. The Office of Population Censuses and Surveys Classification of Interventions and Procedures version 4 codes were used to classify interventions, procedures, and procurement for treatment; and Healthcare Resource Group (HRG) NHS tariff codes were used to define activity-based costs. 11 The HRGs were costed using Payment by Results tariffs. 12
Data extracted included patient demographics at the index date (age, sex, ethnicity, geographic region, index of multiple deprivation), baseline pre-specified comorbidities known to be associated with MG, and baseline Charlson comorbidity index (CCI) score.13,14 The CCI score was calculated using the summary hospital-level mortality indicator with UK-adapted weights 15 (Supplemental Table 1). The proportion of patients with mild (1–2), moderate (3–4), and severe CCI scores (⩾5) were reported. Types and dates of rescue therapies (IVIg, PLEX), rituximab administration, ventilation, and thymectomy procedures provided in the hospital, as well as HCRU during the follow-up period (outpatient visits, inpatient hospitalizations and length of stay, A&E attendances), were extracted. This study also evaluated HCRU based on a history of rituximab treatment, as these patients are more likely to be refractory and thus are expected to require more healthcare needs/costs. Cost of treatment included all activities occurring during an inpatient spell or outpatient visit for a specific treatment, with only the relevant high-cost drugs being included, because administration of other specific treatments (e.g. AChEIs, corticosteroids, or other first-line non-steroidal immunosuppressants) is not available in HES. 16 Acquisition costs of drugs were also not available. 16
Statistical analysis
All analyses were descriptive. Continuous variables were analyzed using mean, standard deviation (SD), median, and interquartile range as appropriate, and categorical variables were presented using frequency counts and proportions. The number of inpatient treatments received by each patient was counted as the number of individual hospital spells, which encompassed the total duration of hospitalization from admission to discharge. To estimate the incidence of MG in the general population, a total adult population of 43 million in England was assumed. 17
HCRU was summarized per patient per year for those with at least one attendance, and the frequency of non-attendances was also reported. The cumulative cost of admission for patients undergoing specific procedures (IVIg, PLEX, and rituximab) was calculated by multiplying the mean cost per patient per year by the number of patients. HCRU and treatment were summarized overall and by subgroups, including sex, age at index diagnosis (<50 and ⩾50), and year of first recorded diagnosis (2015–2020). For the age subgroups, all treatments were aggregated across patients, and age at treatment administration was used rather than age at index diagnosis.
Missing data were reported, but to comply with HES guidance, frequencies estimated in less than five patients were suppressed to preserve patients’ confidentiality, and other related frequencies were rounded to prevent back-calculation of the suppressed numbers. 18
Results
Study participants
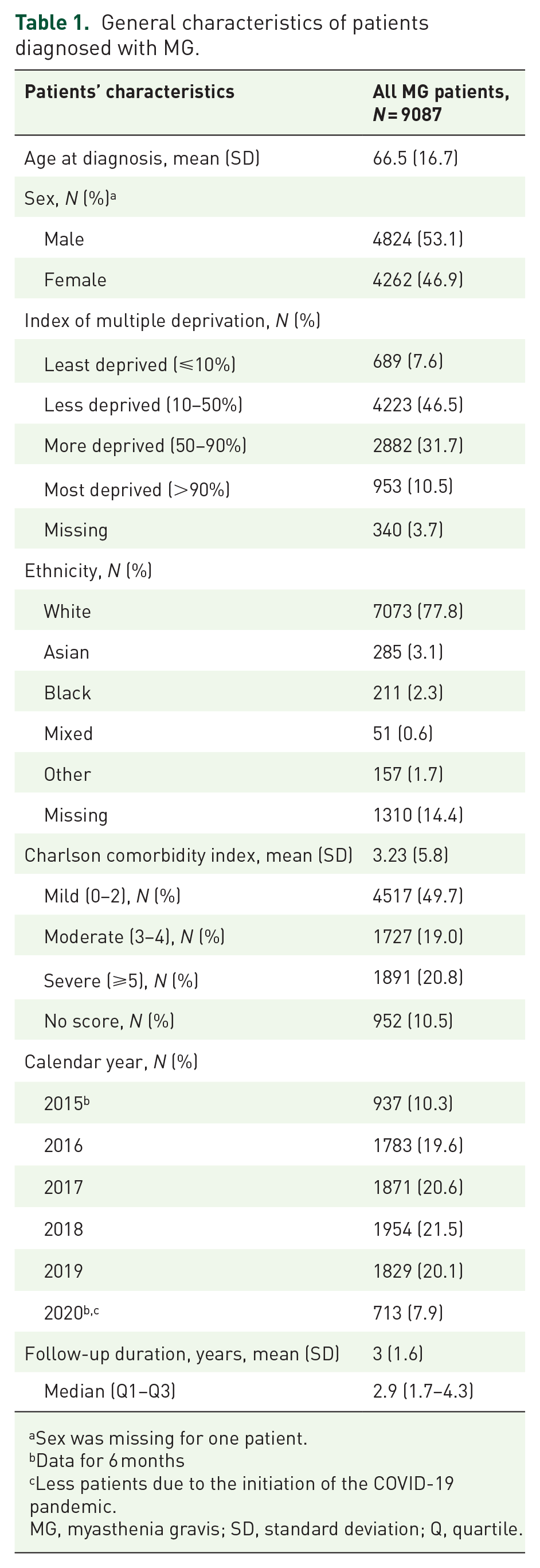
We identified 9087 patients with a diagnosis of MG in HES who were included in the study (Table 1). The majority of patients had their index diagnosis recorded during an inpatient stay (89.5%), with the most common departments being General Internal Medicine (23.2%) and the Neurology department (21.1%; Figure 1).
General characteristics of patients diagnosed with MG.
Sex was missing for one patient.
Data for 6 months
Less patients due to the initiation of the COVID-19 pandemic.
MG, myasthenia gravis; SD, standard deviation; Q, quartile.

Referral pathways in patients diagnosed with MG.
A mean of 1859 patients were diagnosed with MG each year, resulting in an estimated MG incidence of 4.32 (95% CI: 4.13–4.52) per 100,000 population per year. The median follow-up was 2.9 years (Q1–Q3: 1.7–4.3). Of the overall cohort, 8398 (92.4%), 8098 (89.1%), and 6295 (69.3%) patients had data available for at least 6, 12, and 24 months of follow-up period, respectively.
Patients’ characteristics
The mean (SD) age of patients at diagnosis was 66.5 ± 16.7 years; 53.1% were male, 77.8% were white, and 10.5% lived in the most socioeconomically deprived areas in England (Table 1). At diagnosis, 83.8% of patients were aged ⩾50 years (Supplemental Table 2), and females were, on average, 6 years younger than males (mean 63.4 versus 69.4 years).
At baseline, nearly half of the patients (49.7%) had mild CCI scores, 19.0% had moderate scores, and 20.8% had severe scores (Table 1). The most common comorbidities during the baseline period were hypertension (16.2%), diabetes mellitus (7.5%), chronic kidney diseases (3.2%), and chronic obstructive pulmonary disease (3%) (Supplemental Table 2). Neurology and ophthalmology were the most common last specialty outpatient visits prior to MG diagnosis (Figure 1) and 3 months before MG diagnosis (Supplemental Table 2).
Treatments administered in secondary care
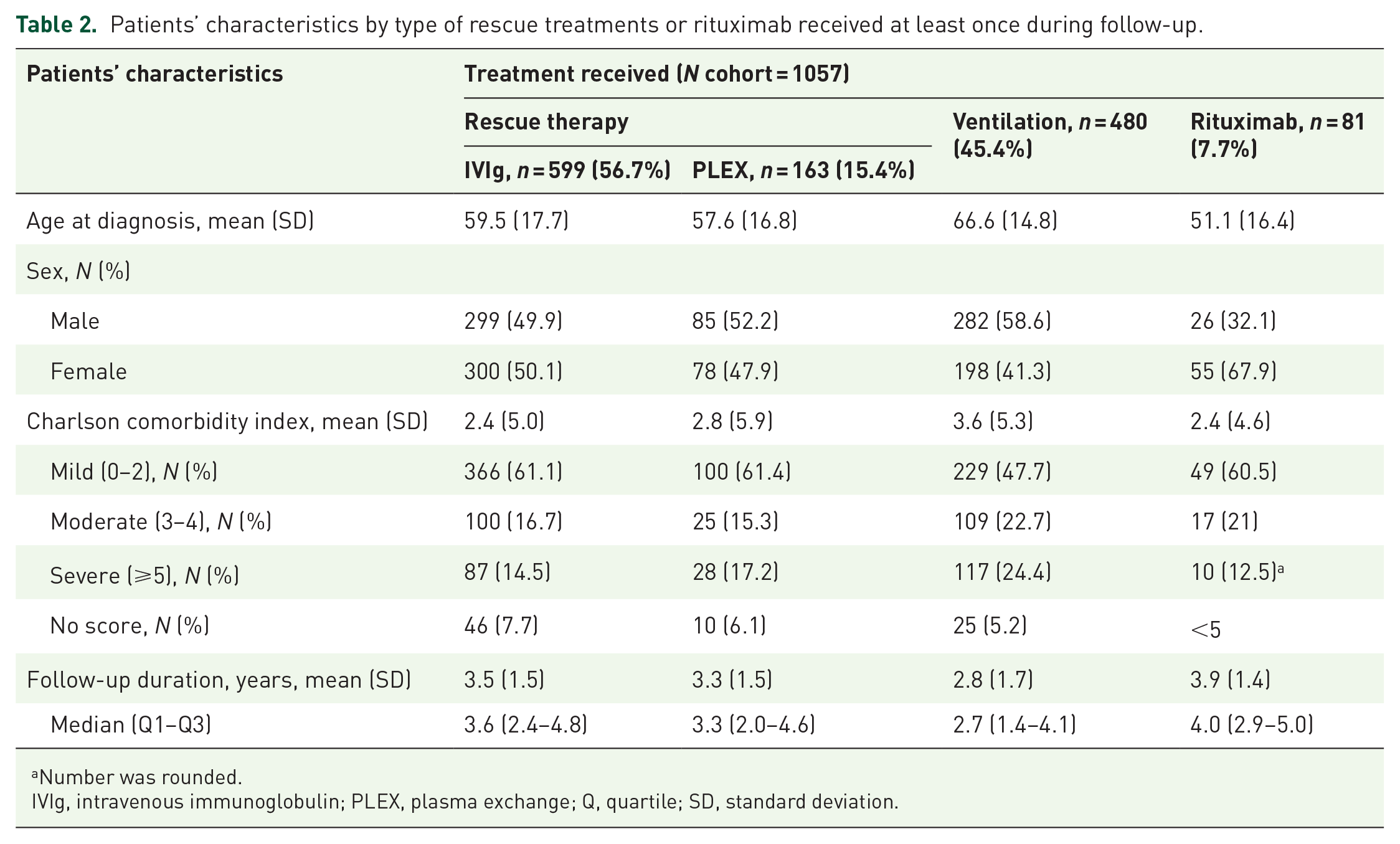
During the full follow-up period, 599 (6.6% of the total cohort) and 163 (1.8%) patients were prescribed IVIg and PLEX, respectively (Table 2). Rituximab was administered to 81 (0.9%) patients, and 480 (5.3%) patients were ventilated. In those treated, the mean number of treatment cycles per patient was 2.3 for IVIg, 2.0 for PLEX, 1.7 for rituximab, and 1.3 for ventilation during the follow-up period (Table 3). The majority of patients receiving PLEX required it as an acute short-term treatment (American Society for Apheresis category I; Table 3). In total, 2.9% (n = 268) of patients underwent thymectomy. For all calendar years between 2015 and 2020, IVIg was the most frequently prescribed treatment (Supplemental Figure 1). Although not tested statistically, we observed a decreasing proportion of patients received IVIg treatment by calendar year; 62.8% of all patients received high-cost treatment and procedures in 2015 versus 43.5% of those in 2020, and similarly for thymectomy; 25.5% of all patients received high-cost treatment and procedures in 2015 versus 12.6% of those in 2020 (Supplemental Figure 1). Few patients received both IVIg and PLEX (n = 70) during follow-up; rituximab and PLEX (n = 25); or IVIg, PLEX, and rituximab (n = 16); however, a considerable subset of those who did receive ⩾3 treatments (32/70, 16/25, and 14/16 patients, respectively) (Table 3).
Patients’ characteristics by type of rescue treatments or rituximab received at least once during follow-up.
Number was rounded.
IVIg, intravenous immunoglobulin; PLEX, plasma exchange; Q, quartile; SD, standard deviation.
Treatments administered in secondary care during follow-up in patients with MG who had a treatment record in HES.

With ⩾1 administration of specified treatments.
85.9% of patients receiving PLEX had 1 (67.5%), 2 (12.3%), or 3 (6.1%) records of PLEX during the entire follow-up period.
Number was rounded.
HES, hospital episode statistics; IVIg, intravenous immunoglobulin; MG, myasthenia gravis; PLEX, plasma exchange.
Equal proportions of males and females received IVIg and PLEX, while approximately twice as many females received rituximab (n = 55 versus n = 26 for males; Table 2) or thymectomy (n = 174 versus 94 for males). IVIg and PLEX treatments occurred in equal proportions across age groups (at treatment time), whereas 12% of all treated patients aged <50 years received rituximab and 5% of those ⩾50 years. Equally, thymectomy occurred more at a younger age (45% of all treated patients aged <50 years versus 15% of those aged ⩾50 years; Supplemental Table 3).
Healthcare resource utilization
The proportion of patients with ⩾1 attendance at 6 months was 85.6% for outpatient visits, 42.5% for inpatient admissions, 4.2% for critical care admissions, and 26.8% for A&E attendances; and for all follow-up was 92.4% outpatient, 72.8% inpatient, 9.7% critical care, and 51% A&E (Table 4).
HCRU and associated costs in MG patients who had HCRU record, during follow-up period.

MG-related inpatient admissions were defined as admissions with a recorded diagnosis of MG.
A&E, accident and emergency; HCRU, healthcare resource use; LOS, length of stay; MG, myasthenia gravis; Q, quartile; SD, standard deviation.
In those with ⩾1 attendance, the mean ± SD number of attendances for all patients’ follow-up was: 7.1 ± 10.5 outpatient visits per patient-year, 1.3 ± 4.0 inpatient admissions per patient-year, 0.4 ± 1.8 critical care admissions, and 0.9 ± 2.7 A&E attendances (Table 4). Mean ± SD of HCRU in those attending hospital was highest at 6 months follow-up and decreased afterward: at 6 months, there were on average 12.0 ± 13.7 outpatient visits (9.9 at 12 months and 8.4 at 24 months follow-up), 4.5 ± 5.9 inpatient admissions (2.8 at 12 months and 1.8 at 24 months follow-up), 3.6 ± 4.3 A&E attendances per patient-year (2.3 at 12 months and 1.5 at 24 months follow-up), and 2.3 ± 2.2 critical care admissions (Table 4).
The distribution of outpatient visits during the follow-up period is shown in Supplemental Table 4. Neurology service (a mean of 6.6 visits per patient) and ophthalmology service (a mean of 5.7 visits per patient) were the most frequently visited outpatient departments in the all-follow-up period in patients with ⩾1 attendance. Myasthenia, gastroenterological, and cardiovascular complications were the most common primary diagnoses in inpatient admissions for patients with MG (Supplemental Table 5).
Patients hospitalized for MG-related complications stayed in the hospital for a mean ± SD of 9.7 ± 29.2 days per patient-year (Table 4) and the most common related complications were myopathy and neuropathy (Supplemental Table 5).
The mean number of A&E attendances, inpatient admissions, or outpatient visits did not differ considerably by sex or age group (Table 5)—.
HCRU in patients diagnosed with MG by sex and age.

A&E, accident and emergency; HCRU, healthcare resource use; MG, myasthenia gravis; Q, quartile; SD, standard deviation.
Costs
The mean ± SD cost of inpatient hospitalization for patients who incurred HCRU was £3226 ± 13,142 per patient-year in all follow-up periods, with the lowest cost incurred by A&E attendance (£140 ± 475 per patient-year; Table 4). The mean ± SD cost per patient-year associated with the treatment administration based on the HRG recorded was highest for PLEX (£4233 ± 8928; Table 6). The cumulative costs of admissions for patients treated with IVIg (n = 599), PLEX (n = 163), and rituximab (n = 81) were £907,072, £689,979, and £146,726, respectively. In patients with a history of rituximab treatment, the mean cost related to all HCRU per patient-year for IVIg, ventilation, and PLEX was substantially higher than in those without a history of rituximab treatment (Supplemental Table 3).
Costs of selected treatment among patients with MG.

Patients with ⩾1 administration of specified treatments.
The reported treatment cost is related to all healthcare resources used during an inpatient spell or outpatient visit for a specific treatment, with only the relevant high-cost drug treatments or procedure being included, as administration of other treatments (e.g. AChEIs, corticosteroids, or other first-line non-steroidal immunosuppressants) is not recorded in HES and presented as cost per patient-year.
Cost of thymectomy presented as per patient-year.
AchEIs, acetylcholinesterase inhibitors; HES, hospital episode statistics; IVIg, intravenous immunoglobulin; MG, myasthenia gravis; PLEX, plasma exchange; Q, quartile; SD, standard deviation.
Discussion
We performed a retrospective analysis of treatment patterns and HCRU for a large cohort of patients with MG diagnosed in secondary care settings in England, using HES data collected between 2014 and 2021. We found that myasthenic crisis and disease exacerbation in MG incur high HCRU and associated costs, with a subset of patients requiring rescue therapy beyond first-line medication.
This study found an estimated annual incidence for MG of 4.32 per 100,000 population. Limitations exist regarding the precision of this estimate due to the absence of direct denominator data in secondary healthcare settings. Notably, certain MG patients, particularly those with ocular or mild MG, may never require hospitalization, potentially leading to a skew toward patients with more severe MG. The lack of mandatory recording of outpatient diagnosis in HES may have led to an underestimate of the true incidence of MG and an over-representation of recorded inpatient MG diagnosis. Our findings align with a recent German study using anonymized insurance claims data, which reported an MG incidence of 4.6 cases per 100,000 individuals between 2015 and 2019. 19 A previous UK study based on primary care data, however, reported a lower incidence rate ratio of 2.46 (CI: 2.34–2.59) per 100,000 person-years, 3 though imprecision may arise from the absence of validated diagnoses in primary care records.
MG diagnoses were primarily documented in neurology or general medicine departments. However, a significant proportion of patients had previously visited neurology and ophthalmology outpatient departments, particularly within 3 months before MG diagnosis (index date). These outpatient visits likely aimed at obtaining a diagnosis, as they occurred in the period leading up to the confirmation of MG.
Among the observed cohort, a subset (11.6%, n = 1057) required additional treatment and procedures, including rescue therapies for myasthenic exacerbation or crisis, as well as rituximab. Only 2.9% (n = 268) of patients were recorded to have undergone thymectomy, similar to 2.6% of MG patients in a previous study of MG patients in England. 9 In a recent study in Germany, 19 29.9% of incident patients experienced exacerbations, and 6.7% experienced myasthenic crises during the follow-up period (mean ± SD: 846.5 ± 528.5 days), most occurring within the first year after diagnosis. The high reported rate of exacerbations and crises in the German study may be attributed to the inclusion criteria, which required patients to have received at least one inpatient and/or two confirmed outpatient diagnoses of MG in the period, or to differences in the healthcare system. In our study, 6.6% of patients received IVIg, 1.8% received PLEX, and 0.9% received rituximab treatment, consistent with a recent real-world HCRU study in the United States. 20 Among the small subset of patients who received multiple therapies during follow-up (i.e. IVIg, PLEX, and rituximab), a significant proportion (87.5%; 14 out of 16) received ⩾3 treatments. These patients likely experience frequent and significant relapses, have an ‘explosive’ disease onset, are unresponsive to conventional rescue treatments, or have inadequate response or intolerance to other immunosuppressive drugs.
In our study, the administration of IVIg was more frequent than PLEX, which is consistent with clinical practice trends. 4 IVIg is often preferred due to its availability and lower risk of adverse events, such as hypotension, coagulopathy, and sepsis, compared to PLEX.21,22 The 599 patients with recorded IVIg use during all follow-ups appear underreported as the National Immunoglobulin Database Report 23 estimates 666 MG patients annually receive IVIg [or 189,534 g, the equivalent of £13 m (based on an average price of £70/g) 24 ]. However, despite its preferred use over PLEX, the use of IVIg has its limitations: it is a human blood product that may be subject to supply issues and there is a risk of passing infections. 25 We observed a slight decrease in the rates of IVIg treatment between 2015 and 2020, which may be due to the introduction of rituximab as an alternative to IVIg for the treatment of refractory MG in the UK in 2018 26 as well as stricter regulation for IVIg treatment. Previous studies showed that rituximab may be used in new-onset MG, MuSK-MG patients, and early treatment initiation.22,27 The higher utilization of rituximab in female patients (11.2%) compared to male patients (4.6%) observed here aligns with the higher prevalence of MuSK-MG in females. 22
The observed decline in thymectomy rates (from 24.5% of patients treated in 2015 to 12.6% in 2020) could be due to reluctance from clinicians to undertake the procedure due to relatively weak evidence of effectiveness; the practice guideline by the Association of British Neurologists published in 2018 6 states thymectomy may induce remission, may prevent generalization of ocular myasthenia, and may reduce corticosteroid requirements. Similar to our study, a recent German study 19 reported a low rate of thymectomy (4.4% of the total MG incident cohort), which the authors interpreted as likely being caused by the refusal of procedures due to older age and the presence of comorbidities. Notably, the rate of thymectomies in the early-onset MG subgroup was higher at 13.3%. 19
A majority of patients were admitted during follow-up (72.8% inpatient admissions and 51.0% A&E attendances), indicating that patients with MG incur significant HCRU. A minority of these attendances are directly linked to rescue therapy treatment. Due to the complexity of MG pathology, a new diagnosis of MG may prompt intervention to address existing symptoms, partially accounting for this healthcare burden. Moreover, a high proportion of patients presenting at the A&E department may suggest the occurrence of acute comorbidities related to MG treatment (e.g. infections and hyperglycemia due to steroids/immunosuppressants) or exacerbations and crises following diagnosis. In addition, due to the lack of outpatient diagnosis recording and the fact that over 80% of patients were included via an inpatient index diagnosis, our study is likely to have an over-representation of severe MG patients; therefore, these findings may not be generalizable to the entire population of MG patients. However, the rate of MG-related hospitalization in our study (1.3 ± 4.0 inpatient admissions per patient-year) aligns with a previous UK study using linked data from the Clinical Practice Research Datalink and HES. 28 The costs incurred by inpatient admissions may be driven by the length of MG-related hospital stays (mean: 9.7 days in the current study), but further investigation of specific cost drivers is required. The reported HCRU patterns, except for inpatient hospitalization, are also consistent with findings from a US study. 20 In those attending the hospital, mean HCRU was highest in the first 6 months after MG diagnosis and decreased during follow-up, in line with previous findings in England, where proportions of patients with myasthenic crises and exacerbations were highest in the first year after the index date, 28 and the United States.20,29
The mean cost of total secondary HCRU for MG patients in our study was £3820 per patient-year (equal to $4849 in 2023), which was lower compared to those reported in a US study. However, a broader definition in terms of healthcare settings was used, and US healthcare costs are generally higher. 20 Published data suggest that IVIg utilization is a major contributor to the total annual cost of MG20,30 and patients receiving chronic IVIg for MG experience a significant economic burden, 31 which aligns with our findings (though IVIg acquisition costs were not included in this study). Recent evidence suggests that PLEX is more cost-effective than IVIg in the treatment of autoimmune neurological conditions like MG, supporting the need for PLEX centers in regional neurology units, particularly considering the constrained supplies of IVIg. 25 However, with limited PLEX centers available and a burdensome treatment administration over several days, a preference to use IVIg may remain. The cumulative cost of admission for IVIg and PLEX was higher compared to rituximab alone, highlighting the unmet need in this subpopulation. It is worth noting that the variability of costs observed was very high, suggesting a small number of extremely high-cost patients. We found that patients with a history of rituximab treatment had higher costs for IVIg, ventilation, and PLEX compared to those without prior rituximab treatment.
A notable strength of this study is the inclusion of a large cohort of MG patients. In addition, the HES data encompass all newly diagnosed MG patients treated within the NHS in England. However, there are several limitations to our study. First, only high-cost drug treatments are recorded in HES, excluding data on non-high-cost MG drugs such as AChEIs, corticosteroids, and non-steroidal immunosuppressant drugs. Second, the current analysis is limited to secondary care settings, omitting information on MG diagnosis, treatment, and HCRU in primary care settings. It is possible that patients were initially diagnosed by their general practitioner, but their diagnosis was only recorded in HES during their first hospital visit, introducing a time lag that may affect estimates of time from diagnosis to end of follow-up. In real-world practice, outpatient visits, particularly in ophthalmology and neurology, often involve suspected MG cases, which may later lead to hospitalization for further investigations or treatment initiation upon confirmation. Consistently, our findings indicate that neurology and ophthalmology departments were the most common specialties visited prior to MG diagnosis. In addition, in certain hospitals, neurology falls under the umbrella of general medicine either as a directorate or due to neurologists practicing within a general internal medicine department. Consequently, reported neurology referrals and subsequent diagnoses may be underestimated. Third, the CCI and baseline comorbidities may have been underestimated as these measures include conditions that can be diagnosed in primary care settings. Fourth, MG patients exclusively treated in private hospitals were not captured in this study. However, since HES covers 98% of the population in England, 10 the number of missed MG patients with inpatient admissions is expected to be minimal. Lastly, HES does not capture clinical outcomes and the coding system for treatments and procedures primarily serves reimbursement purposes, potentially introducing bias in estimates for certain treatments (e.g. low number of patients undergoing thymectomy).
In conclusion, a majority of patients with MG required hospitalization or attending A&E departments, resulting in a significant burden in terms of HCRU and cost. A subset of patients required rescue therapy (including IVIg and PLEX), rituximab administration, ventilation, or thymectomy. Overall, these findings highlight the burden of MG on secondary care settings across England and may help policymakers manage costs and treatment decisions in MG.
Supplemental Material
sj-docx-1-tan-10.1177_17562864241237495 – Supplemental material for A retrospective observational study on characteristics, treatment patterns, and healthcare resource use of patients with myasthenia gravis in England
Supplemental material, sj-docx-1-tan-10.1177_17562864241237495 for A retrospective observational study on characteristics, treatment patterns, and healthcare resource use of patients with myasthenia gravis in England by Jordy van Enkhuizen, Jean Binns, April Betts, Fatemeh Saberi Hosnijeh, Myriam Alexander, Mark McCormack and Saiju Jacob in Therapeutic Advances in Neurological Disorders
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
