Abstract
Background:
Teriflunomide is a once-daily oral disease-modifying therapy (DMT) for the treatment of relapsing-remitting multiple sclerosis (RRMS). Only limited information is available about its real-world use and changes over time.
Objectives:
To collect real-world data on teriflunomide use in clinical routine (and comparison to the previously conducted study TAURUS-MS).
Design:
National, open, non-interventional, prospective, multicenter study.
Methods:
TAURUS-MS II was conducted at 220 German sites between July 2017 and March 2022, including RRMS patients treated with teriflunomide. Data on patient demographics, MS history, previous treatment, therapy satisfaction, and safety were collected.
Results:
In total, 752 patients were included (65% female) with a mean age (±standard deviation) of 43 ± 11 years. Sixty-six percent had DMT before, and 46% had discontinued their last pretreatment ≤6 months prior to study entry. Among the latter, previous DMTs were interferon (21%), glatiramer acetate (11%), and dimethyl fumarate (9%), and reasons for discontinuation were adverse events (AEs; 55%) and insufficient efficacy (16%). Over 24 months, the mean treatment Satisfaction Questionnaire for Medication scores improved by 6 ± 29 points on effectiveness, 8 ± 20 on convenience, and 12 ± 25 on global satisfaction. The mean number of MS relapses decreased from 0.81 ± 0.81 in the 24 months prior to 0.27 ± 0.57 within 24 months after study entry. Non-serious AEs occurred in 423 patients (56%) and serious AEs in 49 patients (7%). Most reported AEs were alanine aminotransferase increase (11%), hypertension (8%), and alopecia (7%). Compared to TAURUS-MS, patients in TAURUS-MS II were younger, had a higher employment rate, and a higher share of treatment-naïve patients.
Conclusion:
Mean number of relapses was significantly reduced. Patient satisfaction was significantly improved compared to previous DMT. Tolerability was comparable to previous trials.
Trial registration:
Bundesinstitut für Arzneimittel und Medizinprodukte public database for non-interventional studies, number 7138.
Introduction
Multiple sclerosis (MS) is an immune-mediated chronic inflammatory disease of the central nervous system and the most common neurological disease with an estimated number of 2.8 million affected people worldwide and up to 223,000 in Germany.1,2 It is characterized by demyelination and axonal damage, leading to permanent disability.3,4
Teriflunomide is an immunomodulatory and anti-inflammatory agent for the treatment of relapsing-remitting MS (RRMS). It selectively and reversibly inhibits the mitochondrial enzyme dihydroorotate dehydrogenase (DHODH), which functionally connects with the respiratory chain and is necessary for de novo pyrimidine synthesis. A consequence of DHODH inhibition is the blockade of highly proliferative cells such as activated lymphocytes.4,5
The efficacy of teriflunomide in MS treatment was shown in two placebo-controlled pivotal trials, TEMSO and TOWER, and was compared with interferon (IFN) beta-1a subcutan in the TENERE study.6–8 In summary, teriflunomide significantly reduced the number of MS relapses, retarded disability progression, and decreased the number of cerebral lesions seen in magnetic resonance imaging (MRI). 9 Teriflunomide was approved in the EU in 2013 for the treatment of adult RRMS patients and in 2021 for pediatric patients of 10 years and older.9,10 Since then, the armamentarium of MS disease-modifying therapies (DMTs) has grown, especially for those that are orally applicable. In addition to fingolimod, dimethyl fumarate (DMF), and teriflunomide, the three oral drugs approved for RRMS treatment at the study start, ozanimod, ponesimod, and cladribine have been approved as further oral treatment options in the meantime.4,11 Moreover, altered diagnostic criteria and growing experience with available DMTs have altered the treatment decision-making process.
Currently, real-world data on the effectiveness, safety, and patient satisfaction of teriflunomide use in daily clinical routines remain limited. This applies especially to the elderly population, leading to a restricted recommendation of teriflunomide use in patients aged ≥65 years by the European Medicines Agency (EMA). 9 Moreover, the treatment patterns may vary from country to country and may change over time.12–15 The previous study TAURUS-MS, performed relatively soon after the market entrance of teriflunomide, contributed to an improved understanding of its use outside of clinical trials in Germany. 16 TAURUS-MS II aimed to complement the existing data by using a similar study design, thus allowing a comparison of the use of teriflunomide at two periods of time (i.e. 2014–2017 in TAURUS-MS versus 2017–2022 in TAURUS-MS II). Objectives of TAURUS-MS II included analyses of the severity of MS relapses, changes in the Expanded Disability Status Scale (EDSS) score, impacts of patient age on therapy outcome, and patient satisfaction and quality of life under treatment with teriflunomide.
Methods
Study design
TAURUS-MS II (Basistherapie mit AUBAGIO® – eine multizentrische, nicht-interventionelle Studie in der Indikation schubförmig-remittierende Multiple Sklerose II) was a national, open, non-interventional, prospective, multicenter observational study conducted in Germany (AUBAGIO = teriflunomide). The study was conducted between 11 July 2017 (first patient in) and 30 March 2022 (last patient out).
Sites
In total, 220 private practices, neurological clinics, or outpatient clinics throughout Germany participated in this study.
Subjects
Patients were eligible for enrolment if they fulfilled the following inclusion criteria: age ≥18 years, diagnosis of RRMS, written and signed patient informed consent, capability of completing questionnaires, and absence of contraindications according to the prescribing information of teriflunomide. The therapy decision had to be independent of the participation in the study. Patient exclusion was considered in case of impediments such as motor impairments, visual impairments, or cognitive impairments. Further explicit exclusion criteria were not defined to avoid selection bias. Administration of 14 mg teriflunomide once daily occurred in accordance with the prescribing information. 9
Documentation
The baseline visit had to be within ±3 months of therapy start, and follow-up visits were scheduled 3, 6, 12, 18, and 24 months after treatment initiation with teriflunomide. Documented parameters included demographics (age, sex, occupational status), MS history (first symptoms and diagnosis of MS, MS disease type, relapses, current EDSS score, 17 symptoms), previous treatment, fatigue according to the Fatigue Severity Scale (FSS), 18 and adverse events (AEs). Therapy satisfaction was assessed from the physician’s perspective and the patient’s perspective regarding effectiveness, convenience, and global satisfaction using the Treatment Satisfaction Questionnaire for Medication (TSQM-9). 19 Questionnaires completed by patients included validated questions from the Short Form 36 (SF-36) Health Survey, TSQM-9, and FSS in German.18–20
Data collection and management
Data collection was performed using electronic case report forms and separate patient questionnaires. Data were validated according to rules previously defined in a data validation plan.
Statistical analysis
For sample size calculation, it was assumed that 80% of the documentation sheets of the 1080 included patients would be evaluable regarding relapse rates. Based on 864 evaluable patients, it was expected that the relapse rates could be estimated with a 95% confidence interval (CI) of width less than 12.3%. In a sample of 1080 patients with teriflunomide therapy, responder rates of binary endpoints could be estimated with 95% CIs of width less than 6.1%. Mean values of normally distributed endpoints could be estimated with 95% CIs whose width does not exceed 0.12 standard deviations (SD). In a sample of 1080 patients with teriflunomide therapy, AEs with an occurrence rate of ≥0.28% (corresponding to 1 patient out of 360) could be observed at least once with a certainty of ≥95%.
Analyses were performed in an exploratory manner using descriptive statistical methods. For continuous variables, the number of patients with missing data, mean, standard deviation, median, 25%, and 75% quantile, minimum and maximum were calculated. For ordinal and categorical variables, frequencies were calculated. Incomplete data sets were included in the analysis. There was no imputation of missing values for any endpoint. No sensitivity analyses were done. All effectiveness analyses were conducted on the modified intention to treat set (mITT) comprising all enrolled patients who signed the informed consent and received at least one dose of teriflunomide within 3 months before or after the baseline visit and with available follow-up data for efficacy. Clinical results were analyzed by visit. For the analysis of relapse rate, the Wilcoxon matched pairs signed-rank test was used because the number of relapses showed no normal distribution. To compare the mean change between the baseline and the 24-month visit, the Wilcoxon matched pairs signed-rank test was used for TSQM-9 and FSS questionnaires. Changes from the baseline were analyzed by repeated measurement analysis for time trends. Subgroup analyses were performed according to previous treatment and age.
The safety analysis set (SAF) was identical to the mITT set. All AEs occurring during this observational study were coded using the Medical Dictionary for Regulatory Activities (MedDRA), Version 25.1 (International Federation of Pharmaceutical Manufacturers and Associations on behalf of the International Conference on Harmonization). The incidence of AEs and adverse drug reactions by MedDRA System Organ Class and Preferred Term was calculated for the SAF set (number, frequency). Analyses were carried out with SAS statistical software, version 9.4 (TS1M3), generally available July 2015, SAS Institute, Cary, NC, USA.
Results
Patient disposition
Of 1003 enrolled patients, 752 were included in the analysis (mITT/SAF set). Of these patients, 239 had a premature study termination, and 513 completed the study regularly (Figure 1).

Study flow chart.
Demographics and MS history
Details on patients’ demographics and MS history are provided in Table 1. The mean age (±SD) was 42.85 ± 11.06 years (87.77% ≤55 years; 12.23% >55 years), 65.43% were female, and 34.57% were male. The majority of patients (50.27%) were regularly full-time employed.
Demographic data at baseline.

In relation to baseline visits.
Patients were only included in the analysis if a date was documented for the cranial MRI performed due to MS. Elapsed time was calculated as follows: a quarter of last cranial MRI − a quarter of baseline visit. Positive values indicate that the last cranial MRI was performed after the baseline visit.
EDSS, Expanded Disability Status Scale; MDD, major depression disorder; MRI, magnetic resonance imaging; MS, multiple sclerosis; SD, standard deviation.
The mean (±SD) time intervals since the first MS symptoms and since MS diagnosis were 9.67 ± 8.47 years (n = 691) and 7.98 ± 7.68 years (n = 726), respectively. The mean (±SD) EDSS score at baseline was 1.99 ± 1.50 (n = 625), with a range from 0.00 to 7.50. Most patients had an EDSS score of ≤3.5 (73.54%). During the last 24 months prior to baseline, the majority of patients had either no MS relapse (36.70%) or one relapse (42.95%).
Main MS associated or induced symptoms at baseline were fatigue (37.77% of total patients), depression (17.95%), bladder disorder (16.49%), cognitive deficits (16.22%), and spasticity (15.29%).
To control for possible selection bias, demographic data of patients in the mITT/SAF set in comparison to total enrolled patients are provided in Supplemental Table S1.
Treatment history
Details on previous treatment are provided in Table 2. For 32.98% of patients (n = 248), no pretreatment was documented. Among patients previously treated with another MS medication (65.82%; n = 495), the most frequently prescribed therapies were IFN (40.56% of total patients), glatiramer acetate SC (23.27%), and DMF (16.89%).
Previous treatment.

The reason for discontinuation of the last pretreatment was assessed among all patients who had discontinued their last pretreatment ≤6 months prior to study entry.
In cases where the order of pretreatments of a patient was not known due to missing dates, the reason for discontinuation of the last pretreatment could not be determined.
Other reasons for the discontinuation of the last pretreatment could be entered in a free-text field.
MS, multiple sclerosis.
Among patients who had discontinued their last pretreatment within 6 months prior to study entry (45.61%; n = 343), the most commonly reported last MS treatments were IFN (21.41% of total patients), glatiramer acetate SC (10.64%), and DMF (8.64%). The most reported reasons for discontinuation of the last pretreatment in these patients were AEs (55.10%), insufficient efficacy (15.74%), wish for therapy interruption (2.62%), and non-compliance (1.75%). A total of 52.79% did not receive any pretreatment within 6 months prior to study entry (unknown: 1.60%).
Treatment with teriflunomide
Treatment of patients with teriflunomide during the study is described in Table 3. The mean (±SD) observation period was 17.69 ± 9.03 months (537.75 ± 274.5 days; n = 708). Treatment continuation was confirmed for 82.18% of patients after 6 months, for 75.4% after 12 months, and 68.22% after 24 months. Among patients with a documented premature study termination (n = 239), the most reported reasons for discontinuation were AEs (33.89%), lack of efficacy (20.92%), loss to follow-up (10.46%), and wish for therapy interruption (10.04%).
Teriflunomide treatment.

The reason for discontinuation was assessed among patients with a documented premature study termination.
SD, standard deviation.
Effectiveness
MS relapses
In the total study population, the mean (±SD) number of MS relapses was 0.81 ± 0.81 within the last 24 months prior to study entry (n = 710), compared to 0.27 ± 0.57 within 24 months after study entry [n = 399; Figure 2(a)]. When considering patient age, the decline in mean relapses after switching to teriflunomide was similar in both age groups (≤55 years; >55 years), but mean numbers of MS relapses for both periods were lower in older patients [Figure 2(b)].
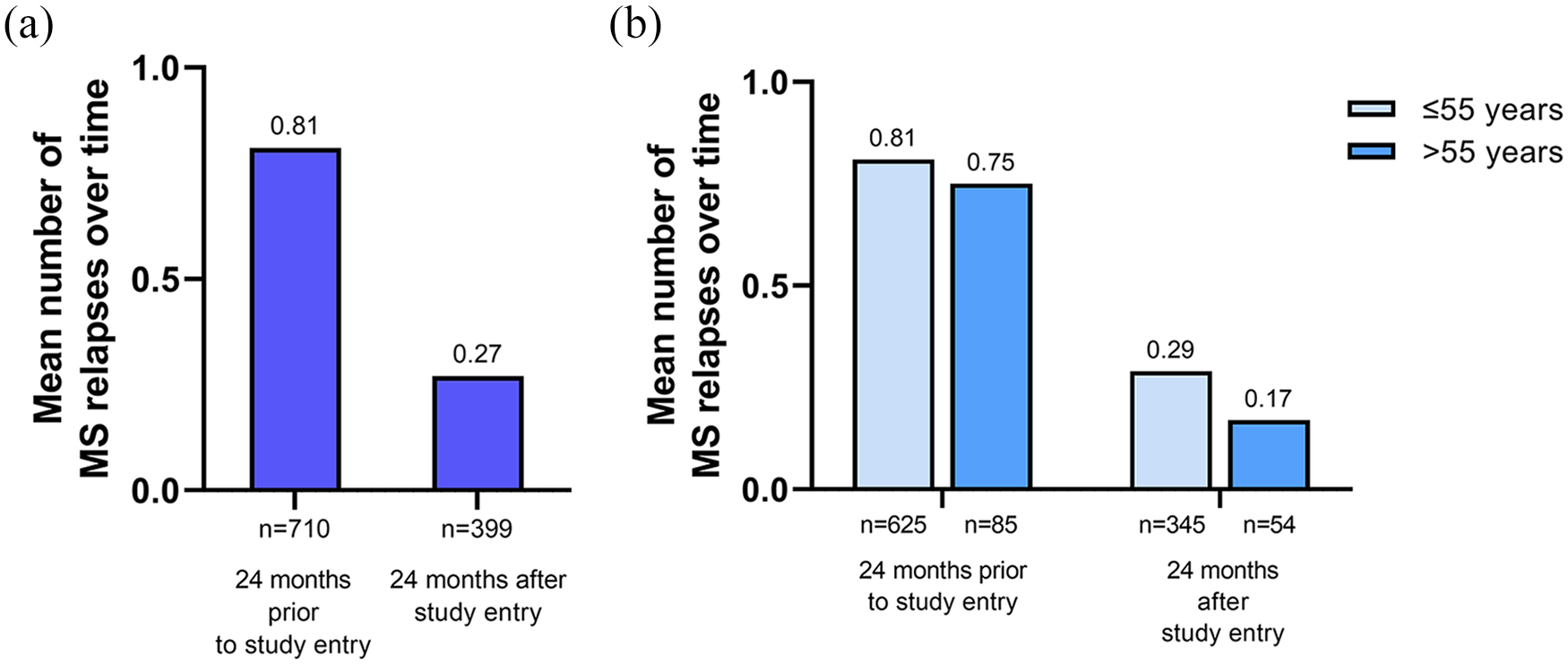
MS relapses. (a) The mean number of MS relapses over time in the total study population and (b) according to patient age. Mean values are indicated above the bars and numbers of patients below the bars, respectively.
The annualized relapse rate (ARR) – defined as the total number of relapses experienced divided by the total number of patient-years within the observational period – was calculated. ARRs for the total population were 0.20 (95% CI: 0.15–0.26; n = 667) for total MS relapses, 0.01 (95% CI: 0.00–0.04; n = 659) for relapses requiring a hospital stay, 0.16 (95% CI: 0.11–0.22; n = 659) for relapses requiring one cortisone pulse therapy, and 0.03 (95% CI: 0.02–0.06; n = 659) for relapses requiring two cortisone pulse therapies. When considering age groups, the ARR for total MS relapses was 0.23 (95% CI: 0.17–0.31; n = 583) for patients aged ≤55 years and 0.10 (95% CI: 0.05–0.20; n = 84) for patients aged >55 years.
EDSS
For patients with EDSS scores documented both at baseline and at the 12-month visit (n = 374), the mean (±SD) EDSS increased from 1.98 ± 1.49 (baseline) to 2.11 ± 1.53 (12 months). For the patients with documented EDSS scores at baseline and the 24-month visit (n = 305), the mean (±SD) EDSS increased from 1.96 ± 1.46 (baseline) to 2.12 ± 1.51 (24 months).
Patient-reported outcomes
Results of the TSQM-9 were evaluated among patients who had discontinued their last pretreatment within 6 months prior to study entry. All three domains of the TSQM-9 scale were improved to a statistically significant degree between study entry and the 24 months visit. In the total study population, the effectiveness scale, convenience scale, and global satisfaction scale improved by a mean (±SD) of 6.01 ± 28.93 (n = 104), 8.29 ± 20.40 (n = 116), and 12.08 ± 25.49 (n = 115), respectively [Figure 3(a)]. In the group of patients aged ≤55 years, the same scales improved by a mean (±SD) of 5.88 ± 28.43 (n = 93), 8.84 ± 20.62 (n = 105), and 12.01 ± 25.6 (n = 102), respectively. Among patients aged >55 years, mean (±SD) changes of the three scales were 7.07 ± 34.34 (n = 11), 3.03 ± 18.15 (n = 11), and 12.64 ± 25.61 (n = 13), respectively [Figure 3(b)]. All three scales were improved to a statistically significant degree in the total population and the group of younger participants, while no statistically significant differences were found in the group of patients aged >55 years, probably due to the low number of patients in this group.

TSQM-9. Mean changes in effectiveness, convenience, and global satisfaction scores from baseline to 24 months in patients who had discontinued their last pretreatment within 6 months prior to study entry, total (a) and according to patient age (b). Paired samples test. Mean values are indicated above the bars and numbers of patients below the bars, respectively.
Only slight changes in the FSS score were observed during treatment with teriflunomide. The mean (±SD) values at baseline (n = 513), 6 months (n = 439), 12 months (n = 386), and 24 months (n = 277) were 4.14 ± 1.81, 3.96 ± 1.80, 4.07 ± 1.73, and 3.98 ± 1.74, respectively. Between baseline and the 24-month visit, the mean (±SD) FSS score changed by 0.05 ± 1.70 points (n = 220; 95% CI: −0.18 to 0.28).
The question on patient satisfaction could be rated from 1 ‘very dissatisfied’ to 5 ‘very satisfied’ by the treating physician at each time point. After 12 and 24 months of treatment, the mean (±SD) score increased by 0.86 ± 1.36 (n = 184) and 0.92 ± 1.41 (n = 153) compared to baseline, respectively.
Safety
Among the 752 study participants, 423 (56.25%) reported a total of 1283 non-serious AEs. Most common AEs were alanine aminotransferase (ALT) increase (10.64% of patients), hypertension (7.58%), alopecia (7.31%), diarrhea (6.78%), and blood pressure increase (6.38%). No substantial differences could be determined between the age groups (≤/>55 years), also considering the small number of patients in the population >55 years. An overview of the incidence of non-serious AEs is provided in Table 4.
Incidence of non-serious adverse events by MedDRA preferred term (≥1.00% of total, patient based, multiple answers possible).

AE, adverse event; MedDRA, Medical Dictionary for Regulatory Activities.
Mean (±SD) levels of ALT in the total study population changed from 23.37 ± 16.93 U/l at baseline (n = 689) to 26.08 ± 20.75 U/l at 6 months (n = 552), 24.63 ± 18.99 U/l at 12 months (n = 474), and 23.17 ± 13.79 U/l at 24 months (n = 356). One hepatobiliary disorder occurred, which was a non-serious case of hyperbilirubinemia and a relation to teriflunomide treatment was not suspected.
A total of 87 SAEs were reported for 49 patients (6.52% of the study population). The most common SAEs were reported as MS relapse (1.99% of patients) and MS (0.4%). SAEs with an incidence of n ≥ 2 are provided in Table 5. A listing of all SAEs is provided in Supplemental Table S2.
Incidence of serious adverse events by MedDRA preferred term (n ≥ 2, patient based, multiple answers possible). a

All SAEs with n = 1 are found in Supplemental Table S2.
One patient died due to an MS relapse.
AE, adverse event; MedDRA, Medical Dictionary for Regulatory Activities; MS, multiple sclerosis.
Three patients died during the study. One patient died due to a myocardial infarction with nausea and arthralgia, and one patient died a natural death. These AEs were all classified as being unlisted according to the company core safety information for teriflunomide and without a causal relationship to teriflunomide treatment. A third patient died due to an MS relapse, which was classified as being listed and with a suspected causal relation to teriflunomide treatment. For the third patient, ‘lack of efficacy’ was documented as the reason for discontinuation instead of ‘death’.
Five cases of neoplasms occurred in four patients: Acoustic neuroma, inflammatory carcinoma of the breast, prostate cancer, renal cancer, and schwannoma (n = 1 each). Two pregnancies occurred during the observation period, in one of which an abortion was conducted.
Discussion
TAURUS-MS II (conducted between 11 July 2017 and 30 March 2022) was a multicenter, prospective, non-interventional study, which aimed to collect real-world data on the use of teriflunomide in daily clinical practice. It observed patients with RRMS over a 24-month period.
In comparison to the previously conducted study TAURUS-MS, where treatment conditions were equivalent, patients included in TAURUS-MS II were younger (mean age 42.85 versus 44.9 years in TAURUS-MS), and had a shorter disease duration since first MS symptoms (9.67 versus 10.6 years in TAURUS-MS). 16 In TAURUS-MS II, a distinctly smaller portion of patients was unemployed (26.46% versus 40.0% in TAURUS-MS). 16 The mean EDSS score at baseline was lower in participants of TAURUS-MS II (1.99 versus 2.3 in TAURUS-MS). 16 Compared to the prior study TAURUS-MS, the proportion of patients with an EDSS ≤3.5 was higher (73.54% versus 70.4% in TAURUS-MS), as was the proportion of patients with either no MS relapse or one relapse in the 24 months prior to study entry (79.65% versus 76.6% in TAURUS-MS). In TAURUS-MS II, 32.98% of participants were treatment-naïve, compared to 24.8% in TAURUS-MS. 16
Comparing the treatment history of patients included in TAURUS-MS and TAURUS-MS II, a change in the clinical routine of MS therapy can be deduced: In TAURUS-MS, 75.2% of all patients had received DMTs before, with different types of IFN (IFNβ-1a SC: 23.8%; IFNβ-1a IM: 22.8%, IFNβ-1b SC: 19.7%) and glatiramer acetate SC (26.9%) being the most common pretreatments. 16 In summary, up to 66.3% of total patients were treated with IFNs, even though the actual percentage might be slightly lower due to potential switching within the IFN DMTs. Among patients included in TAURUS-MS II, 65.82% had received previous MS therapies, mostly IFNs in general (40.56% of patients) or glatiramer acetate SC (23.27%). Hence, comparing TAURUS-MS and TAURUS-MS II, we see a steep decline with the IFNs, but only a modest decline with the comparable glatiramer acetate SC.
Azathioprine oral and immunoglobulin IV have disappeared almost completely from the market (4.5% and 1.2% of total patients in TAURUS-MS, respectively, versus not explicitly listed in TAURUS-MS II). 16
Among patients in TAURUS-MS, DMF was rarely reported as a prior medication (2.48%), 21 whereas it was the third most frequently reported pretreatment in TAURUS-MS II (16.89% of total patients). This reflects the increased share of patients on DMF in recent years.
Previous medications in efficacy categories 2 and 3 were rarely documented for patients included in TAURUS-MS (2.84% fingolimod, 3.10% natalizumab). 21 Interestingly, looking at the combined number of therapies in efficacy categories 2 and 3 in TAURUS-MS II (daclizumab, fingolimod, natalizumab), we observe a total share of 9.84%. The fact that teriflunomide, a medication in efficacy category 1 of 3, according to the German guideline for MS diagnosis and therapy, 22 was subsequently prescribed in these patients, can be considered a treatment de-escalation, but it could also reflect that the growing experience with teriflunomide led to its consideration as an appropriate following therapy after high efficacy treatments.
When considering only the last medication of patients who had discontinued their pretreatment within 6 months, changes in the distribution of pretreatments between TAURUS-MS II and TAURUS-MS are modest, especially for IFN (21.41% versus 22.5% in TAURUS-MS) and glatiramer acetate SC (10.64% versus 10.6% in TAURUS-MS), but also reflect the availability and establishment of new treatment options such as DMF, while also an increase in the share of patients without pretreatment within the last 6 month was observed (52.79% versus 44.7% in TAURUS-MS). 16
Regarding effectiveness, the adjusted ARRs in the teriflunomide 14 mg cohorts of the placebo-controlled registration studies TEMSO and TOWER were 0.37 and 0.32, respectively, while the mean ARR reported in TAURUS-MS was 0.24.6,7,16 In TAURUS-MS II, a lower ARR of 0.20 was observed for the total population.
When considering a 24-month period, the mean number of MS relapses during treatment with teriflunomide in TAURUS-MS II was 0.27, compared to 0.81 prior to study entry. Even though the values for both periods were numerically higher in TAURUS-MS, with 0.35 after and 0.87 before the study entry, the reduction in relapse numbers was similarly evident. 16 The observation that older patients (>55 years) had less relapses, both before and after switching to teriflunomide treatment, is supported by the general observation that MS typically shows its highest activity in young adulthood. 23 A similar trend was observed in age-stratified analyses of data from TEMSO and TAURUS-MS, even though age cohorts were classified slightly differently.24,25 Under teriflunomide treatment, the mean number of MS relapses was reduced similarly in younger as well as older patients (≤/>55 years) compared to the period before study entry, arguing for an effective application possibility independently of patient age.
These outcomes are supported by the observation that both EDSS score and FSS remained stable over the observation period, which is consistent with the disease stabilization that has been reported in the TEMSO and TOWER core- and extension trials.26,27
In recent years, the inclusion of the patient perspective and engagement of patients in their healthcare gained more importance in the treatment of MS and other diseases.28,29 Especially for MS, patient engagement is regarded as a substantial premise for optimizing treatment benefits. 29 The TSQM scale has been applied before to assess factors influencing therapy adherence and satisfaction of MS patients. 30 In patients who had discontinued their last pretreatment within 6 months prior to study entry, all three domains of the TSQM-9, effectiveness, convenience, and global satisfaction, were improved between baseline and the 24-month visit (+6.01, +8.29, and +12.08, respectively), indicating an increased satisfaction under teriflunomide treatment. Results of TAURUS-MS were similar, but the improvement of scores was even higher (+8.1, +17.0, and +15.3 for the corresponding patient subgroup in the same time interval). 16 However, the TSQM scale is not specifically validated for application in MS patients, which hinders an assessment of the actual relevance of the observed changes. 31
Considering age groups, the mean improvement of the effectiveness and global satisfaction scale was more distinct in older compared to younger patients, while the change in the convenience scale was smaller. The distinctly smaller size of the group of patients >55 years may be a limitation for a direct comparison here. However, the observation of positive changes for all three scales of the TSQM-9 in both age cohorts substantiates the notion of teriflunomide being effective independently of patient age. When looking at the individual pre-treatments, the improvements in the TSQM domains failed to reach statistical significance, possibly due to the small number of patients in the various groups (data not shown).
The TSQM-9 results were further confirmed by the assessment of patient satisfaction according to the perspective of the treating physician, which also showed an improved perception during treatment with teriflunomide.
The overall superior perception of teriflunomide in comparison to previous treatments can possibly originate from the increased convenience of oral versus injection or infusion therapy and once-daily versus more frequent dosing.32,33
Our findings confirm other observations and contribute to a mounting body of evidence on teriflunomide usage in the real world. Nevertheless, about a third of patients prematurely discontinued the study. Among these, the most reported reasons for discontinuation were AEs and lack of efficacy. This is in line with the observations from TAURUS-MS, where a discontinuation was documented for one in five patients with the same main reasons and comparable with 60% treatment persistence on teriflunomide in a large register study conducted in France. 34 Discontinuation rates in that range are a common finding in clinical research on MS, according to a meta-analysis on both randomized controlled trials and observational studies on MS DMTs, which found overall discontinuation rates of 17–36% for different DMTs. In line with our findings, AEs and perceived lack of efficacy were the most reported reasons for discontinuation across DMTs. 35 According to a systematic review of patients’ understanding and preferences of DMTs, patients tend to misjudge the risks and benefits of MS therapies, which affects their treatment choice. 36 Especially AEs are a common cause for a shift in patient’s perception of a medication, in such a way that some patients no longer want to be treated. 37
Teriflunomide was generally well tolerated. Non-serious AEs occurred in 423 patients (56.25%), and SAEs were reported for 49 patients (6.52%). Among all study participants, 10.77% discontinued the study due to AEs (n = 81).
According to pooled core safety data of teriflunomide, the most frequent adverse reactions were headache (16% with teriflunomide 14 mg), ALT increased (15%), diarrhea (14%), alopecia (13%), and nausea (11%). The overall discontinuation rate due to AEs was 12.5% in pooled clinical trials. 38 In TAURUS-MS II, no new safety signals occurred, with the most frequently reported AEs being ALT increase (10.64% of patients), hypertension (7.58%), alopecia (7.31%), diarrhea (6.78%), and blood pressure increase (6.38%). The initially increased mean ALT levels (from baseline to month 6) normalized in the course of the study.
In general, teriflunomide was similarly well tolerated in patients below and above 55 years of age. This is supported by recent results of a chart review study that reported a stable safety profile of teriflunomide in patients aged 55 and older. 39
Currently, the EMA advises physicians to use teriflunomide carefully in patients aged ≥65 years due to insufficient availability of data on efficacy and safety. 9 At the same time, the prevalence of MS in older patients is rising, necessitating new and improved treatment recommendations. 40 Thus, TAURUS-MS II adds relevant information regarding the effectiveness of teriflunomide in older patients.
A few limitations should be considered for the interpretation of the study results. The observational character of TAURUS-MS II can potentially lead to unquantifiable bias in the selection of participants. Even though inclusion and exclusion criteria were restricted to a minimum, different types of selection bias, information bias, and confounding cannot be completely eliminated in observational studies. 41 Besides, a connection between treatment adherence and the willingness of patients to participate in clinical trials is known and may lead to a distorted representation of the real-world situation in clinical studies. 42
Comparisons across studies are often complicated due to diverging study designs, whereas the similar design of TAURUS-MS and TAURUS-MS II allows a more reliable comparison. Other strengths of TAURUS-MS II include the distribution of patients across Germany as well as the focus on office-based physicians instead of hospitals or universities (data not shown).
Conclusion
The non-interventional study TAURUS-MS II demonstrates the effectiveness and safety of teriflunomide in the treatment of RRMS over an observational period of 24 months. The mean number of relapses decreased after treatment started, compared to the time before study entry, and patient satisfaction was improved in comparison to pretreatment according to the three TSQM-9 scales. In line with the findings of TAURUS-MS, patients below and above the age of 55 years benefited from the treatment with teriflunomide. The safety profile of teriflunomide was favorable and consistent with previous studies. Comparing the patient population of TAURUS-MS II with that of TAURUS-MS, a shift to younger patients with a higher employment rate and a higher share of treatment-naïve patients has been observed, suggesting that growing experience among neurologists led to broader use of teriflunomide over time.
Supplemental Material
sj-docx-1-tan-10.1177_17562864241252722 – Supplemental material for TAURUS-MS II: real-world use of teriflunomide in Germany and changes in treatment patterns over time
Supplemental material, sj-docx-1-tan-10.1177_17562864241252722 for TAURUS-MS II: real-world use of teriflunomide in Germany and changes in treatment patterns over time by Boris-Alexander Kallmann, Georg zu Eulenburg, Jennifer S. Kullmann and Mathias Mäurer in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-2-tan-10.1177_17562864241252722 – Supplemental material for TAURUS-MS II: real-world use of teriflunomide in Germany and changes in treatment patterns over time
Supplemental material, sj-docx-2-tan-10.1177_17562864241252722 for TAURUS-MS II: real-world use of teriflunomide in Germany and changes in treatment patterns over time by Boris-Alexander Kallmann, Georg zu Eulenburg, Jennifer S. Kullmann and Mathias Mäurer in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-pdf-3-tan-10.1177_17562864241252722 – Supplemental material for TAURUS-MS II: real-world use of teriflunomide in Germany and changes in treatment patterns over time
Supplemental material, sj-pdf-3-tan-10.1177_17562864241252722 for TAURUS-MS II: real-world use of teriflunomide in Germany and changes in treatment patterns over time by Boris-Alexander Kallmann, Georg zu Eulenburg, Jennifer S. Kullmann and Mathias Mäurer in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The authors and Sanofi would like to thank the patients for their participation in the trial, as well as the TAURUS-MS II study team. Medical writing assistance was provided by Madeleine C. Killer, MSc, from Alcedis GmbH, Giessen, Germany, which was funded by Sanofi-Aventis Deutschland GmbH, Frankfurt am Main, Germany. Statistical analyses were done by Alcedis GmbH, Giessen, Germany. This manuscript was reviewed by Robin White, PhD, Jennifer Fine, MD, and Allen Amedume, PhD (Sanofi). All authors provided interpretation of results. All authors reviewed and approved the final version of the manuscript. Submission of the manuscript was done by Madeleine C. Killer, which was authorized by all authors and funded by Sanofi-Aventis Deutschland GmbH.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
