Abstract
Background:
Switching disease-modifying therapies (DMTs) is common in relapsing-remitting multiple sclerosis (RRMS). Vertical switching to higher-efficacy agents generally outperforms horizontal switching within the same efficacy tier, yet horizontal switches remain frequent where escalation is impractical.
Objectives:
To compare real-world outcomes after horizontal versus vertical DMT switches and to identify predictors of successful horizontal switching.
Design:
Retrospective, registry-based observational study.
Methods:
Adults with RRMS who switched DMTs in the MSBase Registry (2010–2023) were analyzed. Horizontal switches were defined as transitions within efficacy tiers, and vertical switches as transitions to a higher tier. Propensity score matching (1:1) generated balanced cohorts. Multivariable mixed-effects models with a random intercept for patients were used to evaluate associations with outcomes. The primary outcome was no evidence of disease activity (NEDA-3) during the treatment period; secondary outcomes included annualized relapse rate (ARR), Expanded Disability Status Scale (EDSS) change, confirmed disability worsening (CDW), confirmed disability improvement (CDI), and progression independent of relapse activity (PIRA). Predictors of successful horizontal switching were explored using logistic regression.
Results:
A total of 4934 matched switches (2467 pairs) were analyzed. Vertical switching achieved higher NEDA-3 rates than horizontal switching (45.8% vs 33.7%) and was associated with lower ARR, reduced CDW risk, and more frequent CDI; differences in EDSS progression and PIRA were not significant. Among horizontal switchers, 33.7% achieved NEDA-3. Success was associated with lower baseline EDSS, fewer prior relapses, and later-line switching. Outcomes varied by destination therapy: anti-CD20 agents had the highest success (≈50%), followed by cladribine (≈43%) and natalizumab (≈41%), whereas interferon and glatiramer acetate performed the poorest. Switches toward anti-CD20 therapies generally yielded better outcomes than other within-tier changes.
Conclusion:
Vertical switching should be preferred when treatment modification is required, particularly for patients with active disease. However, a subset of patients can achieve disease stability after horizontal switching, especially those with lower disability and fewer prior relapses. The dynamics of horizontal switching may further influence outcomes, warranting prospective validation.
Plain language summary
For people living with multiple sclerosis (MS), treatment changes are often necessary due to inadequate response, safety concerns, or personal preferences. These treatment switches can follow different strategies. One option is a “vertical switch,” where patients move to a treatment considered more effective. Another option is a “horizontal switch,” where patients change to a treatment of similar effectiveness but with a different mechanism or formulation. This study analyzed nearly 4,000 patients with relapsing MS from the global MSBase registry who underwent treatment switches. We aimed to better understand the effectiveness of horizontal switching and to identify patient characteristics associated with favorable outcomes. Our findings show that approximately one-third of patients who switched laterally (horizontally) achieved no evidence of disease activity (NEDA-3), meaning they experienced no relapses, disability worsening, or new disease activity on MRI during follow-up. Patients more likely to benefit from a horizontal switch were younger, had lower disability levels, fewer previous relapses, and had been on their prior treatment for longer. When directly compared, vertical switching was associated with superior clinical outcomes, including lower relapse rates and a higher likelihood of achieving NEDA-3. However, horizontal switching still proved to be a reasonable strategy for selected patients with lower disease activity and stable prior treatment exposure. These results can help guide treatment discussions between neurologists and patients, particularly in situations where vertical switching is not possible, preferred, or indicated.
Keywords
Introduction
Multiple sclerosis (MS) is a chronic autoimmune disorder characterized by inflammatory demyelination in the central nervous system, leading to cumulative neurological disability. Disease-modifying therapies (DMTs) are central to MS management, aiming to reduce relapses, prevent disability progression, and achieve disease remission.1,2
Treatment strategies in MS commonly follow two broad approaches: escalation therapy, where treatment intensity increases in response to disease activity, and early intensive therapy, which involves the upfront use of high-efficacy DMT.3–5 In parallel, immune reconstitution therapies (IRTs) have gained attention as a distinct treatment paradigm. 6 Regardless of strategy, DMTs are classified into efficacy tiers based on their impact on clinical and radiological outcomes. 7
While the suppression of inflammatory disease activity is a primary treatment goal, decisions to switch DMTs are often influenced by additional factors such as safety concerns, tolerability, monitoring burden, or access constraints. 8 In such situations, clinicians may opt for a horizontal switch, defined as a transition between DMTs of similar efficacy, rather than a vertical switch to a more potent therapy. Although real-world studies have shown that horizontal switches can maintain disease stability in selected patients,9,10 the clinical profiles of those who benefit most from this strategy remain poorly defined.
In this retrospective cohort study using the international MSBase Registry, we assess the real-world effectiveness of horizontal versus vertical switching among relapsing-remitting MS (RRMS) patients. While it is well established that vertical switches tend to yield superior disease control, our primary objective is to identify baseline factors associated with favorable outcomes following a horizontal switch. This may help refine treatment decisions in scenarios where vertical escalation is not feasible or is deferred due to patient preference, safety, or system-level considerations.
Methods
Study design and data source
This retrospective observational study used data from the MSBase Registry, a global, longitudinal database that captures real-world treatment outcomes in patients with MS. 11 The dataset included individuals with RRMS who underwent a disease-modifying therapy (DMT) switch between January 2010 and December 2023.
Patients were classified based on their treatment change into one of two groups:
Horizontal switchers: transitions between DMTs of comparable efficacy.
Vertical switchers: transitions from a lower-efficacy to a higher-efficacy DMT.
To operationalize DMT efficacy levels, we adopted a predefined three-tier framework reflecting real-world effectiveness and relapse rate reduction, consistent with previous MSBase analyses and regulatory classifications3,5,7,12:
Platform DMTs: interferon beta (IFN-β), glatiramer acetate (GLAT), and teriflunomide (TF).
Moderate-efficacy DMTs: dimethyl fumarate (DMF), fingolimod, and other sphingosine-1-phosphate receptor modulators (S1Ps).
High-efficacy DMTs: cladribine (CLAD), natalizumab (NTZ), alemtuzumab (ALEM), and anti-CD20 agents including rituximab, ocrelizumab, and ofatumumab.
Thus, horizontal switching was defined as a change between DMTs within the same efficacy tier, while vertical switching referred to movement from a lower to a higher efficacy tier. Patients switching from high-efficacy to lower or moderate-efficacy therapies (i.e., de-escalation) were excluded from this analysis.
Study population
Patients included in this study had a confirmed diagnosis of RRMS based on the McDonald criteria13,14 and were aged between 18 and 50 years at the time of their initial DMT. To minimize immortal time bias while ensuring clinically meaningful exposure, patients were required to have received their first DMT for a minimum of 3 months before switching.4,5,15 The start of follow-up was defined as the date of treatment switch. Patients were required to have at least 12 months of follow-up on the second DMT to assess clinical outcomes. For IRTs such as CLAD and ALEM, patients were only included if they had completed the full treatment course and had a minimum of 12 months of follow-up after the final dose. To ensure data quality, patients needed at least two Expanded Disability Status Scale (EDSS) assessments following the switch, spaced a minimum of 6 months apart, and available MRI information, as imaging activity was a core component of outcome definitions. Patients with stem cell transplants, participation in clinical trials, or missing critical data (e.g., baseline EDSS or follow-up) were excluded.
Definitions of outcomes
Primary outcomes included the following: (1) change in annualized relapse rate (ARR), calculated as the difference between ARR under the new treatment and the prior DMT; (2) change in EDSS from the end of the first treatment to the last follow-up (⩾30 days post-relapse); (3) MRI inflammatory activity, measured by new/enlarging T1 and T2 lesions and gadolinium-enhancing lesions; and (4) successful switching, defined as maintaining no evidence of disease activity (NEDA-3) throughout the entire duration of the switching DMT. NEDA-3 comprised the absence of relapses, confirmed EDSS progression, significant pyramidal worsening (pyramidal score ⩾2 and EDSS ⩾4 in the absence of relapse), and new MRI activity during follow-up. This operational definition deviates from the conventional NEDA-3 criteria but was adopted given the availability and reliability of pyramidal functional system data in the MSBase Registry. Prior studies have shown that pyramidal system involvement is a major driver of sustained disability progression in MS, supporting its use as a proxy in disability outcomes analyses.16,17
Secondary outcomes included the reduction in relapse frequency compared to the previous treatment, confirmed disability worsening (CDW, defined as an increase in EDSS by at least 1.0 or 0.5 points if baseline EDSS was above 5.5, confirmed over 6 months without relapse), confirmed disability improvement (CDI, defined as a decrease in EDSS by the same margins), and progression independent of relapse activity (PIRA). PIRA was calculated using the MSOutcomes package, 18 following the definition provided by Dzau et al., 19 in which PIRA was characterized by sustained disability progression in the absence of relapse between the most recent prior EDSS and the EDSS, showing a documented increase.
Statistical analysis
To reduce selection bias and improve comparability between horizontal and vertical switch groups, we performed propensity score matching (PSM) using a logistic regression model. Covariates included the following: age at treatment initiation, sex, treatment order (number of prior DMTs), baseline ARR, baseline EDSS, time from disease onset to treatment switch, treatment period (categorized as: before 2000, 2001–2005, 2006–2010, 2011–2015, or 2016 or later), and geographic region (grouped as North America, Europe, Australia, or Other based on country code). Nearest-neighbor matching was applied in a 1:1 ratio without replacement, using a caliper of 0.1 on the logit of the propensity score. Covariate balance after matching was assessed using standardized mean differences (SMDs), with values below 0.1 considered acceptable. SMDs were used descriptively to assess the balance between groups. Comparisons between successful and unsuccessful horizontal switches were restricted to the matched horizontal cohort to ensure direct comparability with vertical switchers under similar baseline conditions.
For the primary outcome analyses, multivariable mixed-effects models were used to evaluate the association of switch type (horizontal vs vertical) with key clinical outcomes (relapse reduction, ARR change, EDSS change, CDW, CDI, PIRA, and NEDA-3 achievement during DMT duration), accounting for repeated treatment switches per patient (random intercept for patient). These models were adjusted for sex, age, time from symptom onset to switch, baseline ARR and EDSS, prior relapses, treatment level, treatment period, treatment order, and region. In addition, differences in success rates across DMTs were evaluated using chi-squared tests. All analyses were conducted in R (version 4.2.1, The R Foundation for Statistical Computing, Vienna, Austria), using the following packages: dplyr, tidyr, tableone, MatchIt, lmerTest, pROC, caret, ggplot2, and MSOutcomes. A two-sided p-value ⩽0.05 was considered statistically significant.
Results
Baseline characteristics and PSM
Before matching, we identified 4351 horizontal and 4266 vertical switches from 2353 and 3189 unique patients, respectively. The most pronounced imbalance was in treatment distribution (SMD = 2.78) and treatment level (SMD = 1.82). Moderate differences were also observed in treatment period (SMD = 0.77), geographic region (SMD = 0.38), and several clinical outcomes under the switched DMT, including relapses, ARR, change in EDSS, CDI, and NEDA-3 (SMDs 0.29–0.51). By contrast, demographic variables and most baseline disease history variables (ARR, relapses before switch, time from onset to switch) were well balanced (all SMD ⩽ 0.07). Full details are provided in Table 1, baseline and matched cohort. After 1:1 PSM, 2467 matched pairs were generated (Figure 1). In the matched cohort, all prespecified covariates achieved adequate balance (all SMDs ⩽0.05; Supplemental eTable 1). Residual imbalance in individual DMT distribution was expected, given the structural differences between horizontal and vertical switches. The mean follow-up after switching was 4.0 ± 3.1 years.
Patient and treatment characteristics before and after propensity score matching and among horizontal switchers.
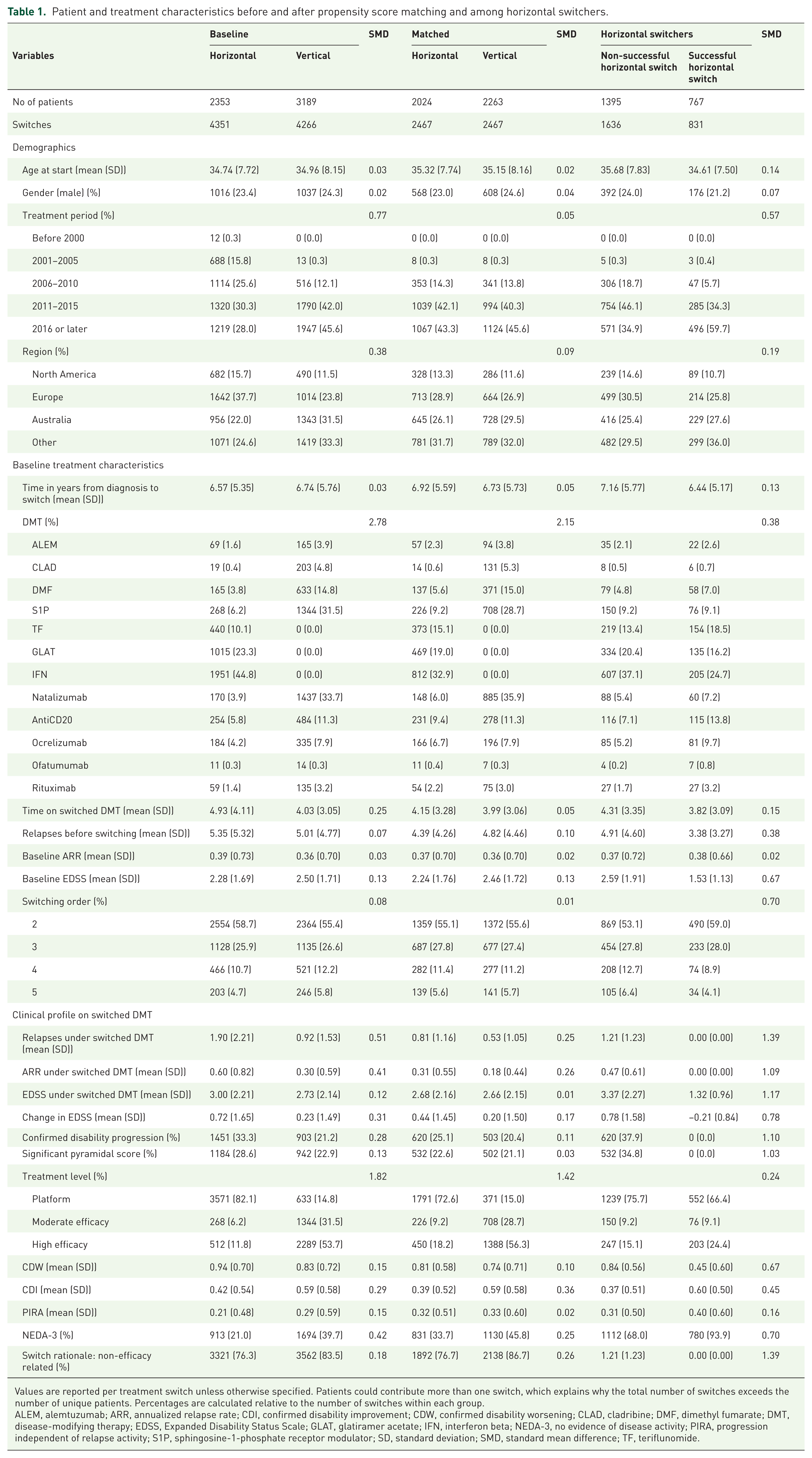
Values are reported per treatment switch unless otherwise specified. Patients could contribute more than one switch, which explains why the total number of switches exceeds the number of unique patients. Percentages are calculated relative to the number of switches within each group.
ALEM, alemtuzumab; ARR, annualized relapse rate; CDI, confirmed disability improvement; CDW, confirmed disability worsening; CLAD, cladribine; DMF, dimethyl fumarate; DMT, disease-modifying therapy; EDSS, Expanded Disability Status Scale; GLAT, glatiramer acetate; IFN, interferon beta; NEDA-3, no evidence of disease activity; PIRA, progression independent of relapse activity; S1P, sphingosine-1-phosphate receptor modulator; SD, standard deviation; SMD, standard mean difference; TF, teriflunomide.

Flowchart of patients’ selection.
In adjusted mixed-effects models (Table 2, (A)), vertical switching was associated with a modest reduction in ARR (OR: 0.94, 95% CI: 0.90–0.99, p = 0.011) and a higher likelihood of relapse reduction (OR: 1.45, 95% CI: 1.15–1.84, p = 0.002). Vertical switching was also associated with a lower risk of CDW (OR: 0.97, 95% CI: 0.94–0.99, p = 0.006) and a greater likelihood of CDI (OR: 1.24, 95% CI: 1.05–1.46, p = 0.010). No significant differences were observed for EDSS progression (OR: 0.92, 95% CI: 0.85–1.01, p = 0.075) or PIRA (OR: 0.93, 95% CI: 0.79–1.10, p = 0.420). Importantly, vertical switching was associated with an increased likelihood of achieving and maintaining NEDA-3 after switching (OR: 1.11, 95% CI: 1.07–1.14, p < 0.001). Additional covariate effects are provided in Supplemental eTable 2, and models additionally adjusted for switching rationale are reported in Supplemental eTable 3.
Mixed-effects models for clinical outcomes by switch type and determinants of successful horizontal switching.

Mixed-effects models assessing outcomes of vertical versus horizontal switching and predictors of successful horizontal switching. Section A compares relapse reduction, EDSS changes, and disability progression between switch types, while Section B identifies factors associated with successful horizontal switching, defined as maintaining NEDA-3 during follow-up. Estimates represent model coefficients with OR and 95% CI, adjusted for clinical variables with unique patients modeled as a random effect.
Adjusted for age at treatment start, gender, time from symptomatic onset to treatment switch, treatment level, treatment order, treatment period (calendar year group), and geographic region, with each unique patient included as a random effect.
ARR, annualized relapse rate; CI, confidence interval; DMT, disease-modifying therapy; EDSS, Expanded Disability Status Scale; NEDA-3, no evidence of disease activity; OR, odds ratio; PIRA, progression independent of relapse activity.
Characteristics of successful horizontal switchers
Among patients with horizontal switches in the matched cohort, 767 (33.7%) achieved NEDA-3 during follow-up and were classified as having a successful horizontal switch (Table 1, horizontal switchers). Compared to non-successful switchers, successful patients were slightly younger at treatment start (mean 34.6 vs 35.7 years; SMD = 0.14) and less often male (21.2% vs 24.0%; SMD = 0.07). They also had a shorter interval from symptom onset to treatment initiation (mean 6.4 vs 7.2 years; SMD = 0.13).
Successful switchers exhibited a lower baseline disease burden, with fewer prior relapses (3.38 vs 4.91; SMD = 0.38) and a lower baseline EDSS (1.53 vs 2.59; SMD = 0.67), while baseline ARR was comparable between groups (0.38 vs 0.37; SMD = 0.02). By definition, successful switchers experienced no relapses during follow-up (ARR 0.00 vs 0.47; SMD = 1.09), no significant pyramidal scores (0.0% vs 34.8%; SMD = 1.03), and no confirmed disability progression (0.0% vs 37.9%; SMD = 1.10). End-of-follow-up EDSS was lower among successful switchers (1.32 vs 3.37; SMD = 1.17), with disability improvement reflected by a negative EDSS change (−0.21 vs +0.78; SMD = 0.78). Longitudinal outcomes also showed a lower rate of CDW events (0.45 vs 0.84; SMD = 0.67) and more CDI events (0.60 vs 0.37; SMD = 0.45). The number of PIRA events was similar between groups (0.40 vs 0.31; SMD = 0.16).
Treatment patterns and rationale differed between groups. Successful switchers more often received high-efficacy therapies (24.4% vs 15.1%; SMD = 0.24) and switched more frequently in recent years (2016 or later: 59.7% vs 34.9%; SMD = 0.57). Non-efficacy-related reasons for switching were also more frequent among successful switchers (93.9% vs 68.0%; SMD = 0.70).
Predictors of successful horizontal switching
In the multivariable logistic regression model (Table 2, (B)), higher baseline EDSS (OR = 0.71, 95% CI: 0.67–0.75, p < 0.001) and a greater number of relapses before treatment switch (OR = 0.90, 95% CI: 0.88–0.93, p < 0.001) were associated with lower odds of achieving a successful horizontal switch. Switching at the third line of therapy or later was associated with higher odds of success (OR = 1.30, 95% CI: 1.08–1.55, p = 0.004). By contrast, switching within non-platform therapies was associated with significantly lower odds of success compared with switching within platform therapies, both for treatment level 2 (OR = 0.26, 95% CI: 0.20–0.34, p < 0.001) and for treatment level 3 (OR = 0.44, 95% CI: 0.37–0.53, p < 0.001; Figure 2).

Multivariable logistic regression model of predictors for successful horizontal switching.
No significant associations were found for age at the time of treatment switch (OR = 1.00, 95% CI: 0.98–1.01, p = 0.455), sex (OR = 0.86, 95% CI: 0.71–1.04, p = 0.124), baseline ARR (OR = 1.07, 95% CI: 0.95–1.20, p = 0.289), or treatment duration prior to switching (OR = 0.98, 95% CI: 0.96–1.01, p = 0.186). Of note, a longer time from symptom onset to treatment switch was associated with slightly higher odds of success (OR = 1.02, 95% CI: 1.00–1.04, p = 0.028).
Success rates of horizontal switching by DMT
Success rates of horizontal switching varied across DMTs when analyzed by the destination therapy (Figure 3). Among high-efficacy therapies, anti-CD20 agents achieved the highest success rate (50%), followed by CLAD (43%), NTZ (41%), and ALEM (39%). Moderate-efficacy therapies showed intermediate results, with DMF (42%) and S1Ps (34%). Low-efficacy therapies were associated with lower success rates, including TF (41%), GLAT (29%), and IFN-β (25%; χ2 = 76.22, p < 0.001). Reductions in relapse activity also varied. Anti-CD20 agents and NTZ were associated with the greatest ARR reductions (−0.19 and −0.12, respectively). CLAD showed a slight increase in ARR (+0.18) despite a relatively high success rate, while IFN-β and GLAT showed minimal reductions (−0.04 and −0.03, respectively). Disability outcomes showed the lowest mean EDSS changes with anti-CD20 agents (+0.06) and CLAD (+0.07), while more pronounced worsening was observed with IFN-β (+0.66) and GLAT (+0.49). Regarding MRI outcomes, suppression of inflammatory activity was most frequently achieved with anti-CD20 agents (100%), NTZ (96%), and ALEM (96%), whereas CLAD showed the lowest proportion of patients with suppressed MRI activity (50%; Table 3).

Rate of outcomes per treatment after horizontal switching.
Efficacy outcomes across matched horizontal switchers.

Efficacy outcomes across different DMTs. The table presents the proportion of sustained NEDA-3, changes in ARR and EDSS after switching, and the percentage of patients with no new MRI activity for each DMT.
ALEM, alemtuzumab; ARR, annualized relapse rate; CLAD, cladribine; DMF, dimethyl fumarate; DMT, disease-modifying therapy; EDSS, Expanded Disability Status Scale; GLAT, glatiramer acetate; IFN-β, interferon beta; NEDA-3, no evidence of disease activity; NTZ, natalizumab; S1P, sphingosine-1-phosphate receptor modulator; TF, teriflunomide.
When success rates were further analyzed by switching dynamics (i.e., the origin and direction of the switch, Figure 4), additional patterns emerged. Low-efficacy therapies demonstrated the lowest overall success (34%), with IFN and GLAT performing the poorest, while TF-origin switches showed slightly higher rates (41%). Moderate-efficacy therapies had intermediate success (DMF 46%, S1P 43%), driven by reciprocal switching between the two. High-efficacy therapies achieved the highest overall success (46%), though outcomes were heterogeneous: switches from ALEM were often successful (55%), as were those from CLAD (50%) and NTZ (48%), whereas switches originating from anti-CD20 therapies were less often effective (30%). Directionality also influenced results: switches toward anti-CD20 therapies or from ALEM were generally more successful than those originating from anti-CD20 (Table 4).

Horizontal switches between DMTs. The inner ring represents the baseline/originating DMT, while the outer ring shows the destination DMT after a horizontal switch. Segment size is proportional to the number of switches. Percentages indicate the proportion of successful switches (achieving NEDA-3) within each DMT pair. Colors distinguish platform, moderate-efficacy, and high-efficacy therapies.
Success rates of horizontal switching by DMT origin and destination.

ALEM, alemtuzumab; CLAD, cladribine; DMF, dimethyl fumarate; DMT, disease-modifying therapy; IFN, interferon; GLAT, glatiramer acetate; NTZ, natalizumab; S1P, sphingosine-1-phosphate receptor modulator; TF, teriflunomide.
Discussion
This study compared the clinical outcomes of horizontal versus vertical switching between DMTs in patients with RRMS. As expected, vertical switching was associated with higher rates of NEDA-3, greater reduction in relapse activity, and less disability progression. The primary aim was to describe the potential role of horizontal switching in patients whose outcomes may be comparable to those achieved with vertical switching. In this context, we identified several clinical variables associated with a higher likelihood of success after a horizontal switch, including lower baseline EDSS, fewer relapses prior to switching, and switching at later lines of treatment. Conversely, male sex and horizontal switching between moderate-efficacy therapies were associated with a lower probability of achieving disease stability. These findings may help clinicians identify patients in whom horizontal switching remains a reasonable option when vertical switching is not feasible or preferred.
Overall, our results confirm that vertical switching is the more effective approach when treatment modification is needed in most patients with MS. In our analysis, vertical switching achieved greater ARR reduction, higher rates of NEDA-3, a lower percentage of disability progression, less increase in EDSS scores, and more frequent CDI events. Our analysis also highlights the nuances of vertical switching. The benefits were evident not only in patients escalating from platform therapies but also in those switching from moderate-efficacy drugs. These findings are consistent with previous reports: the Austrian MS treatment registry 20 showed that vertical switchers were less likely to relapse and had lower ARR than horizontal switchers, while the Swiss National Treatment Registry 21 reported that direct switching to high-efficacy DMTs significantly reduced the risk of relapse compared to stepwise escalation. Collectively, these data suggest that vertical switching is more effective in suppressing inflammatory disease activity; its impact on long-term disability progression and PIRA, however, remains less well established. Nevertheless, several studies indicate that horizontal switching can still be effective in some scenarios. A single-center Italian study 10 found no significant differences in NEDA-3 achievement at 24 months between horizontal and vertical switchers (20.8% vs 18.6%), with similar time to first relapse and time to EDSS milestones. Similarly, data from the Danish MS Registry 22 had outcomes such as ARR after horizontal switching that depend strongly on baseline characteristics, including EDSS and prior DMT history.
In line with these observations, our study shows that while horizontal switching is overall a suboptimal strategy, it can be effective in a subset of patients. Patients with lower EDSS scores, fewer relapses before switching, and relative disease stability were most likely to achieve favorable outcomes, underscoring the importance of patient selection. Moreover, the rationale for switching also appeared relevant: switches undertaken for non-efficacy reasons, such as tolerability or safety, tended to be more successful, reflecting continuity of disease control rather than recovery from breakthrough activity. Our data also suggest that not all horizontal switches between high-efficacy DMTs are equivalent. Anti-CD20 therapies showed comparatively better outcomes, while switches to or from other high-efficacy agents such as CLAD, NTZ, or ALEM were more heterogeneous. These findings imply that sequencing and directionality may influence success rates, refining the interpretation of horizontal switching beyond a uniform strategy. For example, in our analysis, switches toward anti-CD20 therapies or from ALEM were more often successful, while switches originating from anti-CD20 therapies were less likely to maintain stability. Similarly, CLAD demonstrated relatively favorable outcomes when used as a destination therapy but more mixed results when it was the origin of a switch. Such patterns highlight that the clinical context of the switch—whether patients are stepping laterally into a more tolerable therapy within the same tier or moving away from a drug due to emerging risks—can substantially shape outcomes. Recognizing these complexities may help clinicians better anticipate the likelihood of success when horizontal switching is considered. Collectively, this evidence supports the rationale for early initiation of high-efficacy therapies to optimize long-term outcomes,4,5,12 while also providing guidance for treatment selection in resource-constrained settings23,24—such as Mexico—where horizontal switching is often driven by limited access to the full range of DMTs.
This study has several strengths. It is the largest to date, comparing horizontal and vertical switching in RRMS, using a global, real-world dataset from a rigorously curated MS registry. The use of PSM and multivariable modeling helped to reduce confounding and improve the validity of our comparisons. However, limitations must be acknowledged. The retrospective design and use of registry data may introduce selection bias and limit the granularity of available information. A key limitation is that reasons for switching were not systematically recorded for all patients; however, among the subset of patients with documented reasons, we observed that the majority of successful horizontal switches were made for non-efficacy-related reasons, such as safety, tolerability, or patient preference. This suggests that successful horizontal switching is more likely to reflect stability under prior treatment rather than response to recent disease activity. Despite this limitation, additional models adjusted for switching rationale (Supplemental eTable 3) consistently confirmed the superiority of vertical switching in reducing relapses and disability worsening, underscoring that efficacy remains the key driver of long-term outcomes. Another important source of potential bias is missing or incomplete data in the registry. Of more than 22,000 potentially eligible patients, a substantial number were excluded due to missing values in key variables, which were unlikely to be missing at random. This introduces the risk of selection bias that cannot be fully addressed through statistical adjustment. In addition, residual confounding remains possible, as unmeasured factors such as socioeconomic status, adherence, and local treatment practices were not captured in the registry. Misclassification bias may also have influenced our results. Treatment coding errors, variability in EDSS scoring, and incomplete MRI data could have led to misclassification of outcomes or exposures. While we applied strict inclusion criteria to improve data reliability, these issues cannot be fully excluded. Moreover, although we accounted for repeated switches by modeling patients as random effects, this approach may not fully capture dependence across multiple switches contributed by the same individual. Alternative approaches, such as inverse probability weighting or doubly robust estimators, could further validate our findings in future work. Follow-up duration also varied considerably between patients and groups, and not all outcomes were standardized to follow-up time, which may have introduced bias in measures influenced by observation length, such as relapse counts and disability progression. Furthermore, information on concomitant medications was not consistently coded across patients. While we excluded IVIG and other non-MS-specific therapies from the analysis (as shown in Figure 1), the use of symptomatic treatments that may influence relapse occurrence, functional performance, or disability progression was not systematically captured and therefore could not be evaluated. Finally, once patients are already on a high-efficacy therapy, a “vertical” switch is not possible; thus, within-tier switches in this group are necessarily classified as horizontal. While this limits direct comparability with vertical switchers, we consider these dynamics clinically relevant and have provided subgroup analyses to better illustrate these patterns. As registry data are predominantly drawn from specialized MS centers, the findings may be more applicable to referral center populations than to community-based practice settings.
Conclusion
In summary, this registry-based study compared real-world outcomes of horizontal and vertical DMT switching in RRMS. Vertical switching was associated with greater reductions in relapse activity, lower risk of disability worsening, and higher rates of disease stability. However, a subset of patients achieved favorable outcomes following horizontal switching, particularly those with lower baseline disability, fewer prior relapses, and later-line treatment changes. These findings suggest that while vertical switching generally provides superior disease control, horizontal switching may maintain stability in selected patients with milder disease or non-efficacy-related treatment changes. Further studies are warranted to confirm these patterns and clarify their long-term impact on disability progression and PIRA.
Supplemental Material
sj-csv-1-tan-10.1177_17562864251399595 – Supplemental material for Real-world effectiveness of horizontal switching between disease-modifying therapies in multiple sclerosis: a retrospective analysis of the MSBase Registry
Supplemental material, sj-csv-1-tan-10.1177_17562864251399595 for Real-world effectiveness of horizontal switching between disease-modifying therapies in multiple sclerosis: a retrospective analysis of the MSBase Registry by Enrique Gomez-Figueroa, Patricia Orozco-Puga, Cynthia Patricia Corona-Vázquez, Carlos Moreno-Bernardino, Graciela Elizabeth De la Mora-Landín, Amado Jiménez-Ruiz, Christian García-Estrada, Lizeth Zertuche-Ortuño, Sergio Saldívar-Dávila, Roberto Rodríguez-Rivas, Lisette Bazán-Rodríguez, José Flores-Rivera, Tomas Kalincik, Katherine Buzzard, Samia Khoury, Pierre Duquette, Matteo Foschi, Andrea Surcinelli, Bianca Weinstock-Guttman, Riadh Gouider, Saloua Mrabet, Jeannette Lechner-Scott, Helmut Butzkueven, Raed Alroughani, Izanne Roos, Francesco Patti, Bassem Yamout, François Grand’Maison, Daniele Spitaleri, Pamela McCombe, José Luis Sanchez-Menoyo, Serkan Ozakbas, Abdullah Al-Asmi, Nevin John, Elisabetta Cartechini, Anneke Van der Walt, Justin Garber, Emmanuelle Lapointe, Aysun Soysal, Eduardo Aguera-Morales, Joana Guimarães and José Luis Ruiz-Sandoval in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-csv-2-tan-10.1177_17562864251399595 – Supplemental material for Real-world effectiveness of horizontal switching between disease-modifying therapies in multiple sclerosis: a retrospective analysis of the MSBase Registry
Supplemental material, sj-csv-2-tan-10.1177_17562864251399595 for Real-world effectiveness of horizontal switching between disease-modifying therapies in multiple sclerosis: a retrospective analysis of the MSBase Registry by Enrique Gomez-Figueroa, Patricia Orozco-Puga, Cynthia Patricia Corona-Vázquez, Carlos Moreno-Bernardino, Graciela Elizabeth De la Mora-Landín, Amado Jiménez-Ruiz, Christian García-Estrada, Lizeth Zertuche-Ortuño, Sergio Saldívar-Dávila, Roberto Rodríguez-Rivas, Lisette Bazán-Rodríguez, José Flores-Rivera, Tomas Kalincik, Katherine Buzzard, Samia Khoury, Pierre Duquette, Matteo Foschi, Andrea Surcinelli, Bianca Weinstock-Guttman, Riadh Gouider, Saloua Mrabet, Jeannette Lechner-Scott, Helmut Butzkueven, Raed Alroughani, Izanne Roos, Francesco Patti, Bassem Yamout, François Grand’Maison, Daniele Spitaleri, Pamela McCombe, José Luis Sanchez-Menoyo, Serkan Ozakbas, Abdullah Al-Asmi, Nevin John, Elisabetta Cartechini, Anneke Van der Walt, Justin Garber, Emmanuelle Lapointe, Aysun Soysal, Eduardo Aguera-Morales, Joana Guimarães and José Luis Ruiz-Sandoval in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-csv-3-tan-10.1177_17562864251399595 – Supplemental material for Real-world effectiveness of horizontal switching between disease-modifying therapies in multiple sclerosis: a retrospective analysis of the MSBase Registry
Supplemental material, sj-csv-3-tan-10.1177_17562864251399595 for Real-world effectiveness of horizontal switching between disease-modifying therapies in multiple sclerosis: a retrospective analysis of the MSBase Registry by Enrique Gomez-Figueroa, Patricia Orozco-Puga, Cynthia Patricia Corona-Vázquez, Carlos Moreno-Bernardino, Graciela Elizabeth De la Mora-Landín, Amado Jiménez-Ruiz, Christian García-Estrada, Lizeth Zertuche-Ortuño, Sergio Saldívar-Dávila, Roberto Rodríguez-Rivas, Lisette Bazán-Rodríguez, José Flores-Rivera, Tomas Kalincik, Katherine Buzzard, Samia Khoury, Pierre Duquette, Matteo Foschi, Andrea Surcinelli, Bianca Weinstock-Guttman, Riadh Gouider, Saloua Mrabet, Jeannette Lechner-Scott, Helmut Butzkueven, Raed Alroughani, Izanne Roos, Francesco Patti, Bassem Yamout, François Grand’Maison, Daniele Spitaleri, Pamela McCombe, José Luis Sanchez-Menoyo, Serkan Ozakbas, Abdullah Al-Asmi, Nevin John, Elisabetta Cartechini, Anneke Van der Walt, Justin Garber, Emmanuelle Lapointe, Aysun Soysal, Eduardo Aguera-Morales, Joana Guimarães and José Luis Ruiz-Sandoval in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-docx-4-tan-10.1177_17562864251399595 – Supplemental material for Real-world effectiveness of horizontal switching between disease-modifying therapies in multiple sclerosis: a retrospective analysis of the MSBase Registry
Supplemental material, sj-docx-4-tan-10.1177_17562864251399595 for Real-world effectiveness of horizontal switching between disease-modifying therapies in multiple sclerosis: a retrospective analysis of the MSBase Registry by Enrique Gomez-Figueroa, Patricia Orozco-Puga, Cynthia Patricia Corona-Vázquez, Carlos Moreno-Bernardino, Graciela Elizabeth De la Mora-Landín, Amado Jiménez-Ruiz, Christian García-Estrada, Lizeth Zertuche-Ortuño, Sergio Saldívar-Dávila, Roberto Rodríguez-Rivas, Lisette Bazán-Rodríguez, José Flores-Rivera, Tomas Kalincik, Katherine Buzzard, Samia Khoury, Pierre Duquette, Matteo Foschi, Andrea Surcinelli, Bianca Weinstock-Guttman, Riadh Gouider, Saloua Mrabet, Jeannette Lechner-Scott, Helmut Butzkueven, Raed Alroughani, Izanne Roos, Francesco Patti, Bassem Yamout, François Grand’Maison, Daniele Spitaleri, Pamela McCombe, José Luis Sanchez-Menoyo, Serkan Ozakbas, Abdullah Al-Asmi, Nevin John, Elisabetta Cartechini, Anneke Van der Walt, Justin Garber, Emmanuelle Lapointe, Aysun Soysal, Eduardo Aguera-Morales, Joana Guimarães and José Luis Ruiz-Sandoval in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The authors thank the patients and clinicians who contribute data to the MSBase Registry, as well as the MSBase Scientific Leadership Group for oversight of this study.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
