Abstract
Background:
The brain–gut axis has emerged as a potential target in neurodegenerative diseases, including dementia, as individuals with dementia exhibit distinct gut microbiota compositions. Fecal microbiota transplantation (FMT), the transfer of fecal solution from a healthy donor to a patient, has shown promise in restoring homeostasis and cognitive enhancement.
Objective:
This study aimed to explore the effects of FMT on specific cognitive performance measures in Alzheimer’s dementia (AD) patients and investigate the relationship between cognition and the gut microbiota by evaluating changes in gene expression following FMT.
Methods:
Five AD patients underwent FMT, and their cognitive function [Mini-Mental State Examination (MMSE), Montreal Cognitive Assessment (MoCA), and Clinical Dementia Rating Scale Sum of Boxes (CDR-SOB)] was assessed before and after FMT. The patients’ fecal samples were analyzed with 16S rRNA to compare the composition of their gut microbiota. We also assessed modifications in the serum mRNA expression of patients’ genes related to lipid metabolism using serum RNA sequencing and quantitative real-time polymerase chain reaction.
Results:
Significant improvements in cognitive function, as measured by the MMSE (pre- and post-FMT was 13.00 and 18.00) and MoCA were seen. The MoCA scores at 3 months post-FMT (21.0) were the highest (12.0). The CDR-SOB scores at pre- and post-FMT were 10.00 and 5.50, respectively. Analysis of the gut microbiome composition revealed changes via 16S rRNA sequencing with an increase in Bacteroidaceae and a decrease in Enterococcaceae. Gene expression analysis identified alterations in lipid metabolism-related genes after FMT.
Conclusion:
These findings suggest a link between alterations in the gut microbiome, gene expression related to lipid metabolism, and cognitive function. The study highlights the importance of gut microbiota in cognitive function and provides insights into potential biomarkers for cognitive decline progression. FMT could complement existing therapies and show potential as a therapeutic intervention to mitigate cognitive decline in AD.
Keywords
Introduction
Neurodegenerative disorders are a group of conditions characterized by the progressive deterioration and loss of neurons, leading to various abnormalities. Among these, dementia stands out as a significant global cause of mortality, contributing to approximately 1.62 million deaths in 2019 and imposing substantial economic and social burdens.1,2 The impact of dementia extends beyond mortality, affecting the quality of life and leading to heightened morbidity, increased challenges in nursing care, and higher rates of depression. 3 Despite ongoing exploration of diverse treatment modalities, including medication and rehabilitation, the effectiveness of these treatments remains uncertain,4,5 underscoring the necessity for alternative interventions.
An emerging area of research highlights the potential role of the brain–gut axis in neurodegenerative disorders.6,7 This bidirectional communication system between the gastrointestinal and central nervous systems contributes to various cellular mechanisms affecting health and disease, such as immune system regulation and homeostasis. 8 Notably, individuals with neurological disorders, including dementia, exhibit distinct gut microbiota compositions compared to healthy individuals. 9 Moreover, early-stage patients show a different gut microbial composition than those with advanced disease, suggesting the gut microbiota’s potential influence on disease progression.9,10
Fecal microbiota transplantation (FMT), a procedure involving the transfer of a fecal solution from a healthy donor to a patient, has shown promising therapeutic potential.6,7,10 This procedure shifts the recipient’s homeostasis toward a healthier state, restoring the blood–brain barrier and intestinal integrity. 11 FMT’s potential impact on cognitive enhancement through the brain–gut axis has been observed. 12 Despite these potential benefits, many issues still need to be addressed, including safety concerns, lack of standardized protocols, limited understanding of the microbiome, and ethical and regulatory challenges.
Given this context, our study aims to explore whether FMT significantly affects specific cognitive performance measures in dementia patients. We also examined the relationship between cognition and gut microbiota by evaluating patients’ gene expression changes following the FMT procedure.
Methods
Patients recruitment
All patients were screened between April 2020 and July 2022 at the Dementia and Microbiome Centers in the hospital. We enrolled patients aged more than 18 years who were diagnosed with dementia with relapsed Clostridioides difficile infection (CDI) and who required FMT. We came across patients with severe CDI who had previously failed treatment with several antibiotics and had relapsed symptoms with a positive CDI stool test. We also screened patients admitted with a dementia diagnosis made by a neurologist, particularly those with Alzheimer’s dementia (AD). 13 The following patients were considered unfit for FMT and were excluded: patients with known immunosuppression (HIV infection or steroid therapy); other acute infectious diseases except for CDI; pregnant women; breastfeeding women; patients with structural digestive tract abnormalities, such as obstruction, ileus, or perforation; patients requiring intensive care unit care; and patients with ongoing antibiotic therapy. In all, 14 patients were enrolled; however, six were lost to follow-up, and three did not undergo fecal analysis because of the patient’s fecal samples were unsuitable for analysis. Finally, five patients were included in the study.
Fecal microbiota transplantation
Stool donors with no gastrointestinal or other health problems underwent blood and stool tests, including a COVID-19 test. Informed consent was obtained from the physician before screening. Donors were required to answer a questionnaire regarding FMT donor selection. 14 Donor condition was regularly checked using laboratory and stool tests. The donated stool was filtered using a 110 × 10 cm non-woven swab mixed with 40 mL of sterile saline (0.9% NaCl) and stored at −80°C in a stool bank. The stool suspension was stored in a sterile container until FMT.
Before FMT, stool suspensions (60 g/300 mL) were prepared at 37°C for six h. The recipients underwent bowel preparation according to their condition and doctor’s instructions before FMT. Stool suspensions (60 g) were injected into the recipients’ intestinal tracts through colonoscopy by a skilled gastroenterologist, following the FMT guidelines. 15 Recipients were monitored and examined for side effects at the microbiome center after FMT.
Cognitive function and laboratory evaluations
A neurologist evaluated cognitive function individually in all patients with AD for approximately 20–30 min. The Mini-Mental State Examination (MMSE; range, 0–30), Montreal Cognitive Assessment (MoCA; range, 0–30), Clinical Dementia Rating Scale (CDR; range, 0–30), and Glasgow Coma Scale (GCS; range, 3–15) tests were performed before FMT and 1 and 3 months after FMT. Although the test time varied, the test results were confirmed to be as close as possible to those before FMT.
The MMSE is a brief test used to evaluate cognitive function. It is widely used to evaluate dementia severity. 16 However, MMSE is insufficient for assessing executive impairments. 17 MoCA is more sensitive than MMSE for observing mild cognitive impairment and AD. 16 It evaluates a broad range of cognitive domains, including executive and visuospatial functions. Therefore, according to the best practice guidelines, we employed the MMSE and MoCA scores as standardized tools for cognitive testing during clinical practice. 17 CDR is widely used to grade dementia severity and yields the Sum of Boxes (CDR-SOB) scores (range, 0–18) regularly used in clinical and research settings. 18 Higher scores on the MMSE and MoCA and lower scores on the CDR and CDR-SOB suggest better cognitive function (Supplemental Table S1). GCS was the sum of the scores and individual elements (with three being the worst and 15 being the highest).
Laboratory tests were performed, including infection markers, inflammation markers, electrolytes, etc., before FMT and 1 and 3 months after FMT or at admission with cognitive function tests simultaneously (maximum 3 days before and after the cognitive test). The general conditions that affected the cognitive function tests were evaluated. Therefore, we also confirmed whether the patients’ cognition was influenced by their general condition and excluded those patients for whom it was (Supplemental Table S3).
Gut microbiota analysis
Patient feces (approximately 2–5 mL) were collected from a stool carrier before and after FMT. All samples were stored at −80°C until shipping to the Macrogen-Biotech Lab (Seoul, Republic of Korea) for 16S rRNA gene amplicon sequencing and sequencing.
Metagenomic DNA was extracted from fecal samples, and the V3–V4 region of the bacterial 16S rRNA gene was amplified using barcoded universal primers. PicoGreen was used to pool and normalize the amplified products. The quantity and fragment size of the extracted DNA was assessed using an Agilent Technologies 2100 Bioanalyzer equipped with a DNA 1000 chip (Santa Clara, CA, USA). According to the manufacturer’s instructions, sequencing was performed using a MiSeq sequencer on an Illumina platform.
Microbiome profiling was conducted using the 16S-based microbial taxonomic profiling (MTP) platform of EzBio-Cloud Apps (ChunLab Inc., Seoul, Republic of Korea). After taxonomic profiling of each sample, a comparative MTP analyzer from the EzBio-Cloud Apps was used for comparative analysis. On the ChunLab MTP platform, the sequencing reads were preprocessed using the following five steps: (1) filtering of low-quality reads, (2) merging of paired-end reads, (3) removal of barcode and primer sequences, (4) taxonomic assignment of the reads, and (5) removal of chimeric sequences. Taxonomic assignment of the reads was conducted using the ChunLab 16S rRNA database (DB ver. PKSSU4.0). 19
OTU picking was conducted using UCLUST and CD-HIT with a 97% similarity cutoff of 0.49 and Good’s coverage, rarefaction, and alpha-diversity indices were calculated, including the Chao and Shannon indices observed in MTP. Beta diversity was analyzed using the permutational multivariate analysis of variance (PERMANOVA) variance. Overall differences in the microbiome structure were evaluated using principal coordinate analysis of unweighted and weighted UniFrac distances. 20 Linear discriminant analysis effect size (LEfSe) was used to determine the taxonomic differences of pre- and post-FMT. 21 Functional biomarker analysis using the phylogenetic investigation of communities by reconstruction of the unobserved states (PICRUSt) software package was performed between the pre-and post-FMT groups using EzBio-Cloud Apps. 22
RNA extraction and reverse transcription
Total RNA was purified from 200 μL of serum using the miRNeasy Serum/Plasma Kit (Qiagen, Hilden, Germany) according to the manufacturer’s protocols. The RNA concentration and quality were assessed using a NanoDrop spectrophotometer (Thermo Scientific, Wilmington, DE, USA). For mRNA quantitative polymerase chain reaction (qPCR), 250 ng of total RNA was reverse transcribed to cDNA using the RT2 First Strand Kit (Qiagen) according to the manufacturer’s instructions.
Quantitative real-time polymerase chain reaction
Transcriptomes related to lipid metabolism were evaluated via qPCR using the human fatty acid metabolism RT2 Profiler PCR array (Qiagen), which profiled the expression of 84 gene transcripts related to lipid metabolism. Each cDNA sample was mixed with RT2 SYBR Green Master Mix (Qiagen). Two housekeeping genes were selected as references: glyceraldehyde-3-phosphate dehydrogenase and hypoxanthine phosphoribosyltransferase 1. The Ct values were standardized based on gene selection. The fold change/regulation of expression was calculated using the delta/delta Ct method of the data analysis web portal, in which delta Ct was calculated between the gene of interest and the average reference genes, followed by delta–delta Ct calculations. The fold change was calculated using formula 2 (−∆∆Ct).
Statistical analysis
Data are presented as mean, range, median, or number (percentage), as appropriate. Statistical significance among the three groups was determined using the Kruskal–Wallis test, a nonparametric method for deciding whether samples originate from the same distribution. In addition, comparisons among the three groups were conducted using the Friedman test, which is a nonparametric statistical test. Relative gene expression data from the quantitative real-time polymerase chain reaction were analyzed using the comparative Ct method (2−∆∆Ct). Statistical analyses were performed using GraphPad Prism (Version 9.3, GraphPad Software, San Diego, CA, USA), the R statistical package (R Foundation, Vienna, Austria; https://www.r-project.org), and SPSS 27.0, for Windows (SPSS Inc., Chicago, IL, USA). Relative gene expression data were analyzed using the online RT2 Profiler PCR Array data analysis program (https://geneglobe.qiagen.com/cn/analyze). p Values <0.05 were considered significant.
Results
Baseline characteristics
The baseline characteristics of the study groups are presented in Table 1. Patients were diagnosed with AD based on cognitive function tests, serum tests, and brain imaging (Figure 1). Patients presented with CDI symptoms, such as abdominal pain, diarrhea, vomiting, or fever (body temperature >38°C). The major CDI symptoms were abdominal pain and fever in FMT patients. Of the five patients recruited, three were females, and two were males; the median age at the time of FMT was 74 years (range 62–90 years). All the subjects were treated with one or two antibiotics before FMT; two patients (20.0%) were administered oral vancomycin, and two (20.0%) were administered metronidazole.
Clinical characteristics of FMT.

CDI, Clostridioides difficile infection; FMT, fecal microbiota transplantation; IQR, interquartile range; mRS, modified Rankin Scale.

Study flow.
Effects on clinical presentation after FMT
No serious adverse effects were reported after FMT (Table 1). Nausea was reported in one patient 3 h after FMT. However, the patient’s symptoms resolved without intervention. Transient abdominal pain was reported in one patient after the procedure caused by gas insertion for a colonoscopy. No recurrence of CDI symptoms or FMT-related complications was noted.
All five patients showed improved cognitive function post-FMT compared with pre-FMT (Table 2 and Supplemental Table S1). The mean value of the difference in the MMSE scores of five patients before and three months following FMT was 6.0 (p = 0.010). The median MMSE scores were higher after FMT than those before FMT. Notably, cognitive function continued to improve after FMT [pre-FMT; 11.0 (median), 12.8–16.0 (interquartile range, IQR) versus 1-month post-FMT; 16.0 (median), 13.0–18.0 (IQR) versus 3 months post-FMT; 17.0 (median), 12.8–19.0 (IQR)]. Friedman test revealed that the mean value of the difference in MMSE scores of five patients before and three months following FMT was significant (p = 0.005).
Cognitive function difference between before and after FMT.
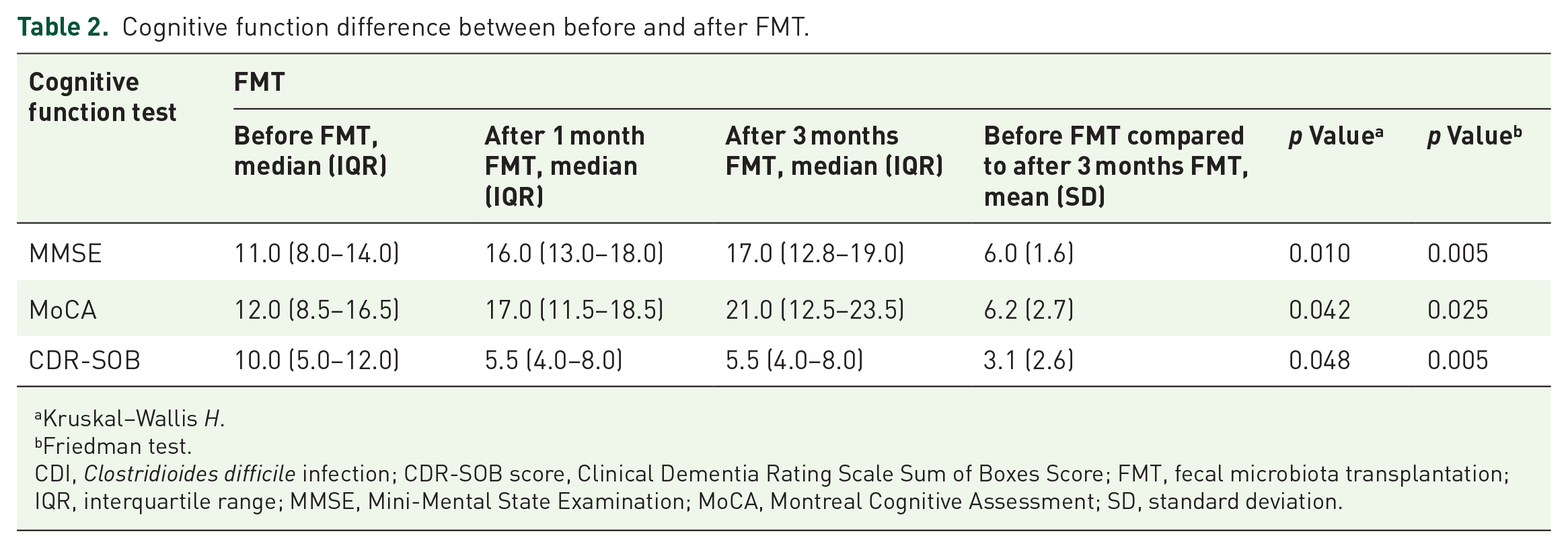
Kruskal–Wallis H.
Friedman test.
CDI, Clostridioides difficile infection; CDR-SOB score, Clinical Dementia Rating Scale Sum of Boxes Score; FMT, fecal microbiota transplantation; IQR, interquartile range; MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment; SD, standard deviation.
The median MoCA scores were similar to the median MMSE scores. Median MoCA scores at three months post-FMT [21.0, 12.5–23.5 (IQR)] were the highest [12.0, 8.5–16.5 (IQR)]. It was confirmed that FMT reduced cognitive impairment, which improved and was maintained over time. The mean difference in MoCA score between pre- and 3 months post-FMT was 6.2 (mean, p = 0.042). In addition, the mean values of MoCA scores were significantly distinct among the five patients (p = 0.025, Friedman test).
The median CDR-SOB score was lesser post-FMT [5.5 (median), 4.0–8.0 (IQR)] than that pre-FMT [10.0 (median), 5.0–12.0 (IQR)]. However, the median CDR-SOB score did not change significantly between 1 and 3 months after FMT. Nevertheless, the mean difference in CDR-SOB scores between pre- and 3 months post-FMT was 3.1 (p = 0.048). The Friedman test of the CDR-SOB was also significantly different between pre- and 3 months post-FMT in five patients (p = 0.005). Therefore, it can be inferred that FMT significantly improved cognitive function.
Comparison of fecal microbiome composition before and after FMT
We compared the paired samples in five patients before and at 1 and 3 months after FMT. The bacterial richness of the gut microbiota increased in patients after FMT (Figure 2). This indicates that the microbiome analysis revealed changes in bacterial taxa before and after FMT. There was an increase in the average bacterial composition, including the abundance of members of the Bacteroidaceae family (from 17% to 33%). By contrast, the abundance of the family Enterococcaceae decreased from 23% to 6%.

Change in microbiome before and after FMT.
An analysis of the abundance of each genus before and after FMT in patient fecal samples revealed that the proportions of Bacteroides, Parabacteroides, Alistipes, Tannerella, and Actinobacteria were relatively enriched in fecal samples after FMT compared to those before FMT (8% versus 27%, 2% versus 10%, 1% versus 5%, 0% versus 4%, and 0% versus 4%, respectively). By contrast, Enterococcus proportions (31% versus 18%) were reduced after FMT compared to before. The taxonomic relative abundance of the Enterococcaceae family was altered after FMT [pre-FMT; median, 6.70 (IQR, 4.60–18.77) versus post-FMT; median, 37.41 (IQR, 21.47–57.87), p = 0.11]. Particularly, Bacteroides genus was altered after FMT [pre-FMT; median, 0.84 (IQR, 0.18–12.9) versus post-FMT; median, 38.18 (IQR, 1.58–76.11), p = 0.047].
The alpha diversity significantly differed before and 3 months after FMT [Figure 3(a)]. The composition of patient feces altered the diversity (Shannon index, p = 0.026) and species richness (Chao index, p = 0.11) after FMT. The beta diversity exhibited a significant difference between pre-and three-month post-FMT according to PERMANOVA [p = 0.046, Figure 3(b)]. LEfSe confirmed a significant taxonomic profile difference between pre-and post-FMT [Figure 3(c)]. After FMT, some bacteria were either enriched or reduced. At the phylum level, Bacteroidaceae was the most enriched taxon post-FMT (LEfSe >2, p < 0.05). At the genus level, Bacteroides, Alistipes, and Odoribacter were enriched after FMT (LEfSe >2, p < 0.05).

Microbiome analysis before and after FMT. (a) The alpha diversity, including composition, and richness of gut microbiome, differed before and 3 months after FMT (Shannon index, p = 0.026, Chao index, p = 0.11). (b) The beta diversity exhibited a significant difference before and 3 months after FMT (PERMANOVA, p = 0.046). (c) LEfSe confirmed a significant taxonomic profile difference between pre- and post-FMT. At the genus level, Bacteroides, Alistipes, and Odoribacter were enriched after FMT (LEfSe > 2, p < 0.05).
Expression of genes involved in lipid metabolism after FMT
We used commercially available PCR arrays to compare the pre-and post-FMT gene expression profiles. These arrays contained primers for 84 genes involved in fatty acid biosynthesis, transport, and metabolism. The results revealed altered expression of all genes (Figure 4). There were statistically significant differences in the expression of 12 mRNA genes between pre- and post-FMT (p < 0.05; Table 3). In all, 11 mRNA genes (ACAA1, ACAD11, ACOT8, BDH1, DECR2, EHHADH, SLC27A2, SLC27A3, SLC27A4, SLC27A6, and B2M) were upregulated and one (FABP6) was downregulated after FMT (more than twofold).

Clustergram before and after FMT.
Expression of genes after FMT.

FMT, fecal microbiota transplantation.
In particular, the expression levels of three mRNA genes (ACAA1, EHHADH, and B2M) increased continuously after FMT (Figure 5). The expression of one mRNA gene (FABP6) expression continuously decreased after FMT. Among the investigated mRNA genes, significant shifts were observed in the expression of ACAA1 (32.51 ± 2.02 versus 29.21 ± 1.63, p = 0.0004), EHHADH (19.33 ± 0.35 versus 27.36 ± 0.58, p = 0.016), and B2M (24.53 ± 0.30 versus 22.33 ± 0.23, p = 0.010, data not shown) after FMT compared to that before FMT. In addition, there were remarkable changes in the expression of genes such as EHHADH after FMT (Table 3 and Figure 5).

Alteration of gene expression after FMT compared to before FMT.
To further explore the association among gene expression, FMT, and cognitive function, we included 11 mRNAs, for which a significant difference between the groups (p < 0.05) was demonstrated to avoid overlooking a relevant relationship with gene expression. A receiver operating characteristic (ROC) curve was generated for cognitive improvement before FMT versus 1 and 3 months after FMT (Table 4 and Figure 6). Furthermore, three months post-FMT showed the highest area under the curve (0.957, p = 0.10) for cognitive improvement compared to 1-month post-FMT (0.591, p = 0.606).
ROC curve analysis of cognitive functions to discriminate gene expression between before and after FMT.

AUC, area under the curve; FMT, fecal microbiota transplantation; ROC, receiver operating characteristic.

ROC curves of cognitive improvement to discriminate gene expression between before and after FMT.
Discussion
The composition of the gut microbiome is known to have a substantial impact on cognitive function. By investigating this interaction through fecal and blood analyses, our study finds that FMT could effectively mitigate cognitive decline in dementia patients. Specifically, our findings indicate a link between alterations in the gut microbiome, gene expression related to lipid metabolism, and cognitive function. Given the essential role of lipid metabolism in brain function, from neuron signaling to maintain the blood–brain barrier, these changes could serve as potential biomarkers for cognitive decline progression.
Our study also discovered a notable correlation between cognitive decline and changes in gut microbiota following FMT. Generally, dementia patients have less diverse gut microbiota compared to healthy individuals.12,23 However, FMT significantly increases the enrichment of Proteobacteria and Bacteroidetes, both positively linked to cognitive function.7,24,25 At the genus level, Bacteroides, Parabacteroides, Alistipes, Tannerella, and Actinobacteria are associated with cognitive function enhancement, while Enterococcus correlates with cognitive dysfunction.26,27 These associations underline the potential of both direct and indirect modifications of the gut microbiota composition to impact cognitive function.
The gut microbiota may impact neuronal function directly or indirectly by producing neuroactive metabolites. 28 This crucial role of the gut microbiota extends to regulating central neurotransmitters and normal brain development, including the hippocampus and microglial morphology.29–31 Our findings corroborate previous studies showing that an absence or insufficiency of gut microbiota is associated with microglial impairment. 13 We further explored the influence of gut microbiota alteration by FMT on host gene expression. Changes in metabolites such as bile acids, amino acids, and short-chain fatty acids induced by alterations in gut microbiota have been found to affect brain function. 32 In particular, changes in lipid metabolism have been linked to cognitive decline, and it is possible that changes in the transcriptional activity of genes involved in lipid metabolism could provide a therapeutic target.33–37 Consequently, our study provides additional evidence linking lipid metabolism to cognitive function, with the discovery that FMT results in the increased expression of three genes (ACAA1, EHHADH, and B2M) associated with lipid metabolism and the downregulation of one gene (FABP6).
In addition, these results had an interesting point where they correlated with the lipid panel results in serum before and after FMT (Supplemental Table S2). After FMT, it was confirmed that triglyceride (TG; 140.25 versus 147.25 mg/dL, mean) and low-density lipoprotein (LDL; 100.75 versus 127.50 mg/dL, mean) levels increased. What is peculiar is that high-density lipoprotein (HDL; 49.75 versus 24.75 mg/dL, mean) levels are lowered after FMT. TG and LDL, bad cholesterol in our body, seem to increase while HDL decreases relatively, so additional research is needed. 38 Therefore, this study provided clinical clues indicating the effect of cognitive function after gut microbiota changes via lipid metabolism.
This study does have limitations. First, our follow-up period was only three months post-FMT, which might only partially capture long-term effects on cognitive function. Future studies should include longer-term follow-ups. Second, the assessment of cognitive function was restricted due to hospital policies. Future studies need to incorporate a broader range of cognitive function tests. Moreover, during our study, we could not evaluate dementia using brain magnetic resonance image (MRI), which would be beneficial for future studies to consider MRI evaluation over extended periods. Third, we check laboratory tests before and after FMT. Although further evaluation is needed, laboratory results showed that patients’ general condition before and after FMT did not significantly affect cognitive function (Supplemental Table S3). However, we could not reflect the required daily energy, including nutrition support status. Therefore, there seems to be a consideration between the patient’s nutritional status and gut microbiome for further study. Lastly, it is necessary to analyze the gut microbiota of patients with cognitive dysfunction in larger patient groups and across multiple centers.
In conclusion, this study suggests that FMT could offer a novel approach to mitigating cognitive decline in dementia patients by influencing the gut microbiota and related lipid metabolism. FMT could serve as an additional treatment option to complement existing therapies, thereby enhancing the overall management of cognitive decline. Furthermore, our findings suggest the potential use of gene expression related to lipid metabolism as a marker for cognitive decline. Future research should delve further into this promising intervention and its potential to improve the quality of life for individuals living with AD.
Supplemental Material
sj-docx-1-tan-10.1177_17562864231218181 – Supplemental material for Effect of altered gene expression in lipid metabolism on cognitive improvement in patients with Alzheimer’s dementia following fecal microbiota transplantation: a preliminary study
Supplemental material, sj-docx-1-tan-10.1177_17562864231218181 for Effect of altered gene expression in lipid metabolism on cognitive improvement in patients with Alzheimer’s dementia following fecal microbiota transplantation: a preliminary study by Jun-Seob Kim, Hyelim Park, Jung-Hwan Lee, Jongbeom Shin, Boram Cha, Kye Sook Kwon, Yong Woon Shin, Yerim Kim, YeoJin Kim, Jong Seok Bae, Ju-Hun Lee, Seok-Jin Choi, Tae Jung Kim, Sang-Bae Ko and Soo-Hyun Park in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The authors are sincerely grateful to the healthy volunteers and patients who voluntarily contributed their dedication to this research. All authors authorized the submission of the article. However, the final submission decision was made by the corresponding author.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
