Abstract
Background:
Generalized convulsive status epilepticus (GCSE) is one of the most challenging life-threatening neurological emergencies. If GCSE becomes super-refractory, it is associated with significant mortality. Although aggressive management of prolonged status epilepticus was conducted, the mortality has not decreased since the late 1990s.
Objectives:
The present study aimed to explore the risk factors for progression to super-refractory in patients with generalized convulsive status epilepticus (GCSE). Moreover, we illustrated the risk factors for mortality in GCSE patients.
Design:
An observational retrospective cohort study.
Methods:
We conducted a retrospective study of patients with GCSE admitted to our neurocritical unit, in Guangzhou, China, from October 2010 to February 2021. The data of sociodemographic information, etiology, laboratory results, treatment, and prognosis were collected and analyzed.
Results:
A total of 106 patients were enrolled; 51 (48%) of them developed super-refractory status epilepticus (SRSE). Multivariate logistic regression analysis demonstrated that patients with autoimmune encephalitis (p = 0.015) and intracranial infection (p = 0.019) are likely to progress to SRSE. The in-hospital mortality was 11.8% and 9.1% for patients in the SRSE and non-SRSE groups, respectively (p = 0.652). Multivariate logistic regression analysis showed that neutrophil-to-lymphocyte ratios (NLR) at admission were independently associated with in-hospital mortality. Up to 31.4% of SRSE patients and 29.1% of non-SRSE patients died within 6 months after discharge (p = 0.798). Multivariate logistic regression analysis showed that plasma exchange (PE) was a protective factor for 6-month mortality. A high NLR at discharge was a risk factor for 6-month mortality.
Conclusion:
In the current study, about 48% of GCSE patients progressed to SRSE. Regarding etiology, autoimmune encephalitis or intracranial infection was prone to SRSE. No significant differences were observed in the in-hospital and 6-month mortality between SRSE and non-SRSE groups. Multivariate logistic regression analysis showed that NLR at admission and discharge was an independent predictor of in-hospital and 6-month mortality, respectively. Moreover, PE significantly reduced the 6-month mortality.
Keywords
Introduction
Generalized convulsive status epilepticus (GCSE) is one of the most challenging life-threatening neurological emergencies. 1 Status epilepticus (SE) in adults and children >5 years old is defined as a seizure duration of 5 min or longer or consecutive seizures without returning to baseline for more than 5 min. 2 Refractory status epilepticus (RSE) occurs if the seizure duration is prolonged and sequential intravenous benzodiazepines followed by second-line anticonvulsants are ineffective. 3 Super-refractory status epilepticus (SRSE) occurs when seizures or recurrences persist after 24 h of continuous intravenous administration of anticonvulsants (including recurrences during tapering or discontinuation of anesthetics). 4
Although GCSE becomes mild after several hours, the excitotoxicity of persistent generalized epileptiform discharge may have deleterious effects on the brain. 5 Therefore, if GCSE becomes refractory or super-refractory, whether overt or mild, it is associated with significant morbidity and mortality. 6 For generalized SE, the mortality within the first 30 min is 3% and thereafter increased to 19%. 7 In a small study, the mortality for SRSE was 57.1% (8/14). 4
Previous studies have investigated the predictive factors for SRSE, while the results were inconsistent.4,8–10 Moreover, the optimal treatment for SRSE is unclear, and the current evidence is mainly based on the observational study and case series.4,11 Since the late 1990s, although aggressive management of prolonged SE was conducted, the mortality has not decreased. 12 Therefore, risk factors for the progression to SRSE and associated with mortality in SE patients need to be investigated further.
Herein, we conducted a single-center retrospective cohort study to investigate the risk factors for the development of SRSE in GCSE patients and illustrate the risk factors for mortality in such patients.
Materials and methods
Study design and setting
In this retrospective study, patients with GCSE who met the inclusion criteria were collected through the electronic medical record system. The sociodemographic data, clinical diagnosis, treatment history, and laboratory test results of the patients were collected. Also, the duration of the neurocritical unit (NCU) stay and the duration of mechanical ventilation (MV) were recorded. Glasgow Coma Scale (GCS) scores and Acute Physiology and Chronic Health Assessment II (APACHE II) scores were collected for each patient within 24 h of NCU admission. Modified Rankin scale (mRS) and GCS score were collected at discharge. Six months after discharge, the patients were followed up by telephone or at the outpatient clinic, and the information, including the mRS score, frequency of residual seizures, and antiepileptic drugs, was collected.
Selection of participants
We screened all patients with generalized SE admitted to the Department of Neurology, Nanfang Hospital, Southern Medical University, Guangzhou, China, from October 2010 to February 2021. The inclusion criteria were as follows: met the diagnostic criteria of GCSE, 2 admitted to NCU, age >16 years, and received long-term follow-up. The exclusion criteria were as follows: incomplete clinical data, mRS score ⩾2 before onset, terminal state, and loss to follow-up.
Standard protocol approvals, registrations, and patient consent
This was a single-center, observational, retrospective cohort study. The study proposal was approved by the Nanfang Hospital’s Ethics Committee on Clinical Research. Informed consent was waived by the review board because of its observational and retrospective design, and all data were fully de-identified. This manuscript adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Statistical analysis
Descriptive statistical analyses were performed on all variables of the patients. The categorical variables are expressed as numbers (%) and compared using a two-sided chi-square test or Fisher’s exact test. Continuous data were presented as mean ± standard deviation (SD) or median (interquartile range [IQR]) and analyzed using an independent t-test or Mann–Whitney U test. The candidate variable with p < 0.1 identified by univariate analysis was included in the multivariate logistic regression model. Risk factors were analyzed by logistic regression. We select the parameters used for the model by maximum likelihood estimation. Logistic regression odds ratios (ORs) with reported 95% confidence intervals (CIs) were calculated by likelihood ratio. All data analyses were conducted using SPSS Statistics for Windows (version 25.0. Armonk, NY: IBM Corp), and p < 0.05 indicated a statistically significant difference.
Data availability
Anonymized data related to the current article are available and will be shared upon request from any qualified investigator. Individuals interested in accessing these data should contact the corresponding author.
Results
Clinical characteristics of the enrolled patients
Of the 146 patients screened, 106 were considered eligible and included in the study (Figure 1). The mean age of the cohort was 37.8 ± 19.4 years, which consisted of 53 (50%) females. The mean APACHE II score at the time of admission was 16 ± 8, and the median NCU stay was 15 (4–31) days. In terms of etiology, 16 (15.1%) cases had electrolyte metabolism disorders, 4 (3.8%) were diagnosed with cerebrovascular disease, 25 (23.6%) presented autoimmune encephalitis, 30 (28.3%) cases had intracranial infections, 13 (12.3%) had epilepsy history, 8 (7.5%) had a history of brain disease, and 10 (9.4%) patients presented unknown reasons. Of the 106 patients enrolled, 82 (77.4%) progressed to RSE, of which 51 (48.1%) progressed to SRSE after 24 h of intravenous anesthesia therapy. Subsequently, 10.4% (11/106) patients died during hospitalization, of whom six patients died in hospital and five died due to discharge against advice because of Chinese tradition, hoping to die at home accompanied by the family. Finally, 30.2% (32/106) patients died during the 6-month follow-up, and 42.5% (45/106) patients achieved a good 6-month prognosis with mRS ⩽ 2.

The inclusion flowchart of the patients.
Demographic and clinical characteristics of SRSE and non-SRSE groups are listed in Table 1, including sociodemographic information, comorbidities, laboratory results, treatment, and outcomes. Compared to the non-SRSE group, the SRSE group was younger (25 versus 46, p = 0.002). SRSE patients had significantly lower GCS scores than non-SRSE patients on admission (5 versus 7, p = 0.002). The percentage of autoimmune encephalitis and intracranial infection was significantly higher in the SRSE than non-SRSE group (p = 0.023 and 0.049, respectively). The use of phenobarbital, glucocorticoid therapy, intravenous immunoglobulin (IVIG) therapy, and the ketogenic diet (Qitong milk; Shenzhen Zeneca Biological Co., Ltd, 16 Baoshan Road, Jinlong Avenue, Shenzhen, Guangdong, China.) was significantly higher in the SRSE than the non-SRSE groups. Compared to non-SRSE patients, SRSE patients had longer hospital and NCU stays (p = 0.000 and 0.001, respectively). However, no significant differences were observed in residual epilepsy or mRS scores at discharge and during the 6-month follow-up. Also, no significant differences were detected in in-hospital and 6-month mortalities between the two groups (p = 0.652 and 0.798, respectively).
Comparisons of clinical features between SRSE and non-SRSE patients.

APACHE II, Acute Physiology and Chronic Health Evaluation II; CSF, cerebrospinal fluid; GCS, Glasgow Coma Scale; IQR, interquartile range; IVIG, intravenous immunoglobulin; LOS, length of hospital stay; mRS, modified Rankin Scale; NCU, neurocritical care unit; NLR, neutrophil-to-lymphocyte ratio; SRSE, super-refractory status epilepticus.
Risk factors for SRSE
To explore the risk factors for SRSE, a multivariate logistic regression model was built by including relevant parameters and considering collinearity (Table 2). Progression to SRSE was more probable when the etiology was autoimmune encephalitis (OR 4.030, 95% CI: 1.314–12.363) and intracranial infection (OR 3.523, 95% CI: 1.232–10.076). Older age was a protective factor for SRSE (OR 0.956, 95% CI: 0.931–0.982). Severe coma (GCS ⩽ 8, on admission) was a risk factor for SRSE (OR 3.332, 95% CI: 1.194–9.293).
Logistic regression analysis of predictors for SRSE.

CI, confidence interval; GCS, Glasgow Coma Scale; OR, odds ratio; SRSE, super-refractory status epilepticus.
Risk factors for in-hospital mortality
In all, 11 (10.4%) patients died during hospitalization. The comparisons of clinical features between survivors and non-survivors are listed in Table 3. No significant differences were observed between the two groups with respect to gender, age, comorbidities, GCS score at admission, etiology, treatment, or SRSE/non-SRSE. APACHE II score at admission was lower among the survivors (p = 0.048). Patients who died in the hospital had relatively higher neutrophil-to-lymphocyte ratios (NLR) (p = 0.067) at admission than survivors. The length of hospital stay in deceased patients was significantly shorter than that in survivors (p = 0.011). Multivariate logistic regression analysis showed that NLR at admission (OR: 1.108, 95% CI: 1.010–1.214) was independently associated with in-hospital mortality (Table 4).
Comparisons of clinical features between in-hospital survivors and non-survivors.

APACHE II, Acute Physiology and Chronic Health Evaluation II; CSF, cerebrospinal fluid; GCS, Glasgow Coma Scale; IQR, interquartile range; IVIG, Intravenous immunoglobulin; LOS, length of hospital stay; mRS, modified Rankin Scale; NCU, neurocritical care unit; NLR, neutrophil-to-lymphocyte ratio; SRSE, super-refractory status epilepticus.
Logistic regression analysis of risk factors for in-hospital mortality.

APACHE II, Acute Physiology and Chronic Health Evaluation II; CI, confidence interval; NLR, neutrophil-to-lymphocyte ratio; OR, odds ratio.
Risk factors for 6-month mortality
Up to 30.2% (32/106) of patients died within 6 months after discharge. The comparisons of clinical features between 6-month survivors and non-survivors are listed in Table 5. No significant differences were observed between the two groups with respect to gender, age, comorbidities, GCS score at admission, APACHE II score at admission, etiology, or SRSE/non-SRSE. However, the utilization rate of plasma exchange (PE) during hospitalization was higher among the long-term survivors. Patients who died within 6 months had higher NLR at discharge than survivors. The length of hospital stay in deceased patients was significantly shorter than that in survivors. Patients in the deceased group had significantly lower GCS scores and higher mRS at discharge than the survivors. None of the patients in the deceased group had a good prognosis at discharge with mRS ⩽ 2.
Comparisons of clinical features between 6-month survived and expired patients.

APACHE II, Acute Physiology and Chronic Health Evaluation II; CSF, cerebrospinal fluid; GCS, Glasgow Coma Scale; IQR, interquartile range; IVIG, intravenous immunoglobulin; LOS, length of hospital stay; mRS, modified Rankin Scale; NCU, neurocritical care unit; NLR, neutrophil-to-lymphocyte ratio; SRSE, super-refractory status epilepticus.
Multivariate logistic regression analysis showed that PE was a protective factor for 6-month mortality in GCSE patients (OR: 0.205, 95% CI: 0.046–0.913). High NLR at discharge was a risk factor for 6-month mortality (OR: 1.093, 95% CI: 1.010–1.184) (Table 6).
Logistic regression analysis of risk factors for 6-month mortality.
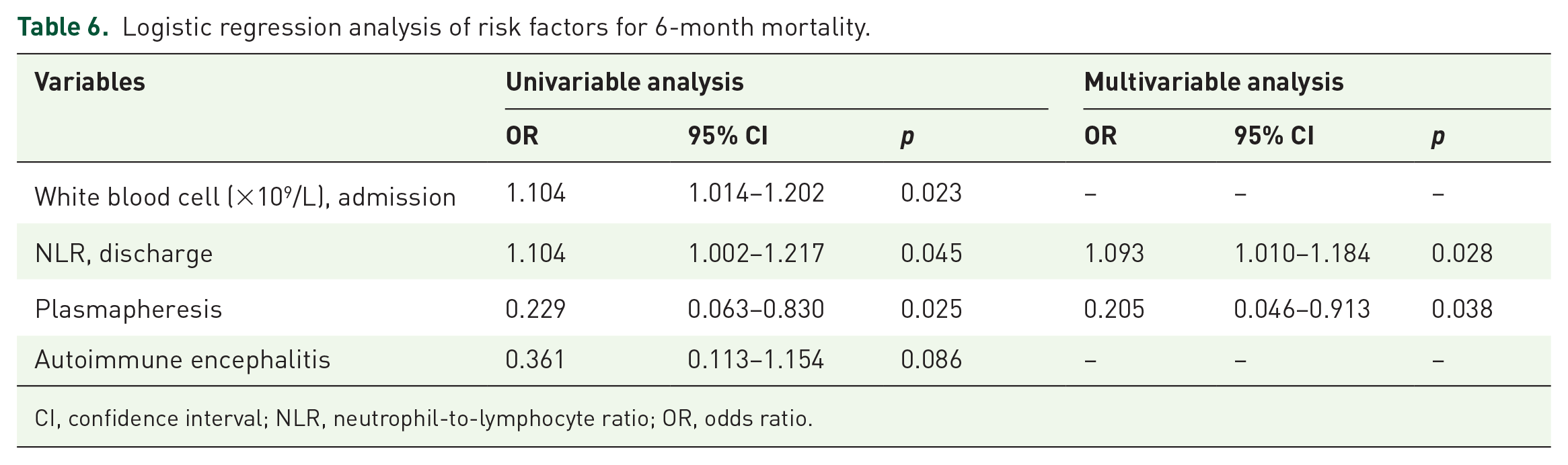
CI, confidence interval; NLR, neutrophil-to-lymphocyte ratio; OR, odds ratio.
Risk factors for 6-month prognosis
A total of 45 (42.4%) patients had favorable outcomes (mRS ⩽ 2) within 6 months after discharge. No significant differences were observed between the good-prognosis and bad-prognosis groups with respect to sex, age, comorbidities, etiology, length of hospital stay, length of NCU stay, treatment, or SRSE/non-SRSE. Patients with a good 6-month prognosis had significantly higher GCS scores and lower mRS scores at discharge (Table 7).
Comparisons of clinical features between patients with good and poor 6-month prognosis.
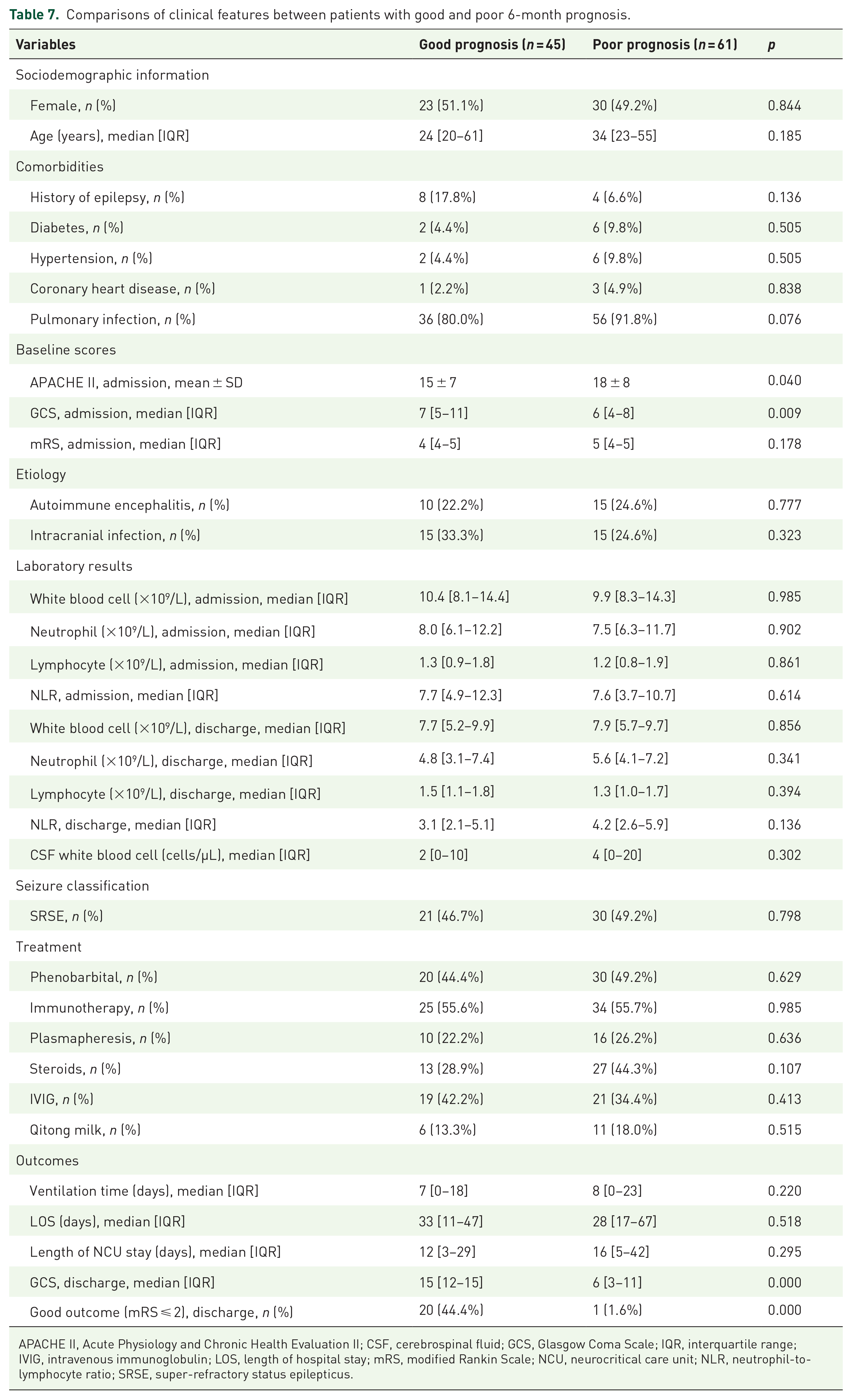
APACHE II, Acute Physiology and Chronic Health Evaluation II; CSF, cerebrospinal fluid; GCS, Glasgow Coma Scale; IQR, interquartile range; IVIG, intravenous immunoglobulin; LOS, length of hospital stay; mRS, modified Rankin Scale; NCU, neurocritical care unit; NLR, neutrophil-to-lymphocyte ratio; SRSE, super-refractory status epilepticus.
Comparisons of clinical features according to etiology
According to the etiology, we further divided the patients into autoimmune encephalitis and non-autoimmune encephalitis groups. Compared to the non-autoimmune encephalitis group, the autoimmune encephalitis group predominantly consisted of females. The duration of MV, hospital stay, and NCU stay were significantly prolonged and percentages of phenobarbital, immunotherapy, and SRSE were significantly higher in the autoimmune encephalitis group than in the non-autoimmune group. Interestingly, the autoimmune encephalitis group patients had higher mRS scores at discharge, while the 6-month prognosis was not different from the non-autoimmune encephalitis group (Table 8).
Comparisons of clinical features between patients with autoimmune encephalitis and non-autoimmune encephalitis.
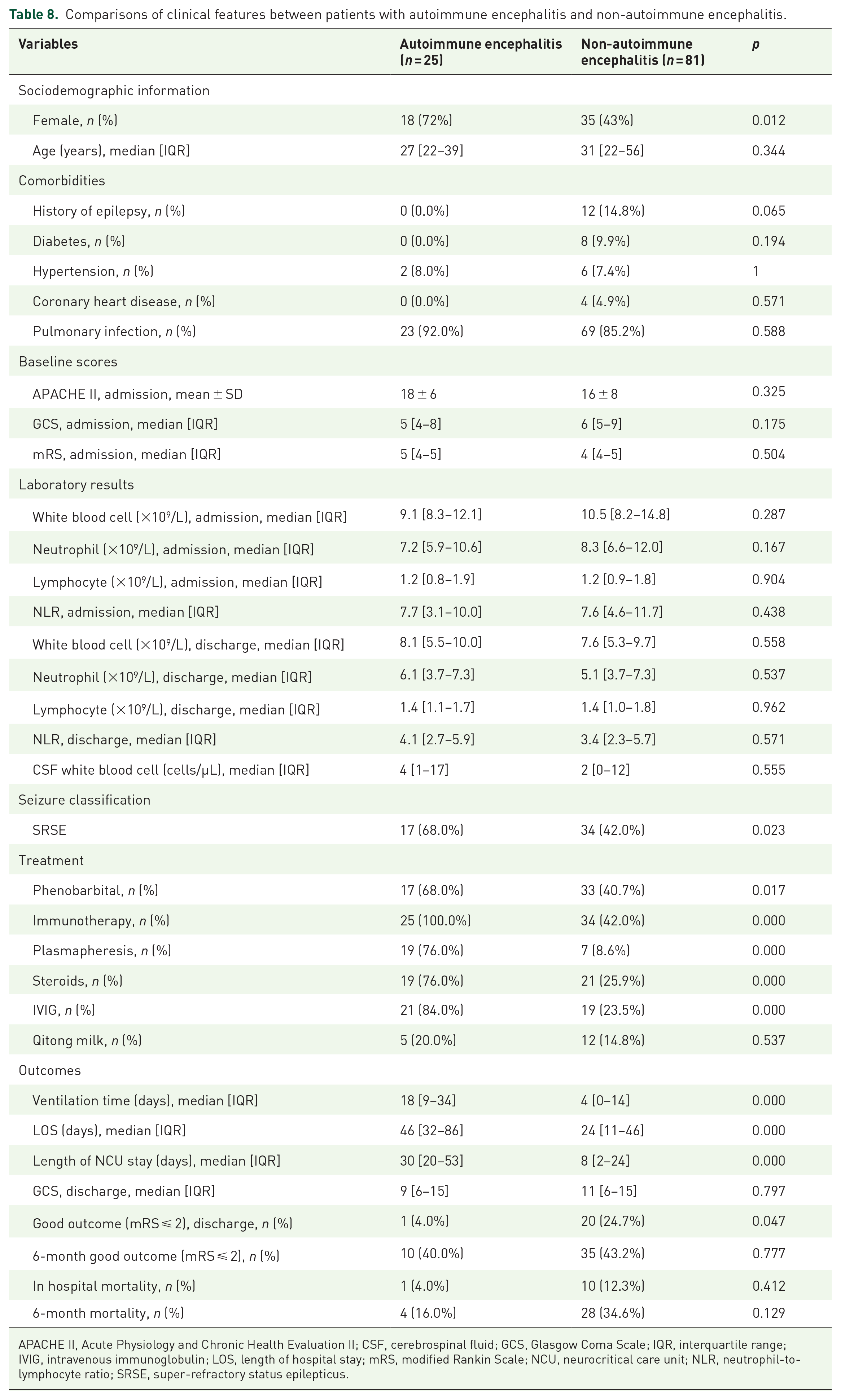
APACHE II, Acute Physiology and Chronic Health Evaluation II; CSF, cerebrospinal fluid; GCS, Glasgow Coma Scale; IQR, interquartile range; IVIG, intravenous immunoglobulin; LOS, length of hospital stay; mRS, modified Rankin Scale; NCU, neurocritical care unit; NLR, neutrophil-to-lymphocyte ratio; SRSE, super-refractory status epilepticus.
Discussion
In this 10-year single-center cohort study in a tertiary teaching hospital in South China, we observed that the incidence of progression to SRSE was 48.1% in GCSE patients. Patients with autoimmune encephalitis and intracranial infection were prone to SRSE. The overall in-hospital and 6-month mortality was 10.4% and 30.2%, respectively. No obvious differences were observed in the short- and long-term mortality between SRSE and non-SRSE groups. Multivariate logistic regression analysis showed that NLR at admission was independently associated with in-hospital mortality. PE significantly reduced 6-month mortality, and NLR at discharge was a predictor of 6-month mortality. We are a tertiary center for critically ill patients in South China. Some of the patients who have been transferred to our center were quite severe. As our center is good at central nervous system infections and autoimmune encephalitis, doctors from nearby hospitals prefer to send such patients to our center. Due to these reasons, we have selection bias in this cohort. As a retrospective single-center study with substantial patient selection bias, all the results may not be representative. Therefore, all the data were based on this cohort and should be interpreted with caution.
In the current study, we found that GCSE triggered by autoimmune encephalitis or intracranial infection was prone to SRSE, which is consistent with previous findings.13–15 Shorvon 4 concluded that the etiology of SRSE is a severe acute brain injury. Tian et al. 13 showed that acute encephalitis is a critical etiology of SRSE. Similarly, in another study, 14 a cohort of SRSE also showed encephalitis as the most common etiology and a major factor in the progress from non-refractory SE to SRSE. A recently published meta-analysis of SRSE 15 revealed that in SRSE patients, acute cerebral events (including acute stroke, intracerebral hemorrhages, infectious disease, and autoimmune encephalitis) and unknown etiologies accounted for 41.6% and 22.3% of all etiologies, respectively. In this study, the percentage of autoimmune encephalitis and intracranial infections accounted for a quite high percentage in the SRSE group with patients’ bias. Therefore, GCSE patients induced by autoimmune encephalitis or intracranial infection tended to progress to SRSE, necessitating caution. In this study, we found that younger patients were more likely to develop SRSE than older patients; however, the results were inconsistent. Some studies revealed that older people had a poor controllable rate of GCSE at 1 h and were inclined to progress to RSE.9,16–18 A few studies showed that age was not a statistically significant factor for this progression.13,19 In the study by Kellinghaus et al., 9 the median age of the GCSE group was 65 years, and the etiology of the patients was mainly acute symptomatic. On the other hand, in our study, the mean age was 38 years, and autoimmune encephalitis or intracranial infection comprised >50% of patients. Therefore, the different results might be caused by the selection bias of the patients and should be interpreted carefully. Some multi-center studies with a large patient number are needed to clarify this issue.
Reportedly, the in-hospital mortality of GCSE was about 20% and could be about 40% if SE remained refractory.3,20–23 A systematic review and meta-analysis of convulsive status epilepticus (CSE) mortality in high-income countries from 1990 to 2017 showed an overall mortality rate of 15.9%. 13 A randomized controlled trial (RCT) involving 66 CSE patients in China showed that 10.6% of them died during hospitalization, and 25.8% had residual symptomatic epilepsy. 24 In the present study, the in-hospital mortality was 10.4% and increased to 30.2% within 6 months after discharge. The whole mortality rate was similar to that reported previously. In the recently published meta-analysis, 15 the in-hospital mortality in SRSE patients was 24.1%. In this study, the mortality of in-hospital mortality and 6 months after discharge in the SRSE group were 11.8% and 31.4%, respectively. In this study, the low in-hospital mortality and high proportion with low mRS after discharge were due to the selection bias of patients we have mentioned before. Second, some critically ill patients were discharged for local cultural reasons. In some rural areas of China, patients prefer to die at home rather than in a hospital, which leads to low in-hospital mortality and a considerably high mortality after discharge.
However, we did not observe any marked differences in the short- and long-term mortality between SRSE and non-SRSE groups, which was contradictory to previous findings.6,14,25,26 These diverse findings could also be ascribed to divergence in the study population. The patients were young, and most had encephalitis. Also, the usage of anesthetics (phenobarbital) and immunotherapy was aggressive in our study, which controlled the condition in most patients after treatment. We suggested that for SRSE patients, doctors should try their best to control the seizures and patients might achieve good prognosis though the seizures were super-refractory.
In a global survey 27 of 488 RSE cases, the most commonly used anesthetic was midazolam (59%), followed by propofol (32%) and barbiturates (8%). According to the guidelines of SE,12,28,29–32 intravenous infusion of midazolam and propofol was an alternative for RSE patients. Phenobarbital infusion was suggested if the above therapy failed. In an RCT 30 of sodium valproate versus phenobarbital, phenobarbital had a higher rate of SE termination than sodium propionate. Due to the high termination rate and low price of phenobarbital, it was widely used in our center. In this study, 50 SRSE patients received phenobarbital of whom only one patient failed treatment. The result was slightly different from the current meta-analysis 15 which revealed that phenobarbital revealed a nonsignificant trend toward worse outcomes at discharge. Based on this study, we suggested that phenobarbital might benefit the patients by the high prevalence of seizure control.
Over the past decades, several immunomodulatory therapies 31 have been suggested as the first-line immunotherapy for SE, including corticosteroids, IVIG, and PE 32 . Recently, international consensus for the management of new onset refractory status epilepticus (NORSE) recommended that with potential immune activation, first-line immunotherapy should be considered within 72 h after seizure. When the first-line immunotherapy response is insufficient, the second-line immunotherapy and ketogenic diet should be considered within 7 days after the seizure. 31 In this study, >50% of RSE patients (32% with PE, 49% with IVIG, and 49% with steroids) received immunotherapy, and we observed that PE significantly reduces 6-month mortality. Previous studies focused on PE in RSE got controversial results. In a systematic review 33 about PE in adult autoimmune RSE, seizure response with the application of PE therapy occurred in 14/27 (51.9%) patients, with 1 (3.7%) and 13 (48.1%) displaying partial and complete responses, respectively. Such results did not support the routine application of PE in adult autoimmune RSE. A previous small cohort study of 18 NORSE patients found that immunotherapy including PE did not change the prognosis of NORSE patients. 34 A RCT 35 of NORSE with immunotherapy (corticosteroids, IVIG, and PE) showed that it improves the prognosis (mRS 0–2) at hospital discharge and during follow-up despite early or late application. In this study, autoimmune encephalitis was the main etiology and PE was the first-line therapy for such patients, which may cause a different result. Therefore, the impact of immunotherapy, especially PE, on the prognosis and mortality of GCSE patients requires further investigation.
Another interesting result in this study was that the NLR at admission and discharge is an independent predictor of in-hospital and 6-month mortality, respectively. Accumulating evidence36,37 suggested that peripheral blood NLR can be used as a marker of systemic inflammation. High NLR is independently associated with 90-day poor outcomes in critically ill stroke patients. 38 We also observed the predictive value of NLR in GCSE mortality, suggesting that clinicians should focus on this index. However, due to the observational study with limited data, more research was warranted to confirm the result.
Nevertheless, the present study has several limitations. First, this was a retrospective, single-center study with unavoidable selection bias of the patients, and the research sample needs to be expanded. Second, as a tertiary referral center, the distribution of the study population was different from the other studies, which could cause bias. Third, as a retrospective study, we can only explore the results in the observational cohort, which was indeed a way of fishing in data. Therefore, the results and conclusions we have obtained should be interpreted carefully.
In summary, about 48% of GCSE patients in the current study developed SRSE. SE triggered by autoimmune encephalitis or intracranial infection was prone to SRSE. The overall in-hospital and 6-month mortality was 10.4% and 30.2%, respectively. Multivariate logistic regression analysis showed that NLR at admission and discharge was an independent predictor of in-hospital and 6-month mortality, respectively. PE significantly reduced the 6-month mortality.
Footnotes
Acknowledgements
The authors thank all the staff of the medical intensive care unit for their support during the study. The corresponding author had full access to all the data in the study and took responsibility for the integrity of the data and the accuracy of the analysis.
