Abstract
Background:
Combination therapy with antiseizure medications (ASMs) is a rational strategy if monotherapy cannot effectively control seizures, thereby aiming to improve tolerance and treatment persistence.
Objectives:
To compare the efficacy of different ASM combinations among patients.
Design:
Patients with epilepsy on monotherapy who had a second ASM added as concomitant two-drug therapy from January 2009 to May 2019 in the Chang Gung Research Database, Taiwan, were included in the analysis.
Methods:
ASM combinations were compared based on their primary mechanism of action (MoA) which are as follows: gamma-aminobutyric acid receptor (G), sodium channel blocker (SC), synaptic vesicle protein 2A (SV2), calcium channel blocker (C), and multiple mechanisms (M). Treatment persistence was compared, and the predictors of persistence were analyzed.
Results:
In total, 3033 patients were enrolled in this study. Combined ASMs with different MoAs had significantly longer treatment persistence than ASMs with similar MoAs, specifically SC and M combinations. Patients receiving combined ASMs with different MoAs were less likely to discontinue treatment [adjusted hazards ratio: 0.83 (95% CI: 0.75–0.93), p < 0.001]. Among all combinations, the SC + SV2 combination had the longest treatment persistence (mean ± SD: 912.7 ± 841.6 days). Meanwhile, patients receiving the G combination had a higher risk of treatment discontinuation than those receiving the SC + SV2 combination. Underlying malignancies were associated with an increased risk of treatment discontinuation across all MoA categories. Male patients receiving the SC, SV2, and M combinations were more likely to discontinue treatment than female patients. Moreover, patients with renal disease were more likely to discontinue treatment with the SV2 combinations.
Conclusion:
ASM combinations with different MoAs had superior efficacy and tolerability to ASM combinations with similar MoAs, particularly SC and M combinations. In our cohort, factors associated with treatment discontinuation included underlying malignancy, male sex, and renal disease. These findings may provide valuable insights into the use of ASM combinations if monotherapy cannot adequately control seizures.
Introduction
Antiseizure medication (ASM) therapy is still the mainstay for seizure control in patients with epilepsy. However, extensive cohort studies have shown that 30–35% of patients who receive ASM therapy are refractory to treatment.1,2 Patients with uncontrolled epilepsy have a reduced quality of life, 3 a substantially high frequency of comorbid illnesses, 4 and an increased risk of mortality. 5 Therefore, the management of drug-resistant epilepsy has gained increasing attention. Besides resection surgery for epileptic foci and electrical stimulation of the nervous system that are only suitable to specific patients with drug-resistant epilepsy,6,7 ASM therapy should be optimized in nearly all patients with epilepsy who are yet to achieve seizure freedom. In a large multicenter observational study, ASM adjustment was associated with seizure freedom in one of five patients with drug-resistant focal epilepsy. 8 Several studies have shown that seizure freedom may be achieved by resolving diagnostic uncertainties and optimizing ASMs in 10–40% of patients with uncontrolled chronic epilepsy.9–11 Furthermore, newer ASMs have additional efficacy and better tolerance. 12
If monotherapy fails to control seizures, ASM combination therapy is considered a rational strategy to improve tolerance and treatment persistence rather than shifting to another ASM monotherapy. A recent clinical study assessed the efficacy of different treatment options after the first failed ASM therapy. ASM combination therapy was superior to increased ASM dose or another ASM monotherapy. 13 Among the methods used to optimize ASM polytherapy, a combination of ASMs with different mechanisms of action (MoAs) is considered optimal. It is the most commonly used strategy to maximize treatment efficacy and decrease adverse events caused by ASMs with overlapping mechanisms. In addition, a synergistic effect leading to increased efficacy compared with the sum of separate drug effects is recommended with valproate–lamotrigine combination compared with phenytoin–carbamazepine–lamotrigine combination. 14 Margolis et al. performed a large population-based study. Results showed that the combinations of ASMs with different MoAs were associated with longer treatment persistence, decreased risk of therapy discontinuation, and lower rates of inpatient admissions and emergency room visits compared with the combinations of ASMs with similar MoAs. 15 Nevertheless, a more detailed classification of MoAs may better identify the benefits of ASM combination treatment with a specific mechanism. For example, gabapentin and pregabalin, classified as gamma-aminobutyric acid (GABA) analogs in the study by Margolis et al., 15 are currently classified as calcium channel blockers. 16 In addition, comorbidities affect the outcomes of patients with epilepsy. Previous studies have reported that different types of comorbidities, including psychiatric disorders, cerebrovascular and ischemic heart diseases, and malignant neoplasms, can affect the degree of seizure control, healthcare utilization, and mortality in patients with epilepsy. 17
Therefore, this study primarily aimed to compare the efficacy of different ASM combinations in treating epilepsy. Furthermore, it secondarily aimed to analyze factors affecting the treatment outcomes of patients with comorbidities who are receiving ASM combination therapy.
Participants and methods
Patient selection and study protocol
We conducted a retrospective review of patients diagnosed with epilepsy based on the electronic medical records of Chang Gung Memorial Hospital (CGMH) (Chang Gung Research Database, CGRD), Taiwan, from January 2009 to May 2019. CGMH is the most extensive hospital system in Taiwan, comprising seven medical institutions from the northeast to southern regions of Taiwan. 18 In the CGRD, all diseases are coded according to the International Classification of Diseases, Ninth or Tenth Revision, Clinical Modification (ICD-9-CM, ICD-10-CM). The cohort included patients diagnosed with epilepsy (ICD-9-CM 345.x, 780.3/ICD-10-CM G40.x, or R56.x) and those who received ASMs, defined as the Anatomical Therapeutic Chemical Classification (ATC) code N03A. The index date (day 0) was defined as the date when the second ASM used as combination treatment was initiated. The following patients were included: (1) those who received ASM monotherapy for >6 months prior to the index date and (2) those who received continuous treatment with concomitant ASM therapy for at least 60 days. This criterion was set because premature withdrawal from the ASM combination could be attributed to intolerable adverse effects or allergic reactions rather than treatment efficacy that may be affected by a lower initial dosage during the titration period. 19
Each ASM was classified into one of the following categories according to the presumed primary MoA: sodium channel blocker (SC), GABA receptor (G), calcium channel blocker (C), synaptic vesicle protein 2A binding (SV2), α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor (AMPA), and multiple mechanisms (M) (Table 1). 20 An alternative classification of ASM into older and newer generations was used to assess the role of newer-generation ASM in treatment persistence (Supplemental Table S1). 21 The exclusion criteria were as follows: patients who received benzodiazepine combinations (e.g. concomitant use of low-dose clonazepam and diazepam) as sleep aids and those receiving ASMs with infrequently used mechanisms (i.e. AMPA). To prevent bias caused by groups with a small sample size, only the top 11 MoA combinations were reported and included for further analysis, accounting for 99% (3033/3065) of all cases in this study. Figure 1(a) shows the process of patient selection.
Taxonomies of ASMs based on primary mechanism of action.

ASM, antiseizure medication.

(a) Study flowchart of patient selection. (b) Combination of antiseizure medications based on the MoA.
Demographic and clinical variables
Data of the baseline (day 0) characteristics of each patient receiving the MoA combination were recorded. Furthermore, information about the following clinical characteristics of each patient was collected: age, sex, and body mass index. Data on comorbid conditions including liver disease (ICD-9-CM 570.x-573.x/ICD-10-CM K70.x-K77.x), renal disease (ICD-9-CM 585.x, 586.x, or 593.9/ICD-10-CM N18.x-N19.x), cerebrovascular disease (ICD-9-CM 430.x-438.x/ICD-10-CM I60.x-I69.x), diabetes mellitus (ICD-9-CM 250.x/ICD-10-CM E08.x-E13.x), dyslipidemia (ICD-9-CM 272.x/ICD-10-CM E78.x), malignancy (ICD-9-CM 140.x-209.x/ICD-10-CM C00.x-C96.x), hypertension (ICD-9-CM 401.x-405.x/ICD-10-CM I10.x-I16.x), ischemic heart disease (ICD-9-CM 410.x-414.x/ICD-10-CM i20.x-I25.x), heart failure (ICD-9-CM 428.x/ICD-10-CM I50.x), psychotic disorders (ICD-9-CM 295.x-299.x/ICD-10-CM F06.0-F06.2, F20.x-F29.x), mood/anxiety disorders (ICD-9-CM 300.x/ICD-10-CM F06.3-F06.4, F30.x-F39.x, F40.x-F48.x), dementia (ICD-9-CM 294.1, 294.2, 331.0, 331.1, 331.82/ICD-10-CM F01.x-F03.x, G30.x, G31.09, G31.83), and intellectual disabilities (ICD-9-CM 317.x-319.x/ICD-10-CM F70.x-F79.x) during physician office visits at baseline were also recorded.
Outcome measurements
The primary study outcome was the efficacy of each combination treatment based on persistence, defined as the number of days from the beginning to the end of the combination treatment or the end of available data (May 2019).
Statistical analysis
Statistical analyses were performed using the SAS software version 9.4 (Copyright © SAS Institute Inc., SAS Campus Drive, Cary, North Carolina 27513, USA). The duration of ASM combination therapy was expressed as mean (standard deviation, SD) and median (Q1–Q3). The Cox proportional hazards models were used to assess the independent predictors of treatment discontinuation during ASM combination therapy. Analysis of variance was used in analyzing treatment persistence among groups. All analyses were two-sided, and a p-value of <0.05 was considered statistically significant. The Center for Big Data Analytics and Statistics (CGMH), Linkou, assisted in statistical and data analysis.
Standard protocol approvals, registrations, and patient consent
The current study protocol was approved by the Institutional Review Board of Chang Gung Medical Foundation, Taiwan. Patient information in the CGRD was deidentified and anonymized before being released to the researchers. The need for informed consent was waived by the Institutional Review Board of Chang Gung Medical Foundation.
Data availability
The data used to conduct the research will be made available by the corresponding author upon reasonable request and subject to approval by the Chang Gung Medical Foundation, Taiwan.
Results
Research samples
We identified 60,140 patients diagnosed with epilepsy during the study period. Among them, 43,539 (72.4%) received ASM treatment. Of these patients, 17,459 (40.1%) received ASM monotherapy, and 26,080 had two or more ASMs. In total, 3033 (1693 men and 1337 women) patients who met the study criteria were included in the final analysis [Figure 1(a)] and were categorized based on their index ASM combination. Table 2 shows the baseline clinical characteristics of the patients.
Patient characteristics.
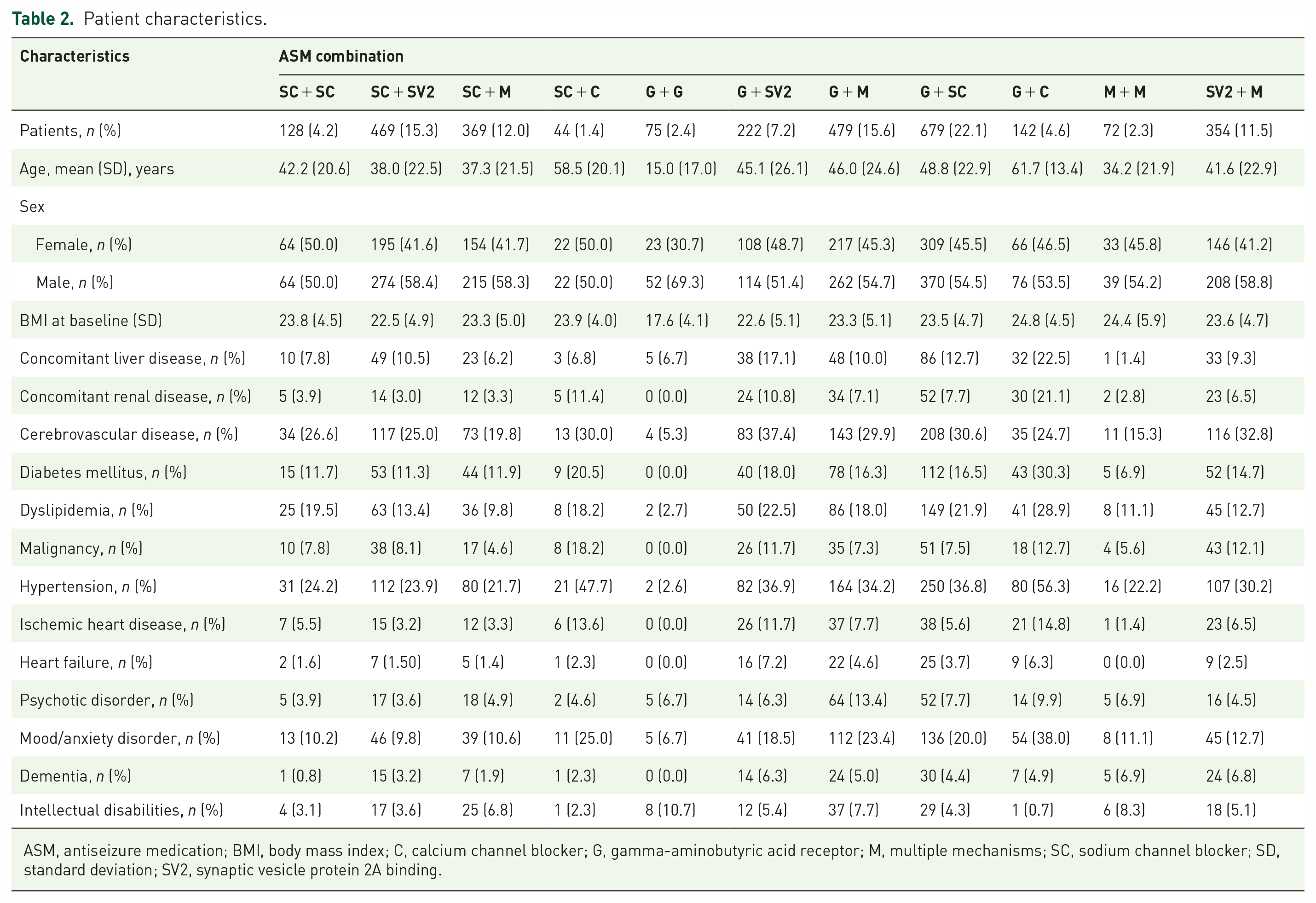
ASM, antiseizure medication; BMI, body mass index; C, calcium channel blocker; G, gamma-aminobutyric acid receptor; M, multiple mechanisms; SC, sodium channel blocker; SD, standard deviation; SV2, synaptic vesicle protein 2A binding.
ASM combinations and characteristics of the patients
SC combinations (55.9%) were the most common treatment, followed by G (52.7%), M (42.6%), SV2 (34.9%), and C (7.1%) combinations. Approximately 91.1% of patients received ASM combinations with different MoAs. Meanwhile, 8.9% were treated with ASMs with similar MoAs.
The G + SC combination (22%) was the most frequently used ASM combination, followed by G + M (16%) and SC + SV2 (15%) combinations [Figure 1(b)]. The mean age ± SD of the patients ranged from 15.0 ± 17.0 to 61.7 ± 13.4 years. The patients who received G ± C combinations were generally older than those who received SC + C combinations (61.7 ± 13.4 years versus 58.5 ± 20.1 years). The patients who received G + G combinations were the youngest, with a mean age of 15.0 ± 17.0 years. Furthermore, they had a lower percentage of female participants (30.7%) than those who received other ASM combinations (41.2–50.0%). Patients who received G + C combinations had the highest percentage of comorbidities at baseline, including concomitant hypertension, mood or anxiety disorders, diabetes mellitus, dyslipidemia, liver disease, renal disease, and ischemic heart disease (Table 2).
Treatment persistence and risk of ASM combination discontinuation
The overall treatment persistence ranged from 466.5 ± 472.3 (G + C combinations) to 912.7 ± 841.6 (SC + SV2 combinations) days (Table 3). Patients who received SC combinations with different MoAs had a longer persistence than those who received combinations with similar MoAs. By contrast, patients who received M combinations with different MoAs, except G + M combinations that had shorter persistence than M + M combinations, had a trend toward longer persistence. However, among patients who received G combinations, although G + SC combinations had the longest persistence (782.1 ± 846.0 days), the persistence of G + G combinations (757.3 ± 772.8 days) was longer than that of G + M (700.8 ± 770.6 days), G + SV2 (647.6 ± 633.2 days), and G + C (466.5 ± 472.3 days) combinations (Supplemental Figure S1). In addition, the combination of older and older; older and newer; and newer and newer-generation ASMs did not exhibit any significant difference in terms of persistence (p = 0.77) (Supplemental Figure S2).
Duration of ASM combination therapy (unadjusted).

ASM, antiseizure medication; C, calcium channel blocker; G, gamma-aminobutyric acid receptor; M, multiple mechanisms; SC, sodium channel blocker; SV2, synaptic vesicle protein 2A binding.
The Cox proportional hazards models were used to evaluate the risk of ASM combination discontinuation. Results showed that the ASM combinations with different MoAs had a lower risk of discontinuation than ASM combinations with similar MoAs [adjusted hazard ratio (HR): 0.83 (95% CI: 0.75–0.93), p < 0.001] [Figure 2(a)]. In patients who received ASM combinations with different MoAs, the risk of discontinuation in various combination categories was further compared with combinations with the longest persistence, with the SC + SV2 combination selected as a reference. Except in patients who received G + SC combinations, those who received other G combinations had a significantly higher risk of discontinuation [G + G-HR: 1.31 (95% CI: 1.12–1.54), p < 0.001; G + M-HR: 1.24 (95% CI: 1.09–1.42), p < 0.001; and G + C-HR: 1.60 (95% CI: 1.31–1.96), p < 0.001] than those who received SC + SV2 combinations. There were no significant differences in the risk of treatment discontinuation between patients who received SC + SC, SC + M, SC + C, M + M, and SV2 + M combinations compared with those who received SC + SV2 combinations [Figure 2(b)].
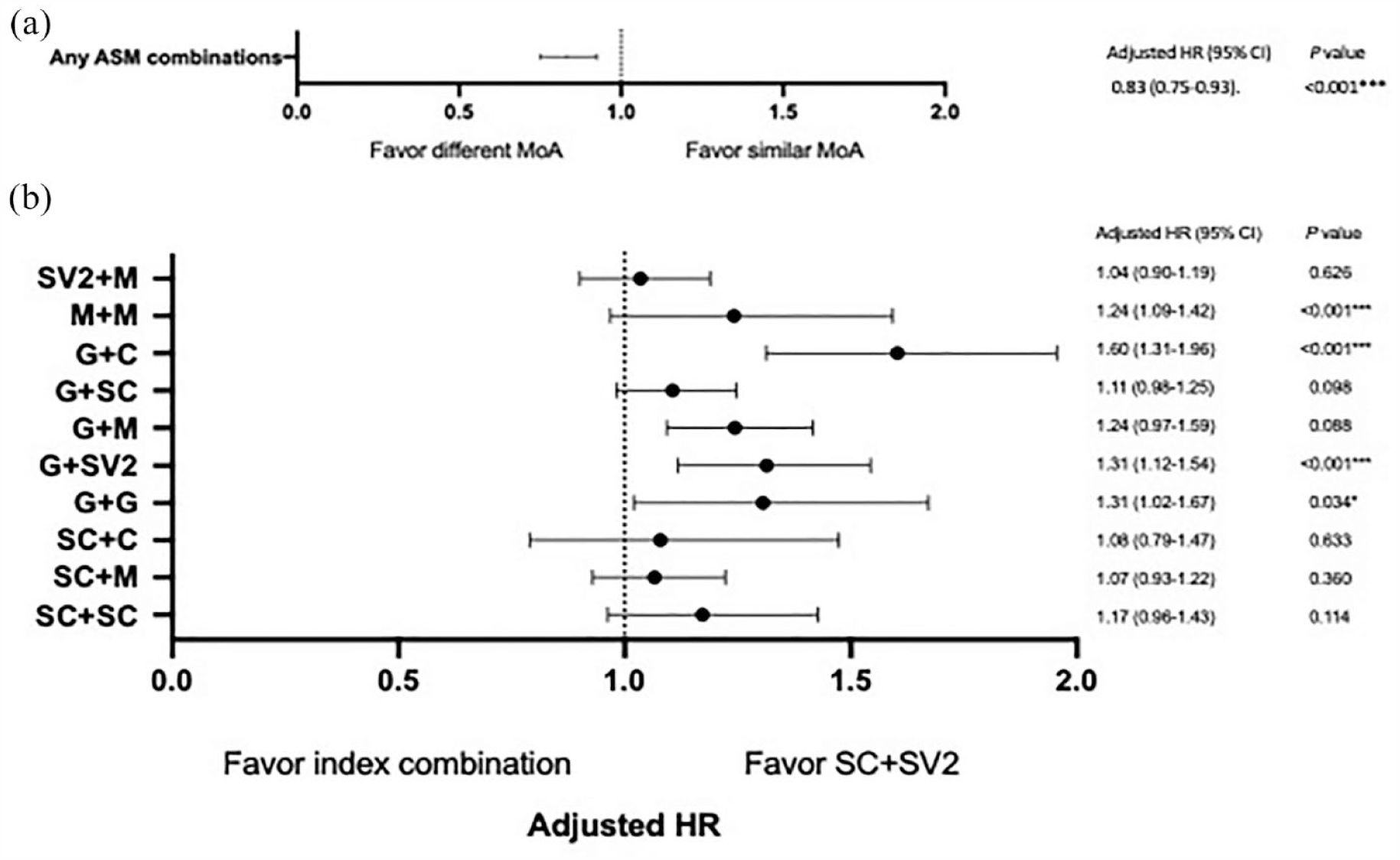
(a) The risk of discontinuation between ASM combinations with different and similar MoAs. (b) The risk of discontinuation between index combinations and SC + SV2 combinations.
According to individual MoA, the hazard models did not show any significant differences between ASM combinations with similar and different MoAs in terms of the risk of discontinuing G, SC, and M combinations. Regarding SV2 combinations, levetiracetam (LEV) was the only SV2 ASM available during the study period. The benefits of specific SV2 (LEV)-based ASM combinations remain unclear. Therefore, we compared the differences among patients who received concomitant SV2 and other MoA ASMs. Patients receiving SC + SV2 and M + SV2 combinations had a lower risk of treatment discontinuation compared with those receiving G + SV2 combinations as a comparator. The risk of discontinuation was similar between SC + SV2 and M + SV2 combinations. As for C combinations, none of the patients in our cohort used the C + C combination. Hence, we compared the differences between patients who received ASMs with C and other MoAs. Patients who received SC + C combinations had a lower risk of treatment discontinuation compared with those who received G + C combinations. In particular, the hazard model further revealed that underlying malignancy increased the risk of treatment discontinuation across all five MoA categories. Male patients were more likely to discontinue combination treatment (SC, SV2, and M combinations) than female patients (all p < 0.05). In addition, patients with renal disease were significantly more likely to discontinue SV2 combinations (Table 4).
Hazard ratios of discontinuation risk in index ASM combination.

All ASM combination variables were included in the table. Model variables that were not statistically significant were not presented in the table (factors taken into analysis include age, male sex, concomitant liver disease, concomitant renal disease, cerebrovascular disease, diabetes mellitus, dyslipidemia, malignancy, hypertension, ischemic heart disease, heart failure, psychotic disorder, mood/anxiety disorder, dementia, and intellectual disabilities).
Hazard ratios were adjusted for age, male sex, and comorbidities.
ASM, antiseizure medication; C, calcium channel blocker; G, gamma-aminobutyric acid receptor; M, multiple mechanisms; SC, sodium channel blocker; SV2, synaptic vesicle protein 2A binding.
p < 0.05. **p < 0.01.
Discussion
Our study showed that patients who received ASM combinations with different MoAs were less likely to discontinue treatment than those who received ASM combinations with similar MoAs, particularly SC and M combinations. Furthermore, patients who received G combinations had a significantly higher risk of treatment discontinuation than those who received SC + SV2 combinations that had the longest persistence. Underlying malignancy was associated with an increased risk of treatment discontinuation across all five MoA categories, and patients with renal disease were more likely to discontinue SV2 combinations.
No randomized controlled trials have investigated the efficacy of different ASM combinations. However, real-life experience in using ASM combinations for treating epilepsy, as in this study, is valuable. Results showed that treatment persistence was generally more extended, and patients receiving combinations of ASMs with different MoAs had a significantly lower risk of treatment discontinuation than those receiving combinations of ASMs with similar MoAs. If ASM monotherapy cannot effectively control seizures, ASM combination therapy with different MoAs is a rational method to achieve better seizure control due to potential synergistic effects while preventing side effects caused by ASM combination therapy with similar MoAs.22,23 This is supported by several clinical studies showing that ASM combinations with different MoAs had superior efficacy or persistence to ASM combinations with similar MoAs.14,15,24 Treatment persistence was used as the primary outcome measure to estimate the efficacy of our study. Persistence is the length of time patients continually receive a similar ASM combination. It has been used in several previous studies on ASM treatment and reflects a composite of optimal seizure control and minimal adverse effects associated with a novel drug regimen.15,25 We found that the patients who received ASM combinations with similar MoAs (SC + SC and M + M) had a shorter persistence than those receiving ASM combinations with different MoAs. This result is consistent with that of a previous study. 15 However, unlike the patients in the study by Margolis et al., the patients who received G + G combinations in this research had a longer persistence than those treated with G + M, G + SV2, and G + C combinations. This could be explained by a difference in MoA categories. G combinations comprised gabapentin and pregabalin in the study by Margolis et al. Meanwhile, these two ASMs were classified as C combinations in our cohort. Therefore, further studies using more detailed MoAs and parameters that were more specific to the efficacy of seizure control, such as reduced seizure frequency, responder rates, and seizure remission, should be conducted to validate our findings.
In this study, the Cox proportional hazards model analysis revealed that patients who received ASM combinations with different MoAs had a lower risk of treatment discontinuation than those who received ASM combinations with similar MoAs. ASM combination with similar MoAs is generally believed to increase the risk of adverse effects and decrease the possibility of achieving effective seizure control compared with ASM combination with different MoAs. 22 Interestingly, patients who received G combinations had a higher risk of treatment discontinuation than those who received combinations with other mechanisms. GABA type A receptor is a significant target of various types of ASMs. However, tolerance induction limits their use for the long-term control of seizures. 26 Due to the increased requirement for high-dose treatment for seizure control, the risk of treatment discontinuation caused by adverse effects, including somnolence, dizziness, gait disturbance, and behavioral problems increases.27,28
In addition, to compare ASM combinations with similar and different MoAs, the beneficial effects of specific ASM combinations with different MoAs or their association with the possibility of treatment continuation were further investigated. Results found that the SV2 + SC combination had the longest persistence. Hence, it had good efficacy/tolerability compared with G combinations in patients with epilepsy. SV2 and SC mechanisms are both distinct targets to decrease neuron hyperexcitation that plays an important role in antiseizure polytherapy. In previous studies, the number of patients who respond to SV2 + SC combinations, such as levetiracetam/lamotrigine and levetiracetam/lacosamide, is higher than that of patients who respond to other combinations. Hence, nonoverlapping mechanisms have a beneficial pharmacodynamic interaction.29,30 However, a supra-additive effect was not observed if levetiracetam was combined with other SC blockers, such as phenytoin and carbamazepine, in the same study. 29 Further prospective randomized studies should be conducted to validate whether the efficacy of SV2 + SC combinations is a class effect or somewhat related to a specific medication.
Furthermore, comorbidities were found to be associated with an increased risk of index ASM combination discontinuation. First, patients with underlying malignancy had a higher risk of treatment discontinuation across all five MoA categories. Malignancy in the central nervous system and noncentral nervous system tumors may affect epilepsy treatment outcomes due to the direct effects of tumors, subsequent infections or metabolic disturbances, and potential interactions between cancer therapy and ASMs, thereby leading to CYP450-dependent hepatic enzyme induction. Several population studies have shown that malignancy is associated with increased mortality in patients with epilepsy.31,32 Second, male patients were more likely to discontinue combination treatment (such as SC, SV2, and M combinations) compared with female patients. The ASM response is generally similar between male and female patients. However, the patients had some minor differences in terms of drug metabolism. For example, male patients exhibit a longer carbamazepine half-life than female patients. 33 Meanwhile, female patients were more likely to have a greater levetiracetam clearance than male patients. 34 However, clinical evidence on ASM response according to sex differences is limited. Some studies have revealed a trend toward different ASM retention rates between sexes. Further research is required to evaluate sex-specific responses to ASM combinations in patients with various epilepsy syndromes.35,36 Third, patients with renal disease were significantly more likely to discontinue SV2 combinations. Levetiracetam, the only ASM with an SV2 MoA in this study, is primarily eliminated via the kidneys. Decreased renal clearance in patients with renal disease may lead to higher serum levetiracetam levels, possibly resulting in an increased rate of adverse effects and a higher risk of intoxication. In summary, underlying malignancy, sex, and renal disease were important factors associated with ASM combination discontinuation. These findings emphasize the need to cautiously consider comorbidities when prescribing and monitoring ASM treatments, particularly in patients with epilepsy and additional medical conditions.
The findings of this study should be interpreted with caution due to the following reasons: First, we did not have data to assess the clinical rationale of initiating or terminating combination treatment, the titration schedule, or the total daily ASM dose administered that may also be relevant to persistence and healthcare resource use.
Second, there was no randomized treatment assignment due to the observational nature of the study. We could not control for the potential influence of unmeasured confounding factors such as syndrome diagnosis, seizure types and severity, ASM dose, structural brain lesions, and medication adherence. Further studies with a prospective design considering these factors can provide a more comprehensive view of factors influencing the efficacy of each ASM combination.
Third, although longer treatment persistence may imply better efficacy and tolerability profile, it may not be the most accurate tool for assessing treatment efficacy. Seizure reduction data can be more robust in evaluating treatment effectiveness. Nevertheless, in previous clinical trials and observational studies on ASMs with larger datasets, treatment persistence is considered a potential marker of efficacy, particularly if seizure reduction data are not readily available.15,37,38 In an evidence-based review conducted by the Commission on Antiepileptic Drugs of the International League Against Epilepsy, treatment persistence is recommended as a primary outcome measure for evaluating the effectiveness of ASMs. 39
Fourth, during the inclusion process, we excluded patients who did not take the second ASM for at least 60 days to ensure that discontinuing the second ASM was not attributed to intolerability but rather inefficacy. Our results showed that 60–80% of the participants were treated for >6 months. Thus, the number of participants excluded at this step could be low. However, selection bias could not be excluded if we assume that ASM inefficacy can lead to treatment discontinuation before 60 days.
Fifth, some subgroups, including those receiving SC + C (n = 44), M + M (n = 72), and G + G (n = 75) combinations had small sample sizes that may limit the possibility of achieving significant statistical outcomes. Nevertheless, the relatively large number of patients in our subgroups from an extensive medical record database with high disease-specific coverage in Taiwan adds strength to our conclusions.
Sixth, the treatment period was defined according to prescription data, and the accuracy might have been affected by noncompliance to ASMs. Finally, to group our patients, all ASMs were classified based on their primary MoA. Future studies on a comprehensive MoA profile of each ASM, more detailed MoA classification (e.g. fast and slow SC blockers), and pharmacokinetic interactions between ASMs should be performed to better elucidate the role of specific MoAs in ASM combination therapy.
Conclusion
This study showed that due to longer treatment persistence and lower risk of treatment discontinuation, SC and M combinations with different MoAs had a better efficacy/tolerability profile for seizure control than combinations with similar MoAs. Among all combinations, the SC + SV2 combination had the longest treatment persistence, and G combinations were associated with a high risk of treatment discontinuation during the treatment period. Underlying malignancy, sex, and renal disease are important factors associated with ASM combination discontinuation. These findings may provide valuable insights into selecting ASM combinations if monotherapy cannot adequately control seizures. Future studies, including more detailed MoA classification and relevant clinical variables, must be performed to investigate the role of specific MoAs in ASM combination therapy.
Supplemental Material
sj-docx-1-tan-10.1177_17562864231207161 – Supplemental material for Optimizing treatment persistence in epilepsy: a comparative analysis of combined antiseizure medications with different mechanisms of action
Supplemental material, sj-docx-1-tan-10.1177_17562864231207161 for Optimizing treatment persistence in epilepsy: a comparative analysis of combined antiseizure medications with different mechanisms of action by Chun-Wei Chang, Wei-En Johnny Tseng, Wey-Ran Lin, Po-Chuan Ko, Chun-Jing Liu and Siew-Na Lim in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The authors thank and wish to acknowledge the support of the Maintenance Project of the Center for Big Data Analytics and Statistics at Chang Gung Memorial Hospital for data analysis and interpretation. The interpretation and conclusions contained herein do not represent the position of Chang Gung Memorial Hospital.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
