Abstract
Background:
Malformation of cortical development (MCD) is one of the most common causes of pharmacoresistant epilepsy. Improving the knowledge of antiseizure medications (ASMs) treatment response in epileptic patients with MCD is crucial for optimal treatment options, either pharmacological therapy or non-pharmacological intervention.
Aim:
To investigate the patterns of medical treatment outcome and the predictors for seizure freedom (SF) with ASM regimens in epilepsy caused by MCD.
Methods:
The epileptic patients with MCD were consecutively enrolled from March 2013 to June 2019. SF was defined as no seizures for at least 12 months or three times the longest pretreatment inter-seizure interval, whichever was longer. Outcomes were classified into three patterns: pattern A: patients achieved SF at one point and remained so throughout follow-up; pattern B: patients’ seizures fluctuated between periods of SF and relapse; pattern C: SF never attained. The terminal SF was defined if the patients remained SF at the last follow-up visit.
Results:
A total of 164 epileptic patients with MCD were included. Pattern A was observed in 22, pattern B in 42, and pattern C in 100 patients. SF was ever achieved in 64 (pattern A and B) patients. Twenty-nine patients had terminal SF after a median follow-up time of 4.3 years. With continuing ASM treatment, seizure relapse risk was very low after a 5-year seizure-free period. The pretreatment seizure frequency was the only independent predictor for pattern A and seizure relapse. Sodium channel blockers monotherapy (33.8%) was more effective than levetiracetam (4.5%) in rendering SF in the initial ASM regimen.
Conclusion:
Medical treatment can be successful in a minority of epileptic patients with MCD, and pretreatment seizure frequency helps to predict the treatment outcome. An unequal efficacy of ASMs in epilepsy caused by MCD suggests etiological evaluation is vital in the management of focal epilepsy.
Introduction
Malformation of cortical development (MCD) is caused by disturbance of corticogenesis, including cell proliferation, neuronal migration, and cortical organization. 1 MCD is considered as one of the common causes of drug-resistant epilepsy, especially in childhood, 2 and is found to be one of the top three pathological findings in resective epilepsy surgery.3 –5 Antiseizure medications (ASMs) are first-line treatment for epilepsy. Improving the knowledge of ASMs treatment response in epileptic patients with MCD is crucial as one can be guided to the optimal treatment options earlier, either pharmacological therapy or non-pharmacological intervention.
Variable ASM treatment response patterns had previously been reported.6 –8 Most epileptic patients with MCD never achieved seizure freedom (SF) despite the optimal ASM regimen; however, some patients attained a period of SF during the early course of epilepsy. About 20–30% of the drug-resistant epileptic patients with focal cortical dysplasia (FCD) reported achieving a seizure-free period longer than 1 year, which was labeled as a “honeymoon” stage, before undergoing resective epilepsy surgery.6,7 During the “honeymoon” stage, most of the patients were found to be on ASM monotherapy. However, the ASM responsiveness in epileptic caused by MCD is still not well understood, and further exploration of this question is necessary.
In recent decades, the knowledge of the clinical and imaging features of MCD is continuously advancing,9,10 and the application of sophisticated magnetic resonance imaging (MRI) techniques continues to improve the detection of the epileptogenic lesions.11,12 Consequently, one can identify MCD lesions, even very subtle ones, in the early course of epilepsy or newly diagnosed epilepsy before it progresses into drug-resistant stages.9,10,13 Predicting whether the epileptic patients with MCD will be drug-responsive or drug-resistant will guide the clinician in making optimal treatment decisions. To address this question, here we reported the treatment response of ASMs in a cohort of epileptic patients with MCD who were consecutively enrolled in our epilepsy center. In addition, the efficacy of different ASMs as well as the clinical features, impacting the patterns of ASM treatment response, were analyzed.
Methods
Patient inclusion
This retrospective observational study was approved by the Medical Ethics Committee of the Second Affiliated Hospital, Zhejiang University School of Medicine (approval number: 2013-032). Written informed consent was obtained from all subjects. The epileptic patients diagnosed with MCD in the comprehensive epilepsy center of our hospital were consecutively enrolled from March 2013 to June 2019. As we are the tertiary epilepsy center, the cohort was a mixture of the newly diagnosed epilepsy patients as well as patients referred to our tertiary epilepsy center for surgical evaluation. The information of age, sex, age at epilepsy onset, family history, perinatal sentinel events, febrile seizure, pretreatment seizure frequency, and a complete detailed record of ASM treatment were collected from epilepsy diaries as well as medical charts. More than one seizure per month was defined as high seizure frequency and low seizure frequency as less than or equal to one seizure per month. 14 All the enrolled patients had focal seizures subdivided into three categories: focal aware seizure, focal impaired awareness seizure, and focal to bilateral tonic–clonic seizure. 15 The enrolled patients met all of the following criteria: (1) a complete medical history with imaging and video-electroencephalography (EEG) materials; (2) confirmed epileptogenic MRI-identified or histological diagnosis of MCD (Figure 1); (3) followed-up regularly for more than 1 year after ASMs were initiated. We excluded patients with epilepsy caused by cerebrovascular diseases, intracranial tumors, neurodegenerative diseases, or other neurological diseases. If the MCD lesion was confirmed non-epileptogenic or there was insufficient evidence that the lesion was epileptogenic, the patients were excluded. For example, a patient with known right-hemispheric heterotopia and confirmed left temporal lobe epilepsy was excluded.

Representative images of malformation of cortical development (MCD) subgroups. Group 1, focal cortical dysplasia (FCD) or focal megalencephaly. (A) The FCD lesion of an 11-year-old girl with drug-resistant sleep-related hypermotor seizures since the age of 6. A1, T1 MRI showing subtle blurring of gray–white matter junction in the right frontal region; A2, T2-FLAIR MRI was unremarkable; A3, focal hypometabolism on PET; the lesion was indicated by the white arrow; postoperative pathology showed the lesion was FCD IIb. (B) FCD lesion of a 16-year-old boy with gelastic seizures since the age of 10. EEG recording showed repetitive and frequent spikes in the right anterior frontal region (FP2, F8, and F4). He was now on a combination of carbamazepine and valproate and remained seizure-free for 5 years. White dashed box, the FCD lesion in the right inferior frontal gyrus. B1, blurring of gray–white matter junction on T1 MRI, which was also indicated by voxel-based morphometry analysis; B2, transmantle sign on T2 MRI; B3, focal hypometabolism on PET. MRI scan was repeated 3 years later and showed the lesion was unchanged. Group 2, heterotopia. (C) A 35-year-old lady experienced the left tongue and face tonic seizures with occasional progression to focal to bilateral tonic and clonic seizures since the age of 28. EEG recorded sharp waves and intermittent focal slowing in the left frontotemporal region (F7, T3). C1–3, MRI identified a subcortical heterotopia overlaid by the face motor cortex (white arrow) in the left hemisphere. Group 3, polymicrogyria or (and) schizencephaly. (D) An 18-year-old boy experienced episodes of clonic jerks of the right face, right head deviation, and flexion of the right arm since the age of 16. EEG recording showed left frontotemporal sharp waves (T3, F7). D1–D3, MRI showing schizencephaly (D1, D2) and polymicrogyria (D3) in the left central region (white arrow).
Using Barkovich classification, 16 MCD were classified into three subgroups, which were corresponding to the disorders in the three major stages of human cerebral cortex development: (1) neural cell proliferation disorders (group 1) including FCD or focal megalencephaly; (2) neuronal migration disorders (group 2) including periventricular nodular heterotopia or band heterotopia; and (3) post-migration remodeling disorders (group 3) including polymicrogyria or schizencephaly.
All the enrolled patients with drug-resistant epilepsy were evaluated by the multi-disciplinary team. During the patient management conference, a recommendation was made by the team on whether to continue medical treatment or to take non-pharmacological therapy including resective surgery, neuromodulation, and ketogenic diet therapy. The epileptic patients with confirmed pathological diagnosis of MCD (mostly FCD) were also included and classified according to the International League Against Epilepsy (ILAE) guidelines. 17
Imaging study and video-EEG evaluation
MRI images were acquired with a 3.0-T Discovery MR750 (GE Healthcare) scanner. The MRI protocol included a high-resolution three-dimensional sagittal T1-weighted brain volume (3D-BRAVO) image (repetition time/echo time = 7.7/2.9 ms, inversion time = 450 ms, flip angle = 12°, matrix = 224 224, voxel size = 0.44 0.44 1 mm3, slice thickness = 1 mm without slice gap), two-dimensional axial (repetition time/echo time = 5000/100 ms, flip angle = 90°, matrix = 512 512, voxel size = .44 0.44 3 mm3, 3-mm slice thickness, and 1 mm interslice gap) and sagittal T2W sequences (repetition time/echo time = 8500/110 ms, flip angle = 110°, matrix = 512 512, voxel size = 0.44 0.44 3 mm3, 3-mm slice thickness, and 0.3 mm interslice gap), and two-dimensional axial (repetition time/echo time = 8000/150 ms, inversion time = 2000 ms, flip angle = 90°, matrix = 512 512, voxel size = 0.44 0.44 3 mm3, 3-mm slice thickness, and 1 mm interslice gap) and sagittal (repetition time/echo time = 8000/150 ms, inversion time = 2000 ms, flip angle = 90°, matrix = 512 512, voxel size = 0.44 0.44 3 mm3, 3-mm slice thickness, and 0.3 mm interslice gap) FLAIR sequences. All patients underwent 24-h video-EEG. The MRI data were reviewed by the experienced neuroradiologists (Jiang B. and Li H.). MRI post-processing and positron emission tomography (PET) co-registration analysis were applied if initial MRI findings were negative or inconclusive.18,19 The indicative features of FCD on MRI were local cortical thickening, blurring of the gray–white matter interface, and focal increased signal of the subcortical white matter on T2-weighted imaging, often tapering toward the underlying ventricle. 20 The cortical changes in dysplastic megalencephaly were severe and consisted of an enlargement of part or all of one hemisphere and included poor differentiation between gray and white matter. 9 Heterotopia was defined as groups of normal neurons that resided in an inappropriate location, 9 including periventricular nodular heterotopias, subcortical heterotopias, and subcortical band heterotopias. Polymicrogyria was characterized by overfolding of the cerebral cortex and abnormal cortical layering. 21 In schizencephaly, abnormal clefts in the gray matter occurred, and the cortex edges seemed to fuse (closed lips) or to stay at a distance (open lips). 9 MRI-identified MCD lesions were carefully reviewed by two neuroradiologists. In addition, the MCD lesions were only classified when the consensus was reached between the two neuroradiologists. Otherwise, a third neuroradiologist was consulted.
ASM regimen and dosage
ASMs were chosen based on seizure types, drug side-effects and drug–drug interaction profiles. 22 An ASM regimen was defined as a trial of either a single (monotherapy) or a combination of two or more drugs. 23 Any changes in ASM after initiation of therapy, such as substituting, adding, or stopping the portion from the original drug combination, were defined as the end of one ASM regimen. For example, switching to another ASM because of severe adverse effects of the initial ASM was considered a second regimen. If the type of drugs remained the same, changes in the dose of a single drug or any drugs in polytherapy were not regarded as switching regimens. In addition, a temporary use of medications in the rescue of status epilepticus was not considered a change in the ASM regimen. The dosage of ASMs was regarded as adequate if SF was achieved or reached 50% of the defined daily dose. 24 In the pediatric population, the dose was referred to as maximal dose for seizure control until signs of clinical or laboratory toxicity became evident under the observation of pediatric neurologists. 25
Type of ASMs
Head-to-head evaluation of ASMs’ response in either monotherapy or adjunctive treatment was not performed in our study. Eslicarbazepine, clobazam, lacosamide, pregabalin, and perampanel were unavailable or not indicated for epilepsy in our region during the study period. Thus, those medications were not included. The most widely used ASMs in our cohort were carbamazepine, oxcarbazepine, lamotrigine, valproic acid, and levetiracetam; 91.3% of patients had tried those ASMs. Hence, we only analyzed the response patterns of ASMs mentioned above. According to the main mechanisms of action, carbamazepine, oxcarbazepine, and lamotrigine were classified as category I (classical sodium channel blockers); valproic acid was classified as category II because it pocesses multiple action mechanisms including potentiation of GABAnergic system, sodium channel blocking, and others; and levetiracetam was classified as category III (it binds to synaptic vesicle protein SV2A).26,27
Definition and patterns of ASM outcome
After initiating an ASM regimen, the treatment outcomes were assessed every 4–6 weeks for the first 6 months, followed by at least every 4 months. SF was defined as no seizures for at least 12 months or three times the longest pretreatment inter-seizure interval, whichever was longer. 28 The main treatment outcome was subdivided into three patterns: pattern A: SF was achieved at one point during the study period and remained so throughout follow-up; pattern B: seizures fluctuated between periods of SF and relapse: pattern C: SF never attained. Patients who underwent non-pharmacological therapy were categorized as seizure relapse, and the last follow-up was the start date of non-pharmacological treatment. Conversely, terminal SF was defined if the patients achieved SF during one of the ASM regimens and remained SF at the last follow-up visit. Thus, in addition to the pattern A group, some patients from pattern B could achieve terminal SF, depending on their seizure frequency at the last follow-up visit.
If the patient ever achieved SF (pattern A or B), epileptic patients with MCD was considered drug-responsive. Those who never achieved SF (pattern C), the epileptic patients with MCD were categorized as drug-nonresponsive. If the patients from the responsive treatment group relapsed, those patients were given the option to switch to the next ASM regimens (Figure 2). After switching to the new ASM regimen, treatment responsiveness was re-evaluated.

Flow chart of drug regimens in epilepsy caused by MCD. Terminal SF, reaching SF at final follow-up (pattern A and parts of pattern B). If seizure relapsed in one drug regimen, a next drug regimen was started except the situations of drug dosage adjustment, satisfaction with the current treatment, or taking non-pharmaceutical treatment.
Statistical methods
All the analyses were conducted with SPSS (version 22.0). The data were analyzed using the Wilcoxon rank-sum, chi-square, and Fisher’s exact tests to compare the data of different subgroups and treatment response patterns of epileptic patients with MCD. Chi-square was also used to compare the response rate of the different categories of ASMs; a post hoc Bonferroni correction was further applied to correct multiple comparisons (p < 0.017,0.05/3). Multiple ordered regression was used to explore the causes of different prognoses of drug treatment.
Among the patients who ever achieved SF, Kaplan–Meier survival analysis was used for calculating the seizure recurrence rates. The endpoint event was set as the first seizure relapse after the SF period. Variables with a significance level of 10% on univariate analysis were then fit in a multivariable Cox proportional hazards regression model with a statistical significance set at the 5% level.
Results
MCD subgroups
A total of 164 patients with a median age of 20 years (interquartile range (IQR): 12–29) and seizure onset age of 9 years (IQR: 3.4–15) were enrolled (Table 1). The median duration of follow-up was 4.3 years (IQR: 1.8–7.2). The median epilepsy duration (from seizure onset to last follow-up) was 8.1 years (IQR: 4–16) and the median duration of ASMs usage was 6 years (IQR: 3.5–12). Eighty-five patients (51.8%) out of the cohort had FCD, including 41 MRI-identified cases and 44 pathologically diagnosed cases (FCD type IIb, 26 cases; FCD type IIa, 13 cases; FCD type I, 5 cases). The patients in group 1 were younger at seizure onset (p < 0.001) and at the time of evaluation (p < 0.001) than the other two groups; they also had a higher percentage of pretreatment high seizure frequency (p = 0.002) and epileptiform discharges (p = 0.021) than group 2. Group 3 had the highest percentage of patients with history of perinatal sentinel events (p = 0.024) than the other two groups.
Demographic profiles of the patients in each subgroup of MCD.

EEG, electroencephalography; FBTCS, focal to bilateral tonic-clonic seizures; FCD, focal cortical dysplasia; IQR, interquartile range; MCD, malformation of cortical development; MEG, megalencephaly; PMG, polymicrogyria; PNH, periventricular nodular heterotopia; SBH, subcortical band heterotopias; SCH, subcortical heterotopias; SCHI, schizencephaly.
Statistically significant difference between the group and the other two groups.
Statistically significant difference between the two groups with the marker.
Fourteen out of 26 patients (53.9%) with schizencephaly belonged to open lips–type schizencephaly.
p < 0.05, **p < 0.01. ***p < 0.001.
ASMs treatment outcome over time
Pattern A (i.e. patients achieved SF throughout follow-up) was observed in 22 cases (13.4%). Forty-two cases (25.6%) were classified as pattern B (i.e. SF and relapse fluctuated), and the remaining 100 cases (61.0%) were classified as pattern C (i.e. SF never attained; Table 2 and Figure 2).
Different patterns of ASM response in epilepsy caused by MCD.
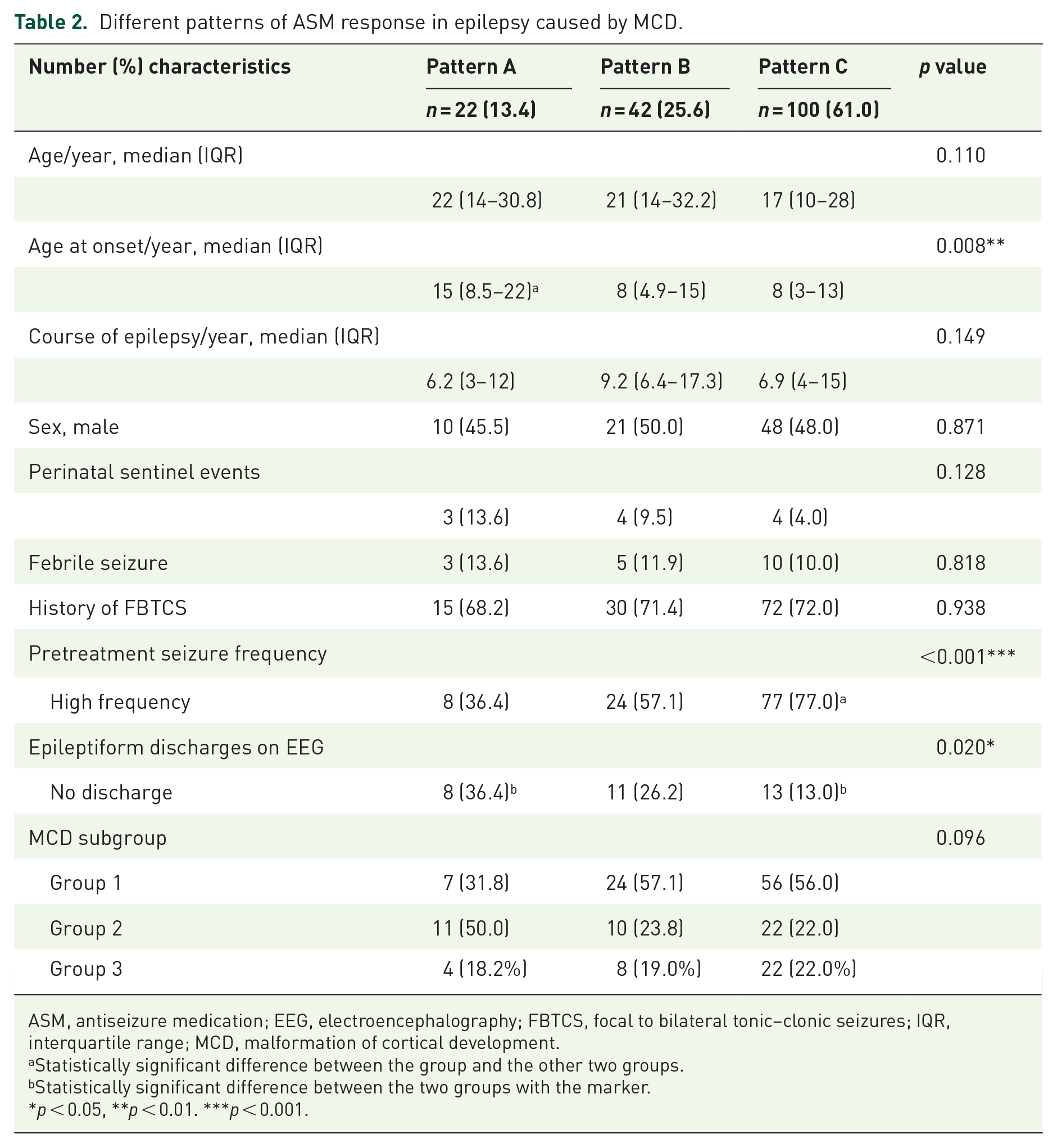
ASM, antiseizure medication; EEG, electroencephalography; FBTCS, focal to bilateral tonic–clonic seizures; IQR, interquartile range; MCD, malformation of cortical development.
Statistically significant difference between the group and the other two groups.
Statistically significant difference between the two groups with the marker.
p < 0.05, **p < 0.01. ***p < 0.001.
ASMs treatment outcome at last follow-up
Sixty-four patients (39%, pattern A and pattern B) achieved SF at one point (median duration of SF 2.4 years (IQR: 1.5–4.0)), but only 29 patients (17.7%, 22 from pattern A (100%) and 7 from pattern B (16.7%)) achieved terminal SF at the last follow-up visit. Most of the patients (23/29, 79.3%) achieved terminal SF during the first two regimens (Figure 2). For 135 patients who did not achieve terminal SF, 50 patients underwent non-pharmacologic therapies (resective surgery, 44 patients with histological confirmed FCD; VNS, 5 patients; ketogenic diet therapy: 1 patient).
Response to ASM regimen
After the initial ASM regimen, 25.6% (42/164) of patients achieved SF, and 91.5% received monotherapy. During the second and third ASM regimens, 15.4% (21/136) and 10.1% (9/89) of patients achieved SF, respectively. Nine (5.5%) patients in the initial, six (4.4%) patients in the second, and five (5.6%) patients in the third ASM regimen discontinued the current ASM regimens due to unsustainable adverse drug reactions.
Predictors of three treatment patterns after initiation of ASM
Univariate analysis showed that the seizure onset age (p = 0.008), pretreatment seizure frequency (p < 0.001), and the presence of epileptiform discharges on EEG (p = 0.020) were predictors found to have a statistically significant association with different patterns of ASM treatment response (Table 2). The ordered multiple regression model showed that the pretreatment seizure frequency (p = 0.012, risk ration = 2.52, 95% confidence interval (CI): 1.22–5.22) was the only independent factor affecting the treatment response.
Predictors of seizure relapse after seizure-free period
If a patient had several seizure-free periods, the risk of seizure relapses after the first seizure-free period was analyzed. The Kaplan–Meier survival analysis was used to calculate the median probability of SF among 64 patients who ever achieved SF (Figure 3). MCD subgroups (p = 0.048) and pretreatment seizure frequency (p = 0.014) had a significant impact on the risk of seizure relapse (Figure 3(a) and (b)). The predictive variables of seizure onset age, pretreatment seizure frequency, and MCD subgroups were put into the multivariable COX proportional hazards regression model. We found that pretreatment seizure frequency was the only independent factor for the risk of seizure relapse (p = 0.022, risk ratio = 2.13, 95% CI: 1.12–4.61).
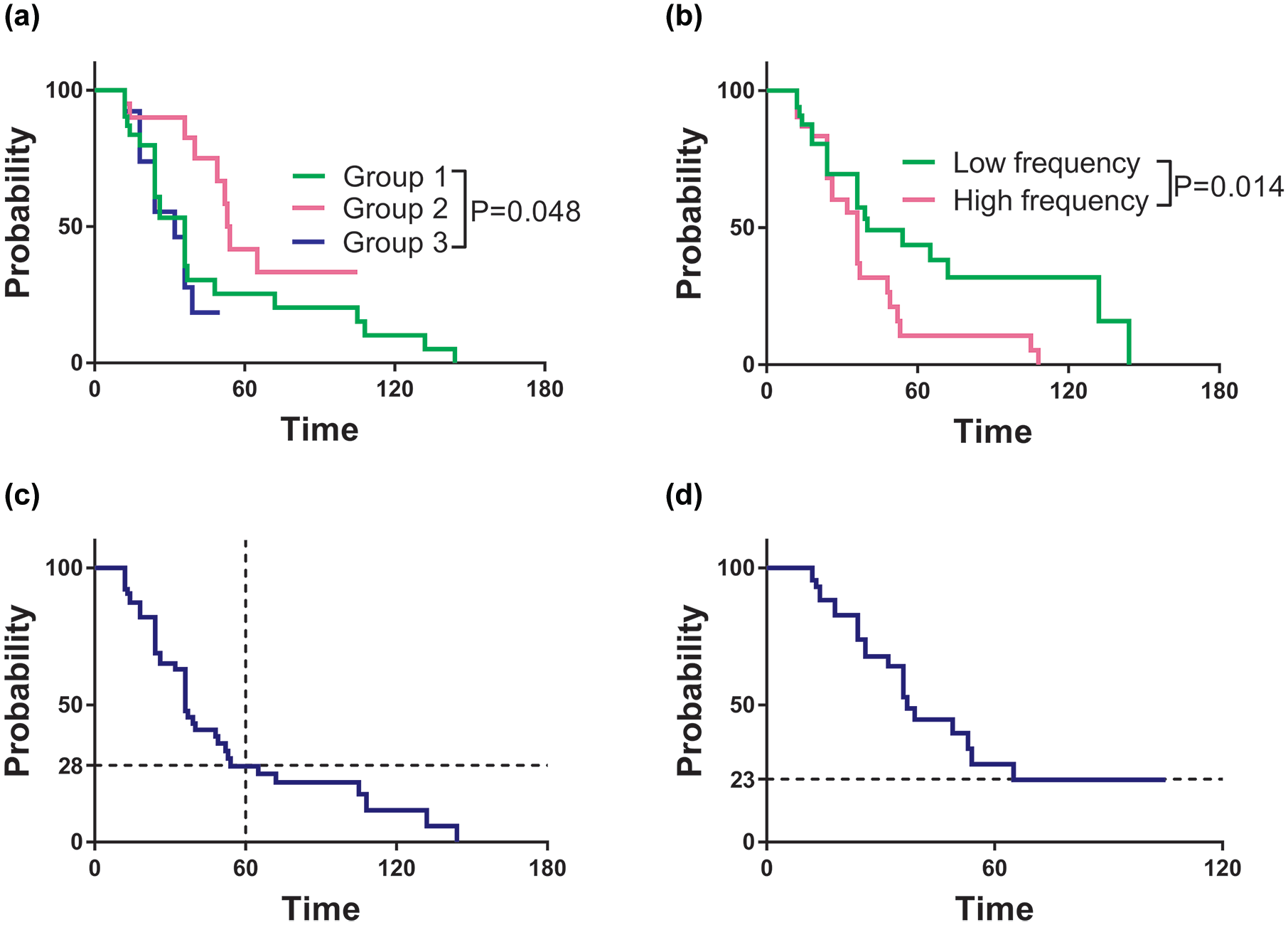
The probability of remaining seizure-free in epileptic patients with MCD: (a) cumulative probability of remaining seizure-free by the time since the last seizure and MCD subgroup. (b) Cumulative probability of remaining seizure-free by the time since the last seizure and pretreatment seizure frequency. (c) Trend chart of remaining seizure-free by the time since the last seizure. (d) Trend chart of remaining seizure-free by the time since the last seizure in the patients without ASM withdrawal. The endpoint event was set as the first seizure relapse after seizure-free period.
Among the 42 patients (pattern B) with seizure relapse, almost half of patients (20/42, 47.6%) had recurrence during the drug weaning or withdrawal period, either for pregnancy planning or for long-term side effects of ASMs. Although weaning or withdrawal of ASMs was supervised by the clinicians, seizure relapse invariably occurred within the first 12 months of the initiation of ASMs reduction.
Survival analysis revealed that after obtaining the first SF, 72% of patients were likely to have seizure relapse within 5 years (Figure 3(c)). Among 13 patients who had seizure-free periods longer than 5 years, six patients (five from MCD group 1 and one from MCD group 2) had seizure relapse during the drug withdrawal period. Thus, if the patients had been SF for more than 5 years, seizure relapse occurred only after the drug withdrawal. Conversely, 23% of patients who continued ASM treatment with the initial ASM doses (without ASM weaning off or withdrawal) were likely to have sustained SF and had no risk of seizure relapse after a 5-year seizure-free period (Figure 3(d)).
The treatment response of different ASMs
Since pharmacoresistance gradually develops over time in epileptic patients with MCD, the drug-responsiveness of ASM was evaluated only in the initial drug regimen. During this period, 14 patients were on combination therapy, and the remaining 150 patients were on monotherapy. There was no significant difference (p = 0.558) in treatment response between monotherapy (37/150, 24.7%) versus combination therapy (5/14, 35.7%). During the initial regimen, patients taking levetiracetam were less likely to achieve SF (1/22, 4.5%) than the patients taking classical sodium channel blockers (24/71, 33.8%, p = 0.007, Figure 4). There were no statistically significant differences in the clinical characteristics of patients with different ASMs monotherapy (Supplemental Table S1).
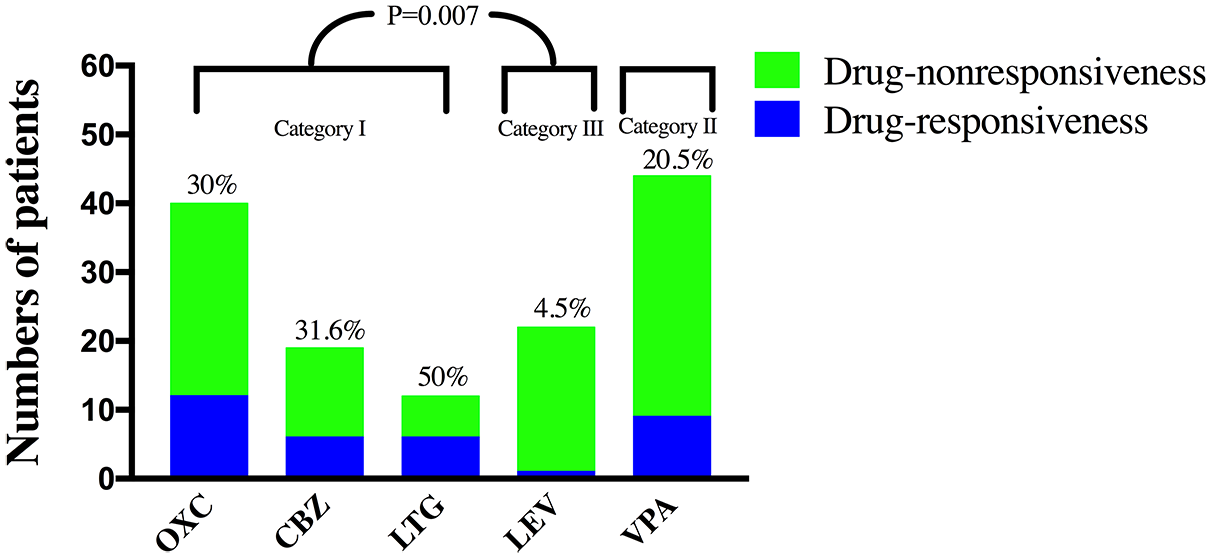
Treatment responsiveness of ASMs in 164 patients during the initial regimen. The percentile above each bar indicates the proportion of drug-responsiveness. A post hoc Bonferroni correction was used.
Interestingly, after the initiation of oxcarbazepine and levetiracetam in the initial ASM regimen, one patient experienced a 150% increase in seizure frequency within the first 2 months. After discontinuation of the levetiracetam, the patient had an 80% reduction in seizure frequency. On the contrary, levetiracetam was noted to be effective as an adjunctive ASM in the combination therapy during the later regimens. In the second and third regimens, levetiracetam was added or used to substitute other ASMs in 48 and 28 patients, respectively. Nine (18.8%) patients from the second regimen and six (21.4%) patients from the third regimen achieved SF.
Discussion
Epileptic seizures are one of the common presenting symptoms of MCD. In our cohort, a small portion of patients attained terminal SF (17.7%) at a median follow-up time of 4.3 years. Pretreatment seizure frequency was the only independent predictor for treatment response and terminal SF. Although incidence of drug-resistance was high, a period of drug-responsiveness was possible and ever achieved in 39% of the cohort. After a 5-year seizure-free period, the chance of seizure relapse was uncommon if the patients remained on ASMs. Notably, for monotherapy, there were different treatment response patterns with different ASMs groups. In the initial regimen, sodium channel blockers were noted to be distinctly superior to render SF compared with levetiracetam.
Many studies, including ours, revealed initial seizure frequency was perhaps the most important predictor for ASM treatment outcome.29,30 Thus, seizure frequency in the early course of epilepsy was the strongest risk factor for the worst outcome. In our cohort, patients with pretreatment low seizure frequency were more likely to achieve SF than those with high seizure frequency. When attaining SF, the patients with pretreatment high seizure frequency were more likely to relapse than those with low seizure frequency. This correlation between initial seizure frequency and pharmacoresistance suggested they might share a common neurobiological basis.
In our cohort, the treatment response in epileptic patients with MCD during the initial and second treatment regimen were 25.6% and 15.4%, respectively. Brodie et al. 23 reported that the first two ASM regimens’ treatment response were about 70% and 50% at 5 years follow-up period in the general population of newly diagnosed epilepsy. In our cohort, 39% of the cohort achieved a seizure-free period, mostly in the first two regimens, whereas 61% of the patients never attained SF. At the last follow-up, 82.3% belonged to drug-resistant epilepsy, suggesting pharmacoresistance was more prevalent in epileptic patients with MCD and developed in the early course of epileptic patients with MCD. Thus, in epileptic patients with MCD after failing on the initial and second ASM regimens, non-pharmacological treatments should be considered timely, especially for those with high pretreatment seizure frequency. Our data showed that ASM treatment could be successful in a minority of epileptic patients with MCD.
The treatment response of ASMs was examined in the initial regimens among which most patients received monotherapy. In monotherapy, the rate of treatment response was 50% for lamotrigine, 31.6% for carbamazepine, 30% for oxcarbazepine, 20.5% for valproic acid, and 4.5% for levetiracetam. There was a statistically significant difference in drug-responsiveness between levetiracetam and sodium channel blockers (carbamazepine, oxcarbazepine, and lamotrigine). Levetiracetam acts by targeting the synaptic vesicle protein receptors and is one of the most commonly prescribed ASMs. 31 However, its SF rate was extremely low in epileptic patients with MCD and even caused seizure aggravation in one patient with FCD. This paradoxical effect of levetiracetam in FCD was reported previously. 32 Cvetkovska et al. 33 found that levetiracetam caused seizure aggravation in five out of 139 patients, and four of the five patients showed MRI abnormality highly suggestive of FCD. Therefore, one might need to be cautious in using levetiracetam as monotherapy in newly diagnosed epileptic patients with MCD. However, adding levetiracetam was found to be useful as adjunctive therapy in later regimens. Thus, our data highlighted that etiology of epilepsy should be considered in choosing ASMs. Supporting this viewpoint, Feyissa et al. 34 also reported that sodium channel blockers might have an advantage in autoimmune epilepsy, which is usually characterized by a high seizure burden. Future multi-center prospective studies are warranted to better understand the efficacy of different types of ASMs in epileptic patients with MCD.
ASMs are known to be related to long-term adverse effects, such as cognitive and behavioral alterations. 35 Withdrawal of ASMs might be advantageous for individuals with long-term remission of seizures if the benefits of withdrawal surpass the harm caused by the usage of ASM. 36 Currently, there are limited data on determining whether or when this discontinuation can safely occur in epileptic patients with MCD. In our study, nearly one-third of patients who had weaned off or withdrew ASMs during SF period, seizure relapse invariably occurred within 12 months from the initiation of ASMs. Out of them, six patients were seizure-free for longer than 5 years. In addition, after a 5-year seizure-free period, we observed that no seizure relapse was reported if patients remained on continuous ASM treatment. It had been reported that the possibility of SF after ASM withdrawal was about 60% in the general population with focal epilepsy, who had been seizure-free longer than 5 years. 37 Our data showed that the chance of SF after ASM withdrawal was very slim for epileptic patients with MCD. Therefore, the clinician may need to be very cautious in reducing the ASM dosage in epileptic patients with MCD, especially after achieving SF.
This real-world study was based on an enrolled cohort of epileptic patients with MCD, but it had several limitations. First, the cohort was a mixture of the newly diagnosed epilepsy patients and patients who were referred to our tertiary epilepsy center for surgical evaluation. Parts of the medical history were retrospectively collected in the transferred patients of epilepsy surgical candidates. Thus, the rate of SF in the general population of epileptic patients with MCD might not be accurately reflected. Second, FCDs may be hard to be identified by imaging if MRI results are atypical or unremarkable, especially type I. In such situations, FCD can only be confirmed in surgical specimens in those patients who already have drug-resistant epilepsy. Therefore, a considerable proportion of MRI-negative FCD could be missed. Third, in our cohort, we only study the efficacy in the initial regime. Thus the efficacy of combination therapy in epileptic patients with MCD required further evaluation. Last but not least, in our study, we did not investigate the impact of other neurological comorbidities and neuropsychological profiles in epileptic patients with MCD. These factors need to be evaluated in future studies based on newly diagnosed epilepsy.
Conclusion
Our study showed that medical treatment could be successful in a minority of epileptic patients with MCD. Although the selection of ASM is typically based on seizure type, our data suggested that etiological evaluation was important. Our study paved the way to optimize the medical treatment in order to facilitate the clinical decision in the management of epileptic patients with MCD.
Supplemental Material
sj-docx-1-tan-10.1177_17562864211050027 – Supplemental material for Response to antiseizure medications in epileptic patients with malformation of cortical development
Supplemental material, sj-docx-1-tan-10.1177_17562864211050027 for Response to antiseizure medications in epileptic patients with malformation of cortical development by Wei Chen, Bo Jin, Thandar Aung, Chenmin He, Cong Chen, Shan Wang, Yao Ding, Fang Ding, Chao Wang, Hong Li, Biao Jiang, Zhe Zheng, Haibin Dai, Junming Zhu, Yu Geng, Meiping Ding and Shuang Wang in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The authors thank all the staff involved in this study, as well as the patients and their families for their participation and cooperation. WC and BJ contributed equally to this paper.
Author contributions
WC and BJ drafted the manuscript. FD, YD, and SW contributed to conception and design of the study. CMH, CC, MPD, CW, HL, and BJ contributed to acquisition of data. ZZ, HBD, JMZ, and YG contributed to analysis of data. TA and SW revised the manuscript.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (grant nos: 82001366 and 81971207), the Natural Science Foundation of Zhejiang Province (grant no: LQ20H090019), China and Zhejiang Medical Science and Technology Project (grant no: 2021KY020), China.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
