Abstract
Background:
Cladribine is an effective immunotherapy for people with multiple sclerosis (pwMS). Whilst most pwMS do not require re-treatment following standard dosing (two treatment courses), disease activity re-emerges in others. The characteristics of pwMS developing re-emerging disease activity remain incompletely understood.
Objectives:
To explore whether clinical and/or paraclinical baseline characteristics, including the degree of lymphocyte reduction, drug dose and lesions on magnetic resonance imaging (MRI) are associated with re-emerging disease activity.
Design:
Service evaluation in pwMS undergoing subcutaneous cladribine (SClad) treatment.
Methods:
Demographics, clinical, laboratory and MRI data of pwMS receiving two courses of SClad were extracted from health records. To assess associations of predictor variables with re-emerging disease activity, a series of Cox proportional hazards models was fitted (one for each predictor variable).
Results:
Of n = 264 pwMS 236 received two courses of SClad and were included in the analysis. Median follow-up was 4.5 years (3.9, 5.3) from the first, and 3.5 years (2.9, 4.3) from the last SClad administration. Re-emerging disease activity occurred in 57/236 pwMS (24%); 22/236 received further cladribine doses (SClad or cladribine tablets) at 36.7 months [median; interquartile range (IQR): 31.7, 42.1], and 22/236 other immunotherapies 18.9 months (13.0, 30.2) after their second course of SClad, respectively. Eligibility was based on MRI activity in 29, relapse in 5, both in 13, elevated cerebrospinal fluid neurofilament light chain level in 3, deterioration unrelated to relapse in 4 and other in 3. Only 36/57 of those eligible for additional immunotherapy had received a reduced dose of SClad for their second treatment course. Association was detected between re-emerging disease activity and (i) high baseline MRI activity and (ii) low second dose of SClad.
Conclusion:
Re-emerging disease activity was associated with baseline MRI activity and low dose second course of SClad.
Keywords
Introduction
Cladribine tablets (CladT) is an immunotherapy for people with relapsing MS (pwRMS).1,2 Post hoc analysis of the pivotal phase III trial showed that pwRMS with high disease activity experienced disability risk reduction, measured using the expanded disability status scale (EDSS) score, of 82%, and an annualized relapse rate of 67–68%, respectively, versus placebo over 2 years. 2 This data, alongside reports further exploring the safety and efficacy of CladT,3–5 underpinned the licence of CladT (Mavenclad®) in Europe. 6
Whilst most people with multiple sclerosis (pwMS) do not require re-treatment following the standard dosing schedule of two treatment courses, that is, 5 days of weight-adjusted CladT daily in weeks 1 and 5, repeated in year 2, disease activity resumes in others. However, there is limited evidence to instruct on the need and scope of subsequent immunotherapy.7,8
We recently reported our real-world data on the use of subcutaneously injected cladribine (SClad, Litak®) as an off-label immunotherapy in 208 people with MS (pwMS) from 2014 onwards, 9 that is, 3 years prior to licensing of Mavenclad® in Europe. Given the extended follow-up of our cohort, we investigated clinical, magnetic resonance imaging (MRI) and dosing characteristics that may relate to re-emerging disease activity.
Methods
From the cohort of pwMS using the MS service (BartsMS) at our centre, we identified pwMS treated with SClad between 1 October 2014 and 31 May 2022 using a previously reported dosing schedule (Figure 1).9–12

The dosing schedule of SClad was adjusted to body weight and total lymphocyte count (TLC). Treatment course 1: Following injections of SClad 10 mg on 3–4 consecutive days in week 1, TLC was measured at week 4 during full blood count testing. If TLC was ⩾1.0 × 109 L−1, further injections of SClad 10 mg were given on three consecutive days in week 5. If TLC was 0.8–0.9 × 109 L−1, further injections of SClad 10 mg were given on two consecutive days in week 5. If TLC was 0.5–0.7 × 109 L−1, one further injection of SClad 10 mg was given in week 5. If TLC was <0.5 × 109 L−1, no further injection of SClad was given during this treatment course. Treatment course 2 (option A): If TLC at week 44 was ⩾1.0 × 109 L−1, injections of SClad 10 mg were given on three consecutive days in week 48. TLC was then measured at week 51. If TLC was ⩾1.0 × 109 L−1, further injections of SClad 10 mg were given on three consecutive days in week 52. If TLC was 0.8–0.9 × 109 L−1, further injections of SClad 10 mg were given on two consecutive days in week 52. If TLC was 0.5–0.7 × 109 L−1, one further injection of SClad 10 mg was given in week 52. If TLC was <0.5 × 109 L−1, no further injection of SClad was given. Treatment course 2 (option B): If TLC at week 44 was 0.8–0.9, injections of SClad 10 mg were given on two consecutive days in week 48. If TLC was 0.5–0.7 × 109 L−1, one further injection of SClad 10 mg was given in week 48. If TLC was <0.5 × 109 L−1, no further injection of SClad was given.
SClad (Litak®) had been offered irrespective of whether patients were eligible for National Health Service-funded disease-modifying treatment (DMT). Patients considering SClad were provided with comprehensive information to aid their understanding and decision. 13 Between October 2014 and February 2016, treatment decisions were based on clinical judgement (chronic disability accrual, relapses), although gadolinium-enhancing (Gd+) lesion(s) and new/enlarging T2 lesion(s) on MRI were considered. From February 2016 onwards, disease activity based on MRI and/or cerebrospinal fluid (CSF)–neurofilament light chain (NfL) levels, over and above clinical activity, was mandatory for eligibility. 14 Treatment with SClad in all cases was approved by the neuroinflammation multidisciplinary team of Barts Health.
Patient demographics, clinical, laboratory and MRI variables were extracted from the electronic health record.
Patients were reviewed at baseline and annually thereafter, although some were seen more frequently. MRI disease activity was defined as the presence of gadolinium-enhancing (Gd+) lesions on T1-weighted MRI and/or new lesion(s), or increased lesion volume, compared to a reference scan, on T2-weighted MRI. CSF-NfL levels were quantified using a commercial enzyme-linked immunosorbent assay (UmanDiagnostics NF-Light ELISA) 15 and age-related reference levels were applied.9,16
Of those who received two courses of SClad, we analysed pwMS with (i) no new disease activity and no further immunotherapy, (ii) new disease activity and yet no further immunotherapy, (iii) new disease activity who received a third course of cladribine (either CladT or SClad) and (iv) new disease activity switching to different immunotherapies as a result.
Descriptive summary tables were produced to illustrate the difference between groups with evidence of disease activity after their second course of SClad (‘activity’ group) and those without (‘no activity’ group). To assess the association with time to disease activity, we fitted a series of Cox models, one for each predictor variable. We used multiple imputations by chained equations to account for missing data, using predictive mean matching for numeric variables, and logistic regression for binary ones. For treatment courses one and two, further models adjusting for the weight (kg) were fitted. The coefficient for both courses is for an increase of 10 mg. The total lymphocyte count (TLC) ratio was found by dividing TLC at 20 weeks by TLC at baseline. Results are expressed as hazard ratios (HRs) with corresponding 95% confidence intervals. The large proportion of missing data precluded imputation or formal statistical testing for some clinical indices. 17
Results
The pathways of 264 pwMS treated with at least one course of SClad are shown in the Consolidated Standards of Reporting Trials (CONSORT) flow diagram (Figure 2). In all, 21 pwMS who received only one course of SClad were excluded for the purpose of this analysis, leaving 243 pwMS. Of these, a further seven patients were excluded due to follow-up of less than 2 years after the start of their first course of SClad.

CONSORT flow diagram of treatment pathways in 264 patients with MS receiving at least one course of SClad.
Age at the first course of SClad was 39.1 years [median; interquartile range (IQR): 35.6, 46.9]. Disease duration from first symptoms until data cut-off was 11.0 years (median; IQR: 8.2, 15.5). The reasons why 21 pwMS received only one course of SClad were as follows: personal choice (n = 6), difficulties travelling to our centre (1), haematopoietic stem cell transplantation (HSCT) (1), switch to fingolimod (1), planned pregnancy (2), death (1), 18 low TLC prior to planned second treatment course (2), intensive treatment unit (ITU) admission following surgery (1), non-specific health issues (1) and not yet due second treatment course at the cut-off date of our cohort (5). Thus, there were 236 pwMS for further analysis (Table 1). Median follow-up was 4.5 years (3.9, 5.3) from the first, and 3.5 years (2.9, 4.3) from the last SClad administration. The baseline median EDSS prior to receiving SClad was 6.0 in 205/236 pwMS. Median EDSS at the last follow-up (mean 43 months after the first SClad administration) was 6.0 in 127/236 pwMS.
Demographics, clinical outcome measures and TLC in 236 patients with multiple sclerosis grouped according to disease activity status a median of 4.5 years after starting treatment with SClad.
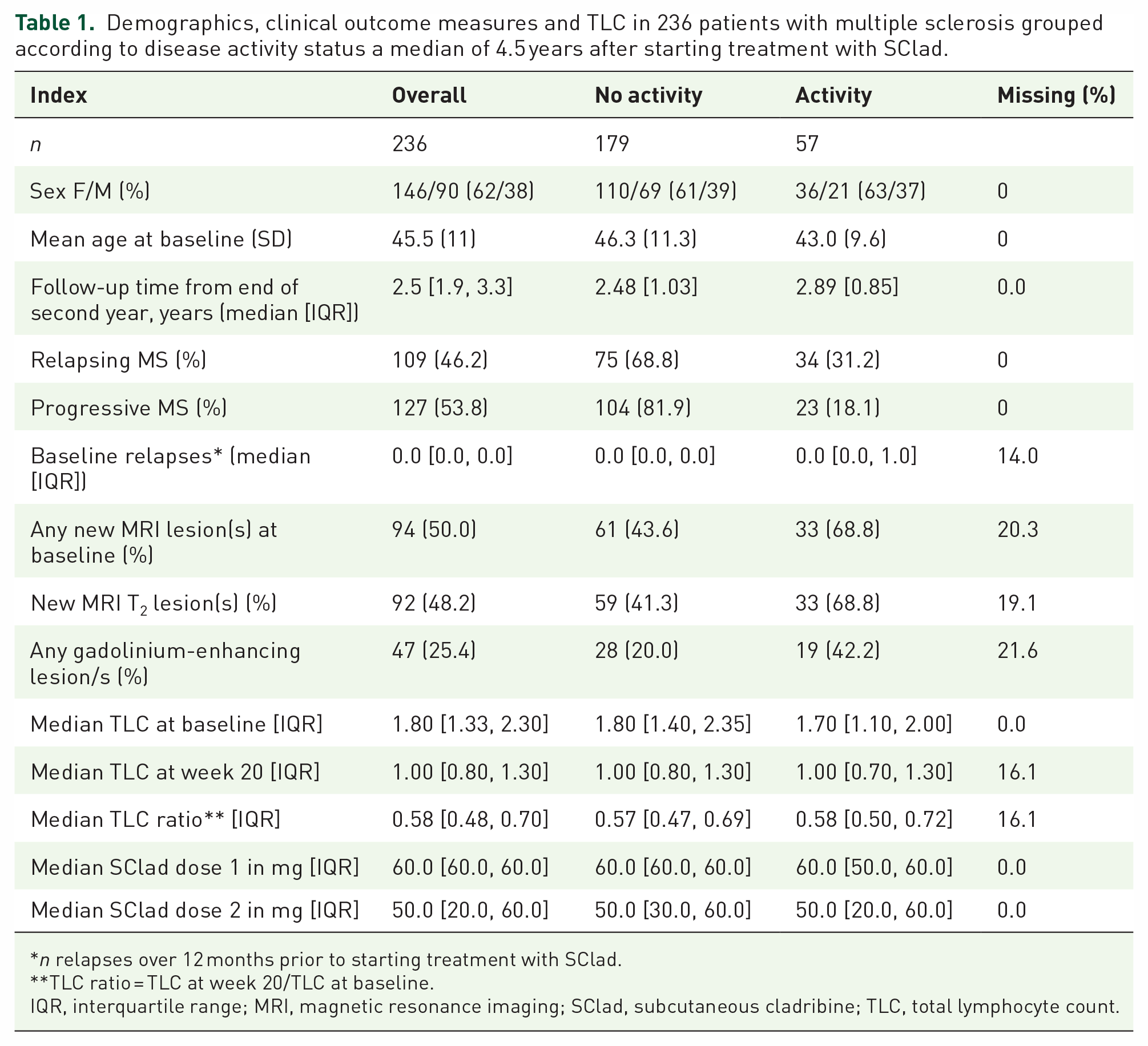
n relapses over 12 months prior to starting treatment with SClad.
TLC ratio = TLC at week 20/TLC at baseline.
IQR, interquartile range; MRI, magnetic resonance imaging; SClad, subcutaneous cladribine; TLC, total lymphocyte count.
In all, 7/236 pwMS died during the follow-up period, having received two courses of SClad (one three courses). Causes of death were encephalitis (1), advanced MS (2), aortic aneurysm and dissection (1), COVID-19 (1), hypoxic brain injury due to choking (1) and pneumonia (1). In total, 2/236 pwMS included in the analysis emigrated 5 and 12 months after completing their second course of SClad, respectively. Since the frequency of reviews in these nine pwMS (seven deaths and two emigrants) was not significantly different from the remainder of the cohort, this small number of patients lost to follow-up was included in the analysis. Excluding these patients from the analysis did not materially affect the results. One pwMS started CladT 5 months after their fourth ocrelizumab infusion. The indication to start ocrelizumab had been a relapse with incomplete recovery 2 months after completing the second course of SClad. This case is included in the ‘received a third course of cladribine’ category (Figure 2). One pwMS received one course of SClad, but not the second course due to prolonged lymphopenia. They then went on to commence CladT treatment 46 months after completing their single course of SClad, however, included among the n = 21 pwMS who did not receive a second course of SClad.
Of 236 pwMS, 179 did not develop new disease activity (Tables 1 and 2). In 57/236 pwMS who did develop new disease activity after two courses of SClad, the decision to offer further immunotherapy was based on relapses in 5 (9%), MRI activity in 29 (51%), both relapses and MRI activity in 13 (23%), deterioration unrelated to relapse activity in 4 (7%), elevated CSF-NfL levels in 3 (5%) and other reasons in 3 (5%). Twenty-two/236 pwMS (14 women, 8 men) who developed new disease activity received further doses of cladribine, a median of 36.7 months (median; IQR: 31.7, 42.1) after completing their second course of SClad. In 11/22 patients (50%), a third course of SClad was administered, and the other 11/22 (50%) received CladT. For the 22 patients who received a third course of SClad or CladT, the median time to ‘reason for re-treatment’ in those with MRI activity and/or relapse activity was 30 months (IQR: 20.4, 36) after completing the second course of SClad.
Clinical characteristics of patients with MS before receiving any SClad.

EDSS, expanded disability status scale; 9HPT, nine-hole peg test; SDMT, symbol digit modality test; T25FW, timed 25 foot walking test.
Twenty-two/236 pwMS (14 women, 8 men) who developed new disease activity received immunotherapies other than cladribine, 19/22 of which received one and 3/22 two subsequent immunotherapies. One patient started ocrelizumab 33.0 months after rituximab, one started natalizumab 14.1 months after ocrelizumab and one underwent autologous HSCT 33.5 months after alemtuzumab.
For those pwMS receiving immunotherapy other than SClad or CladT, the median time to ‘reason for re-treatment’ in those with MRI activity and/or relapse activity was 14.4 months (IQR: 8.4, 28.8) after completing the second course of SClad. For those who had not (yet) started further immunotherapy, the median time to ‘reason for re-treatment’ in those with MRI activity and/ or relapse activity was 42 months (IQR: 33.6, 46.8) after completing the second course of SClad. The median lapse between completing the second course of SClad and commencing subsequent immunotherapy was 18.9 months (IQR: 12.99, 30.21). Specifically, there was a delay of 24.6 months in two/22 pwMS receiving alemtuzumab, 13.2 months in 14/22 receiving ocrelizumab, 34.9 months in 2/22 receiving rituximab and 11.5 months in 2/22 starting siponimod. Two patients started stem cell therapy a median of 11.5 months after their second course of SClad. In all, 13/236 pwMS (eight women, five men) who developed new disease activity had not (yet) started to undergo further immunotherapy. These patients’ further immunotherapy was approved at our multidisciplinary team meeting; however, they had either yet to start (n = 11) or declined further treatment (n = 2). In all, 6/236 pwMS who had undergone SClad treatment were participants in the MS-STAT2 trial of simvastatin in secondary-progressive MS (NCT03387670), and 2/236 were participants in the SIZOMUS trial of ixazomib as an (add-on) immunotherapy (NCT03783416).
Since our dosing schedule of SClad was adjusted not only to body weight but also to individual TLC, we explored the association between the total dose received and re-emerging disease activity. In those eligible for further immunotherapy due to disease activity following two treatment courses of SClad, 42/57 (74%) received the standard dose (60 or 70 mg), depending on body weight 9 ) of SClad for their first, and 21/57 (37%) for their second course. During the third treatment course, 6/22 (27%) received a standard dose of either SClad or CladT. The total tablet dosages were converted to the bioavailable dose. 19 Except for four patients, those who started CladT as follow-on immunotherapy had only received the first of two scheduled courses. Cox proportional hazard models are shown in Table 3. There was strong evidence for the association between each MRI index at baseline and the time to re-emerging disease activity following two courses of SClad with an approximate doubling of the HR (between 2.28 and 2.11; p < 0.02).
Cox proportional hazard models of association between resuming disease activity and demographic, clinical and MRI indices in 236 patients with multiple sclerosis.
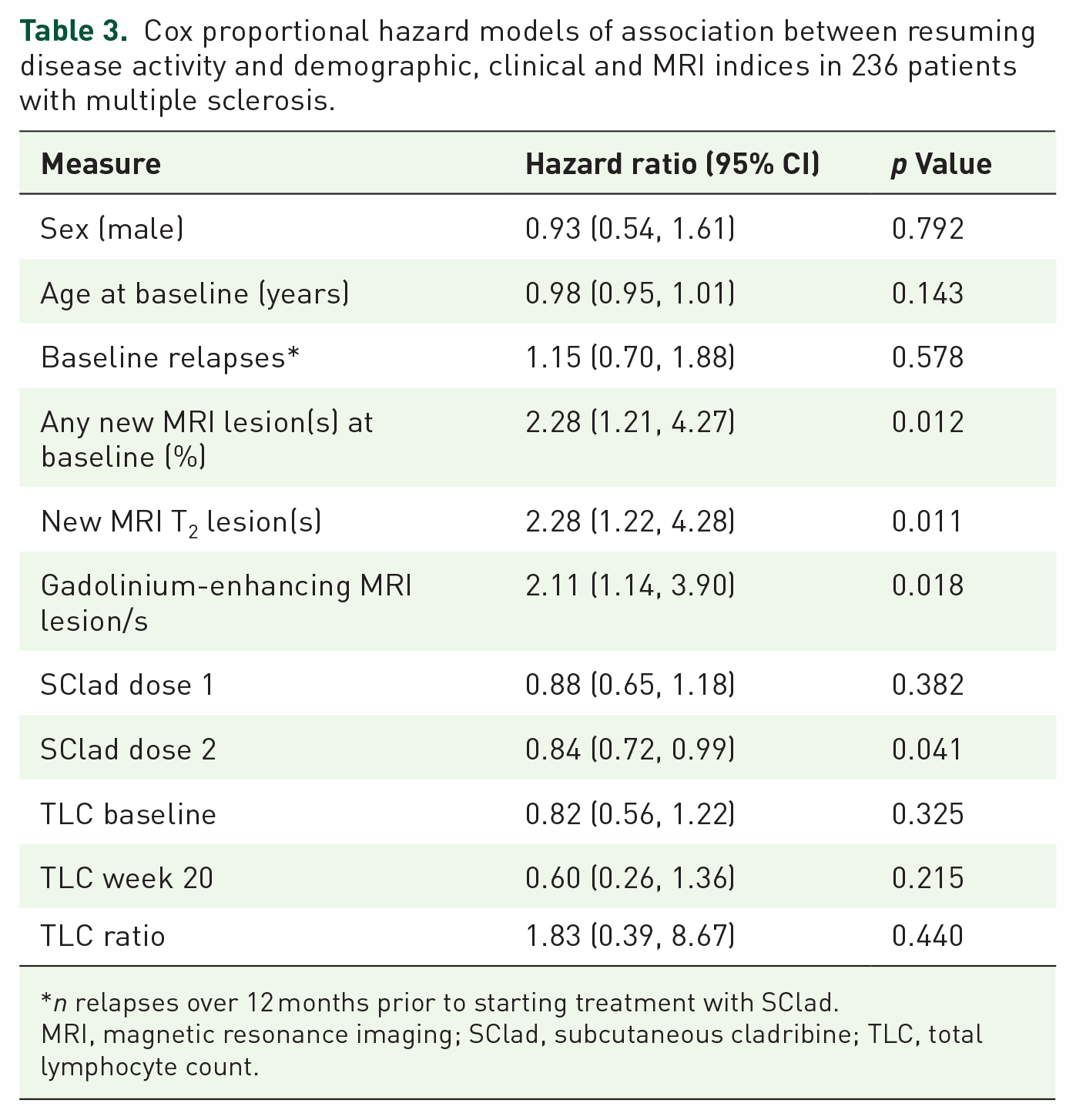
n relapses over 12 months prior to starting treatment with SClad.
MRI, magnetic resonance imaging; SClad, subcutaneous cladribine; TLC, total lymphocyte count.
There was evidence that the amount of SClad administered during the second course was associated with the hazard: each increase of 10 mg of the second dose was associated with a lower hazard of disease activity of approximately 16% (HR: 0.84, 95% CI: 0.72, 0.99). When adjusted for weight in an additional model, this association was even stronger, with a reduction in hazard of 24% for each increase of 10 mg for dose two (HR: 0.76, 95% CI: 0.59, 0.96; Supplemental Table 1). Neither age, sex, the number of relapses over 12 months prior to starting SClad nor the TLC ratio (week 20/baseline) was associated with disease activity. There was no evidence for an association between baseline EDSS and hazard of disease activity (HR: 0.96, 95% CI: 0.84, 1.11; p = 0.598) (Figure 3).

Sankey flow diagram illustrating outcomes of SClad treatment, including (re-)treatment with cladribine (SClad or CladT) and transfer to other immunotherapies, in 264 patients with multiple sclerosis.
Further models for dose, adjusted for weight, are shown in Supplemental Table 1. The association between the second dose and the hazard of disease activity is still present, and the p value is somewhat reduced (HR: 0.76, 95% CI: 0.59, 0.96; p = 0.025).
Discussion
Starting from the regulatory approval of natalizumab in 2006, highly effective immunotherapy (HEI) has increasingly become available to pwMS. 20 Evidence suggests using HEI as a first-line DMT leads to better outcomes than escalation approaches, where HEI is given once disease activity emerges whilst on treatment with moderately active DMT.21,22 Access to HEI, however, remains limited for many pwMS due to cost-effectiveness guidelines in public healthcare systems, 23 let alone lower- and middle-income (LMI) healthcare settings.12,24 Using SClad enabled us to make treatment decisions based on the biology of MS, rather than cost-effectiveness, 25 such that, for example, pwMS with an EDSS beyond 6.5 were able to receive treatment. 9
SClad is available as a generic compound for people with hairy cell leukaemia. Its bioavailability is 100% compared to 42% with CladT. 26 Other than the required dose adjustment, however, CladT is bioequivalent with SClad. 9 Whilst different incipients associated with the two preparations might lead to variations in allergic responses,27,28 the pharmacological effects of equivalent doses are not affected. Cladribine undergoes rapid absorption into plasma, irrespective of the route of administration, and it is the intracellular accumulation and metabolism of cladribine in lymphocytes that is key for its main pharmacological effect. 26
Thus, the high efficacy and comparably low associated risk of cladribine, demonstrated using CladT, 29 renders SClad also an attractive HEI for pwMS in LMI healthcare settings. 12
Mechanistically, cladribine leads to significant alteration of adaptive immune cell subsets. The strong association between (i) inhibition of new lesions on MRI, reduction in relapses and disability accrual and (ii) the time course of B-cell subtype depletion following cladribine treatment, with sustained and significant reduction in memory B cells over at least 24 months,30,31 suggests an important role of this specific B-cell subset for inducing sustained disease control in pwMS.32–34
Monoclonal B-cell-depleting antibodies, such as ocrelizumab, 35 ofatumumab, 36 or rituximab,37,38 lead to similar patterns of B-cell depletion followed by reconstitution of B-cell subsets, except for memory B cells. However, whilst these compounds are given at regular intervals, the standard with cladribine is to administer two courses followed by surveillance for new disease activity.39,40 Supportive evidence for the latter strategy comes from an interim analysis of the CLASSIC-MS (NCT03961204) trial, which revealed that 63.3% of pwRMS did not receive any further immunotherapy for 10 years after their last phase III parent study dose. 41
However, given that any new disease activity may be detrimental to long-term outcomes, there is concern about such a ‘wait and see’ approach,22,42 although current clinical guidelines on the management of pwRMS after completing two courses of CladT are based on expert opinion,7,43 rather than direct evidence. Given our use of SClad in clinical practice dates back nearly 8 years, we were able to explore factors associated with re-emerging disease activity in our cohort of currently 264 pwMS, who received at least one course of SClad.
In line with results from CladT, our data using SClad indicate more than three-quarters of pwMS do not develop new detectable disease activity after a median of 4.5 years from their first treatment course. 44 However, two factors predicted new disease activity: (i) an active MRI at baseline and (ii) a low dose of SClad administered during the second treatment course.
As reported previously,9–12 our dosing regime was driven (i) by European Medicines Agency/Food and Drug Safety Authority concerns relating to lymphopenia and (ii) the mechanism of action guiding our understanding of the potentially major importance of specific lymphocyte subsets. As hypothesized by others 45 and our group,30,32,46 once depleted, memory B cells may not recover for years to re-establish disease activity. The absence of detectable inflammatory disease activity in more than 3/4 of our cohort underpins this. However, in some cases, lymphocyte modulation may not have been of the magnitude and/or duration required to prevent the recurrence of disease activity. We can only speculate that the current dosing regimen of CladT may confer longer-lasting efficacy given the full dose is being administered once patients have recovered to a TLC of 0.8 × 109/L, with no further adjustment as used in our dosing schedule.
Given the prescribing guidelines for CladT require MRI activity for treatment eligibility, at least in some healthcare settings, 47 it is important to recognize that, compared to pwMS with no MRI activity, such activity confers a doubling of risk for new disease activity approximately 4.5 years after commencing treatment. This result is in line with the apparent tailing off of the treatment effect over time 44 and should caution against a ‘wait and see’ approach beyond 4.5 years from starting cladribine treatment.
As described previously, our dosing schedule with SClad was personalized not only for body weight, similar to pwRMS receiving CladT, but also for individual TLC at the first and second treatment courses, leading to a considerable number of pwMS not receiving the full dose of SClad at year 2.9,10 These elements were originally introduced as a safety measure to limit severe lymphopenia which was considered to be an adverse effect, rather than part of the mechanism of action of cladribine. 1 However, our data indicate that there is a stepwise increase in risk of disease activity with such dose adjustment. Thus, in some pwMS, reducing the cladribine dose for safety reasons comes at the cost of incomplete long-term disease control.
Our findings also confirm that TLC and its drop following drug administration are not helpful predictors of treatment response in pwMS receiving cladribine. Whilst evidence suggests lymphocyte subset kinetics following cladribine treatment are more closely associated with the onset of clinical effects, the correlation of re-emerging disease activity with specific subset(s) remains to be demonstrated. 33 Since lymphocyte subsets were not analysed in the cohort reported here, we can only speculate that the apparent dose effect may indicate incomplete long-term suppression of the autoaggressive cell pool. Other variables such as age, sex and number of relapses at baseline were not predictive of re-emerging disease activity. MS immunotherapies used prior to starting SClad are unlikely to impact re-emerging disease activity after two courses of SClad have been completed unless those prior therapies were immune reconstitution therapies (IRTs). As described previously, however, less than 4% of our cohort had most recently been treated with either alemtuzumab or drugs that have IRT properties including mitoxantrone and cyclophosphamide. 9
Limitations
Limitations of this work include the relatively low frequency of investigations used to detect disease activity, particularly during the peak phase of the COVID-19 pandemic, including its impact on MRI scanning capacity. Moreover, some clinical data were missing in this real-world cohort.
Conclusion
In pwMS treated with SClad, MRI activity at baseline, as well as the dose administered at the second treatment time point, significantly impact the likelihood of re-emerging disease activity. Given the current eligibility criteria for CladT requiring MRI activity at baseline, and against the backdrop of the need for ongoing suppression of MS disease activity, our results support the case for re-treatment in pwRMS receiving CladT, after the regular interval of 4 years.
Supplemental Material
sj-docx-1-tan-10.1177_17562864231200627 – Supplemental material for Disease activity 4.5 years after starting cladribine: experience in 264 patients with multiple sclerosis
Supplemental material, sj-docx-1-tan-10.1177_17562864231200627 for Disease activity 4.5 years after starting cladribine: experience in 264 patients with multiple sclerosis by Kimberley Allen-Philbey, Stefania De Trane, Amy MacDougall, Ashok Adams, Lucia Bianchi, Thomas Campion, Gavin Giovannoni, Sharmilee Gnanapavan, David W. Holden, Monica Marta, Joela Mathews, Benjamin P. Turner, David Baker and Klaus Schmierer in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The authors are grateful for support by the clinical BartsMS team based at The Royal London Hospital including Ana Jaramillo, Anastasiia Meryndia, Charles Tugwell, Charlotte Sellers, Emma Ridgway, Freya Edwards, Grace Anjorin, Tatiana Sayali, and Thamanna Begum.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
