Abstract
Background and objectives:
Alemtuzumab demonstrated superior efficacy versus subcutaneous interferon (IFN) beta-1a in participants with relapsing-remitting multiple sclerosis in the 2-year CARE-MS I and II trials. Efficacy was maintained in the 4-year CARE-MS extension, during which alemtuzumab-treated participants (‘alemtuzumab-only’) could receive additional courses upon disease activity, and IFN-treated participants switched to alemtuzumab (‘IFN-alemtuzumab’). Participants who completed the CARE-MS extension could enroll in the open-label TOPAZ study which assessed safety and efficacy for 5–7 years (11–13 years after alemtuzumab/IFN initiation).
Methods:
Participants received additional alemtuzumab courses as needed. Assessments included adverse events (AEs; primary outcome), annualized relapse rate (ARR), 6-month confirmed disability worsening [CDW; ⩾1.0-point Expanded Disability Status Scale (EDSS) score increase or ⩾1.5 if baseline EDSS = 0], and 6-month confirmed disease improvement [CDI; >1.0-point EDSS decrease (baseline score ⩾2.0)].
Results:
43.5% of alemtuzumab-only participants from CARE-MS II and 54.2% from CARE-MS I received no additional alemtuzumab courses; 30.0% and 20.9%, respectively, received one additional course (the median). Incidences of AEs, including thyroid AEs and infections, declined over time. The safety profile of alemtuzumab was similar for participants who received zero, one, or two additional courses. For CARE-MS II participants, who had inadequate response to previous treatment, ARR remained low during Years 3–13 for the alemtuzumab-only [0.17; 95% confidence interval (CI) 0.15–0.20] and IFN-alemtuzumab (0.14; 0.11–0.17) groups. At Year 11, the proportions of participants who were either free from CDW or who had CDI were higher in the alemtuzumab-only group (58% and 49%, respectively) than in the IFN-alemtuzumab group (51% and 37%). For CARE-MS I participants, who were previously treatment-naïve, clinical outcomes remained improved, and no between-group differences were apparent.
Conclusion:
Safety risks associated with alemtuzumab treatment declined over time. Clinical benefits were maintained up to 11–13 years, and most participants did not require more than one additional course.
Clinicaltrials.gov identifiers:
NCT00530348; NCT00548405; NCT00930553; NCT02255656
Keywords
Introduction
Relapsing-remitting multiple sclerosis (RRMS) is a chronic inflammatory neurodegenerative disease that involves demyelination, axonal loss, and disability accumulation over time. 1 Although disease duration can typically last for decades, 2 most clinical trials assess the effects of disease-modifying therapies (DMTs) over a relatively short period of time (typically less than 3 years), 1 which limits the evaluation of long-term disability. 3 It remains vital to establish the enduring benefit-risk balance of DMTs, particularly high-efficacy DMTs, which have the most complex safety profile, but are likely to provide the greatest protection against disability accumulation. 4
Alemtuzumab (Lemtrada®; Sanofi, Cambridge, MA) is a high-efficacy DMT for RRMS that targets CD52-expressing B and T cells for depletion, enabling subsequent repopulation of B and T cells with lower pathogenicity.5,6 Compared with most other DMTs for RRMS that require life-long dosing, 7 treatment with alemtuzumab requires brief and infrequent dosing, with two courses administered 12 months apart and additional courses provided only upon evidence of disease activity. 8 The phase III CARE-MS trials compared the efficacy and safety of IV alemtuzumab versus subcutaneous interferon (IFN) beta-1a (SC IFNB-1a) over 2 years in treatment-naïve participants with RRMS (CARE-MS I; NCT00530348) 9 and participants with inadequate response to prior therapy (CARE-MS II; NCT00548405). 10 Both studies demonstrated significantly greater improvements in clinical and magnetic resonance imaging (MRI) outcomes at Year 2 with alemtuzumab versus SC IFNB-1a, including reductions in relapses, gadolinium (Gd)-enhancing lesions, new or enlarging T2 lesions, and brain volume loss.9,10 A reduction in disability accumulation was also observed for alemtuzumab in CARE-MS II. 10 The most important adverse events (AEs) reported in the CARE-MS studies were infusion-associated reactions, autoimmune conditions (including thyroid disorders), and infections. In addition, several rare but serious AEs have been reported in post-marketing studies, including hemorrhagic stroke, pulmonary alveolar hemorrhage, myocardial infarction, and cervicocephalic arterial dissection, possibly related to infusion. 11
Extension studies support a favorable long-term benefit-risk balance of alemtuzumab. People who completed CARE-MS I and II could enroll in the 4-year CARE-MS extension study (NCT00930553), in which participants who received alemtuzumab in the core studies could receive as-needed additional courses (the ‘alemtuzumab-only’ group), and participants who received SC IFNB-1a in the core studies switched to alemtuzumab (the ‘IFN-alemtuzumab’ group).12–14 People who completed the CARE-MS extension study were invited to enroll into TOPAZ (a long-
This report presents the final data from TOPAZ (up to 13-years of follow-up), with a focus on participants who had an inadequate response to prior therapy and were initially enrolled in the CARE-MS II core study.
Methods
Study design
The study designs for the CARE-MS I and II core studies, the CARE-MS extension study, and the TOPAZ study are illustrated in Supplemental Figure 1.
Results from the 2-year CARE-MS core studies and 4-year CARE-MS extension study have been published previously.9,10,12 In brief, CARE-MS I and II were randomized, rater-blinded, active-controlled trials of IV alemtuzumab 12 mg/day (for five consecutive days at baseline and for three consecutive days 12 months later) versus SC IFNB-1a 44 µg three times/week for 2 years.9,10 The CARE-MS extension study was a single-arm study in which participants who received alemtuzumab in the core study were eligible to receive additional alemtuzumab courses (12 mg/day for three consecutive days ⩾12 months after the previous alemtuzumab course) upon evidence of disease activity (⩾1 relapse or ⩾2 new/enlarging T2 hyperintense and/or Gd-enhancing MRI lesions). 12 Participants who received SC IFNB-1a in the core study discontinued that treatment at the beginning of the extension study and received alemtuzumab 12 mg/day (for five consecutive days and an additional 3-day course ⩾12 months later), with as-needed additional courses as described above. Participants were allowed to receive other licensed DMTs at the discretion of their treating neurologist. The CARE-MS extension also enrolled participants who received alemtuzumab 24 mg/day in CARE-MS II and participants from a phase II study of alemtuzumab; those participants were excluded from these analyses.
TOPAZ was a phase IIIb/IV, international open-label study of participants who completed the CARE-MS extension study at 142 centers (academic medical centers and clinical practices) in 21 countries (Europe, America, Australia, and Israel). For 5 years, participants could receive additional alemtuzumab courses (12 mg/day for three consecutive days ⩾12 months after the previous alemtuzumab course) at the investigator’s discretion upon clinical or radiological evidence of disease activity. Participants could also receive other licensed DMTs at the investigator’s discretion; however, these participants were required to discontinue treatment with other DMTs before receiving an additional alemtuzumab course and were not included in efficacy analyses. The first participant was enrolled into TOPAZ on 7 January 2015, and the last participant completed the study on 15 July 2020.
Participants
Eligibility criteria for the core and extension studies have been published previously.9,10,12 In brief, adults (18–50 years old for CARE-MS I and 18–55 years old for CARE-MS II) with RRMS were eligible to enroll in the core studies. Participants with previously untreated disease were enrolled to CARE-MS I. 9 Participants who had been previously treated (most commonly with IFNB-1a and/or glatiramer), but still had relapse activity, were enrolled into CARE-MS II. 10 Participants who completed the core studies were eligible to enroll into the CARE-MS extension study. 12 Participants could continue into TOPAZ if they had completed at least 48 months in the extension study.
Primary endpoint (Safety)
The primary endpoint of TOPAZ was long-term safety of alemtuzumab, evaluated via treatment-emergent AEs (TEAEs), serious TEAEs (defined as per Good Clinical Practice guidelines), AEs of special interest (AESIs), and clinically significant changes in laboratory parameters. AESIs included autoimmune-mediated conditions, malignancy, serious infections, and infusion-associated reactions. Infusion-associated reactions were defined as any AE with onset during or within 24 h of an alemtuzumab infusion. AEs were reported at least every 6 months. Laboratory parameters were measured monthly for 48 months after an alemtuzumab course.
Secondary endpoints (Efficacy)
The Expanded Disability Status Scale (EDSS) was assessed by qualified practitioners during scheduled 6-monthly study visits and at the time of a suspected relapse. Relapses required objective signs on neurological assessment lasting at least 48 h and present at normal body temperature, and were verified at each 6-monthly study visit. Brain MRI scans were performed every 12 months and read by blinded assessors at NeuroRx Research (Montréal, QC Canada; for lesion-based analyses) and the Cleveland Clinic [Cleveland, OH, USA; for brain parenchymal fraction (BPF) analysis].
Clinical efficacy endpoints included the proportions of participants free of 6-month confirmed disability worsening [CDW; ⩾1.0-point increase in EDSS from core study baseline (or ⩾1.5-point increase if baseline EDSS was 0)] and with 6-month confirmed disability improvement (CDI; >1.0-point decrease in EDSS from baseline in participants with core study baseline EDSS score ⩾2.0); change from baseline in EDSS score; the proportions of participants with stable (⩽0.5 points change) or improved (⩾1.0-point decrease) EDSS score relative to baseline; annualized relapse rate (ARR); and the proportion of participants who remained relapse free.
MRI outcomes included the cumulative proportion of participants without MRI disease activity [no new Gd-enhancing lesions, new/enlarging T2 hyperintense lesions, or new T1 (and new hypointense T1) lesions since the last scan]; the cumulative proportion of participants without new Gd-enhancing lesions; the cumulative proportion of participants without new/enlarging T2 lesions; and annual brain volume loss assessed via BPF. Distribution-free estimates for annual BPF were obtained for the CI of the median.
Statistical analyses
Analyses were based on all available data (without imputation) for all alemtuzumab-only and IFN-alemtuzumab participants. All analyses were stratified by core study (CARE-MS I or II). Yearly interim analyses were performed.
Analyses of TEAEs included the exposure-adjusted incidence rates (EAIRs), calculated as the number of participants with the first TEAE in the time interval divided by the total follow-up duration in years for all participants in that time interval (censoring at the time of TEAE for participants counted in the numerator), multiplied by 100. The proportions of participants with CDW or CDI and the proportion who remained relapse free were estimated using Kaplan–Meier estimates. ARR was calculated using negative binomial regression with robust variance estimation adjusted for geographic region.
Exploratory post hoc analyses of participant subgroups, according to the number of alemtuzumab courses received (0 to 5+) and the time since last infusion (⩽30 days to 4+ years), were conducted for participants receiving alemtuzumab 12 mg/day during CARE-MS II who subsequently entered the extension trials.
Safety and efficacy endpoints were analyzed using summary statistics (n, mean, median, range, SD, and standard error for continuous variables; frequencies and percentages for categorical variables). No formal hypothesis testing was done, although the two-sided 95% CI was calculated where appropriate.
Standard protocol approvals, registrations, and patient consents
The study was conducted in accordance with the principles laid out in the Declaration of Helsinki and all applicable local, national, and international regulations and guidelines. The approving ethics boards are listed in the Supplemental Material. All participants signed an informed consent form before enrolling in the study. This trial is registered with ClinicalTrials.gov (NCT02255656).
Results
Participants
CARE-MS II
Among participants who enrolled in the CARE-MS II study, 207/435 (48%) initially randomized to alemtuzumab, and 78/202 (39%) initially randomized to SC IFNB-1a continued in the study extensions through Year 11 (Figure 1). Follow-up continued for up to two additional years for some participants. In the alemtuzumab-only group, 61 participants completed Year 12 and 8 completed Year 13; in the IFN-alemtuzumab group, 24 completed Year 12 and 3 completed Year 13.

Disposition of participants from the 2-year core CARE-MS II study through to the CARE-MS and TOPAZ extension studies.
In the extension studies, 222/393 (56.5%) participants in the alemtuzumab-only group and 61/143 (42.7%) in the IFN-alemtuzumab group received at least one additional course of alemtuzumab [i.e. three or more courses; Figure 2(a)]; 30.0% (alemtuzumab-only) and 28.0% (IFN-alemtuzumab) of participants received exactly three courses. A total of 150/393 (38.2%) participants in the alemtuzumab-only group and 74/143 (51.7%) in the IFN-alemtuzumab group did not receive additional alemtuzumab courses or other DMT. During the first year of TOPAZ (Year 7), 31/336 (9.2%) participants in the alemtuzumab-only group and 18/123 (14.6%) in the IFN-alemtuzumab group received an additional course [Figure 2(b)–(c)]. Participants received a median (interquartile range) of 1 (1–2) additional alemtuzumab courses during the extension and TOPAZ studies. A total of 37.7% of participants in the alemtuzumab-only group who completed TOPAZ received no additional courses, with 31.4% receiving one, 18.4% receiving two, 9.7% receiving three, and 2.9% receiving four or more.
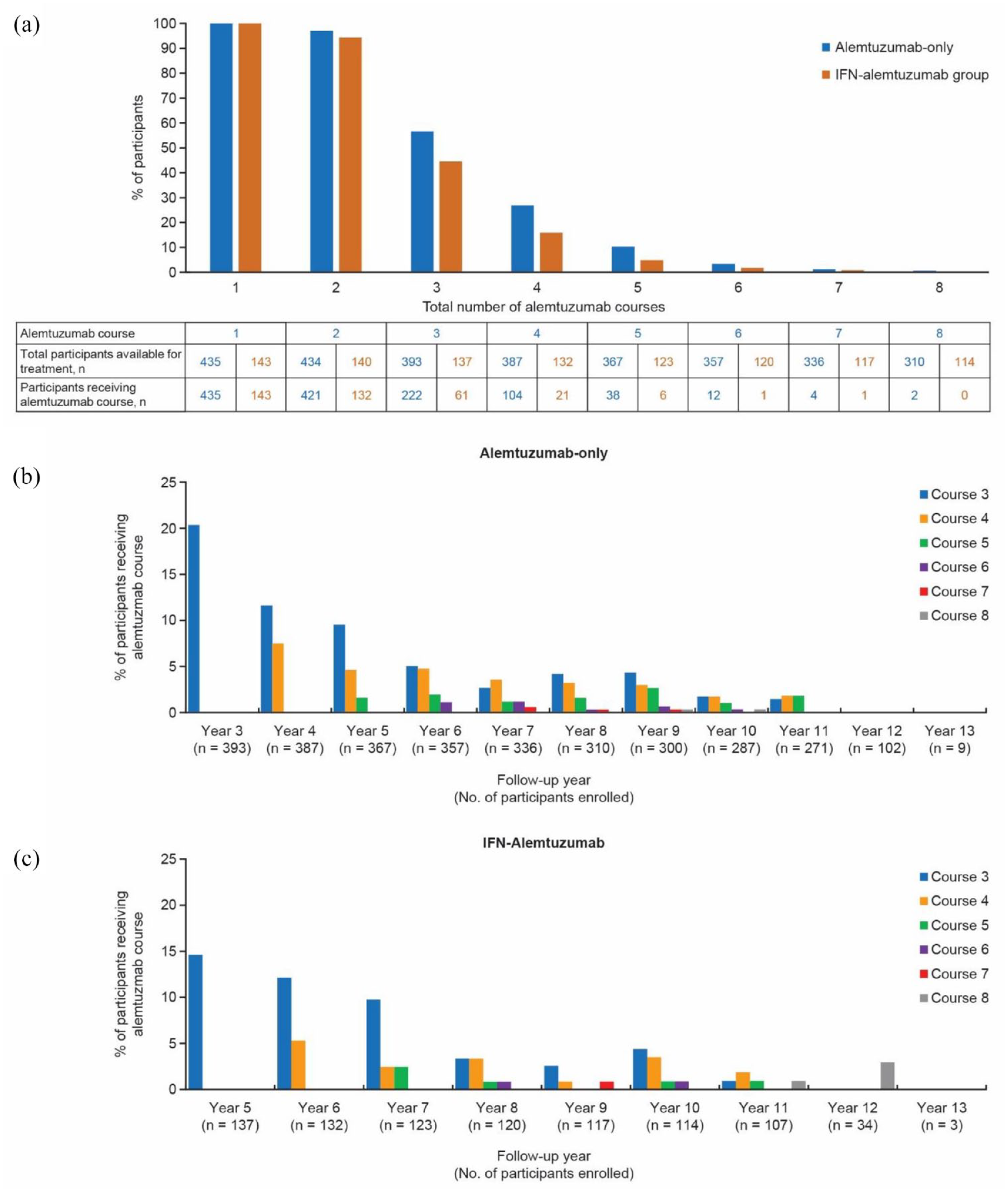
(a) Total number of alemtuzumab courses during the CARE-MS II core study and CARE-MS and TOPAZ extension studies. Participants in the IFN-alemtuzumab group were eligible to receive SC IFNB-1a during Years 1–2 and alemtuzumab during Years 3–11. (b and c) Receipt of alemtuzumab courses 3–8 by study year for participants in the CARE-MS (Years 3–6) and TOPAZ (Year 7–13) extension studies in the (b) alemtuzumab-only group (n = 393) and (c) IFN-alemtuzumab group (n = 143). SC IFNB-1a, subcutaneous interferon beta-1a.
Characteristics of participants at CARE-MS II baseline and at entry to TOPAZ are presented in Table 1. Participants who received at least one additional alemtuzumab course during the extension were slightly younger than participants who did not receive additional courses, whereas participants who received three or more additional courses had higher mean Gd-enhancing lesion counts 2 years after treatment initiation (Supplemental Table 1). There were no notable differences in baseline characteristics between participants who completed Year 11 and those who enrolled in TOPAZ (data not shown).
Demographics and disease characteristics for participants from TOPAZ at CARE-MS II core study baseline and at the start of TOPAZ.

Participants in the IFN-alemtuzumab group were eligible to receive SC IFNB-1a during Years 1–2 and alemtuzumab during Years 3–11.
Values shown are mean (SD) except where stated.
EDSS, Expanded Disability Status Scale; Gd, gadolinium; SC IFNB-1a, subcutaneous interferon beta-1a.
CARE-MS I
Among participants who initially enrolled in CARE-MS I, 257/376 (68%) assigned to alemtuzumab, and 109/139 (78%) assigned to SC IFNB-1a, continued through Year 11 (Supplemental Figure 2). Follow-up continued for up to two additional years for some participants. In the alemtuzumab-only group, 140 completed Year 12 and 17 completed Year 13; in the IFN-alemtuzumab group, 49 completed Year 12 and 4 completed Year 13.
In the extension studies, 160/349 (45.8%) participants in the alemtuzumab-only group and 63/139 (45.3%) in the IFN-alemtuzumab group received at least one additional course of alemtuzumab during the study extensions [Supplemental Figure 3(a)]; 20.9% (alemtuzumab-only) and 26.6% (IFN-alemtuzumab) of participants received exactly three courses. A total of 181/349 (51.9%) participants in the alemtuzumab-only group and 73/139 (52.5%) in the IFN-alemtuzumab group did not receive additional alemtuzumab courses or other DMT. During the first year of TOPAZ (Year 7), 38/321 (11.8%) participants initially randomized to alemtuzumab and 13/122 (10.7%) initially randomized to SC IFNB-1a received an additional course [Supplemental Figure 3(b)–(c)].
Characteristics of participants at CARE-MS I baseline and at entry to TOPAZ are presented in Supplemental Table 2.
Safety endpoints
CARE-MS II
Alemtuzumab had a consistent safety profile over 13 years (Table 2). During the overall time period of alemtuzumab treatment, TEAEs were reported for 434/435 (99.8%) of participants in the alemtuzumab-only group (Years 0–13) and for 140/143 (97.9%) in the IFN-alemtuzumab group (Years 3–13), with the incidence of TEAEs and AESIs generally declining over time. Serious TEAEs were reported for 208/435 (47.8%) and 57/143 (39.9%) of participants, respectively, during the same period. Among all participants who received at least one course of alemtuzumab, the cumulative incidence of thyroid TEAEs was 44.6% and the cumulative incidence of immune thrombocytopenia was 3.7%. Infusion-associated reactions were reported for 399/435 (91.7%) participants in the alemtuzumab-only group and for 130/143 (90.9%) in the IFN-alemtuzumab group. TEAEs led to treatment withdrawal for 22/435 (5.1%) and 8/143 (5.6%) of participants in the alemtuzumab-only and IFN-alemtuzumab groups, respectively.
Incidence (%) of AEs for participants from CARE-MS II.

Includes 9 CARE-MS II participants who were randomized to receive alemtuzumab 24 mg, but were administered the 12 mg dose.
EAIR was calculated as follows: (number of participants with first AE in the time interval)/(total follow-up duration (years) of all participants within the time interval, censoring at the time of AE for participants counted in the numerator) × 100.
First occurrence of AE for a patient.
Participants in the IFN-alemtuzumab group were eligible to receive subcutaneous interferon beta-1a (SC IFNB-1a) during Years 1–2 and alemtuzumab during Years 3–11.
AE, adverse event; EAIR, exposure-adjusted incidence rate; ITP, immune thrombocytopenia.
In exploratory subgroup analyses, the AE profile of alemtuzumab was similar between participants who received zero, one, or two additional courses during the extension phase (Supplemental Table 3). The 38 participants who received a third additional course generally experienced fewer TEAEs. Infusion-associated reactions were the most common TEAEs in the first 30 days post-infusion, with infections and MS relapse most common in the 31 days to 2 years after infusion. Considerably fewer TEAEs were observed more than 2 years after infusion.
Participants who received their third alemtuzumab course within 2 years of the previous two courses experienced similar safety events for the remainder of the study to those who received their third course 2 or more years later, with somewhat higher frequency (Supplemental Table 4).
CARE-MS I
During the overall time period of alemtuzumab treatment, TEAEs were reported for 370/376 (98.4%) of participants in the alemtuzumab-only group (Years 0–13) and for 136/139 (97.8%) in the IFN-alemtuzumab group (Years 3–13) (Supplemental; Table 5). Serious TEAEs were reported for 152/376 (40.4%) and 53/139 (38.1%) of participants, respectively, during the same time period. TEAEs led to treatment withdrawal for 6/376 (1.6%) and 2/139 (1.4%) of participants, respectively.
A total of 11 deaths occurred during TOPAZ, including two from cancer, three from septic shock, one from suicide, one acute and organizing bronchopneumonia, one from atrioventricular block during an unrelated medical procedure, and three from unknown causes. The two cancer deaths, which occurred in men who participated in CARE-MS I, were deemed related to alemtuzumab by the treating physician. One participant was diagnosed with metastatic rectal cancer 9 years 8 months after the initial course of alemtuzumab, and died 8 months later at 55 years of age due to this cancer. The other participant was diagnosed with metastatic carcinoma of unspecified localization 2 years 7 months after the first course of alemtuzumab, and died 7 weeks later at 52 years of age due to this cancer.
Efficacy endpoints
CARE-MS II
ARRs across the study extension period (Years 3–13) were 0.17 (95% CI 0.15–0.20) in the alemtuzumab-only group and 0.14 (0.11–0.17) in the IFN-alemtuzumab group [Figure 3(a)]. During the extension studies, ARRs decreased from 0.22 (95% CI 0.18–0.27) at Year 3–0.10 (0.06–0.15) at Year 11 in the alemtuzumab-only group and from 0.12 (0.07–0.19) at Year 3–0.09 (0.04–0.19) at Year 11 in the IFN-alemtuzumab group.

Changes in clinical outcomes among participants from CARE-MS II. (a) ARR was estimated using a negative binomial model. (b) Mean EDSS score from baseline to Year 11 and the percentage of participants for whom EDSS score was stable (⩽0.5-point change in either direction) or improved (⩾1.0-point decrease) relative to baseline. (c) Proportion of participants free of 6-month CDW, defined as ⩾1.0-point increase in EDSS score or ⩾1.5-point increase if the baseline EDSS score was 0, confirmed over 6 months. (d) Proportion of participants with 6-month CDI, defined as ⩾1.0-point decrease in EDSS score from baseline (assessed only in participants with baseline EDSS ⩾2.0), confirmed over 6 months.
Mean changes in EDSS score from baseline to Year 11 were +0.28 (95% CI 0.03–0.54) points in the alemtuzumab-only group and +0.08 (95% CI 0.22–0.38) points in the IFN-alemtuzumab group [Figure 3(b)]. Over this time period, 45.9% of participants in the alemtuzumab-only group and 47.0% of participants in the IFN-alemtuzumab group had stable EDSS scores (i.e. ⩽0.5-point change from baseline); 20.5% and 23.5%, respectively, had improved EDSS scores [i.e. ⩾1.0-point increase; Figure 3(b)]. The mean (SD) EDSS score at baseline was slightly higher for participants who had improved EDSS scores at Year 11 [3.0 (1.2)] compared with participants who had stable [2.5 (1.1)] or worsened [2.6 (1.6)] EDSS scores at Year 11. At Year 11, 58% and 51% of participants in the alemtuzumab-only and IFN-alemtuzumab groups, respectively, were free of 6-month CDW; 49% and 37%, respectively, had 6-month CDI [Figure 3(c)–(d)].
A Sankey plot was used to visualize EDSS score trajectories in participants with stable, improved, or worsened EDSS scores at Year 2 (Supplemental Figure 4). Of the 87.5% of participants with stable or improved EDSS scores at Year 2 who were assessed at Year 10 or later, 76.2% remained stable or improved relative to baseline at follow-up. Of the 12.5% of participants with worsened EDSS scores at Year 2 who were assessed at Year 10 or later, 93.8% had worsened EDSS scores relative to baseline at follow-up.
Exploratory post hoc analyses of ARR and EDSS data over time for participants who received four or more (4+) and, separately, five or more (5+) alemtuzumab courses were performed. ARRs were reduced by 86.6% and 42.9% 12 months after course 4 and 5, respectively, compared with 12 months prior [Supplemental Figure 5(a)]. EDSS remained stable following 4+ courses, though sample sizes were small and estimates more variable for 5+ courses [Supplemental Figure 5(b)].
Freedom from MRI disease activity during Years 3–11 was observed for 29.3% (95% CI 20.0–38.7%) of participants in the alemtuzumab-only group and 23.1% (9.9–36.3%) in the IFN-alemtuzumab group (Figure 4). During Years 3–11, similar proportions (95% CI) of participants in the alemtuzumab-only and IFN-alemtuzumab groups remained free of Gd-enhancing lesions [58.7% (48.6–68.8%) and 64.1% (49.0–79.2%)], new/enlarging T2 hyperintense lesions [28.4% (19.4–37.5%) and 23.1% (9.9–36.3%)], and new T1 hypointense lesions [59.8% (49.8–69.8%) and 59.0% (43.5–74.4%)].

MRI lesion outcomes through Year 11 among participants from CARE-MS II. (a and b) Freedom from MRI disease activity each year for participants who received (a) alemtuzumab-only and (b) SC IFNB-1a in Years 0–2, then alemtuzumab in Years 3–11. (c) Freedom from MRI disease activity, Gd-enhancing lesions, and new/enlarging T2 lesions. MRI disease activity was defined as new Gd-enhancing T1 lesions on current MRI or new/enlarging T2 hyperintense lesions since the last MRI. The percentage of participants free of MRI disease activity was based on the average percentages and the distribution range (minimum, maximum) during both the core study (Years 1–2) and extension studies (Years 3–11). CI, confidence interval; Gd, gadolinium; SC IFNB-1a, subcutaneous interferon beta-1a; MRI, magnetic resonance imaging.
The annual extent of brain atrophy, measured as annual percentage change in BPF, stabilized over time (Figure 5). At Year 11, the median percent change from baseline in BPF was −1.58% (95% CI −1.92 to −1.30%) in the alemtuzumab-only group and –1.68% (–2.30 to –1.22%) in the IFN-alemtuzumab group.

Brain volume loss through Year 11 in participants from CARE-MS II who received (a) alemtuzumab-only and (b) SC IFNB-1a in Years 0–2, then alemtuzumab in Years 3–11.
Participants who discontinued the extension studies early had similar or slightly lower ARRs compared with the overall population (Year 11: 0.071 versus 0.097, respectively), reduced CDI (Year 9 onwards: 37.0% versus 42.7%), and higher baseline EDSS (2.9 versus 2.7), albeit with comparable CDW by the end of the extension (36.3% versus 35.4%) and a similar proportion of participants experiencing at least one relapse during the extension (51.5% versus 56.5%).
An exploratory Kaplan–Meier analysis of incident relapse after the last alemtuzumab course received by the number of additional courses received during the extension studies was conducted (Supplemental Figure 6). The percentage of participants without relapse was similar for zero to two additional courses until approximately 36 months, after which a higher percentage of participants who received no additional courses remained relapse free than participants who received one or two additional courses. Sample sizes for three or more additional courses were insufficient for further comparison.
CARE-MS I
ARRs across the study extension period (Years 3–13) were 0.14 (95% CI 0.12–0.17) in the alemtuzumab-only group and 0.14 (0.11–0.17) in the IFN-alemtuzumab group [Supplemental Figure 7(a)]. During the extension studies, ARRs were 0.19 (95% CI 0.15–0.25) at Year 3 and 0.11 (0.07–0.17) at Year 11 in the alemtuzumab-only group, and 0.10 (0.06–0.17) at Year 3 and 0.15 (0.09–0.26) at Year 11 in the IFN-alemtuzumab group.
At Year 11, there was a marginal accumulation of disability with mean changes in EDSS score from baseline of +0.22 (95% CI 0.05–0.40) points in the alemtuzumab-only group and +0.50 (95% CI 0.21–0.79) points in the IFN-alemtuzumab group [Supplemental Figure 7(b)]. At Year 11, 65% and 64% of participants in the alemtuzumab-only and IFN-alemtuzumab groups, respectively, were free of 6-month CDW; 42% and 39%, respectively, had 6-month CDI [Supplemental Figure 7(c)-(d)].
Freedom from MRI disease activity during Years 3–11 was observed for 27.7% (95% CI 20.7–34.6%) of participants in the alemtuzumab-only group and 33.8% (23.2–44.3%) in the IFN-alemtuzumab group (Supplemental Figure 8). Similar proportions of participants in the alemtuzumab-only and IFN-alemtuzumab groups remained free of Gd-enhancing lesions and, separately, new/enlarging T2 lesions.
The median annual percent change from the previous year in BPF stabilized over time (Supplemental Figure 9). At Year 11, the median percent change from baseline in BPF was −2.20% (95% CI −2.51 to −2.06) in the alemtuzumab-only group and −2.30 (95% CI −2.45 to −1.78) in the IFN-alemtuzumab group.
Discussion
To our knowledge, this report represents the longest prospective follow-up of people with RRMS who received alemtuzumab and is the most comprehensive description of alemtuzumab’s benefit-risk profile for RRMS. Over 13 years, approximately one-half of participants received additional alemtuzumab courses after their first two courses, with 30% of CARE-MS II participants and 21% of CARE-MS I participants receiving only one additional course. Safety risks associated with alemtuzumab treatment declined over time in people with RRMS who had failed to attain disease control on another DMT (i.e. CARE-MS II participants) or were previously treatment-naïve (i.e. CARE-MS I participants), and were similar for participants who did or did not receive additional courses. Efficacy findings suggest that the clinical benefit of alemtuzumab is maintained for at least 11 years after the initial course in most people with RRMS, with disability improvement or stabilization being more common for CARE-MS II participants who initiated alemtuzumab treatment 2 years earlier than participants who received alemtuzumab after 2-year treatment with SC IFNB-1a.
Potential AEs associated with alemtuzumab treatment are well characterized, detectable with regular monitoring, and generally manageable if identified early. 17 Data from long-term trials, including TOPAZ and post-marketing studies, show that the benefit-risk profile of alemtuzumab becomes more favorable over time as AE incidence declines.18–20 In a previous analysis of TOPAZ data, participants receiving additional courses of alemtuzumab had a consistent safety profile up to Year 8 compared with those who received two courses and the overall study cohort. 20 There was no evidence of increased rates of infections, malignancies, or autoimmune AEs with additional alemtuzumab courses. The present analysis supports and extends these findings by showing that participants who received their third course 2 or more years after their second course experienced fewer AEs than participants who received their third course within 2 years. It is important to note that these results may be influenced by a possible lead bias resulting from AEs with delayed onset, particularly thyroid AEs which are most commonly reported within 2 years after a course of alemtuzumab.11,19 Further insights on the timing of additional alemtuzumab courses and other best practices for maximizing safety are expected to arise from ongoing, prospective real-world studies.18,21
Efficacy results from the present phase III/IV studies support previous findings. An analysis of pooled data from two open-label studies of 87 participants who received alemtuzumab over a median follow-up of 7 years reported that about one-half of participants required additional alemtuzumab courses. 22 In agreement with the present results, this previous analysis reported that alemtuzumab treatment was associated with low mean ARR (0.16), stable mean EDSS score, and 6-month CDI and CDW in 44% and 32% of participants, respectively. In several large, real-world studies, 44–67% of people with RRMS achieved no evidence of disease activity (NEDA-3) status at 2–3 years after initiation of alemtuzumab treatment.23–29 Results from TOPAZ indicate that alemtuzumab’s efficacy is durable beyond 7 years. Particularly notable is the consistently higher percentage of alemtuzumab-only than IFN-alemtuzumab participants from CARE-MS II with 6-month CDI during TOPAZ, suggesting that early switching to alemtuzumab treatment has the greatest probability of reversing preexisting disability in people with RRMS who have inadequate disease control on another DMT.
There is debate about whether high-efficacy DMTs, such as alemtuzumab, should be used as first-line RRMS therapies for people with or without highly active disease or only after unsuccessful treatment with other DMTs that carry fewer safety concerns.30,31 These different treatment strategies are currently being examined in two randomized clinical trials. 32 Available evidence from real-world studies indicates that early versus delayed initiation of high-efficacy DMTs is associated with improved long-term disability outcomes.33–35 However, in the USA, the prescribing information for alemtuzumab indicates that treatment should generally be reserved for people with MS who have had an inadequate response to two or more approved therapies. In Europe, the European Medicines Agency has issued a provisional measure that new treatment with alemtuzumab should only be initiated in adults with highly active RRMS who have received at least two other DMTs, or in adults with highly active RRMS where all other DMTs are contraindicated or otherwise unsuitable. The present work does not provide direct evidence in support of or against these regulatory guidelines since efficacy comparisons for participants from CARE-MS I and II are limited due to differences in baseline characteristics. Nevertheless, participants who initially randomized to alemtuzumab in CARE-MS I versus CARE-MS II were less likely to require additional alemtuzumab courses or other DMT (14% point difference) and were more likely to remain free from 6-month CDW (7% point difference at Year 11).
Strengths of this work include a long duration of follow-up, a large population, and broad inclusion criteria which maximize the generalizability of the findings. In addition, this study had a high retention rate relative to follow-up duration (54.3% of participants from the core trials completed TOPAZ), possibly due to effective control of disease activity. Considering the limitations of this study, it is possible that a selection bias exists since participants who discontinued during the extension period had slightly higher baseline disability than the overall population, which may have skewed EDSS results. Selection bias may also apply for analyses grouped by additional alemtuzumab courses as participants with mild disease would have required fewer alemtuzumab courses than participants with more aggressive disease. However, participants were allowed to continue in the extension studies even if they did not receive additional alemtuzumab courses, though they were required to discontinue other DMTs prior to receiving additional alemtuzumab courses.
In conclusion, this analysis supports a positive, long-term benefit-risk balance of alemtuzumab for people with RRMS who had an inadequate response to other DMTs or were previously treatment-naïve. Over 11 years, alemtuzumab’s therapeutic effects on ARR, disability, and MRI outcomes, including annual brain volume loss, were maintained and most participants either did not require additional courses or needed just one additional course. Risks accompanying the beneficial therapeutic effects of alemtuzumab reduced over time, and no new safety signals emerged with additional courses.
Supplemental Material
sj-docx-1-tan-10.1177_17562864231194823 – Supplemental material for Safety and efficacy with alemtuzumab over 13 years in relapsing-remitting multiple sclerosis: final results from the open-label TOPAZ study
Supplemental material, sj-docx-1-tan-10.1177_17562864231194823 for Safety and efficacy with alemtuzumab over 13 years in relapsing-remitting multiple sclerosis: final results from the open-label TOPAZ study by Alasdair J. Coles, Anat Achiron, Anthony Traboulsee, Barry A. Singer, Carlo Pozzilli, Celia Oreja-Guevara, Gavin Giovannoni, Giancarlo Comi, Mark S. Freedman, Tjalf Ziemssen, Debora Shiota, Andreea M. Rawlings, Alana T. Wong, Magdalena Chirieac and Xavier Montalban in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The authors and Sanofi thank the participants and their caregivers for their participation in the trial. Writing assistance, including assistance drafting and editing of the article text, figures, and tables, as directed by the authors; data checking and incorporation of comments from reviewers; and assisting with the submission process was provided by Autumn Kelly, MPC, and Conor F. Underwood, PhD, of Envision Pharma Group.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
