Abstract
Background:
ADVANCE was a phase III trial of the efficacy and safety of subcutaneous peginterferon beta-1a 125 µg every 2 or 4 weeks in patients with relapsing-remitting multiple sclerosis (RRMS). ATTAIN was a 2-year extension study of ADVANCE. The aim was to evaluate the long-term safety, tolerability, and efficacy of peginterferon beta-1a 125 µg every 2 or 4 weeks in ATTAIN.
Methods:
ADVANCE dosing schedules were maintained in ATTAIN, except that every-4-weeks dosing patients were switched to every-2-weeks dosing after conversion of the study to an open-label protocol. ATTAIN was considered complete when the last patient completed the 96-week extension study. Primary endpoints included adverse event (AE) and serious AE (SAE) incidence. Secondary endpoints included relapse, magnetic resonance imaging, and disability outcomes.
Results:
Of the 1512 patients randomized in ADVANCE, 1076 (71%) continued treatment in ATTAIN; of these, 842 (78%) completed the open-label extension study. During ATTAIN, 478 patients (87%) in the every-2-weeks group and 471 patients (89%) in the every-4-weeks group experienced an AE; SAEs were reported in 90 patients (16%) in the every-2-weeks group and 113 patients (21%) in the every-4-weeks group. The most frequent AEs reported were injection site reactions and flu-like symptoms, both of which numerically decreased over time. Peginterferon beta-1a every 2 weeks versus every 4 weeks significantly reduced the adjusted annualized relapse rate over 6 years (0.188 versus 0.263, p = 0.0052) and the risk of relapse over 5 years (36% versus 49%, p = 0.0018). Fewer new T1, new/newly enlarging T2, and gadolinium-enhancing magnetic resonance imaging lesions were observed with every-2-weeks dosing than every-4-weeks dosing over 4 years.
Conclusions:
Results from the ADVANCE extension study, ATTAIN, confirm the favorable long-term safety and tolerability profile of peginterferon beta-1a in patients with RRMS and provide additional evidence for the clinical and radiological benefits associated with this therapy.
Introduction
Peginterferon beta-1a, a pegylated form of interferon (IFN) beta-1a, is approved for the treatment of relapsing forms of multiple sclerosis (RMS). It has a prolonged half-life that permits a reduced dosing frequency compared with other beta IFNs and is administered at a subcutaneous (SC) dose of 125 µg every 2 weeks.1–3
In the first year of the 2-year pivotal phase III ADVANCE study, patients receiving SC peginterferon beta-1a every 2 weeks or every 4 weeks demonstrated significant reductions in both clinical and magnetic resonance imaging (MRI) outcomes compared with placebo. 4 Peginterferon beta-1a had a similar safety profile compared with established IFNs, and the most common adverse events (AEs) described were injection site erythema and influenza-like illness. 4 After 1 year, patients receiving placebo were re-randomized to treatment with peginterferon beta-1a every 2 weeks or every 4 weeks, and patients receiving active treatment in year 1 continued on the same dosing regimen. 5 Efficacy was maintained over 2 years in the continuous peginterferon beta-1a groups, with greater efficacy observed with every-2-weeks versus every-4-weeks dosing. Moreover, peginterferon beta-1a continued to be well tolerated. 5
Post-hoc analyses of ADVANCE data revealed that peginterferon beta-1a every 2 weeks significantly reduced confirmed disability worsening (CDW) over 2 years compared with delayed treatment (placebo in year 1 followed by peginterferon beta-1a every 2 or 4 weeks in year 2). The every-2-weeks dosing regimen also significantly reduced the proportion of patients experiencing relapses associated with 24-week CDW over 2 years compared with the every-4-weeks regimen, indicating that the beneficial effect of peginterferon beta-1a on risk of CDW may be attributable to both an overall reduction in the risk of relapses and an improvement in recovery from relapses.6,7 In addition, compared with the delayed-treatment group, there were significantly more patients in the peginterferon beta-1a every-2-weeks treatment group who had no evidence of disease activity, including both MRI and clinical components, at 2 years. 8 Treatment with peginterferon beta-1a every 2 weeks also improved health-related quality-of-life measures in patients with 12-week CDW or a recent relapse. 9
The ATTAIN study was an open-label extension study of ADVANCE. The objectives of the ATTAIN study were to evaluate the long-term safety, tolerability, and efficacy of SC peginterferon beta-1a 125 µg in patients originally treated in the ADVANCE study who continued treatment, and to describe long-term multiple sclerosis (MS) outcomes in these patients.
Materials and methods
Study design and participants
ADVANCE was a 2-year, multicenter, randomized, double-blind, parallel-group phase III study with a 1-year placebo-controlled period conducted at 183 sites in 26 countries. During year 1 of the study, patients were randomized (1:1:1) to self-administered SC injections of placebo or peginterferon beta-1a 125 µg every 2 weeks or every 4 weeks. 4 Patients who received peginterferon beta-1a in year 1 are referred to as the continuous-treatment group. There was no crossover from placebo to peginterferon beta-1a prior to the end of the 1-year placebo-controlled study, even in the event of relapse or CDW. At the end of year 1, patients treated with placebo were re-randomized to receive peginterferon beta-1a 125 µg every 2 weeks or every 4 weeks (referred to as the delayed-treatment groups). All patients were maintained on their assigned dosage schedules through year 2. The ATTAIN study was a dose-frequency blinded extension study of ADVANCE (Supplementary Figure S1). Implementation of a protocol amendment changed ATTAIN to an open-label study, in which all ongoing patients on peginterferon beta-1a every-4-weeks dosing were switched to every-2-weeks dosing; the original dosing schedules remained blinded. The study was considered complete when the last patient completed 96 weeks in the ATTAIN study. At this time point, some patients had received up to nearly 6 years of treatment.
Inclusion and exclusion criteria for the ADVANCE study have been described previously.4,5 Key eligibility criteria were a diagnosis of relapsing-remitting MS as defined by the 2005 McDonald criteria, age 18–65 years, a score of 0.0–5.0 on the Expanded Disability Status Scale (EDSS), and at least two clinically documented relapses in the previous 3 years, of which at least one occurred within 12 months of screening visit. Key exclusion criteria were progressive forms of MS, prespecified laboratory abnormalities, and previous treatment with IFNs for MS for more than 4 weeks or discontinuation of treatment less than 6 months before baseline visit.
Study procedures and endpoints
Study procedures have been reported in detail previously. 4 Subjects continued to be treated with peginterferon beta-1a at the same dosing regimen received in their second year of ADVANCE, either every 2 weeks or every 4 weeks. To ensure blinding between dose-frequency groups, each patient received one injection of either placebo or peginterferon beta-1a every 2 weeks.
AEs were summarized with the use of descriptive statistics for all patients who received at least one dose of active study treatment, excluding data obtained after patients switched to alternative MS medications. Relapse was defined as new or recurrent neurological symptoms not associated with fever or infection, lasting for at least 24 hours, and separated from the onset of other confirmed relapses by at least 30 days. Disability progression was defined as an increase in EDSS score of ⩾1.0 point for patients with a baseline score of ⩾1.0 or an increase of ⩾1.5 points for patients with a baseline score of 0.0, confirmed after 24 weeks. Immunogenicity was measured by an analytically validated cell-based assay to characterize neutralizing antibodies (NAbs) to IFN beta-1a. 10
Primary endpoints for the ATTAIN study were the incidence of AEs, serious AEs (SAEs), discontinuations of study treatment due to an AE, and laboratory abnormalities. Secondary endpoints included MS relapse outcomes, MRI outcomes, disability outcomes, and immunogenicity.
Statistical analysis
The safety population was the ATTAIN intent-to-treat (ITT) population and was defined as all patients who received at least one dose of treatment while enrolled in ATTAIN. Adjusted annualized relapse rate [ARR; adjustments were for baseline EDSS score (<4.0 versus ⩾4.0), baseline relapse rate, and age (<40 versus ⩾40 years)], mean number of gadolinium-enhancing (Gd+) lesions, new/newly enlarging T2 lesions, and new T1 hypointense lesions were evaluated in each study year by peginterferon beta-1a dosing frequency in the ATTAIN ITT population for patients who received peginterferon beta-1a treatment continuously (up to nearly 6 years). The every-2-weeks continuously dosed analysis group included only those patients who received peginterferon beta-1a every 2 weeks from the start of ADVANCE; it did not include patients who switched to every-2-weeks dosing at the time of the protocol amendment. Due to the limited number of patients completing 6 years, clinical outcomes reported are over 5 years. MRI data were collected only out to 4 years. Risk of relapse and CDW are shown across all treatment groups, including delayed-treatment groups. The adjusted relapse rate was obtained by negative binomial regression.
Standard protocol approvals, registrations, and patient consents
The protocol was approved by the institutional review board at each site, and the study was conducted according to International Conference on Harmonisation Guidelines for Good Clinical Practice and the Declaration of Helsinki. Every patient provided written informed consent before entering the study. This trial is registered with ClinicalTrials.gov (identifier: NCT01332019). Patients were eligible to enter the ATTAIN extension study if they completed the study treatment and visit schedule through week 96 of ADVANCE.
Results
Patients
Patient disposition across the ADVANCE and ATTAIN studies is presented in Supplementary Figure S2. Of 1512 patients randomized and dosed in ADVANCE, a total of 740 received peginterferon beta-1a every 2 weeks (512 received continuous treatment, and 228 received treatment beginning in year 2) and 728 received peginterferon beta-1a every 4 weeks (500 received continuous treatment, and 228 received treatment beginning in year 2). Of the 1332 patients who completed ADVANCE, 1076 (81%) were dosed in ATTAIN, and 842 patients (78% of those dosed in ATTAIN; 56% of the original ADVANCE study population) completed the ATTAIN study. During ATTAIN, 234 patients withdrew from the study, 122 in the peginterferon beta-1a every-2-weeks group (22 due to an AE) and 112 in the peginterferon beta-1a every-4-weeks group (13 due to an AE). Reasons for study withdrawal included AEs, patient consent withdrawn, patient lost to follow-up, investigator decision, and death. None of the five deaths occurring during the study (car accident, myocardial infarction, bacterial meningitis, MS relapse with recurrent pneumonia, and ventricular fibrillation) were considered related to study treatment. Reasons given for withdrawal of patient consent included logistics and personal reasons; no reason was provided in some cases. The mean duration of exposure to peginterferon beta-1a for the ATTAIN ITT population (n = 1076) across all treatment groups throughout the study was 106 weeks. Patient demographics and baseline disease characteristics were generally well balanced across the treatment groups (Table 1).
Demographics and baseline characteristics of patients receiving continuous peginterferon beta-1a during ADVANCE and ATTAIN.

ATTAIN ITT population; only the continuous every-2-weeks and continuous every-4-weeks treatment groups are shown. Data are presented as mean (standard deviation) unless otherwise stated.
Relapses occurring before peginterferon beta-1a treatment in ADVANCE.
Relapses occurring during peginterferon beta-1a treatment in ADVANCE.
n = 352.
n = 374.
n = 353.
n = 375.
n = 375.
n = 353.
EDSS, Expanded Disability Status Scale; Gd+, gadolinium-enhancing; ITT, intent to treat; MS, multiple sclerosis; NA, not available; SC, subcutaneous.
Safety
The overall incidence of AEs, SAEs, and discontinuations due to AEs by treatment group during the first 2 years (ADVANCE; includes patients who received peginterferon beta-1a at any time) and years 3–6 (ATTAIN) is shown in Table 2. During the ATTAIN study, the most commonly reported treatment-related AEs (combined every-2-weeks and every-4-weeks dosing) included influenza-like illness (43%), injection site erythema (41%), headache (29%), pyrexia (26%), myalgia (12%), and chills (12%) and were similar across treatment groups. A total of 26 patients (5%) in the every-2-weeks treatment group and 18 patients (3%) in the every-4-weeks treatment group discontinued treatment due to an AE. Most AEs were categorized as mild or moderate. The incidence of any single AE that led to study discontinuation was <1%. The most common AEs leading to study discontinuation were MS relapse (n = 5), hepatic toxicity (n = 3), influenza-like illness (n = 3), and alanine aminotransferase increase (n = 3).
Incidence of AEs and SAEs in patients who received peginterferon beta-1a at any time during ADVANCE and ATTAIN.

Individual AEs affecting ⩾10% of patients in either peginterferon beta-1a treatment group in ATTAIN. Numbers in parentheses are percentages; for patients who switched to alternative MS medications, data after switch and 14 days after last dose of study treatment are excluded. A patient was counted only once within each preferred term.
Events over 2 years in all patients treated with peginterferon beta-1a at any time (continuous and delayed treatment) in ADVANCE, as previously reported. 5
Events in years 3–6 in patients in peginterferon beta-1a treatment groups; duration of exposure ranges from 2 to 4 years in ATTAIN.
Includes MS relapse, which was reported as an SAE for 57 patients (10%) in the every-2-weeks group and 82 patients (16%) in the every-4-weeks group.
AE, adverse event; MS, multiple sclerosis; SAE, serious adverse event.
The incidence of SAEs, while low in both groups, was slightly higher in the peginterferon beta-1a every-4-weeks group [113 patients (21%)] than in the peginterferon beta-1a every-2-weeks group [90 patients (16%); Table 2]. This difference primarily resulted from the higher incidence of MS relapse in the every-4-weeks group [82 patients (16%)] than the every-2-weeks group [57 patients (10%); Table 2]. The incidence of treatment-related SAEs was low (1% overall; <1% for any individual SAE in both treatment groups).
The incidence of flu-like symptoms (FLS) and injection site reactions (ISRs), the most common AEs, is shown in Table 3. The most common FLS were influenza-like illness and pyrexia, and the most common ISR was erythema. Of patients who experienced ISRs, most reported them as mild or moderate. The proportion of patients experiencing FLS or ISRs during each year of the study in each continuous-treatment group decreased over time, as shown in Figure 1.
Incidence of FLS and ISRs in patients who received peginterferon beta-1a at any time during ADVANCE or ATTAIN.
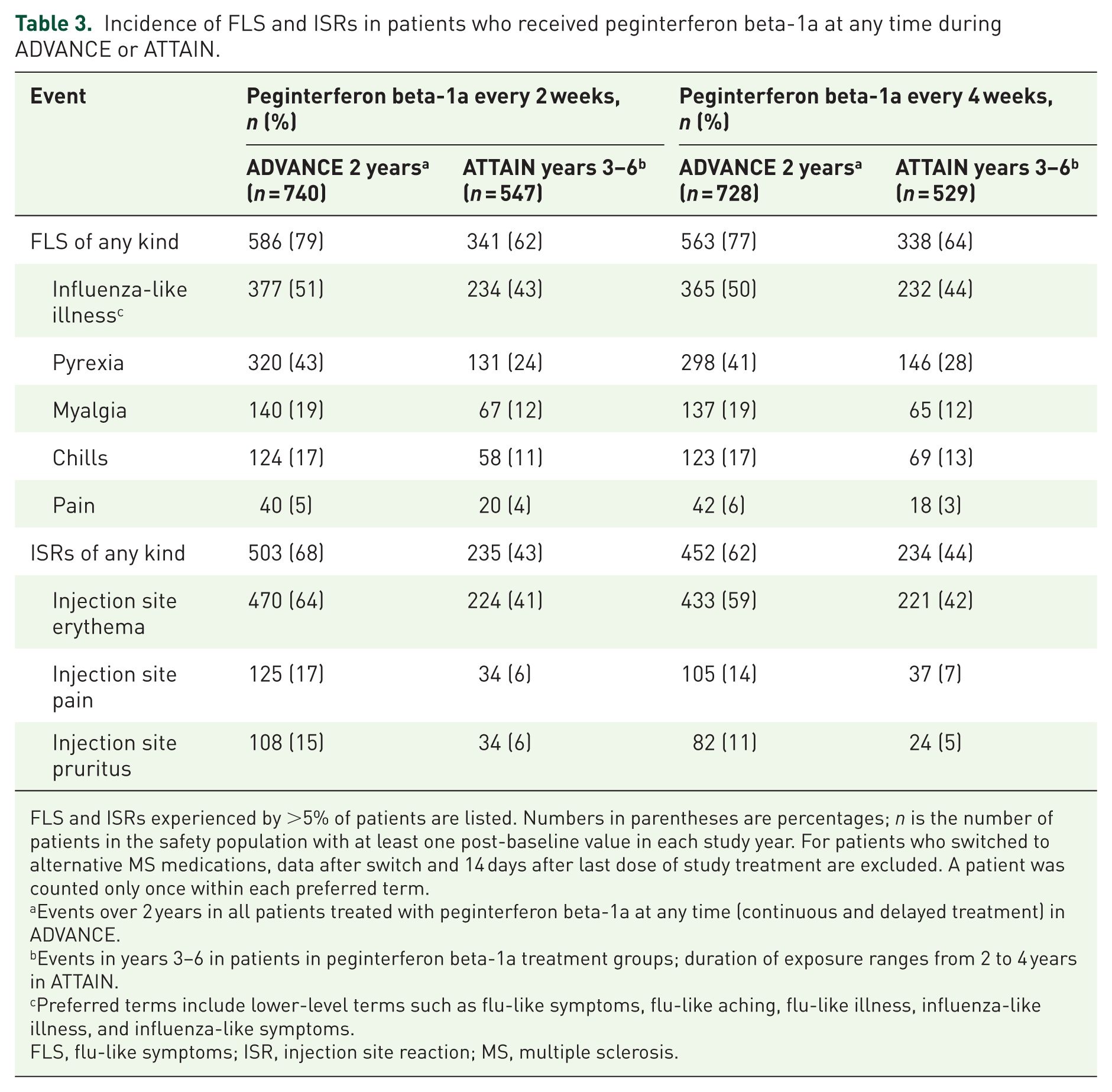
FLS and ISRs experienced by >5% of patients are listed. Numbers in parentheses are percentages; n is the number of patients in the safety population with at least one post-baseline value in each study year. For patients who switched to alternative MS medications, data after switch and 14 days after last dose of study treatment are excluded. A patient was counted only once within each preferred term.
Events over 2 years in all patients treated with peginterferon beta-1a at any time (continuous and delayed treatment) in ADVANCE.
Events in years 3–6 in patients in peginterferon beta-1a treatment groups; duration of exposure ranges from 2 to 4 years in ATTAIN.
Preferred terms include lower-level terms such as flu-like symptoms, flu-like aching, flu-like illness, influenza-like illness, and influenza-like symptoms.
FLS, flu-like symptoms; ISR, injection site reaction; MS, multiple sclerosis.

Patients reporting (
As in the ADVANCE study, the incidence of binding anti-IFN antibodies (BAbs; every-2-weeks group, 8%; every-4-weeks group, 6%), NAbs (every-2-weeks group, 1%; every-4-weeks group, <1%), and anti-PEG antibodies (every-2-weeks group, 2%; every-4-weeks group, 3%) at any time during ATTAIN continued to be low. Laboratory abnormalities were typically consistent with ADVANCE, except for a reduced white blood cell (WBC) count in the every-2-weeks group. WBC counts were <3.0 × 109/l in 86 patients (16%) in the every-2-weeks group and 28 patients (5%) in the every-4-weeks group. Red blood cell counts were <3.3 × 1012/l in seven patients (1%) in the every-2-weeks group and one patient (<1%) in the every-4-weeks group. Total absolute neutrophil counts were ⩽1 × 109/l in 15 patients (3%) in the every-2-weeks group and 8 patients (2%) in the every-4-weeks group. Lymphocyte counts were <0.8 × 109/l in 62 patients (11%) in the every-2-weeks group and 41 patients (8%) in the every-4-weeks group. Hemoglobin levels were ⩽100 g/l in 35 patients (6%) in the every-2-weeks group and 33 patients (6%) in the every-4-weeks group. Platelet counts were ⩽100 × 109/l in 11 patients (2%) in the every-2-weeks group and 3 patients (<1%) in the every-4-weeks group.
Efficacy
Over years 0–6, the adjusted ARR was significantly improved in the continuous peginterferon beta-1a every-2-weeks group compared with the continuous-every-4-weeks group (0.188 versus 0.263; p = 0.0052). Year-over-year adjusted ARRs were generally reduced in the every-2-weeks group (Figure 2).

ARR, based on objective relapses, for study patients by year in the placebo (year 1), continuous peginterferon beta-1a every-2-weeks, and continuous peginterferon beta-1a every-4-weeks treatment groups.
Compared with continuous dosing of peginterferon beta-1a every 4 weeks, continuous dosing of peginterferon beta-1a every 2 weeks significantly reduced the risk of relapse over 5 years, from 49% to 36% (p = 0.0018; Supplementary Figure S3). It is notable that patients in the peginterferon beta-1a every-2-weeks group who received delayed treatment (those who received placebo during year 1 of ADVANCE) did not exhibit a similar risk reduction. The risk of relapse in the every-2-weeks and every-4-weeks delayed-treatment groups (47% and 50%, respectively) was comparable to the risk of relapse in the every-4-weeks continuous-treatment group.
Over 5 years, there was a trend toward a reduced risk of 24-week CDW in patients receiving continuous peginterferon beta-1a every 2 weeks compared with continuous peginterferon beta-1a every 4 weeks (15% versus 20%, p = 0.0526; Figure 3). The delayed every-2-weeks treatment group displayed a similar trend toward a reduced risk of 24-week CDW (assessed at week 216) compared with the delayed every-4-weeks group (13% versus 22%; p = 0.1646).

24-week confirmed disability worsening over ADVANCE and ATTAIN.
The mean number of MRI lesions continued to be significantly lower in the peginterferon every-2-weeks treatment group compared with the every-4-weeks group. In year 4, patients treated with peginterferon beta-1a every 2 weeks displayed 53% fewer new T1 lesions (p < 0.0001), 61% fewer new/newly enlarging T2 lesions (p < 0.0001), and 75% fewer Gd+ lesions (p = 0.0067) compared with patients treated every 4 weeks (Figure 4).

MRI lesions by year in the placebo (ADVANCE ITT at year 1), continuous peginterferon beta-1a every-2-weeks, and continuous peginterferon beta-1a every-4-weeks treatment groups. (
Discussion
The safety and tolerability of SC peginterferon beta-1a 125 µg every 2 weeks remains favorable for up to 5 years in patients with RMS. Furthermore, the overall safety profile of ATTAIN is similar to that of ADVANCE, with no marked changes in event rates for any AEs. The proportion of patients reporting any AE during years 3–6 of ATTAIN (87% in the every-2-weeks group; 89% in the every-4-weeks group) was similar to that reported by the year 1 placebo patients during ADVANCE (83%). 4 As in the ADVANCE study, the most frequently reported AEs were ISRs and FLS, both of which were numerically decreased in later years of the study. Although a greater proportion of patients who experienced AEs choosing to withdraw from the study cannot be ruled out, only 4.2% of patients in the peginterferon beta-1a every-2-weeks group and 2.6% in the every-4-weeks group withdrew due to AEs, which would not account for the observed reductions in AE incidence rates.
Tolerability improved over time in ATTAIN, with observed decreases in both FLS and ISRs. Both are common AEs associated with SC IFN beta therapies. A Delphi-consensus generating study of 374 patients treated by clinicians in ADVANCE found that both FLS and ISRs generally decreased in incidence and duration after 3 months of treatment and had a minimal impact on daily activities in their typical patient. Patient education was important to set expectations regarding the characteristics and management of FLS and ISRs. 11 In COMPARE, injection site erythema and injection site pain AEs were similar or less severe for patients treated with peginterferon beta-1a than for those treated with SC IFN beta-1a therapy. However, the incidence of injection site erythema was 2.5 times greater in patients receiving SC IFN beta-1a 44 µg three times per week (TIW). 12
Peginterferon beta-1a 125 µg every 2 weeks led to significantly improved clinical outcomes (ARR in most individual years, as well as proportion of patients relapsed) and MRI outcomes compared with every-4-weeks treatment, demonstrating sustained and more robust efficacy with long-term treatment. It is notable that at week 216, the proportions of patients who relapsed in both delayed-treatment groups (those who received placebo in year 1) were similar to the proportion of patients who relapsed in the every-4-weeks continuous-treatment group (those who received peginterferon every 4 weeks continuously from ADVANCE baseline). This observation supports early use of every-2-weeks peginterferon beta-1a, as the patients receiving the every-4-weeks dose or placebo for 1 year remained at a higher risk of relapse compared with patients receiving peginterferon beta-1a every 2 weeks.
The long-term clinical efficacy reported here is in the range of other IFN therapies for RMS. Sustained efficacy over time, combined with every-2-weeks dosing frequency, makes peginterferon beta-1a an effective and viable treatment option for many patients. Other IFN beta therapies require injections up to four times per week, and the less frequent dosing regimen of peginterferon beta-1a could lead to improved adherence. Furthermore, decreased frequency of dosing is not associated with lower drug exposure. In the phase I COMPARE pharmacokinetics study of healthy subjects, drug exposure over 2 weeks (area under the concentration-time curve from time 0 to 336 hours) was 60% higher with SC peginterferon beta-1a compared with SC IFN beta-1a 44 µg TIW (117.4 versus 73.1 ng/mL/h; p < 0.0001). Safety profiles of both treatments were consistent with prior observations, and the higher overall drug exposure during peginterferon beta-1a dosing was not associated with an increased rate of adverse effects.
The treatment effect of peginterferon beta-1a in patients with RMS is not expected to be attenuated by immunogenicity. In an analysis of immunogenicity in the ADVANCE study, 6%, <1%, and 7% of patients developed anti-IFN BAbs, NAbs, and anti-PEG antibodies, respectively, over 2 years, and there was no discernible clinically meaningful effect of antibody status on any evaluated efficacy or safety endpoint. 10
Open-label extension studies have limitations that should be considered in the interpretation of their results. Specifically, such studies are prone to patient self-selection bias. Patients who withdrew from the original study are not available for the follow-up study, and those who experienced favorable outcomes during the main study are more likely to agree to participate in an extension study than those who had less favorable experiences.
In conclusion, patients continuing peginterferon beta-1a treatment for up to 5 years showed a safety profile similar to that of the year 1–2 ADVANCE patients, indicating that no new safety signals are associated with continued therapy. In addition, ATTAIN patients saw reductions over time in the most common AEs, as well as in the risk of MS relapse and MRI indications of disease progression. This study confirms the safety, tolerability, and efficacy of SC peginterferon beta-1a in patients with RMS.
Supplemental Material
Newsome_ATTAIN_2Y_TAND_Resubmit_Supp_Material_051418b – Supplemental material for Long-term outcomes of peginterferon beta-1a in multiple sclerosis: results from the ADVANCE extension study, ATTAIN
Supplemental material, Newsome_ATTAIN_2Y_TAND_Resubmit_Supp_Material_051418b for Long-term outcomes of peginterferon beta-1a in multiple sclerosis: results from the ADVANCE extension study, ATTAIN by Scott D. Newsome, Thomas F. Scott, Douglas L. Arnold, Gereon Nelles, Serena Hung, Yue Cui, Shulian Shang, Maria L. Naylor and Marcelo Kremenchutzky in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The authors thank the patients who volunteered for this study and the many site staff members who helped to conduct the study. The study was sponsored by Biogen (Cambridge, MA, USA). Oksana Mokliatchouk (Biogen, Cambridge, MA, USA) provided assistance in interpretation of the statistical analyses. The authors were assisted in the preparation of the manuscript by Jenna Steere and Nicholas White of Ashfield Healthcare Communications (New York, NY, USA). Writing and editorial support, including editing and creation of figures and tables, were funded by the study sponsor. All authors were involved in reviewing the manuscript critically for important intellectual content, had full editorial control of the manuscript, and provided final approval of the submitted version.
Funding
The ADVANCE and ATTAIN studies were funded by Biogen (Cambridge, MA, USA).
Conflict of interest statement
SDN has received research support (paid directly to institution) from Biogen, Genentech, the National MS Society, and Novartis, and has participated in scientific advisory boards for Biogen and Genentech.
TFS has received research support from Biogen and Novartis and has received honoraria for participation in scientific advisory boards and speaking from Acorda Therapeutics, Biogen, Genentech, Genzyme, Novartis, and Teva Neurosciences.
DLA reports an equity interest in NeuroRx during the conduct of the study and personal fees from Acorda, Biogen, EMD Serono, Genentech, Genzyme, Hoffman-La Roche, Innate Immunotherapy, MedImmune, Mitsubishi, Novartis, Receptos, Sanofi-Aventis, and Teva outside the submitted work, as well as grants from Biogen and Novartis.
GN has participated in scientific advisory boards for Bayer HealthCare, Biogen, Genzyme, and Merck, and received personal compensation for speaking activities from Bayer HealthCare, Biogen, Genzyme, Merck, Novartis, and Teva.
SH, YC, and SS were full-time employees of Biogen and holders of Biogen stock and/or stock options at the time of the analysis.
MLN is a full-time employee of Biogen and holder of Biogen stock and/or stock options.
MK has received research grants, research support, and personal compensation for consulting or speaking activities from Biogen, the Canadian Institute of Health Research, Genzyme, the MS Society of Canada, Novartis, Sanofi, Teva, and Wellesley Therapeutics. MK’s institution has received research grants from Biogen and the Canadian Institute of Health Research and research support from the Chapman Chair in MS Clinical Research at London Health Sciences Centre.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
