Abstract
Background:
Dementia has a crucial impact on the quality of life of elderly patients and their caregivers. Proton-pump inhibitors (PPIs) are the most frequently prescribed treatment, but they have been shown to be associated with dementia. The data are inconsistent, however.
Objective:
To investigate the association between PPIs use and Alzheimer’s disease (AD) or all-cause dementia in six observational Korean databases using a Common Data Model (CDM) and to perform a distributed network analysis.
Methods:
Subjects aged over 18 years between 1 January 2004 and 31 December 2020. Among 7,293,565 subjects from 6 cohorts, 41,670 patients met the eligibility criteria. A total of 2206 patients who were included in both cohorts or with a history of dementia were excluded. After propensity matching, 5699 propensity-matched pairs between the PPIs and histamine-2 receptor antagonist (H2RA) users were included in this study. The primary outcome was the incidence of AD at least 365 days after drug exposure. The secondary outcome was the incidence of all-cause dementia at least 365 days after drug exposure.
Results:
In the 1:1 propensity score matching, the risk of AD or all-cause dementia was not significantly different between the PPIs and H2RA groups in all six databases. In the distributed network analysis, the long-term PPI users (⩾365 days) were unassociated with AD [hazard ratio (HR) = 0.92, 95% confidence interval (CI) = 0.68–1.23;
Conclusion:
In the distributed network analysis of six Korean hospital databases using Observational Medical Outcomes Partnership (OMOP)-CDM data, the long-term use of PPI was not associated with a statistically significantly increased risk of AD or all-cause dementia. Therefore, we suggest that physicians should not avoid these medications because of concern about dementia risk.
Introduction
Proton-pump inhibitors (PPIs) are the most frequently prescribed classes of drugs for the treatment of gastroesophageal reflux disease and peptic ulcers. 1 PPIs are preferred over histamine-2 receptor antagonists (H2RA) because PPIs have been shown to have superior efficacy at reducing gastric acid compared with H2RAs.1,2 In fact, PPI use rapidly increased from 0.2% in 1990 to 14.2% in 2018, whereas the usage of H2RAs has remained relatively low (1.2%–3.4%) in the United Kingdom. 3
Presumed adverse events of PPIs, however, have been reported in the clinical field including
Therefore, we investigated the association between PPIs use and Alzheimer’s disease (AD) or all-cause dementia in six observational Korean databases using a Common Data Model (CDM) and performed a distributed network analysis.
Methods
Data source
This study included patient-based retrospective cohort data from six medical centers including Ajou University Medical Center (AUMC,
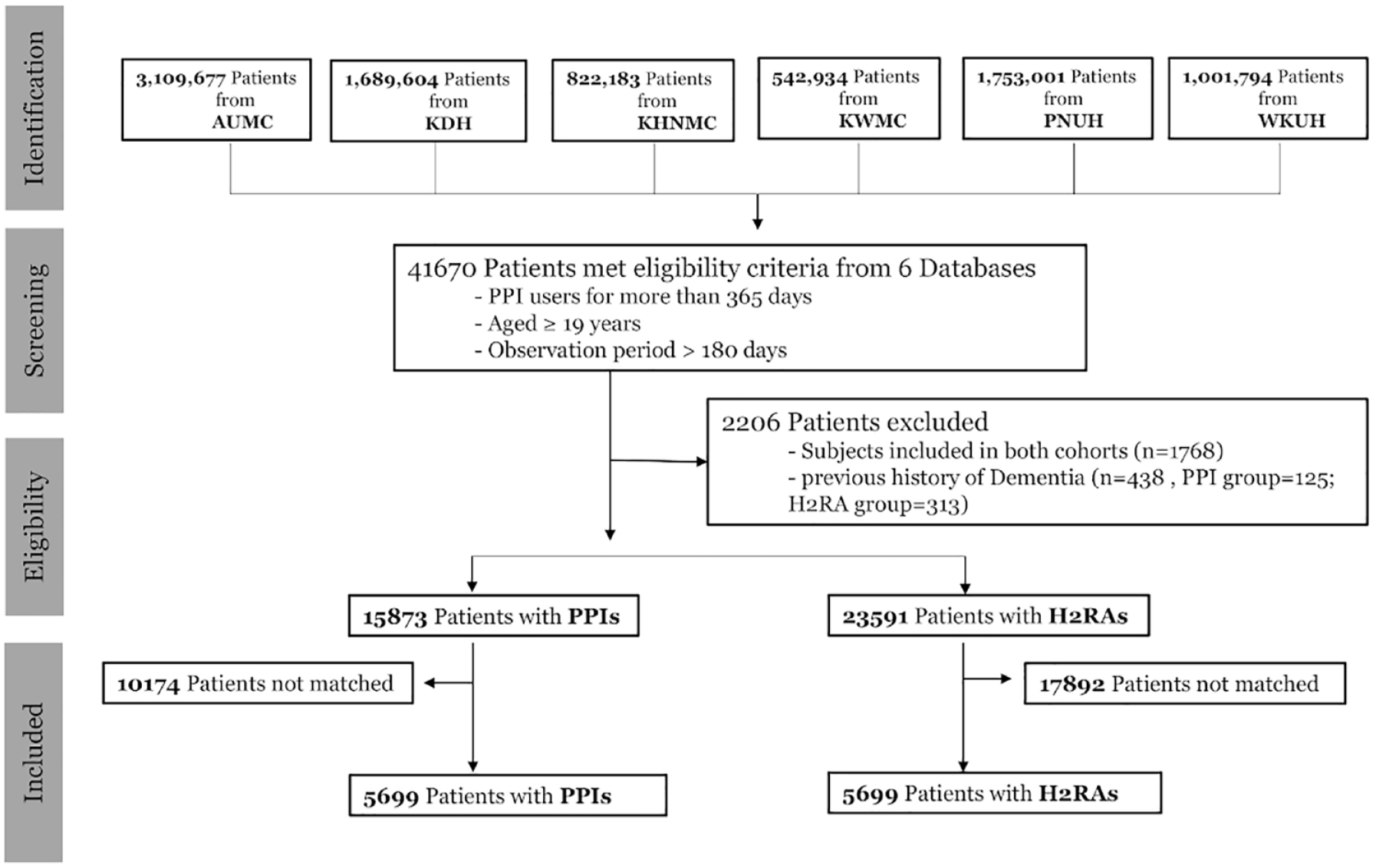
Study flowchart of included patient-based retrospective cohort data from six medical centers.
Observational Health Data Sciences and Informatics (OHDSI) is an international collaborative consortium aimed at facilitating the generation of high-quality evidence by generating and applying open-source data analysis solutions to a large network of health databases worldwide. 15 Most Korean hospitals use electronic health record (EHR) systems; however, numerous Korean codes for diagnosis, medications, and procedures are not compatible with international coding systems. Since 2016, data from Ajou University and the Korean nationwide cohort database were successfully transformed into the OMOP-CDM model and validated.16,17 EHR data from each hospital were converted to the CDM version, which is potentially applicable for collaborating OHDSI networks worldwide. This study was approved by the Institutional Review Board of the study institution (Institutional Review Board number 2018-05-013) and conformed to the tenets of the Declaration of Helsinki.
Study design and cohort definitions
We conducted a retrospective, observational, comparative cohort study of all outpatient-based subjects aged over 18 years between 1 January 2004 and 31 December 2020. The study flowcharts of the included patients were shown in Figure 1 and Supplementary Figure 1, and diagram of study construction from six databases was depicted in Figure 2.
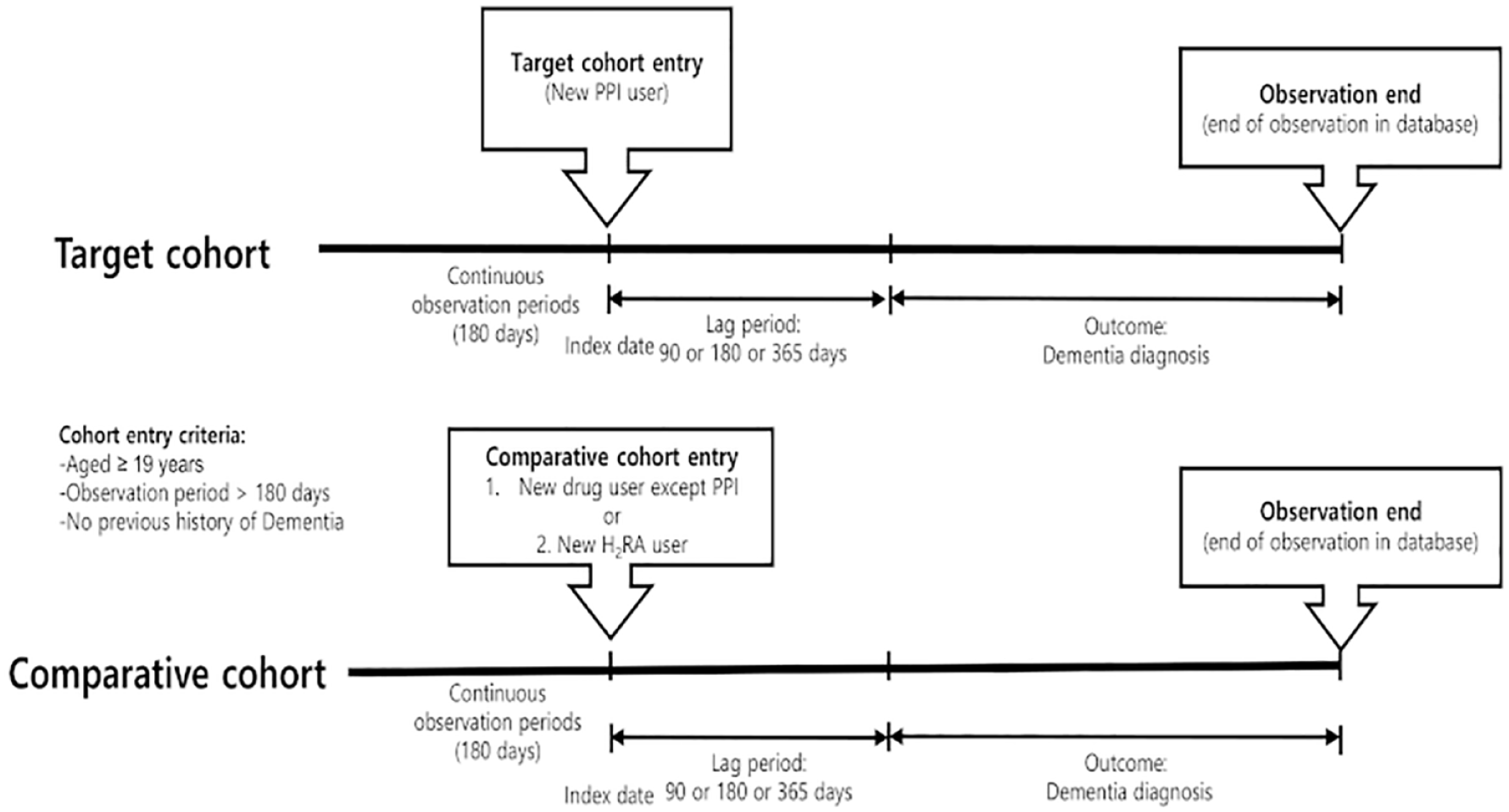
Diagram of cohort construction.
The index date of the target and comparative cohorts was defined as the first date of drug prescription. To avoid immortal time bias and duplication, both cohorts had continuous observation periods of 180 days before cohort entry as in our previous study. 18 Both cohorts were censored or end of database at the time of the identification of dementia. PPI exposure was defined as prescription for more than 90, 180, or 365 days. In this study, TAR start was defined as the period from 1 day after the index date. We used TAR start of 180 days or 365 days from the cohort start date. Because the TAR end was set to 99,999 days, it means that the subject is followed up until the end of the observation (Figure 2).
Subjects who met at least one of the following criteria were excluded from the target and comparative cohorts: (1) history of dementia before cohort entry, (2) an observation period of less than 180 days before cohort entry, (3) history of drug use, and (4) aged <19 years. In the PPIs group, H2RAs users of 180 days before PPI exposure and all H2RAs users after PPI exposure were excluded, and PPIs users of the same period were excluded from the H2RAs group.
The target cohort was defined as new PPIs users who were prescribed PPIs for more than 90 consecutive days. In the subgroup analysis, the PPI group was further divided into subgroups by the duration of PPI exposure (⩾180 or ⩾365 days). All PPIs on the market in Korea were included in these analyses (esomeprazole, pantoprazole, lansoprazole, rabeprazole, omeprazole, ilaprazole, and dexlansoprazole). Continuous drug exposures were restricted by allowing less than 30-day gaps between the drug prescriptions. The comparative cohort was defined as subjects prescribed H2RA for more than 90, 180, or 365 consecutive days. H2RAs included ranitidine, nizatidine, cimetidine, famotidine, and lafutidine.
Outcomes
The primary analysis was performed to compare the risk of AD between PPIs and H2RAs according to the duration of PPI exposure. The secondary analysis was performed to compare the risk of all-cause dementia including AD, Parkinson’s disease dementia, Lewy body dementia, frontotemporal dementia, and vascular dementia between PPIs and H2RAs. Furthermore, we conducted the same analyses using another comparative cohort of non-PPIs users.
The primary outcome was the incidence of AD at least 365 days after drug exposure. AD was defined by the diagnosis codes F000 to F002 in the 10th version of the
Covariates
We performed large-scale propensity score matching using the OMOP-CDM tool. The following covariates were used between the target and comparative cohorts: age, sex, all recorded comorbidities, prescribed drugs 365 days before the index date, and Charlson’s comorbidity index.
The distribution of preference scores between groups receiving PPIs and those receiving H2RAs before and after matching and covariate balance was summarized with mean values for all baseline covariates in the target and comparator cohorts, with the associated standardized mean difference computed for each covariate. Standardized mean differences were lower than 0.1 after propensity score matching (Supplementary Figure 2).
Statistical analysis
Most OHDSI analysis tools are embedded in the ATLAS platform and the OHDSI Methods Library R packages. The open-source software in OHDSI is publicly available at the GitHub repository (https://github.com/OHDSI/). ATLAS ver. 2.7.3 was used herein, and we analyzed the platform of FEEDER-NET, a health big-data platform based on OMOP-CDM supported by the Korean national project. We conducted the Cox regression analysis to examine the hazard ratio (HR) of two cohorts for AD and all-cause dementia. The Kaplan–Meier method was used to estimate the cumulative incidence rate (IR) of AD and all-cause dementia between the two groups. IRs were determined per 1000 person years by dividing the number of dementia events by the total number of person years at risk and multiplying the results by 1000. The cumulative incidence between the two groups was compared using the log-rank test. Two-sided
The assessment for statistical heterogeneity was calculated using the Chi-square and
Sensitivity analysis
To assess the robustness of the results, several sets of sensitivity analyses were performed using different definitions of TAR, propensity score matching, comparatives, and outcomes. First, in addition to 1:1 propensity score matching, 1:4 propensity score adjustments were performed. Second, two TAR periods (180 or 365 days) were applied. Third, we conducted this study using two control groups (H2RA users or non-PPIs users). Fourth, we added logistic regression analyses.
Results
Among 7,293,565 subjects from 6 cohorts, 41,670 patients met the eligibility criteria. Because 2206 patients were included in both cohorts or had a history of dementia were excluded, 15,873 subjects who were PPIs users and 23,592 subjects who were H2RAs users were left. After propensity matching, 5699 propensity-matched pairs between the PPIs and H2RAs users were included in this study (Figure 1). The baseline characteristics are shown in Table 1.
Distribution of baseline characteristics across six databases between the PPI ⩾365 days group and the comparative group (H2RA) in the overall population before and after propensity score matching.

H2RA, histamine-2 receptor antagonists; PPIs, proton-pump inhibitors; PS, propensity score; SMD, standardized mean difference; NaN, not a number.
The mean follow-up time was different from the six cohorts (AUMC = 1457 days, KDH = 1630 days, KHNMC = 1564 days, KWMC = 1758 days, PNUH = 1207 days, and WKUH = 1930 days). The number of subjects, follow-up time, number of outcome events, and event IR per 1000 patient years are presented in Table 2.
Incidence rates and risk of Alzheimer’s disease between long-term PPI users (⩾365 days) and comparatives (H2RA) in the six medical centers.

AUMC, Ajou University Medical Center; H2RA, histamine-2 receptor antagonists; KDH, Kangdong Sacred Heart Hospital, Hallym University College of Medicine; KHNMC, Gangdong Kyung Hee University Hospital; KWMC, Kangwon National University Hospital; PNUH, Pusan National University Hospital; PPIs, proton-pump inhibitors; WKUH, Wonkwang University Hospital.
Association of PPIs and risk of AD and dementia compared with H2RA
We conducted the Kaplan–Meier analyses for the risk of AD and all-cause dementia between PPIs and H2RAs after propensity score matching.
In the 1:1 propensity score matching, the risk of AD was not significantly different between the PPIs and H2RAs groups in all six databases [AUMC = 0.89 (95% confidence interval (CI) = 0.46–1.73), KDH = 0.80 (95% CI = 0.31–2.06), KHNMC = 1.00 (95% CI = 0.53–1.90), KWMC = 1.00 (95% CI = 0.54–1.84), PNUH = 0.67 (95% CI = 0.27–1.66), and WKUH = 1.00 (95% CI = 0.46–2.18)] (Figure 3).
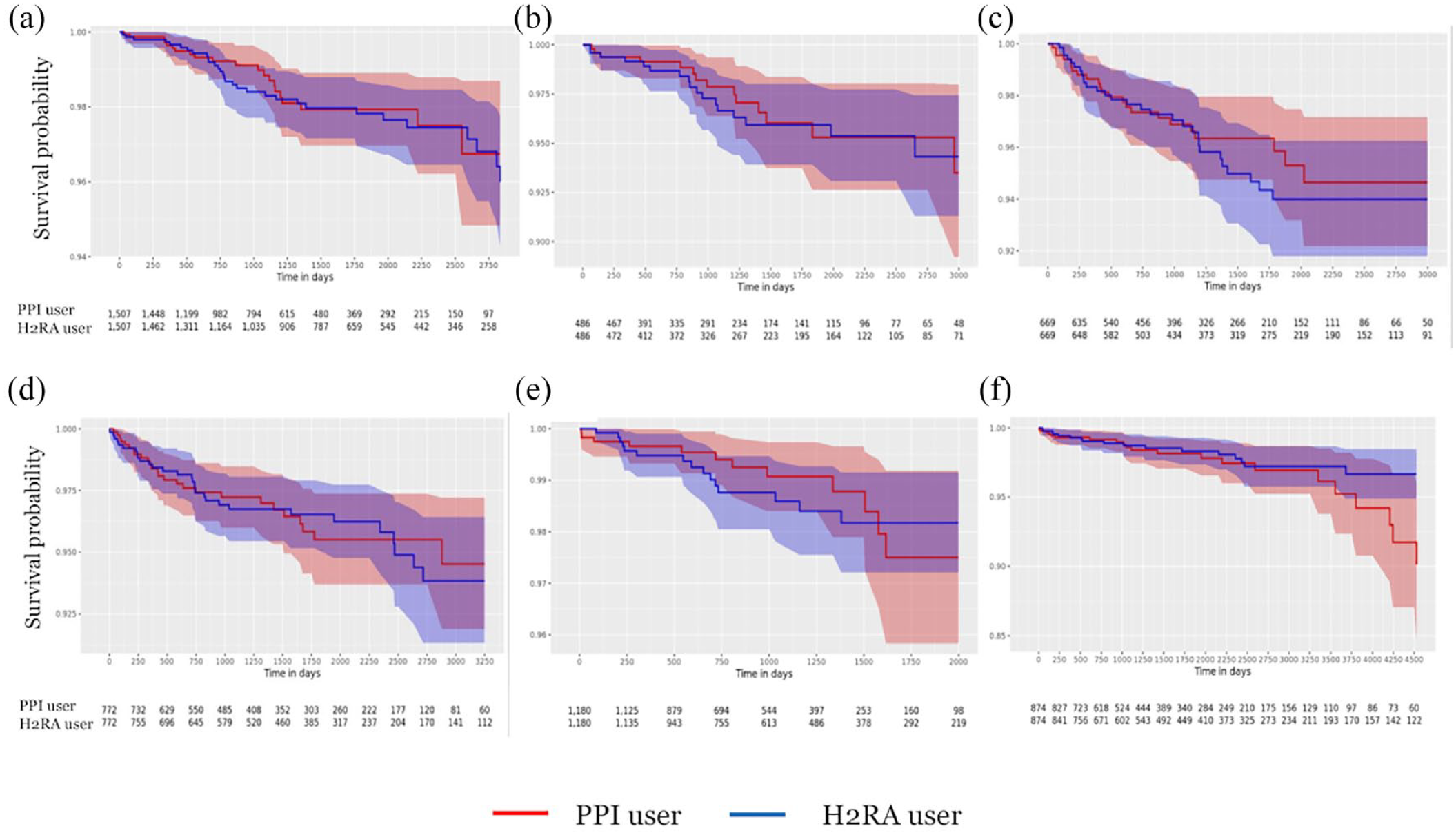
Kaplan–Meier plots for the risks of Alzheimer’s disease on-treatment comparisons of proton-pump inhibitors
In the 1:1 propensity score matching, the risk of all-cause dementia was not significantly different between the PPIs and H2RAs groups in all six databases [AUMC = 1.20 [95% CI = 0.60–2.40], KDH = 1.00 (95% CI = 0.47–2.12); KHNMC = 0.6 (95% CI = 0.31–1.14), KWMC = 1.18 (95% CI = 0.79–1.75), PNUH = 1.00 (95% CI = 0.43–2.34), and WKUH = 1.13 (95% CI = 0.69–1.84)] (Figure 4).
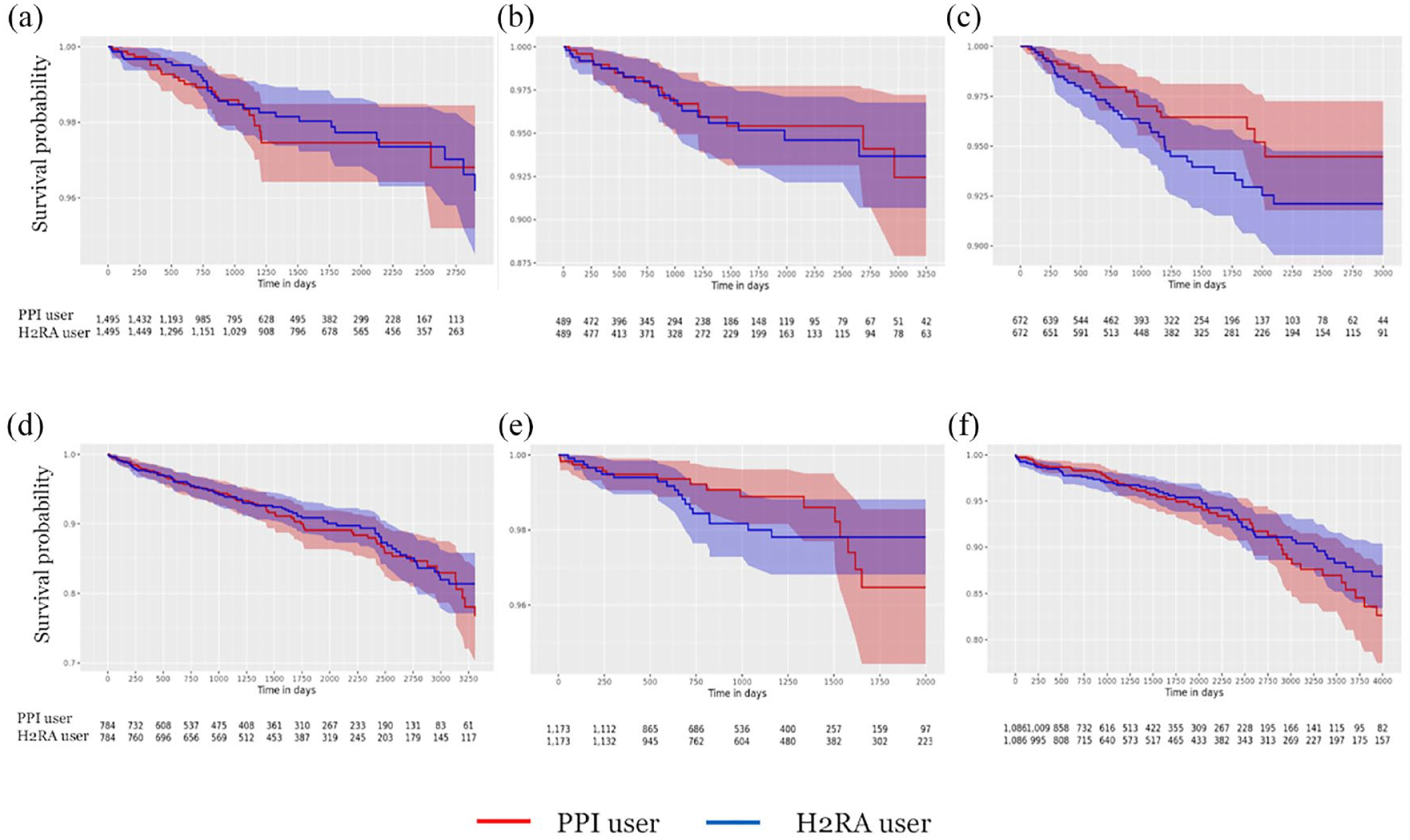
Kaplan–Meier plots for the risks of all-cause dementia on-treatment comparisons of proton-pump inhibitors
In the distributed network analysis with 1:1 propensity score matching, the long-term PPIs users (⩾365 days) were unassociated with AD [HR = 0.92, 95% CI = 0.68–1.23;

Meta-analysis of risk of PPIs on Alzheimer’s disease and all-cause dementia compared with histamine-2 receptor antagonists. In the distributed network analysis with 1:1 propensity score matching, the long-term PPI users (⩾365 days) were unassociated with AD [HR = 0.92, 95% CI = 0.68–1.23;
To assess the robustness of the results, in addition to 1:1 propensity score matching, 1:4 propensity score adjustment and 1:1 propensity score matching with an additional TAR period (365 days) were applied. No differences, however, were observed between PPIs and H2RAs in all analyses (Table 3).
Risk of Alzheimer’s disease or all-cause dementia in the overall population between PPI users and comparatives (H2RA or non-PPIs).

H2RA, histamine-2 receptor antagonists; HR, hazard ratio; PPIs, proton-pump inhibitors; PS, propensity score.
Association of PPIs and risk of AD and all-cause dementia compared with non-PPIs
In addition, to clarify the effect of long-term PPI use on cognitive decline, we compared PPI users and non-PPI users.
In the distributed network analysis with 1:1 propensity score matching, the long-term PPI users (⩾365 days) were unassociated with AD [HR = 1.08, 95% CI = 0.59–1.99;

Meta-analysis of Risk of PPIs on Alzheimer’s disease and all-cause dementia compared with non-PPIs. In the distributed network analysis with 1:1 propensity score matching, the long-term PPI users (⩾365 days) were unassociated with AD [HR = 1.08, 95% CI = 0.59–1.99;
Sensitivity analysis
To support the robustness of the results, we also conducted the logistic regression analyses. In the distributed network analysis with 1:1 propensity score matching, the long-term PPIs users (⩾365 days) were unassociated with AD [odds ratio (OR) = 0.87, 95% CI = 0.72–1.06;
In addition, we performed sensitivity analysis to identify the influence of PPI duration on subjects with PPI use exceeding 90 and 180 days. Similar to the long-term PPI users (⩾365 days), the risks of AD or all-cause dementia in these subgroups (PPI user ⩾90 or ⩾180 days) were unassociated with PPI use (Table 3).
Two propensity score adjustments (1:1
Discussion
In the distributed network analysis of six Korean hospital databases using OMOP-CDM data, the long-term use of PPIs, compared with H2RA or non-PPIs, was not associated with an increased risk of AD or all-cause dementia. There was no significant difference in the risk for AD or all-cause dementia according to the duration of PPI use. These results are consistent with other analyses using different propensity score matching and time windows. The findings are consistent with those of a recent meta-analysis of 10 studies involving 642,305 patients. 13
To confirm a causal relationship, but not association, of the long-term complication of drugs, we should conduct well-designed randomized controlled trials (RCTs). This type of RCT, however, is very difficult to perform because of the long observation periods, huge cost, and ethical problems. Instead, causal inference from observational databases can be evaluated as a try to emulate a particular target trial.19,20 Thus, we can evaluate the causality of drug complications indirectly by satisfying the Hill criteria including strength of association, consistency, specificity, temporality, biological gradient, biological plausibility, coherence, experiment, and analogy.21,22
These study results should be reliable as the design of our research was based on a review of previous studies. First, regarding the strength of the association, the dementia risk of PPIs in previous reports had an HR of <2. This means that more clinical trials are needed to examine this connection because an HR <3 has a likelihood of potential bias, although it can also suggest biological plausibility. Second, this study showed consistent results in the extensive sensitivity and subgroup analyses. We conducted several sensitivity analyses using different drug exposure periods (⩾90, ⩾180, and ⩾365 days), TAR windows (⩾180 and ⩾365 days), and propensity score matching (1:1 and 1:4). Third, regarding the specificity, because patients with dementia may have multiple comorbidities, there may be many drug–drug interactions. This study, however, was conducted using OMOP-CDM, which allowed large-scale propensity score matching including multiple drugs. Fourth, regarding the temporality, it is unknown how long dementia takes to develop. Dementia may be diagnosed at late stages or be undiagnosed. Therefore, to overcome this limitation, we used continuous observation periods of 180 days before cohort entry. After drug exposure, we used two different TAR windows (⩾180 and ⩾365 days) to clarify the drug effect on dementia occurrence. Fifth, regarding the biological gradient, although we conducted three different drug exposures (⩾90, ⩾ 180, and ⩾365 days), we did not find a dose–response relationship in this study. Sixth, regarding analogy, a systematic review reported that cognitive function was improved after bariatric surgery, which is a situation similar to vitamin B12 deficiency. 23
The association between PPI use and dementia, however, is still vague. Regarding plausibility and experiments, some plausible mechanisms such as the increased production of β-amyloid (Aβ), 24 vitamin B12 deficiency, 10 and blocking acid secretion by binding to H+/K+-ATPase 10 might be related. We cannot explain the exact mechanisms for the discrepancy between our findings and those of the prior studies. Given the widespread use of PPIs, however, these findings will be a significant step in better understanding the safety of long-term use of PPIs.
This study had some strengths. First, to define outcomes, other health claims data should depend on the medical code of diagnosis, and this always has a limitation of uncertainty. In Korea, however, physicians are required to demonstrate a clinical diagnosis of dementia from the medical history, as well as medical scores using the Mini-Mental State Examination, Clinical Dementia Ratings, or Global Deterioration Scale in order to prescribe the antidementia medications. 24 To improve the diagnostic accuracy of AD, we classified patients with AD when they had been prescribed antidementia medications with documented dementia codes. Second, this study was designed to focus on robustness and we performed various sensitivity analyses, which were consistent with our main analysis. Third, to clarify the impact of PPIs on AD or all-cause dementia, we compared PPI users and non-PPIs users, as well as PPI uses and H2RA users.
Despite these strengths, there were some limitations in this study. First, we could not determine the dementia status of patients or those with undiagnosed dementia. Physicians in South Korea, however, are required to provide strict diagnostic criteria before prescribing antidementia medications. Therefore, among all the diagnoses in the health claims data, the diagnosis of dementia is established relatively accurately. Second, we could not determine whether the prescribed dose was the actual dose taken. This, however, is a fundamental
Conclusion
In conclusion, long-term PPI use was not significantly associated with an increased risk of AD or all-cause dementia. These results are consistent with other analyses using different propensity score matching and time windows. Given some of the conflicting results, further researches will be needed. However, we suggest that physicians should not avoid these medications because of concern about dementia risk.
Supplemental Material
sj-docx-1-tan-10.1177_17562864221135700 – Supplemental material for Long-term use of proton-pump inhibitor on Alzheimer’s disease: a real-world distributed network analysis of six observational Korean databases using a Common Data Model
Supplemental material, sj-docx-1-tan-10.1177_17562864221135700 for Long-term use of proton-pump inhibitor on Alzheimer’s disease: a real-world distributed network analysis of six observational Korean databases using a Common Data Model by Yerim Kim, Seung In Seo, Kyung Joo Lee, Jinseob Kim, Jong Jin Yoo, Won-Woo Seo, Hyung Seok Lee and Woon Geon Shin in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-tif-2-tan-10.1177_17562864221135700 – Supplemental material for Long-term use of proton-pump inhibitor on Alzheimer’s disease: a real-world distributed network analysis of six observational Korean databases using a Common Data Model
Supplemental material, sj-tif-2-tan-10.1177_17562864221135700 for Long-term use of proton-pump inhibitor on Alzheimer’s disease: a real-world distributed network analysis of six observational Korean databases using a Common Data Model by Yerim Kim, Seung In Seo, Kyung Joo Lee, Jinseob Kim, Jong Jin Yoo, Won-Woo Seo, Hyung Seok Lee and Woon Geon Shin in Therapeutic Advances in Neurological Disorders
Supplemental Material
sj-tif-3-tan-10.1177_17562864221135700 – Supplemental material for Long-term use of proton-pump inhibitor on Alzheimer’s disease: a real-world distributed network analysis of six observational Korean databases using a Common Data Model
Supplemental material, sj-tif-3-tan-10.1177_17562864221135700 for Long-term use of proton-pump inhibitor on Alzheimer’s disease: a real-world distributed network analysis of six observational Korean databases using a Common Data Model by Yerim Kim, Seung In Seo, Kyung Joo Lee, Jinseob Kim, Jong Jin Yoo, Won-Woo Seo, Hyung Seok Lee and Woon Geon Shin in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The authors thank Dr Rae Woong Park (Department of Biomedical Informatics, Ajou University School of Medicine, Suwon, Korea), Dr Seng Chan You (Department of Preventive Medicine and Public Health, Yonsei University College of Medicine, Seoul, Korea), Dr Jae Myung Cha (Department of Internal Medicine, Kyung Hee University School of Medicine, Seoul, South Korea), Dr Sang Hoon Lee (Department of Internal Medicine, Kangwon National University Hospital, Kangwon National University School of Medicine, Chuncheon-si 24289, South Korea), Dr Gwang Ha Kim (Department of Internal Medicine, Pusan National University College of Medicine, and Biomedical Research Institute, Pusan National University Hospital, Busan, Korea), and Dr Suck Chei Choi (Department of Internal Medicine, Digestive Disease Research Institute, Wonkwang University College of Medicine, Iksan, Korea) for their feedback and comments.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
