Abstract
Background
Headaches resulting from proton pump inhibitor (PPI) use could cause discontinuation of PPI in as many as 40% of patients who experience such headaches. Previous studies focusing on acute headache risk from PPI use are rare and limited to clinical trials of a single PPI.
Objectives
To investigate the association between PPI use and headache with a nationwide population-based case-crossover study.
Methods
Records containing the first diagnosis of any headache, including migraine and tension-type headaches, were retrieved from Taiwan National Health Insurance Database (1998–2010). We compared the rates of PPI use for cases and controls during time windows of 7, 14, and 28 days. The adjusted self-matched odds ratios (ORs) and 95% confidence intervals (CIs) from a conditional logistic regression model were used to determine the association between PPI use and headache.
Results
Overall, 314,210 patients with an initial diagnosis of any headache during the study period were enrolled. The adjusted ORs for headache risk after PPI exposure were calculated for three time periods (within 7 days = 1.41,
Conclusion
PPI usage is associated with an increased risk for acute headache. Female patients and use of lansoprazole or esomeprazole present the greatest risks of headache.
Introduction
Headache is a common adverse event associated with medications and may cause poor patient adherence to treatment regimens (1). Drug-induced headache complicates the diagnosis and treatment of patients who concurrently have other headache disorders.
Proton pump inhibitors (PPIs) are widely used to treat acid-related diseases, such as peptic ulcers and gastroesophageal reflux disease. Omeprazole was the first PPI and was introduced in 1989. There are currently five available PPIs (omeprazole, esomeprazole, lansoprazole, pantoprazole, and rabeprazole). Recently, some safety concerns about PPIs have emerged, such as osteoporosis, increased risk of certain infections, hypergastrinemia, hypomagnesemia, and reduced efficacy of clopidogrel (2–5). However, short-term PPI treatment is still considered relatively safe and tolerable. In 2006, the total expenditure on brand-name PPIs was more than $10 billion in the USA (5).
Despite their widespread use, PPIs still have some adverse effects, such as nausea, diarrhea, and headache (6). A study showed as many as 40% of PPI-related headache sufferers discontinued use of PPIs (7). Headache incidence among PPIs users has been reported in randomized clinical trials (8–12). In these trials, headaches were reported in 2.9–6.9% of omeprazole users, 3.8–8.8% of lansoprazole users, 1.3% of pantoprazole users, 2.4–6.0% of rabeprazole users, and 5.8–7.8% of esomeprazole users. In prescription-event monitoring (PEM) studies in England, headache was the fourth or fifth most frequently reported adverse event during PPI use. The incidence density of headaches per 1000 patient-months of exposure was 4.6 for lansoprazole, 3.7 for pantoprazole, 2.5 for omeprazole, and 2.1 for esomeprazole (13–15).
Characteristics of PPI-related headache and predisposing factors for headache occurrence were reported in only one study (7), a nested case-control study that focused on lansoprazole users. The report showed that women, patients with previous use of analgesics, and patients reporting other adverse events were at risk of developing headaches. About two-thirds of PPI-related headaches were tension-type headaches, and approximately one-third were migraines. However, the response rate of this survey was low (44.6%).
In addition, little is known about the differences in headache risk among PPIs. Only one study indicated that lansoprazole and pantoprazole had greater headache risks than omeprazole (6). Therefore, we conducted a nationwide population-based case-crossover study to explore associations between the use of different PPIs and headaches.
Method
Data source
In 1995, Taiwan launched the National Health Insurance (NHI) program that covers approximately 99% of the Taiwanese population. In 1999, the Bureau of National Health Insurance made patient data available electronically through the National Health Insurance Research Database (NHIRD) project. Various extracted datasets are available to researchers, and hundreds of published studies have analyzed these data. The NHIRD contains comprehensive patient information, including demographic characteristic, diagnoses, medical expenditures, and prescription claims that include the date and duration of prescriptions, type of medications, dosages. Diseases are coded according to the International Classification of Diseases (ICD) -9-CM, 2001 edition. The diagnoses of several diseases, such as diabetes mellitus, acute coronary syndrome, and stroke, have been validated in addition to the accuracy of comorbidity by the Charlson score in the NHIRD (16–20).
In this study, we used the Longitudinal Health Insurance Database (LHID) from 1995 to 2010 obtained from the NHIRD. The LHID contains 1,000,000 beneficiaries randomly sampled from the original NHIRD. The NHRI has reported that there were no statistically significant differences in age, sex, or healthcare costs between the sample group and all enrollees. The NHIRD consists of personally unidentifiable secondary data released for research purposes; therefore, this study did not require ethics approval.
Study subjects
In the current study, we defined patients with an incident headache event as those with an outpatient or emergency room visit for a primary diagnosis of headache that was noted under ICD-9-CM codes 346.x (migraine), 784.0 (headache), or 307.81 (tension headache) (21–23). The index date for this dataset was defined as the date of the first visit when the patient was diagnosed with headache. Patients with an initial incident headache occurring between 1 January 1998 and 31 December 2010 were enrolled. Three groups of patients were excluded: those who had diagnoses of headache who were younger than 18 years of age; those who had diagnoses of acquired immune deficiency syndrome, human immunodeficiency virus, cancer, or cerebrovascular disease prior to the index date; and those who were hospitalized within 1 month before the diagnosis of headache because the dates of medication use during hospitalization could not be identified accurately.
Case-crossover design and exposure to proton pump inhibitors
The case-crossover design is a thoroughly validated research method to investigate the transient effects of acute events (24). The influence of unmeasured confounding factors, such as race, smoking status, and lifestyle, is reduced in the case-crossover design because each patient serves as his or her own control. The odds ratio (OR) for headache risk after exposure to a specific drug can be estimated by the ratio of patients who were exposed to that particular drug during the 7-day case period (1–7 days before the index date) to the patients who were exposed only during the 7-day control period (8–14 days before the index date). The ORs of other time windows, set at 14 days (1–14 days compared with 15–28 days before the index date for case and control periods, respectively) and 28 days (1–28 days and 29–56 days before the index date for case and control periods, respectively) were chosen for a sensitivity analysis and to determine the maximal risk window for headache after PPI exposure.
In this study, the exposure variable was PPI use. PPI exposure was defined as a prescription for PPIs for at least 1 day during the case or control time period. Concomitant medications were defined and accounted for in the same manner as PPIs. Two drugs were chosen as control drugs, nitrates as positive control and domperidone as negative control (1,25,26).
Data analyses
For case-crossover analyses, conditional logistic regression models were used to estimate the ORs and their 95% confidence intervals (95% CIs). Adjusted odds ratios (aORs) of headaches from PPI use were estimated after controlling for exposure to confounding medications, including angiotensin-converting-enzyme inhibitors, angiotensin II receptor blockers, beta-blockers, cilostazol, clopidogrel, dipyridamole, H2-receptor antagonists, sex hormones, nitrates, selective serotonin reuptake inhibitors, statins, ticlopidine, and warfarin.
Subgroup analyses were performed according to age and gender of the patients. Tests of interactions were performed for these subgroups by the likelihood ratio test. The
Results
Study population
During the 12-year study period, 422,978 patients with incident headache were identified. We excluded patients who were younger than 18 years of age (67,626 patients), had antecedent acquired immune deficiency syndrome or human immunodeficiency virus (194 patients), cancer (14,515 patients), cerebrovascular disease (19,680 patients), or were admitted to a hospital less than 1 month before the diagnosis of headache (6753 patients). Consequently, we enrolled 314,210 patients with a first diagnosis of any headache in the study period.
Demographic data.

Headache risks
Risk of headache associated with current use of proton pump inhibitor.
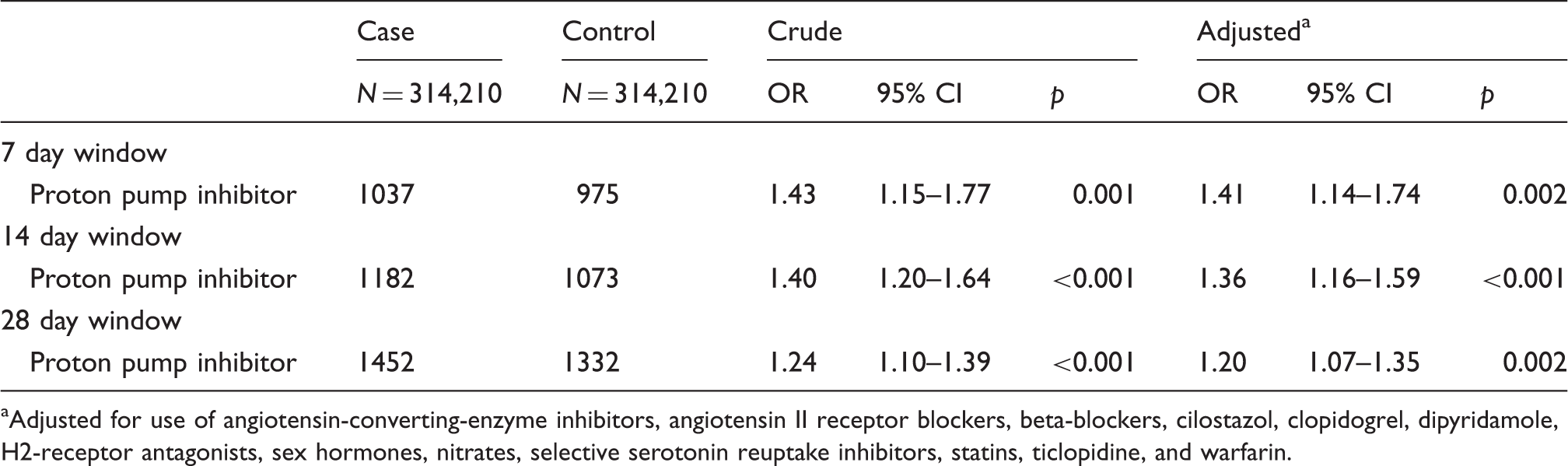
Adjusted for use of angiotensin-converting-enzyme inhibitors, angiotensin II receptor blockers, beta-blockers, cilostazol, clopidogrel, dipyridamole, H2-receptor antagonists, sex hormones, nitrates, selective serotonin reuptake inhibitors, statins, ticlopidine, and warfarin.
Risk of headache associated with different proton pump inhibitor for 7-day window.
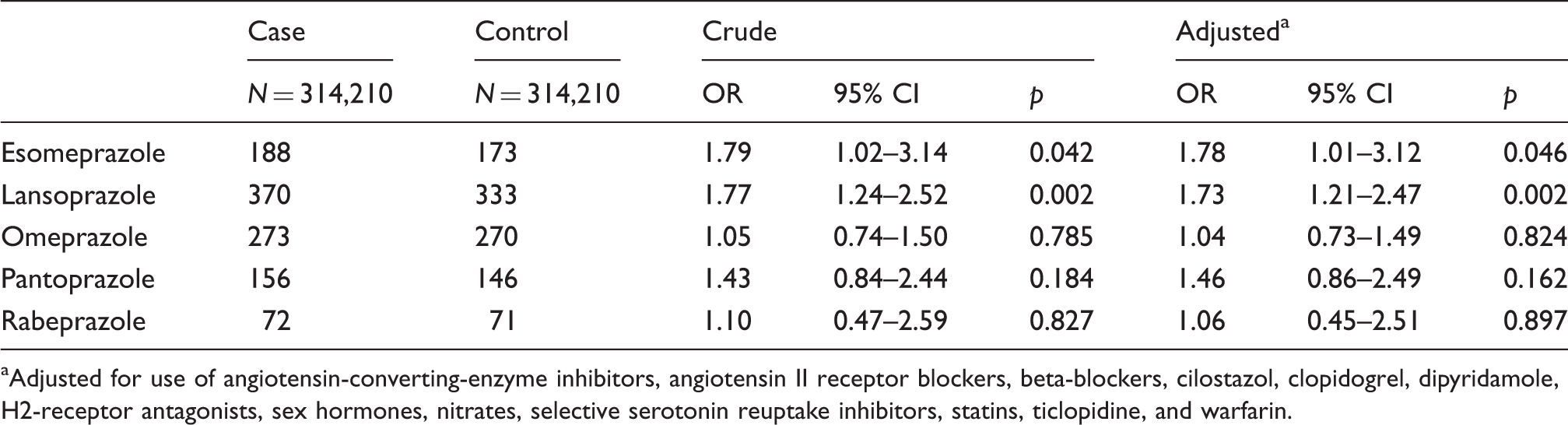
Adjusted for use of angiotensin-converting-enzyme inhibitors, angiotensin II receptor blockers, beta-blockers, cilostazol, clopidogrel, dipyridamole, H2-receptor antagonists, sex hormones, nitrates, selective serotonin reuptake inhibitors, statins, ticlopidine, and warfarin.
Risk of different headache associated with current use of proton pump inhibitor for 7-day window.

Adjusted for use of angiotensin-converting-enzyme inhibitors, angiotensin II receptor blockers, beta-blockers, cilostazol, clopidogrel, dipyridamole, H2-receptor antagonists, sex hormones, nitrates, selective serotonin reuptake inhibitors, statins, ticlopidine, and warfarin.
Subgroup analyses, stratifying by age and gender
Subgroup analyses for risk of headache associated with current use of proton pump inhibitor for 7-day window.
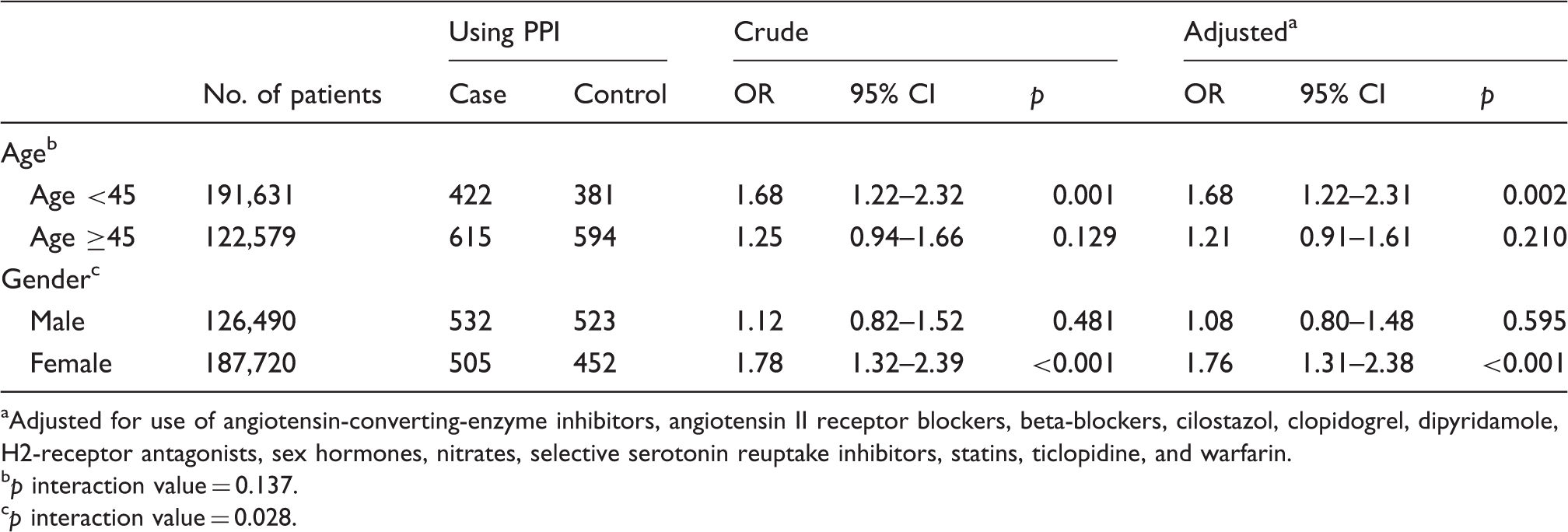
Adjusted for use of angiotensin-converting-enzyme inhibitors, angiotensin II receptor blockers, beta-blockers, cilostazol, clopidogrel, dipyridamole, H2-receptor antagonists, sex hormones, nitrates, selective serotonin reuptake inhibitors, statins, ticlopidine, and warfarin.
Discussion
The data in this study reveal several major findings. First, headache risk was temporarily increased after PPI use. The overall headache risk increased up to 1.41-fold within 7 days after PPI use. Second, the risk of headache differed between different PPIs, with esomeprazole and lansoprazole presenting the greatest risk that was similar to that of nitrates. Third, female patients were more susceptible to PPI-related headache. Fourth, the coding of PPI-induced headache was mainly nonspecific.
There are several strengths of this study. First, this is a population-based study that relied on a comprehensive medical utilization claims database that included all headache patients who visited physicians between 1998 and 2010 in Taiwan. The large sample size provided sufficient power for statistical analyses. Second, all five oral PPIs were included in the analysis and compared individually. We additionally controlled for the use of the most well-known headache-inducing drugs to prevent potential confounding effects. Third, the occurrence and reporting of headache are often influenced by other factors, such as personality traits, history of concurrent headache disorders, work stress, or values. These factors can be difficult to control for among groups of patients (27,28). The use of a case-crossover study design minimizes the confounding factors between subjects. Finally, we compared the risks of acute headache for PPIs to medications with known headache risks, such as nitrates (positive control) and domperidone (negative control). These controls demonstrate that the headache risk estimation for PPIs in our study was reliable.
Our study showed that PPIs were associated with an increase in headaches (ICD 784.0). Although not significant, the risk of tension headaches was also increased. In contrast, the risk of migraines was not increased. Our results somewhat agree with a previous study that reported that PPI-induced headaches were mainly tension-type headaches (75%) or unclassified headaches (7). However, the diagnoses of headaches in our study were based on ICD coding, and the proportion of non-specific headaches was high. These non-specific headache patients might fulfill the diagnosis of migraine or tension-type headache (29). The lack of statistical significance in tension headache or migraine might be a result of small sample size. Our data also showed that female patients are more susceptible to PPI-induced headache. This finding agrees with previous clinical trials and post-marketing surveys (7). The reasons for gender discrepancies remain unknown, but women are more susceptible to certain headache disorders, such as migraines, tension-type headaches, and nonspecific headaches (30,31). Moreover, the nocebo effect is more common in female patients and should be considered in medication-related side effects (32,33).
Compared with the other PPIs, lansoprazole and esomeprazole were more likely to induce headache. This result is similar to one cohort study that included only omeprazole, lansoprazole, and pantoprazole (6). The reason for the discrepancy in headache risk among different PPIs remains speculative, but certain biological and physiological characteristics are different between PPIs, such as bioavailability, metabolism, and drug interactions (34,35). For example, lansoprazole and esomeprazole were metabolized mainly by liver enzyme cytochrome P450(CYP) 3A4, whereas omeprazole and pantoprazole PPIs were metabolized mainly by CYP 2C19, and rabeprazole by both renal and liver (CYP 2C19 and 3A4) pathways (8,34–36). Lansoprazole and esomeprazole also have the highest bioavailability. Another study showed lansoprazole had the highest incidence of adverse effects stemming from drug interactions among the five PPIs (37). Therefore, further studies are needed to determine the mechanisms of PPI-induced headaches.
Our study has clinical implications. First, as PPIs increased the risk of acute headache, especially in female patients, physicians should be cautious when prescribing these medications to susceptible patients. Second, headache patients are more likely to be comorbid with peptic ulcers or gastroesophageal reflux disease and need PPI treatment (38,39). PPI-induced headache should be considered when headaches worsen after treatment. Third, certain PPIs are more likely to induce headache than the others. Physicians may select different PPIs to prevent or improve headaches in susceptible patients.
Some methodological issues should also be addressed. First, the occurrence of headache in this study was based on physician visits in the administrative database. The headache incidence could be underestimated because patients with headaches might not visit physicians. Nevertheless, this bias might not be clinically significant because patients who did not seek medical treatment for headaches might have milder or tolerable headaches because the accessibility to the healthcare system is high in Taiwan. Second, the NHIRD dataset is an administrative database that lacks thorough clinical data, such as headache characteristics, severity, neuroimaging, or other laboratory results. Therefore, the precise diagnosis of headache disorders was not available. Besides, there was no validation study of these diagnostic codes for headache disorders in the NHIRD in Taiwan. However, the estimation of overall headache should be reliable because the diagnosis of headache was based on patient complaint rather than a diagnostic algorithm. In this study, most headache diagnoses were unclassified. Third, there may be deviations in the claims data, as coding by physicians may purposefully correspond to their prescriptions or other factors. However, the data remain representative. The Bureau of the NHI routinely and randomly samples a fixed percentage of claims from every contracted medical institution, and the accuracy of coding is audited by an independent group of physicians. Fourth, the case-crossover design does not account for within-person confounding such as the acute indication of PPI use or fluctuations in disease severity. As mentioned above, migraine and GERD are comorbid disorders to each other. Fifth, our study could not delineate whether the clinic visits for headache were because of headache exacerbation or new onset headache based on the claim data. Finally, although we controlled for the most well-known and frequently used medications that are known to induce headaches, there are still unidentified headache-inducing drugs that would not have been included in this study.
In conclusion, PPI use was associated with an increased risk of acute headache. The headache risks differed between populations based on gender and specific PPI. Clinicians should consider the association of PPIs and headache when prescribing PPIs.
Clinical implications
PPIs increased the risk of acute headache, especially in female patients. Lansoprazole and esomeprazole had the highest risks of headache, similar to that of nitrates. PPI-induced headache should be considered in patients with other headache and acid-related disorder concurrently.
Footnotes
Funding
This study was supported in part by grants from the National Science Council of Taiwan (100-2314-B-010-019-MY2, 100-2314-B-010-018-MY3, 102-2321-B-010 -030 -), Taipei-Veterans General Hospital (VGHUST102-G7-6-1, V102C-118, V102E9-001), NSC support for Center for Dynamical Biomarkers and Translational Medicine, National Central University, Taiwan (NSC 102-2911-I-008-001-), Brain Research Center, National Yang-Ming University and a grant from Ministry of Education, Aim for the Top University Plan. No additional external funding was received for this study.
Conflict of interest
None declared.
