Abstract
Background:
Systematic data are lacking on the immune response toward SARS-CoV-2 mRNA vaccination in SPMS patients on disease-modifying therapies (DMTs).
Objective:
The AMA-VACC clinical trial was designed to characterize immune responses to SARS-CoV-2 mRNA vaccines in siponimod-treated SPMS patients.
Design:
AMA-VACC is an ongoing three-cohort, multicenter, open-label, prospective clinical study.
Methods:
The study included patients at risk for SPMS or patients with SPMS diagnosis. Patients received SARS-CoV-2 mRNA vaccine as part of their clinical routine during ongoing siponimod treatment (cohort 1), during siponimod treatment interruption (cohort 2), or while on dimethyl fumarate, glatiramer acetate, beta-interferons, teriflunomide, or no current therapy (cohort 3). SARS-CoV-2-specific neutralizing antibodies and T-cell responses were measured 1 week and 1 month after the second dose of vaccination.
Results:
In total, 17 patients, 4 patients, and 20 patients were recruited into cohorts 1, 2, and 3, respectively. The primary endpoint of seroconversion for SARS-CoV-2-neutralizing antibodies at week 1 was reached by 52.9%, 75.0%, and 90.0% of patients in cohorts 1, 2, and 3, respectively. For 64.7% of patients in cohort 1, all patients in cohort 2, and 95% of patients in cohort 3, seroconversion was observed at either week 1 or month 1 or both time points. After 1 week, 71.4% of cohort 1, 75.0% of cohort 2, and 85.0% of cohort 3 were positive for either SARS-CoV-2-neutralizing antibodies or SARS-CoV-2-specific T-cells or both. After 1 month, the rates were 56.3%, 100.0%, and 95.0%, respectively.
Conclusion:
The study shows that the majority of siponimod patients mount humoral and cellular immune response under continuous siponimod treatment. The data do not sufficiently support interruption of treatment for the purpose of vaccination.
Registration:
EU Clinical Trials Register: EudraCT 2020-005752-38 (www.clinicaltrialsregister.eu); ClinicalTrials.gov: NCT04792567 (https://clinicaltrials.gov).
Keywords
Introduction
Limited data are available on the efficacy of SARS-CoV-2 mRNA vaccines in patients with secondary progressive multiple sclerosis (SPMS) receiving disease-modifying therapies (DMTs). Some evidence suggests an impaired antibody response in DMT-treated patients.1,2
The response to SARS-CoV-2 infection is characterized by the development of IgG, IgA, and IgM antibodies, including neutralizing antibodies (NAbs) able to block viral entry. 3 In addition, T-cells play a central role. Major histocompatibility complex (MHC) I class presentation of intracellular viral peptides activates CD8+ cytotoxic T-cells. Upon destruction of the host cells, viral peptides are released and presented by MHC class II antigen-presenting cells. This triggers CD4+ T-cell expansion and differentiation to T-helper 1 (Th1) and T-helper 2 (Th2) cells. CD4 + Th2-cells then stimulate B-cells to produce virus-specific antibodies, while CD4+ Th1-cells activate T-cell-mediated antiviral response.4–6
SARS-CoV-2 mRNA vaccines mimic the natural antiviral reaction, including initial cellular mRNA uptake and subsequent production, presentation, and release of the viral spike protein. The mRNA vaccine by Moderna (mRNA-1273) and the BioNTech/Pfizer mRNA vaccine (BNT162b2) both encode the full-length spike protein that has been stabilized in its prefusion conformation (S-2P).7,8
BNT162b2 and mRNA-1273 have both demonstrated efficacy in preventing COVID-19 in persons not receiving immunosuppressive or immunomodulating medications.9,10 Both have been shown to induce anti-spike protein antibodies and NAbs in all patients after the second dose of vaccination as well as Th1-based CD4+ and CD8+ T-cell reactivity against the spike protein.8,11 The initial response to mRNA vaccines therefore involves CD8+ cytotoxic T-cells, while Th1- and Th2-cells mediate the secondary response including antibody production.8,11
It is therefore essential to investigate both humoral and cellular immune reactivity toward mRNA vaccines in patients receiving DMTs like siponimod, which inhibits S1P1 and S1P5 receptors on lymphocytes inducing their retention in lymph nodes. 12 The summary of product characteristics (SmPC) for siponimod suggests considering temporary treatment interruption for vaccination. 13 Protein-based influenza vaccines have proven adequate reactivity in siponimod patients. 14 However, it remains unclear whether siponimod impacts the immune response to the novel mRNA-based vaccines and whether treatment interruption is necessary in this particularly vulnerable patient population.
This study was designed to characterize the cellular and humoral immune response to SARS-CoV-2 mRNA vaccines in siponimod-treated SPMS patients with and without treatment interruption, and in a control group receiving dimethyl fumarate, glatiramer acetate, beta-interferons, teriflunomide, or no DMT. With these data, we aim to offer a guidance to treating physicians and patients for the coordination of multiple sclerosis (MS) therapy and vaccination.
Materials and methods
Study design, participants, and treatments
[An Open-label Multicenter Study to Assess Response to SARS-CoV-2 modRNA VACCines in Participants With Secondary Progressive Multiple Sclerosis Treated With Mayzent (Siponimod)] (AMA-VACC) is an ongoing three-cohort, multicenter, open-label, prospective clinical study (EudraCT 2020-005752-38; NCT04792567) over 6 months in Germany. The study population consists of patients with SPMS diagnosis and patients with relapsing-remitting multiple sclerosis (RRMS) at risk of developing SPMS. No criteria for RRMS at risk of SPMS transition were applied. Selection of patients of this category was at the discretion of the physician. Patients had to be eligible and planning to receive an SARS-CoV-2 mRNA vaccine as part of clinical routine. Patients with acute [assessed by polymerase chain reaction (PCR)] or previous SARS-CoV-2 infection (assessed by IgA levels ⩾0.8 index and IgG levels ⩾50 AU/ml) at screening were excluded.
SARS-CoV-2 vaccinations were administered according to the German vaccination guidance at dedicated sites outside this study. Booster vaccinations are allowed as part of the clinical routine at the physician’s discretion.
Patients are treated with siponimod, glatiramer acetate, dimethyl fumarate, interferons, or teriflunomide or currently receive no DMT as part of their clinical routine. The first cohort consists of participants vaccinated during ongoing siponimod treatment. The second cohort includes participants interrupting their siponimod therapy for vaccination. Physician and patient decided upon the preferred option, which are both according to the siponimod SmPC. The third cohort (control group) receives vaccination while on dimethyl fumarate, glatiramer acetate, beta-interferons, teriflunomide, or no DMT.
Administrative procedures
The trial is conducted in accordance with the International Conference on Harmonisation guidelines for Good Clinical Practice and the principles of the Declaration of Helsinki. The protocol was approved by the ethics committee ‘Technische Universität Dresden’ (AMG ff-EK-34012021). All patients or their legal representatives provided written informed consent before commencing trial-related procedures.
Outcomes and assessments
The primary endpoint is the proportion of participants achieving seroconversion as defined by detection of SARS-CoV-2 NAbs 1 week after the second dose of vaccine. Secondary endpoints are SARS-CoV-2 serum NAb levels and SARS-CoV-2-specific T-cell reactivity.
NAbs are analyzed utilizing the cPassTMSARS-CoV-2 Neutralization Antibody Detection Kit from GenScriptUSA Inc (L00847) and the assay specific cut-off was used for interpretation. SARS-CoV-2-specific T-cell reactivity is assessed ex vivo by enzyme-linked immunosorbent spot (ELISpot) assays measuring the release of interleukin-2 (IL-2) or interferon-gamma (IFN-γ) from isolated peripheral blood mononuclear cells (PBMCs, 2 × 105) upon antigen stimulation with SARS-CoV-2 peptide mix. The CoV-iSpot Interferon-γ plus Interleukin-2 (ELSP 7010 strip format) from GenID is used. Cross-reactivity with other coronaviruses is assessed using the same ELISpot assay after PBMC stimulation with a homologous coronavirus family peptide mix (pan-corona peptide mix).
Assessments are performed at 1 week, 1 month, and 6 months after the second dose of the vaccination cycle. If booster vaccinations are performed, an additional study visit is planned 1 month after this booster vaccination. A final follow-up call to assess the occurrence of COVID-19 infections is intended 12 months after the second dose of the vaccination cycle.
Statistical methods
The results of a pre-planned interim analysis, scheduled after all participants have completed the study visit 1 week after the second vaccination, are presented (data cut-off: 31 January 2022). This analysis constitutes the primary analysis of the study. The primary endpoint results, month 1 seroconversion data and secondary data on T-cell reactivity at week 1 and month 1 are presented along with safety data. Month 6 data and antibody levels will be presented separately with the final study results.
No formal statistical testing was applied. A sample size of 20 participants per arm was selected based on the need for early availability of results and the feasibility to recruit sufficient participants. The number and the proportion of participants per group achieving seroconversion were analyzed descriptively and presented as frequencies and percentages with a 95% confidence interval (exact Clopper–Pearson). All secondary endpoints were analyzed descriptively and presented as frequencies and percentages, mean and standard deviation (SD), or median and range. Statistical analyses were performed with SAS version 9.2.
Results
In total, 41 MS patients were enrolled at 10 sites in Germany from 19 April 2021 to 4 August 2021, no screening failures were reported. Of these, 17 patients, 4 patients, and 20 patients were recruited into cohorts 1, 2, and 3, respectively. All patients were tested negative for previous or acute SARS-CoV-2 infection by IgA/IgG assessment and PCR. All patients (100%) had completed week 1 visit, and 40 patients (97.6%) had completed month 1 visit at the time of the interim analysis. For one patient, the assessment at month 1 was not performed.
Patient characteristics per cohort for this first interim analysis are shown in Table 1. Briefly, median age of participants was 51–56 years and MS history was 9–18 years, with higher age and longer disease history in the siponimod cohorts (cohorts 1 and 2) (Table 1). The mean (±SD) duration of interruption of siponimod for the purpose of vaccination in cohort 2 (n = 4) was 76.7 ± 15.0 days. Siponimod treatment was stopped at a mean 15.3 ± 9.1 days before first vaccination until a mean 29.7 ± 2.9 days after second vaccination, reflecting the suggested treatment break according to the SmPC.
Patient characteristics.

DMF, dimethyl fumarate; GA, glatiramer acetate; IFN, interferon-beta; MS, multiple sclerosis; RRMS, relapsing-remitting multiple sclerosis; SPMS, secondary progressive multiple sclerosis; TF, teriflunomide.
If not indicated otherwise, data are presented as median (min; max).
The primary endpoint of seroconversion for SARS-CoV-2 NAbs at week 1 was reached by 9 of 17 patients (52.9%) in cohort 1 with continuous siponimod treatment, by 3 of 4 patients (75.0%) in cohort 2 with interrupted siponimod treatment, and by 18 of 20 patients (90.0%) in cohort 3, that is the control group. Seroconversion at month 1 was observed in 9 of 16 patients (56.3%) in cohort 1, in 4 of 4 patients (100.0%) in cohort 2, and in 19 of 20 patients (95.0%) in cohort 3 (Figure 1(a)). In 11 of 17 patients (64.7%) in cohort 1, in 4 of 4 patients (100%) in cohort 2, and in 19 of 20 patients (95.0%) in cohort 3, seroconversion had been observed at either week 1 or month 1 or both time points after full vaccination.

(a) Development of SARS-CoV-2 neutralizing antibodies; (b) SARS-CoV-2-specific T-cell responses; and (c) combined immune responses.
One week after vaccination, 7 of 14 patients (50.0%) continuously treated with siponimod, 3 of 4 patients (75.0%) interrupting their siponimod treatment, and 12 of 20 patients (60.0%) in the control group mounted an SARS-CoV-2-specific T-cell response. One month after second vaccination, T-cell reactivity was observed in 0 of 16 patients (0.0%), in 1 of 4 patients (25.0%), and in 14 of 20 patients (70.0%) of cohorts 1, 2, and 3, respectively (Figure 1(b)). Of note, T-cell response could not be assessed in three patients with continued siponimod treatment at the 1-week visit and one patient with continued siponimod treatment at the 1-month visit because of insufficient cell counts after PBMC isolation. Due to its mode of action, siponimod treatment reduces the proportion of CD3+ T-lymphocytes in the blood, which was also observed in this study (Table 2). This resulted in a reduced proportion of CD3+ cells in isolated PBMCs and thus a lower absolute number of T-cells among the 2 × 105 PBMCs plated in ELISpot assays in the continuously treated siponimod cohort.
Proportion of CD3+ T-lymphocytes of total PBMCs.
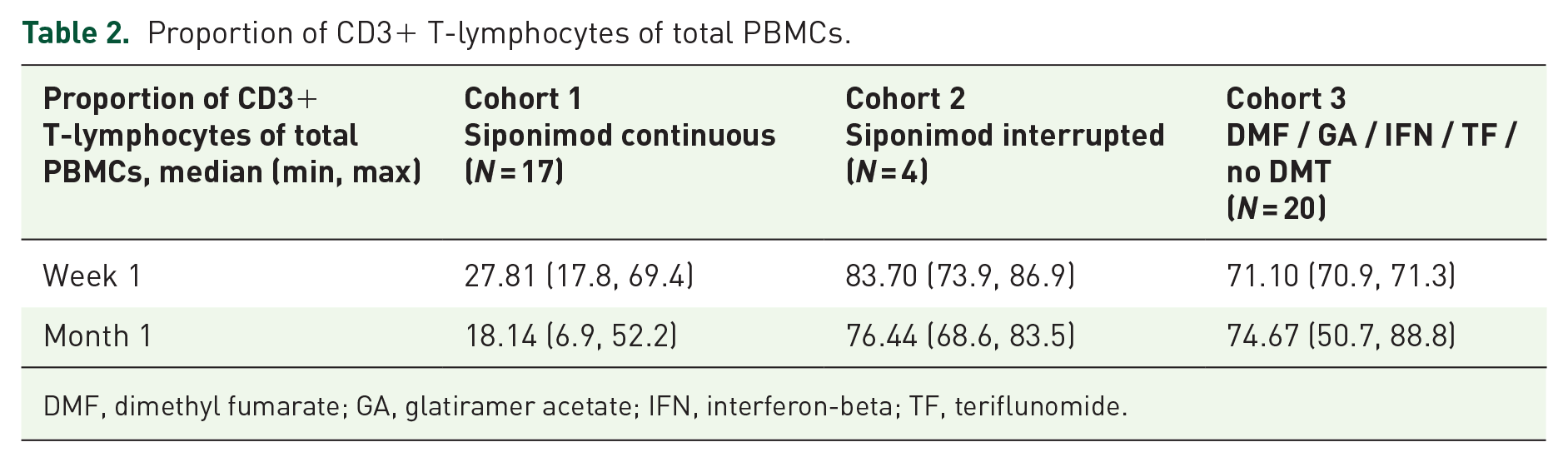
DMF, dimethyl fumarate; GA, glatiramer acetate; IFN, interferon-beta; TF, teriflunomide.
Analysis of combined immune response (development of SARS-CoV-2-specific NAbs or T-cell reactivity or both) showed that 1 week after the second dose of vaccine, 10 of 14 patients in cohort 1 (71.4%), 3 of 4 patients in cohort 2 (75.0%), and 17 of 20 patients in cohort 3 (85.0%) were positive for either humoral or cellular response or both. One month after the second dose of vaccine, 9 of 16 patients in cohort 1 (56.3%), 4 of 4 patients in cohort 2 (100.0%), and 19 of 20 patients in cohort 3 (95.0%) were positive for either humoral or cellular response or both (Figure 1(c)); percentages refer to patients with evaluable assessment].
To analyze the level of cross-reactivity with other coronaviruses, an ELISpot assay measuring IFN-γ or IL-2 secretion of PBMCs after stimulation with a pan-coronavirus peptide mix representative for multiple other coronaviruses was performed. None of the patients with T-cell response against SARS-CoV-2 at week 1 or month 1 in the siponimod cohorts (cohorts 1 and 2) had IFN-γ or IL-2 reactivity against other coronaviruses. For cohort 3, the results showed no IL-2-reactivity but were equivocal for IFN-γ in one patient, suggesting possible previous immune response against other coronaviruses. None of the patients negative for SARS-CoV-2-specific T-cell reactivity at week 1 or month 1 had IFN-γ or IL-2 reactivity against other coronaviruses at screening.
Until the cut-off date of this interim analysis, two relapses occurred during the study, both more than 5 months after the last vaccination in cohort 1. No relapses were observed in cohorts 2 and 3. Overall, 25 patients (61.0%) reported adverse events (AEs) during the study. Of these, 20 AEs were related to DMTs or SARS-CoV-2 vaccines (Table 3). One patient from cohort 3 reported serious AEs (acute sinusitis and gastroenteritis rotavirus). One patient from cohort 1 discontinued study medication (siponimod) due to AEs until the cut-off date. No COVID-19 infection was reported until the cut-off date. No deaths occurred.
Overview on adverse events.

In case of multiple AEs, a patient is counted only once in the respective category.
DMF, dimethyl fumarate; GA, glatiramer acetate; IFN, interferon-beta; TF, teriflunomide.
MS relapse, leg pain after injection of glatiramer acetate, liver transaminase elevation.
Discussion
According to the AMA-VACC interim results, about two-third of patients under continuous siponimod treatment, all patients with siponimod treatment interruption, and almost all patients of the control group developed NAbs within 1 month after vaccination. T-cell response was developed by 50–75% of patients in the different cohorts. Taken together, more than 70% of patients with continuous siponimod treatment, 75% with siponimod interruption, and 85% in the control group developed SARS-CoV-2-specific humoral or cellular response or both as soon as 1 week after full vaccination. SARS-CoV-2-reactivity in all cohorts was neither elicited by prior SARS-CoV-2 infection nor impacted by coronavirus cross-reactivity.
Adaptive immune response to SARS-CoV-2 has been shown to involve IgG antibodies as well as CD4+ and CD8+ T-cell reactivity.4–6,8,11 NAbs, a subset of specific antibodies, have been shown to prevent binding of virus particles to the host cells and interrupt viral entry. 3 They are considered a more stringent correlate of protective immunity than total anti-SARS-CoV-2 antibodies. 3 The presence of NAbs in combination with CD4+ and CD8+ T-cell reactivity have been proposed as suitable predictors of a protective immune response. 15 Therefore, in contrast to other studies, the present study analyzes the development of SARS-CoV-2-specific NAbs instead of total anti-SARS-CoV-2 antibodies. In addition, SARS-Cov-2-specific T-cell reactivity is analyzed. The latter is measured by antigen-stimulated release of IFN-γ and IL-2, which are cytokines released by activated CD4+ and CD8+ T-cells. 16 The results of this study provide important information about whether siponimod-treated patients are able to mount potentially protective immunity after vaccination.
It has previously been hypothesized that both humoral and cellular immune responses are functional in patients treated with S1PR modulators as the majority of patients recovers unremarkably from COVID-19. 15 According to a case series, 86% of siponimod patients reported asymptomatic, mild, or moderate infection, and the majority of patients completely recovered or were recovering at the time of data collection. 17 As according to the authors of the case series, their data are subject to potential underreporting of less severe cases typical for post-marketing settings, 17 these results should not be generalized, but at least allow to assume sufficient immunocompetence in siponimod-treated patients. The present findings from AMA-VACC further support this hypothesis and highlight that it is important to consider both humoral and cellular immune responses. 15 T-cells are a prerequisite for B-cell activation and antibody development after vaccination with mRNA vaccines. 16 The development of NAbs suggests initial T-cell reactivity together with functional T-cell–B-cell interaction in the majority of patients even if T-cell responses were not detectable in all patients with NAbs.
Up to now, evidence regarding immune reactivity toward SARS-CoV-2 vaccines in siponimod-treated patients was mainly limited to humoral response. A study including 13 patients receiving siponimod and 11 healthy controls detected antibodies in 85% and 100% of patients, respectively. 18 Bar-Or et al. 19 and Conte 20 found similar seroconversion rates for siponimod-treated patients (80% and 88%, respectively). Neutralization activity was not assessed. Results seen with other S1PR modulators are quite diverse with seroconversion rates ranging from 3.8% to 85.7%.21,22 Due to differences in pharmacokinetics and pharmacodynamics, the results cannot be transferred to siponimod. 23 Antibody titers in patients receiving siponimod (n = 13) were found to be significantly lower than in healthy controls (n = 11). 18 However, it should be noted that no clinical relevance threshold for antibody titers has yet been established. To what extent lower titers in patients with seroconversion under siponimod might impact the efficacy of the vaccination, if at all, cannot be answered so far.
The AMA-VACC study is the first to systematically analyze both humoral and cellular immune responses to SARS-CoV-2 vaccines in patients receiving siponimod. In line with previous publications recommending SARS-CoV-2 vaccination for patients currently receiving DMTs,24,25 the present results support vaccination of siponimod-treated patients.
The SmPC of siponimod recommends temporary discontinuation 1 week prior until 4 weeks after vaccination. 13 While AMA-VACC shows that SARS-CoV-2 vaccination of siponimod-treated patients induced relevant immune reactivity, the results regarding treatment interruption for vaccination need to be interpreted carefully. It has to be pointed out that patients could be included in either cohort 1 or 2 at the investigators’ discretion. Most physicians prioritized ongoing therapy, probably because of the increased risk of disease activity and progression associated with treatment interruption. 26 This contrasts with a higher immune response rate in the cohort with paused siponimod. However, results from this very small-sized cohort (n = 4) are insufficient to support siponimod interruption. The potential marginal benefit does not outweigh the associated risks. Given that an immune response can be achieved under continued siponimod treatment and a third vaccine dose has meanwhile been recommended to increase vaccination efficiency, treatment interruption is even less favorable. Booster vaccinations are allowed in the AMA-VACC study. The results regarding these booster vaccinations are not available yet and will be described together with the results of the final analysis.
Concomitant siponimod use during vaccination is supported by a double-blind, placebo-controlled study with siponimod in healthy volunteers. It was observed that concomitant siponimod was associated with no relevant effect on antibody response to pneumococcal polysaccharide vaccine. Furthermore, most of the patients showed seroconversion for a T-cell-dependent influenza vaccine although titers were lower in comparison with placebo. 14 Furthermore, safety results agreed with previous safety data for both, DMTs 27 and SARS-CoV-2, vaccines.9,10 This suggests that vaccination during continuous siponimod treatment is safe and induces SARS-CoV-2-specific immune response.
Despite these encouraging results, the present study bears some limitations. First, the study included a small sample size only. Confirmation of the results in further studies is thus necessary. Nevertheless, the results of AMA-VACC allow for the assumption that an immune response toward SARS-CoV-2 mRNA vaccines is elicited in siponimod-treated patients. Second, participants in cohorts 1 and 2 are older and have a longer MS history than participants in cohort 3. Based on recently published data, higher age is negatively correlated with SARS-CoV-2 NAb titers after vaccination and could potentially confound this analysis.28,29 Although patients in cohorts 1 and 2 can be considered representative for patients currently treated with siponimod in general clinical practice, the need for earlier diagnosis of SPMS might rejuvenate the typical siponimod population in future. 30 It can be hypothesized that immune response rates in earlier diagnosed and thus younger SPMS patients might be even higher.
Noticeably, siponimod treatment in cohort 1 reduced the proportion of CD3+ T-lymphocytes in the blood and the absolute number of plated T-cells in ELISpot assays. This possibly minimized the overall chance of IFN-γ or IL-2 release. This technical problem due to the mode of action of siponimod limits the meaningfulness of this assay in siponimod-treated patients and potentially underestimates the T-cell response in this cohort. Regarding cohort 2, it has to be pointed out that the very small size impacts the meaningfulness of the results and they should not be the basis for rushed treatment decisions.
In summary, the results of the pre-planned interim analysis of the AMA-VACC study show that the majority of siponimod-treated patients mounts humoral and cellular immune responses under continuous siponimod therapy. The presented interim analysis data are insufficient to support a general recommendation for an interruption of treatment for the purpose of vaccination. Further results from AMA-VACC on the effect of booster vaccinations, the maintenance of the immune response, and the clinical efficacy regarding COVID-19 in siponimod-treated patients will be published together with the final analysis of the study.
