Abstract
Background and Purpose:
Considering the highly time-dependent therapeutic effect of endovascular treatment in patients with large vessel occlusion–associated acute ischemic stroke, prehospital identification of large vessel occlusion and subsequent triage for direct transport to a comprehensive stroke center offers an intriguing option for optimizing patient pathways.
Methods:
This prospective in-field validation study included 200 patients with suspected acute ischemic stroke who were admitted by emergency medical service to a comprehensive stroke center. Ambulances were equipped with smartphones running an app-based Field Assessment Stroke Triage for Emergency Destination scale for transmission prior to admission. The primary measure was the predictive accuracy of the transmitted Field Assessment Stroke Triage for Emergency Destination for large vessel occlusion and the secondary measure the predictive accuracy for endovascular treatment.
Results:
A Field Assessment Stroke Triage for Emergency Destination ⩾4 revealed very good accuracy to detect large vessel occlusion–related acute ischemic stroke with a sensitivity of 82.4% (95% confidence interval = 65.5–93.2), specificity of 78.3% (95% confidence interval = 71.3–84.3), and an area under the curve c-statistics of 0.89 (95% confidence interval = 0.85–0.94). Field Assessment Stroke Triage for Emergency Destination ⩾4 correctly identified 84% of patients who received endovascular treatment [73.5% specificity (95% confidence interval = 66.4–79.8)] with an area under the curve c-statistics of 0.82 (95% confidence interval = 0.74–0.89). In a hypothetical triage model of an urban setting, one secondary transportation would be avoided with every fifth patient screened.
Conclusion:
A smartphone app-based stroke triage completed by emergency medical service personnel showed adequate quality for the Field Assessment Stroke Triage for Emergency Destination to identify large vessel occlusion–associated acute ischemic stroke. We demonstrate feasibility of the use of a medical messaging service in prehospital stroke care. Based on these first results, a randomized trial evaluating the clinical benefit of such a triage system in an urban setting is currently in preparation.
Clinical Trial Registration: https://clinicaltrials.gov Unique identifier: NCT04404504.
Introduction
The introduction of endovascular treatment (EVT) in addition to intravenous thrombolysis (IVT) has vastly improved treatment of patients with acute ischemic stroke (AIS) caused by large vessel occlusion (LVO). However, while IVT can be applied in any hospital with specialized stroke care, EVT requires a much more complex infrastructure and mostly is limited to Comprehensive Stroke Centers (CSCs). In consideration of the highly time-dependent therapeutic effect of the reperfusion therapy in AIS,1,2 many prehospital patient pathways still focus on transportation to the nearest stroke unit. On the contrary, patients with LVO-related AIS admitted to centers without EVT capability must be referred to CSCs after initiation of IVT by secondary interhospital transfer (drip-and-ship). This procedure is not only expensive and causes inefficient use of emergency medical service (EMS) resources, but also delays the possible use of EVT, resulting in a poorer clinical outcome. 3 Therefore, prehospital identification of patients with LVO and subsequent triage for direct transport to a CSC offers an intriguing option for optimizing patient pathways. This is especially useful in urban settings, where delay to first hospital contact is negligible when bypassing a nearby non-EVT center. In this context, several clinical scores were developed to estimate LVO risk in patients with AIS in the emergency setting. Among them, the simple and short Field Assessment Stroke Triage for Emergency Destination (FAST-ED) scale yields high sensitivity by adding cortical symptoms to the regular FAST-test. 4 Previous studies have shown superior prediction quality in comparison to other LVO recognition scores; 5 however, in-field validation has not yet been studied. In addition, very limited experience with prehospital smartphone-assisted assessment of stroke patients by paramedic EMS personnel is available 6 and further proof of practicability is urgently needed. 7
In this prospective study, we performed in-field validation of the smartphone-based FAST-ED scale that was digitally transmitted by paramedic EMS personnel prior to hospital arrival. In addition, we aimed to evaluate the potential impact of prehospital triage of patients with suspected stroke.
Methods
Study setting
This was a prospective in-field validation study for an app-based prehospital stroke triage for patients with suspected acute stroke. The study included 200 consecutive patients who were admitted by EMS to the CSC of the Neurological University Hospital Essen, Germany, from March 2019 to August 2020 and met the following inclusion criteria: suspected acute stroke, age above 18 years, and a digitally transmitted FAST-ED by EMS prior to admission. Patients with confirmed onset of stroke symptoms beyond 24 h were excluded.
EMS personnel of the city of Essen, which is managed by the local fire department (Feuerwehr der Stadt Essen), was systematically trained to use an app-based FAST-ED score. In addition, all ambulances were equipped with a robust smartphone (Caterpillar, CAT S60) running a customized German version of a FAST-ED triage app and a medical messaging service (JoinTriage and Join; Allm, Inc. https://play.google.com/store/apps/details?id=net.allm.fasted&hl=en https://apps.apple.com/us/app/jointriage/id1099779970). EMS personnel used this messaging platform to digitally transmit the examination results to the hospital stroke team prior to arrival.
EMS is designed as a two-tiered system including paramedic staffed ALS-Ambulances and physician staffed response units. In suspected stroke without signs of severe respiratory distress, the EMS dispatch center protocol leads to a single ALS-Ambulance response. Thus, in majority of suspected stroke, only paramedic EMS personnel is involved.
FAST-ED is a simple scale that adds clinical symptoms predicting cortical involvement (gaze deviation, denial, neglect) to the commonly used FAST-test, which already includes the evaluation of facial palsy, arm weakness, and speech disturbances. The app-based FAST-ED scale omits the need to examine symptoms of neglect and denial, if the patient suffers from aphasia or in the absence of arm weakness. The score ranges from zero to eight points (Figure 1) and FAST-ED items as well as basic clinical information (time of symptom onset, age) are entered into the Triage App, which then automatically predicts the likelihood of LVO based on previous data. 8 Assessment results and the estimated time of arrival are digitally transferred to our hospital via the smartphone App.
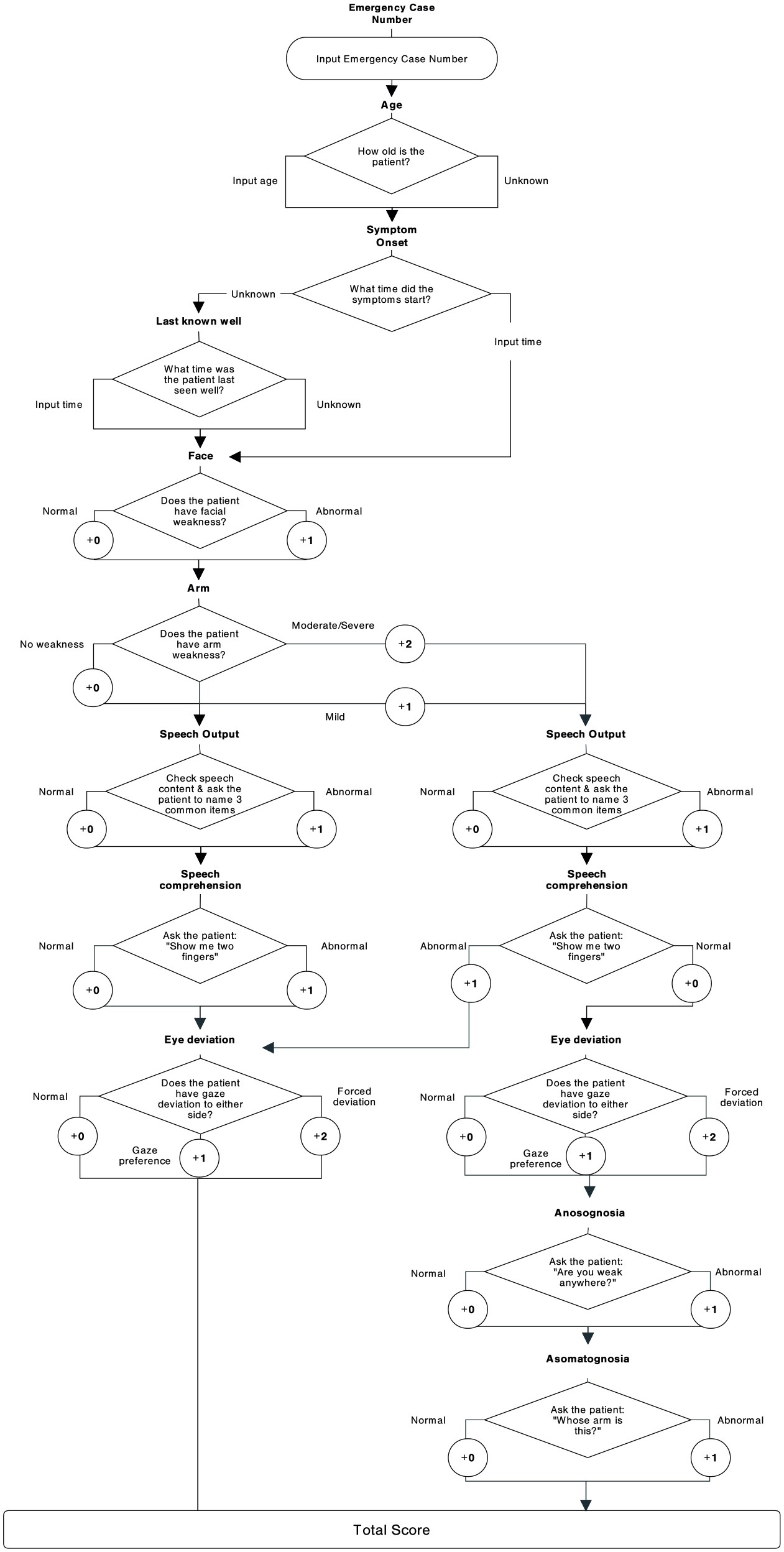
Flow chart of the algorithm used in JoinTriage.
The assessment of age was modified from a dichotomized (⩾80 years) to a continuous variable after analyzing the first 40 patients) and information of prior oral anticoagulation was excluded from the assessment due to the lack of validity (the respective data are presented in the result section). The implementation of these changes was completed after the inclusion of 53 patients. The sequence of the FAST-ED items itself remained unchanged. 8 The final app display and algorithm are presented in Figure 1 and Supplemental Figure S1.
Outcome measures and statistical analysis
The primary measure was the predictive accuracy of the prehospital FAST-ED scale as computed by EMS personnel for LVO-associated AIS, defined as symptomatic occlusion of the internal carotid artery, the first segments of the middle cerebral artery (M1, M2), or the basilar artery. Additional analysis was performed using a stricter definition of LVO that excludes the M2-middle cerebral artery segments. Overall, patients were grouped into four categories: (1) ischemic stroke with LVO, (2) ischemic stroke without LVO, (3) intracranial hemorrhage (ICH), and (4) stroke mimics. The detection of LVO in patients with AIS was performed by computed tomography angiography (CTA), which is the standard emergency diagnostic tool in our center for patients with ischemic stroke eligible for reperfusion treatment. All patients with AIS received vascular imaging. In patients with AIS clinically not eligible for IVT or EVT (e.g. due to non-disabling symptoms or large infarction on non-contrast CT), and who did not receive CTA on admission, emergency extracranial Doppler/duplex and transcranial Doppler ultrasound was used to detect LVO (n = 47 patients, 33.8%). 7 The secondary outcome measure was the predictive accuracy of FAST-ED used by EMS personnel regarding EVT.
The predictive accuracy for the primary and secondary outcome measures were evaluated using receiver operating curve (ROC) analysis and the area under the curve (AUC) c-statistics. Values are presented together with the respective 95% confidence intervals (CIs). To compare the FAST-ED scale used by EMS personnel with the results obtained from the examination of stroke neurologists in hospital, a FAST-ED score was calculated using the National Institute of Health Stroke Scale (NIHSS) score at admission, as described previously, 4 and analyzed using the nonparametric Spearman coefficient. Rater agreement for basic clinical information (age, intake of oral anticoagulation, symptom onset) assessed by EMS personnel and emergency neurologists was calculated using Cohen’s Kappa statistics.
Based on the nature and distribution of the data, descriptive statistics are presented as numbers and percent, median with interquartile range (IQR), or mean with standard deviation. Chi-square tests were used for categorical variables, and based on the distribution of the data, t-tests or Mann–Whitney U tests were used for continuous variables. Statistical analyses were performed with SPSS (version 25.0; IBM Inc) and a two-sided p < 0.05 was considered the minimal level of statistical significance.
Results
Overall, we included 200 patients with suspected acute stroke. Final discharge diagnosis was AIS in 139 patients (69.5%) and ICH in 12 patients (6.0%). In 49 patients (24.5%), other diagnoses were found which mimicked acute stroke in the prehospital setting (Figure 2).

Flow chart with description of subgroups based on final discharge diagnosis and FAST-ED scale dichotomized between 3 and 4.
Assessment quality for clinical information
Assessment of prior anticoagulation by EMS personnel revealed poor inter-rater reliability with emergency neurologists (Cohen’s Kappa = 0.189; sensitivity = 88.2%, specificity = 40.0%). Even though oral anticoagulation was explained on the app screen by listing the specific drugs, the false-positive rate was 60%, which was mainly due to an incorrect classification of antiplatelet drugs as oral anticoagulation. In contrast, the assessment of age showed high inter-rater agreement (Cohen’s Kappa = 0.780; n = 137): there was no difference between the assessment by EMS personnel and emergency neurologists in 107 cases (78.1%), a difference of 1 year in 20 cases (14.6%), and 2–10 years in the remaining 10 cases (7.3%).
The time from symptom onset or the time last seen well to admission showed fair inter-rater agreement between the assessment by EMS personnel and emergency neurologists (Cohen’s Kappa = 0.378). Symptom onset or last seen well was documented in 130/200 cases, both by EMS personnel and emergency neurologists. In 51 cases (25.5%) there was no difference, in 31 cases (15.5%) a difference of 1–30 min, in 18 cases (9.0%) a difference of 31–60 min, and in 32 cases (16.0%) a difference of 61 min to 16 h.
Predictive quality of FAST-ED for LVO
LVO-associated AIS was detected in 34 patients (17.0%). Site of occlusion was internal carotid artery in 7 patients (20.6%), proximal middle cerebral artery (M1) in 9 patients (26.5%), second segment of the middle cerebral artery (M2) in 14 patients (41.2%), and basilar artery in 4 patients (11.8%). CTA was performed for LVO detection in 92/139 (66.2%) patients with AIS and in 43/48 (89.5%) patients with AIS and prehospital FAST-ED score ⩾4. The five patients with AIS with prehospital FAST-ED score ⩾4 showed spontaneous, complete cessation of stroke symptoms upon arrival at hospital and therefore did not receive CTA; none of these patients had LVO emergency Doppler/duplex ultrasound examination.
The FAST-ED score documented by EMS showed strong correlation with the NIHSS score evaluated at hospital arrival (r = 0.755, p < 0.001) as well as with the FAST-ED score calculated based on the NIHSS score at hospital arrival (r = 0.701, p < 0.001). Median FAST-ED score was significantly higher in patients with AIS with LVO [5 (4–6)] and in patients with ICH [5 (4–6)] than in patients with AIS without LVO [2 (1–3)] or stroke mimics [1 (0–3)] (both p < 0.001).
In line with that, the area under ROC (c-statistics) showed very strong prediction of LVO by the prehospital FAST-ED [0.89 (95% CI = 0.85–0.94)]. A FAST-ED cut-off at 4 correctly identified the presence or absence of LVO in 158/200 patients (accuracy = 79.0%). 28/64 (43.8%) patients with FAST-ED score ⩾4 had LVO, compared to 6/136 (4.4%) patients with FAST-ED score < 4 (Table 1, Figures 3 and 4). For a cut-off at 4, exclusion of M2-occlusion from the LVO-definition resulted in lower sensitivity (80.0% versus 82.4%) and specificity (73.3% versus 78.3%; for details see Supplemental Table 1).
True negatives (TN), false negatives (FN), true positives (TP), false positives (FP), sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) of different cut-off values of the FAST-ED scale for the detection of large vessel occlusion (defined as occlusion of the internal carotid artery, first two segments of the middle cerebral artery (M1, M2), and basilar artery).
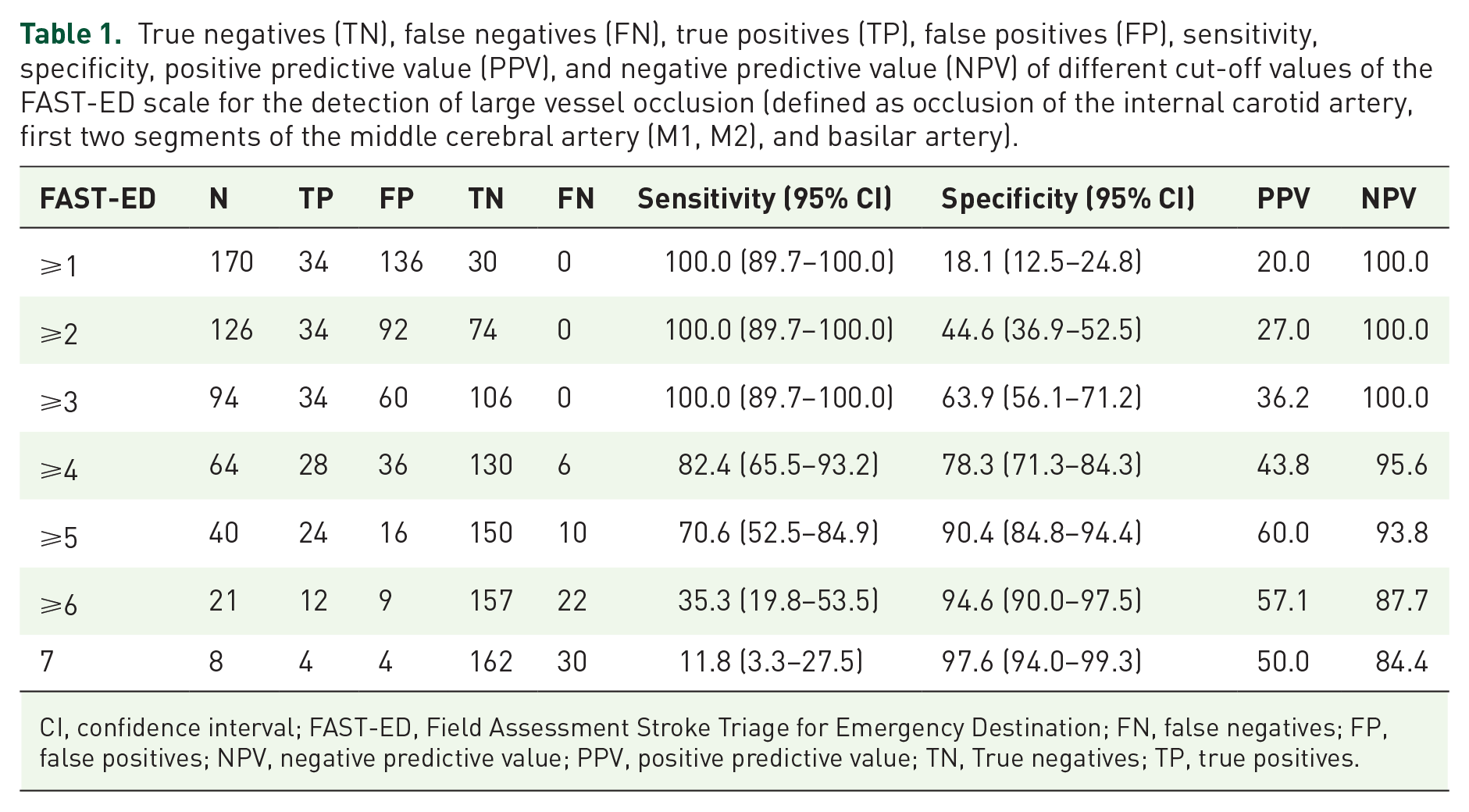
CI, confidence interval; FAST-ED, Field Assessment Stroke Triage for Emergency Destination; FN, false negatives; FP, false positives; NPV, negative predictive value; PPV, positive predictive value; TN, True negatives; TP, true positives.

Sensitivity (squares), specificity (circles), and positive predictive value (triangles) of different cut-off values of the FAST-ED scale for the detection of large vessel occlusion.

Distribution of acute ischemic stroke with large vessel occlusion (AIS with LVO; black), without LVO (AIS without LVO; gray), intracranial hemorrhage (ICH; dashed), and stroke mimics (mimic; white) across FAST-ED scores.
The various reasons for misclassification of patients when FAST-ED was used are presented in Figure 2. False positives were caused by the entry of neglect or speech disturbances, although no cortical involvement was detectable during imaging (n = 5), spontaneous recanalization of LVO because clinical symptoms resolved during transport (n = 4), pre-existing dementia with impaired speech comprehension/production (n = 4), or residual stroke symptoms (n = 4). In addition, 9/12 patients with ICH and 7/42 stroke mimics reached a FAST-ED of ⩾4. On the contrary, false-negative AIS-associated LVOs (6/34) were explained by very good collateral blood supply according to computed tomography perfusion imaging, occlusion in the M2-segment, or occlusion of their basilar artery (each n = 2).
Predictive value of FAST-ED for EVT
Overall, 19/200 patients underwent EVT. FAST-ED revealed very good prediction of EVT with an area under ROC (c-statistics) of 0.82 (95% CI = 0.74–0.89). A FAST-ED cut-off at 4 correctly classified 149/200 (74.5%) patients for EVT. 16/64 (25.0%) patients with FAST-ED score ⩾4 received EVT, compared to 3/136 (2.2%) patients with FAST-ED score < 4. As a result, sensitivity was 84.2% (95% CI = 60.4–96.6), specificity was 73.5% (95% CI = 66.4–79.8), positive predictive value was 25.0% (95% CI = 19.6–31.3), and negative predictive value was 97.8% (95% CI = 94.0–99.2).
Hypothetical prehospital stroke triage model
On the basis of our data, we calculated the expected benefit of prehospital stroke triage in an urban setting with multiple stroke units in the surrounding area. Thus, differences in transportation time to CSCs and stroke units without the possibility for EVT and neurosurgical care (referral centers) were considered negligible. In our sample, median transportation time from emergency site to hospital was 7.8 min (IQR = 5.9–11.6). For this model, we assumed that stroke symptoms started in the catchment area of a referral center. We compared the following two scenarios: (A) All patients with acute stroke symptoms would be transferred to a referral center without considering initial FAST-ED score. Patients with LVO and patients with major ICH (defined as ICH and FAST-ED ⩾4) would be secondarily transferred to a CSC. (B) Patients with FAST-ED ⩾4 would be directly transported to the CSC and only patients with a FAST-ED < 4 would be admitted to the referral center. In this model, only patients with LVO and FAST-ED < 4 would be secondarily transferred to a CSC.
In scenario A, 43/200 patients (21.5%) would require secondary transportation (ICH = 9 and AIS with LVO = 34). In scenario B, 64/200 (32.0%) patients would be directly transported to CSC of whom 28/200 patients with LVO (14.0%) could receive EVT immediately. Among the remaining 36 non-LVO cases transported to the CSC, 9 major ICH should be expected. Only 6/200 patients (3.0%) would require secondary transportation, resulting in an absolute risk reduction of 18.5% for secondary transportation. Thus, the number needed to screen in order to prevent secondary transportation would be 5.
Discussion
We describe the first results of a smartphone app-driven prehospital FAST-ED examination of patients with suspected stroke by EMS personnel. In a novel approach, we successfully combined the smartphone app-guided assessment with a medical smartphone messaging service. This concept allows digital transmission of the assessment results by EMS personnel before arrival at the hospital. Since the results of the prehospital assessment are sent to a group chat involving stroke physicians and neurointerventionalists, the simultaneous alarming of the stroke team is assured.
The implementation of the FAST-ED scale in a real-world urban setting showed very good accuracy in identifying stroke patients with LVO (sensitivity 82% and specificity 78%) with an AUC of 0.89. Our results are in line with prior validation studies, in which FAST-ED scores were calculated based on NIHSS results on admission (0.64–0.91).4,5,9 In contrast to these prior retrospective validation studies of FAST-ED, the examination of neglect and denial was omitted, if the patient had either aphasia or in the absence of arm weakness. Despite faster assessment, the predictive accuracy remained consistent. Together with the already in-field-validated Rapid Arterial Occlusion Evaluation (RACE) scale, test quality of the app-based FAST-ED is highest among LVO recognition scores. 5 In a large prehospital EMS validation study, the RACE scale detected LVO at a cut-off ⩾5 with a sensitivity of 84% and specificity of 60% (AUC = 0.77). 7 Specificity of the FAST-ED in our study might have been higher than that of the RACE scale, as it includes less items to test motor function (i.e. leg movement), which are less specific for LVO than cortical signs. For the purpose of prehospital triage scores, a certain amount of false-positive results is acceptable though, as a relevant proportion of non-LVO patients with FAST-ED ⩾4 are attributed to ICH (25% in our sample). These patients are known to benefit from direct transport to a CSC, as they are more likely to receive neurosurgical and endovascular treatments. 10
In our study, sensitivity to identify patients for EVT was satisfying. FAST-ED ⩾4 correctly identified 84% of the patients who finally received EVT and thus was comparable with prior EMS validation studies for LVO recognition scores.7,9
As in this non-randomized prospective study the impact on prehospital patient pathways could not be tested directly, a hypothetical prehospital stroke triage model is presented. Under the assumption that LVO-associated stroke and major ICH would require special care of a CSC, prehospital stroke triage with FAST-ED and a cut-off at 4 could decrease the rate of secondary transportation from 21.5% to 3.0%. Every fifth patient that is screened would avoid one unnecessary secondary transport to a CSC (number needed to screen).
While the recently completed randomized Spanish RACECAT trial (NCT02795962) and the ongoing Danish TRIAGE-STROKE trial (NCT03542188) have aimed to investigate the potential benefit of directing EVT candidates directly to CSCs in rural areas, prehospital stroke triage in areas with a high density of stroke units and CSCs has been neglected so far. In contrast to rural areas with an inherent risk of prolonged treatment delay for IVT by bypassing smaller stroke units, in urban areas like the German Ruhr area (Ruhrgebiet, Germany), the additional transportation time for direct transport to the CSCs is negligible: In the city of Essen, the mean transportation time for patients with AIS from the emergency site to the hospital is markedly below 10 min, and CSCs and referral centers are usually located in the immediate vicinity. Data from a prior prospective observational study from our region showed a median difference of 83 min from symptom onset to groin puncture for direct transfer to CSC for EVT in comparison to admission to a referral center and subsequent secondary transportation. 11 However, the relatively high number of stroke patients in urban areas should be taken into account, so that the redirection of patients with AIS is associated with the risk of unnecessary flooding of CSCs. In our model, the rate of patients without LVO-associated AIS or ICH redirected to CSC was acceptably low with a rate of 13.5%.
Limitations of our analysis are related to the non-randomized design of this single-center observational study and might reduce the generalizability of our data. Even though we assessed the test quality of FAST-ED in a patient cohort of a university CSC, at the time of trial conduct patients were transported to our hospital based on the site of emergency and not based on the level of stroke severity. Finally, our data are not able to give information about a possible improvement in time metrics or clinical outcome. Thus, a randomized trial is necessary to evaluate the clinical and economic impact of prehospital stroke triage in our urban setting.
Conclusion
These first results of the use of a smartphone app-guided stroke triage by EMS personnel showed good quality of the FAST-ED to identify patients with LVO. In addition, we could prove the feasibility for the use of a medical messaging service in prehospital stroke care. On this basis, a randomized trial evaluating the clinical benefit of triage performed by EMS personnel in patients with suspected acute stroke is in preparation.
Supplemental Material
sj-docx-1-tan-10.1177_17562864211054962 – Supplemental material for FAST-ED scale smartphone app-based prediction of large vessel occlusion in suspected stroke by emergency medical service
Supplemental material, sj-docx-1-tan-10.1177_17562864211054962 for FAST-ED scale smartphone app-based prediction of large vessel occlusion in suspected stroke by emergency medical service by Benedikt Frank, Thomas Lembeck, Nina Toppe, Bastian Brune, Bessime Bozkurt, Cornelius Deuschl, Raul G. Nogueira, Marcel Dudda, Joachim Risse, Clemens Kill, Michael Forsting, Christoph Kleinschnitz and Martin Köhrmann in Therapeutic Advances in Neurological Disorders
Footnotes
Author contributions
BF and MK conceived the original idea, supervised the project, performed the analysis, and drafted the manuscript with input from all authors; all authors had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. All authors provided critical feedback and helped shape the research, analysis, and manuscript.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: No authors received any payments for work on the submitted manuscript. B.F. and M.K. report modest fees for advisory roles with Allm Inc. R.G.N. reports consulting fees for advisory roles with Anaconda, Biogen, Cerenovus, Genentech, Imperative Care, Medtronic, Phenox, Prolong Pharmaceuticals, and Stryker Neurovascular and stock options for advisory roles with Astrocyte, Brainomix, Cerebrotech, Ceretrieve, Corindus Vascular Robotics, Vesalio, Viz-AI, and Perfuze. The other authors refer no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by an unrestricted research grant by Allm, Inc. The company had no influence on the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Ethics statement
The study protocol was reviewed and approved by the ethics committee of the medical faculty of the University Duisburg-Essen (approval number 18-8162-BO) and data protection authority of the University Hospital Essen. The study was performed in accordance with the principles of Helsinki Declaration. The need for written informed consent was waived by the ethics committee.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
