Abstract
Background and Purpose:
Large vessel occlusion (LVO) recognition scales were developed to identify patients with LVO-related acute ischemic stroke (AIS) on the scene of emergency. Thus, they may enable direct transport to a comprehensive stroke centre (CSC). In this study, we aim to validate a smartphone app-based stroke triage with a shortened form of the Field Assessment Stroke Triage for Emergency Destination (FAST-ED).
Methods:
This retrospective validation study included 2815 patients with confirmed acute stroke and suspected acute stroke but final diagnosis other than stroke (stroke mimics) who were admitted by emergency medical service (EMS) to the CSC of the Neurological University Hospital Essen, Germany. We analysed the predictive accuracy of a shortened digital app-based FAST-ED ( ‘FAST-ED App’) for LVO-related AIS and yield comparison to various other LVO recognition scales.
Results:
The shortened FAST-ED App had comparable test quality (Area under ROC = 0.887) to predict LVO-related AIS to the original FAST-ED (0.889) and RACE (0.883) and was superior to Cincinnati Prehospital Stroke Severity (CPSS), 3-Item Stroke Scale (3-ISS) and National Institute of Health Stroke Scale (NIHSS). A FAST-ED App ⩾ 4 revealed very good accuracy to detect LVO related AIS (sensitivity of 77% and a specificity 87%) with an area under the curve c-statistics of 0.89 (95% CI: 0.87–0.90). In a hypothetical triage model, the number needed to screen in order to avoid one secondary transportation in an urban setting would be five.
Conclusion:
This validation study of a shortened FAST-ED assessment for a smartphone-app guided stroke triage yields good quality to identify patients with LVO.
Introduction
Rapid treatment of patients with acute ischemic stroke (AIS) is crucial to improve functional outcome. While intravenous thrombolysis (IVT) is available in a large number of hospitals, Comprehensive Stroke Centres (CSCs) providing endovascular treatment (EVT) for patients with large vessel occlusion (LVO) are limited.
At present, the pre-hospital identification of LVO-related ischemic stroke and direct transport of those patients to a CSC is an untapped opportunity to shorten time to revascularization and has the potential to improve outcome. Secondary interhospital transfer to CSC after start of IVT (drip-and-ship) is known to delay treatment initiation of EVT. 1 Even though the other also important component of recanalization treatment, the IVT,2–5 can be administered without delay, the drip-and-ship concept is associated with less favourable outcome in EVT-patients. 1 In addition, direct transport would reduce the overall number of secondary interhospital transfers, thus saving precious resources in the prehospital ambulance service. 6
To optimize allocation for patients with suspected stroke, several LVO recognition scales that easily can be used by Emergency Medical Services (EMS) have been proposed. Among these, the Rapid Arterial Occlusion Evaluation (RACE) and the Field Assessment Stroke Triage for Emergency Destination (FAST-ED) have shown to be superior to other LVO recognition scales. 7 For both scales smartphone app versions are available. 8 This provides the opportunity to digitally transmit the results to the target hospital stroke team prior to hospital admission. However, the algorithm of the app based FAST-ED differs from the validated FAST-ED version, as the app interactively adjusts the assessment so that neglect and denial are only evaluated, if the patient has arm-weakness and no disturbance in speech comprehension. Thus, app version of FAST-ED is slightly shorter and ranges from 0 to 8 possible points instead of 0 to 9 points. In this study, we aim to validate the app version of FAST-ED and compare test quality with available LVO recognition scales.
Methods
Patient selection
This was a retrospective validation study for LVO-recognition scores in patients with suspected acute stroke. We included adult stroke patients directly admitted by EMS to the CSC of the Neurological University Hospital Essen from January 2017 to March 2020. We excluded patients with final diagnosis of ischemic stroke who received no vascular diagnostics and excluded all patients with witnessed onset of symptoms more than 24 h before admission.
Patients with acute stroke are prospectively collected in a local stroke registry database. The core data set is entered by the attending physician on admission and completed during hospitalization. Data for this analysis were validated by a senior stroke neurologist (BF).
Patients with EMS-suspected stroke but final diagnosis other than stroke (i.e. stroke mimics) were identified and added to the analysis. For this purpose, we analysed all EMS protocols of patients admitted to our stroke centre during this period.
Ethics statement
The study protocol was reviewed and approved by the ethics committee of the medical faculty of the University Duisburg-Essen (approval number 18-8162-BO) and data protection authority of the University Hospital Essen. The study was performed in accordance with the principles of Helsinki Declaration. The need for written informed consent was waived by the ethics committee.
LVO recognition scales
In our emergency department, the stroke neurologist assesses and records the NIHSS in every stroke patient. In most patients with stroke mimics, the NIHSS needed to be scored retrospectively with a reliable algorithm 9 from physical examination records by a senior stroke neurologist (BB). The NIHSS was used for calculating the following LVO recognition scales as described previously:10,11 Field Assessment Stroke Triage for Emergency Destination (FAST-ED), Rapid Arterial Occlusion Evaluation (RACE), Cincinnati Prehospital Stroke Severity (CPSS), and 3-Item Stroke Scale (3-ISS). A feature of the app-based assessment of FAST-ED is to omit examination of neglect and denial, if either the patient has aphasia or no arm weakness 8 (Figure 1). Thus, we calculated a modified FAST-ED, in this work called ‘FAST-ED App’, ranging from zero to eight points.
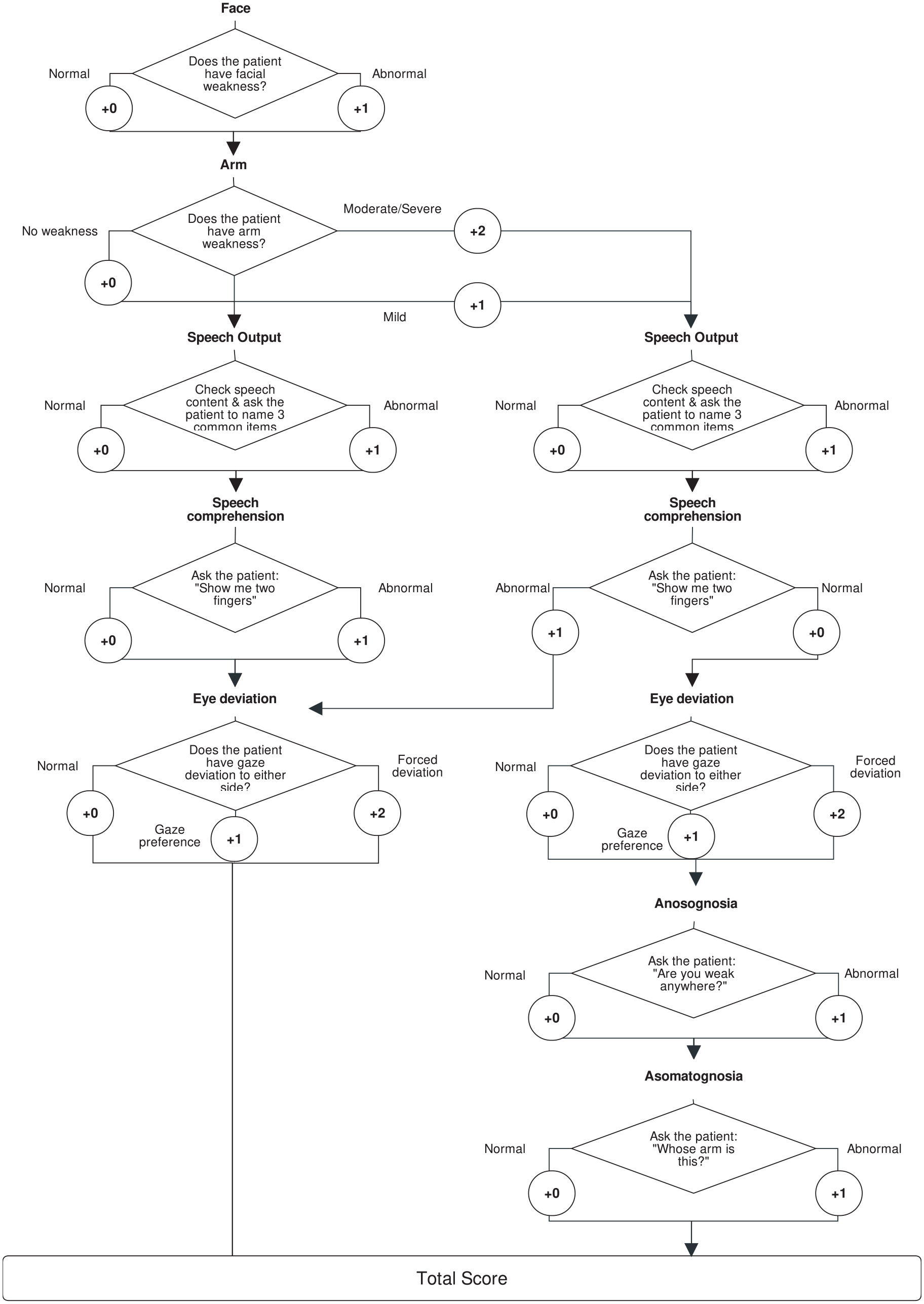
Flow chart of the FAST-ED App algorithm.
Definition of LVO, hypothetical prehospital stroke triage model and statistical analysis
We analysed the predictive accuracy of different LVO recognition scales for LVO-related acute ischemic stroke (AIS) in patients with suspected stroke. LVO was defined as symptomatic occlusion of the internal carotid artery, the first segments of the middle cerebral artery (M1, M2), or the basilar artery. Additional analysis was performed using a stricter definition of LVO that excludes the distal middle cerebral artery segments (M2). Overall, patients were grouped into four categories: AIS with LVO, AIS without LVO, intracranial haemorrhage (ICH) and stroke mimics. The detection of LVO in patients with AIS was performed by computed tomography angiography (CTA), which is the standard emergency diagnostic tool in our centre for AIS patients eligible for reperfusion treatment. In AIS patients clinically not eligible for IVT or EVT (e.g. due to non-disabling symptoms or large infarction on non-contrast CT), and who did not receive CTA on admission, emergency transcranial doppler (TCD) ultrasound was used to detect LVO. 12
The predictive accuracy of each LVO recognition scale to identify LVO-related AIS was evaluated with receiver operating curve (ROC) analysis and the area under ROC (c-statistics). Values are presented together with the respective 95% confidence intervals (CI). Difference of area under ROC of LVO recognition scales was compared with DeLong test. Predictive quality to identify LVO-related AIS and patients receiving EVT was calculated. Prespecified published thresholds of all LVO recognition scales were used for comparison. 13
We present a hypothetical prehospital stroke triage model calculating from our data the expected benefit of prehospital stroke triage for an urban region with multiple nearby stroke units. Thus, differences in transportation time to CSCs and stroke units without the possibility for EVT and neurosurgical care (PSC, resp. referral centres) would be negligible. For this model, we assume that stroke symptoms started in the catchment area of a referral centre. We assume that patients with LVO and patients with severe ICH (defined for this model as ICH with FAST-ED App ⩾ 4) would require treatment in a CSC. Based on the nature and distribution of the data, descriptive statistics are presented as numbers and percent, median with interquartile range or mean with standard deviation. Chi-square tests were used for categorical variables, and based on the distribution of the data t-tests or Mann–Whitney-U tests were used for continuous variables. Statistical analyses were performed with SPSS (version 25.0; IBM Inc) and SAS 9.2 (SAS Institute Inc) and a 2-sided p < 0.05 was considered the minimal level of statistical significance.
Results
Data of 3359 patients directly admitted by EMS with stroke or stroke mimic were available. According to predefined inclusion- and exclusion criteria, we excluded 60 patients without vascular evaluation and 484 patients with witnessed onset of symptoms more than 24 h before admission. A total of 2815 patients were included into the analyses. Final discharge diagnosis was AIS in 2038 patients (72.4%), ICH in 350 patients (12.4%) and 427 patients (15.2%) had stroke mimics.
LVO was diagnosed in 442 patients with AIS. Site of occlusion was internal carotid artery in 136 patients (30.8%), first segment of the middle cerebral artery (M1) in 149 patients (33.7%), second segment of the middle cerebral artery (M2) in 108 patients (24.4%), and basilar artery in 49 patients (11.1%).
The FAST-ED App scale (Area under ROC = 0.887) had comparable test quality to predict LVO-related AIS to the original FAST-ED (0.889) and RACE (0.883) and was superior to the NIHSS, CPSS, and 3-ISS (compare Figure 2(a)). Similar results were derived for the more strict definition of LVO (excluding M2-occlusions; Figure 2(b)). Detailed test quality parameters for each LVO recognition scale are presented in the online supplement. Comparison of established thresholds for the different scales showed highest accuracy for patients with FAST-ED App score of ⩾ 4 (85.5%; Table 1).
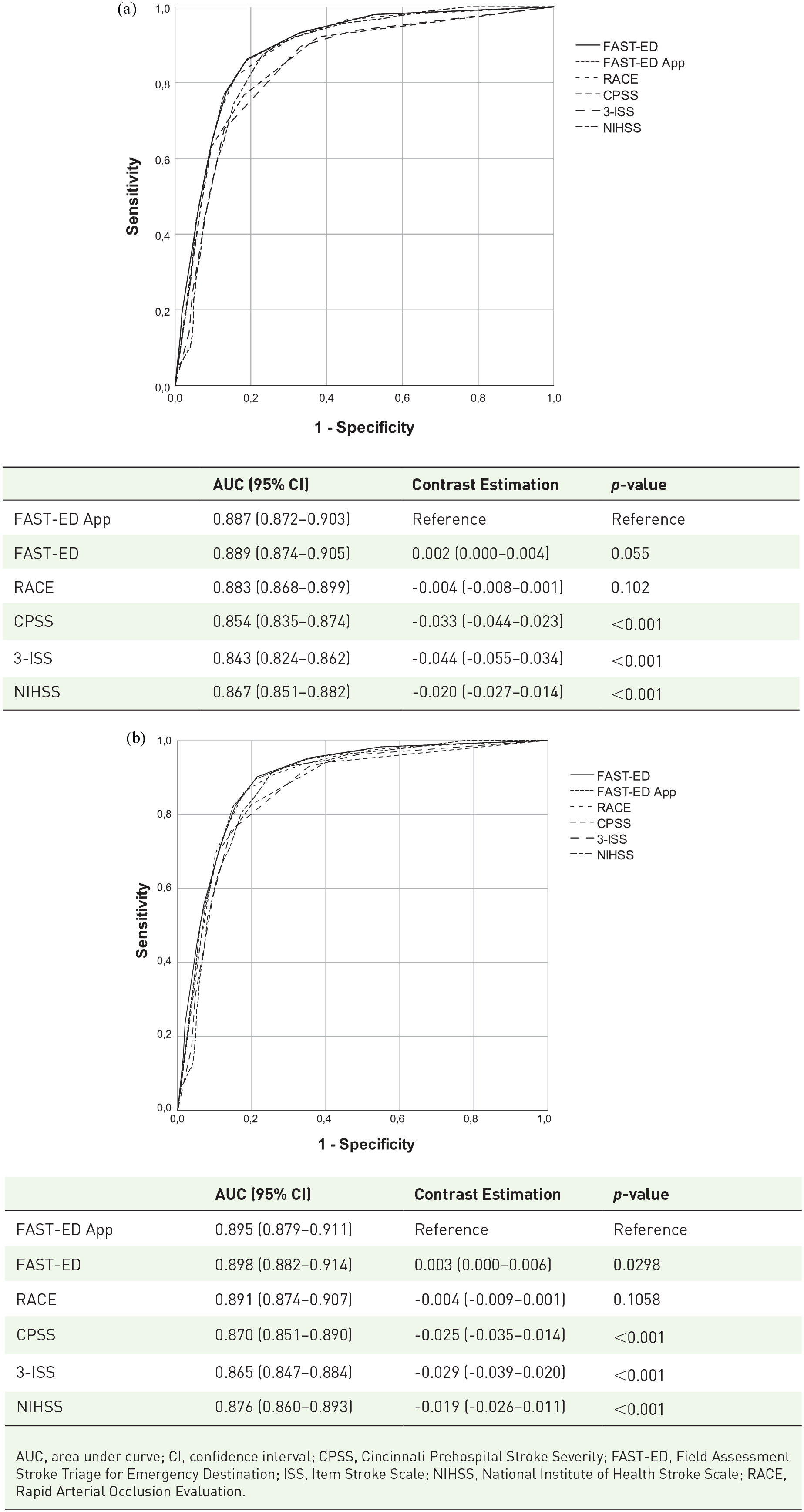
Receiver operating characteristic curve comparing the discrimination of FAST-ED, Field Assessment Stroke Triage for Emergency Destination; RACE, Rapid Arterial Occlusion Evaluation; CPSS, Cincinnati Prehospital Stroke Severity Scale; 3-ISS, 3-Item Stroke Scale; NIHSS, National Institutes of Health Stroke Scale for the detection of large vessel occlusion (LVO) related ischemic strokes. LVO defined as (a) as occlusion of the internal carotid artery, first (M1) and second (M2) segment of the middle cerebral artery, and basilar artery; (b) excluding the second segment of the middle cerebral artery (M2). AUC indicates area under the curve. p values presented refer to the contrast estimation comparing each scale with the FAST-ED App. All individual curves presented a p value < 0.001.
Comparison of test parameters of LVO recognition scales at different thresholds.
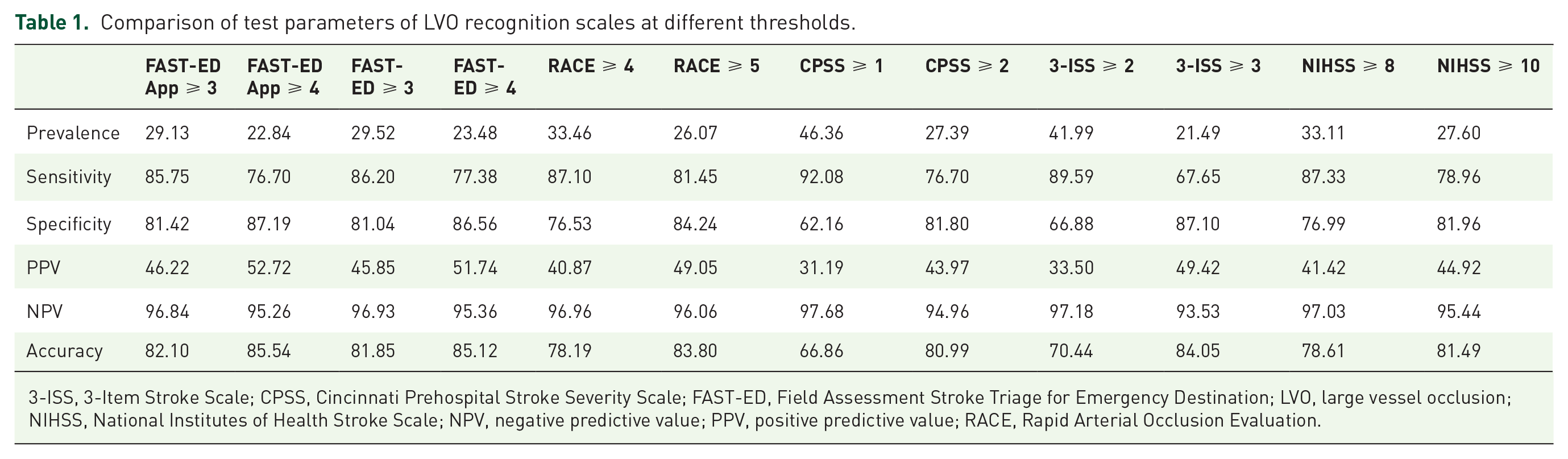
3-ISS, 3-Item Stroke Scale; CPSS, Cincinnati Prehospital Stroke Severity Scale; FAST-ED, Field Assessment Stroke Triage for Emergency Destination; LVO, large vessel occlusion; NIHSS, National Institutes of Health Stroke Scale; NPV, negative predictive value; PPV, positive predictive value; RACE, Rapid Arterial Occlusion Evaluation.
A FAST-ED App score of ⩾ 4 correctly identified 76.7% of AIS with LVO. 6.5% of AIS without LVO had a score of ⩾ 4, as well as 3.5% of patients with stroke mimic, and 52.9% of patients with ICH (3.7%, 0.5%, and 6.6% of the cohort). The latter are later referred to as ‘severe ICH’. The distribution of the different diagnoses across the FAST-ED App score is shown in Figure 3.
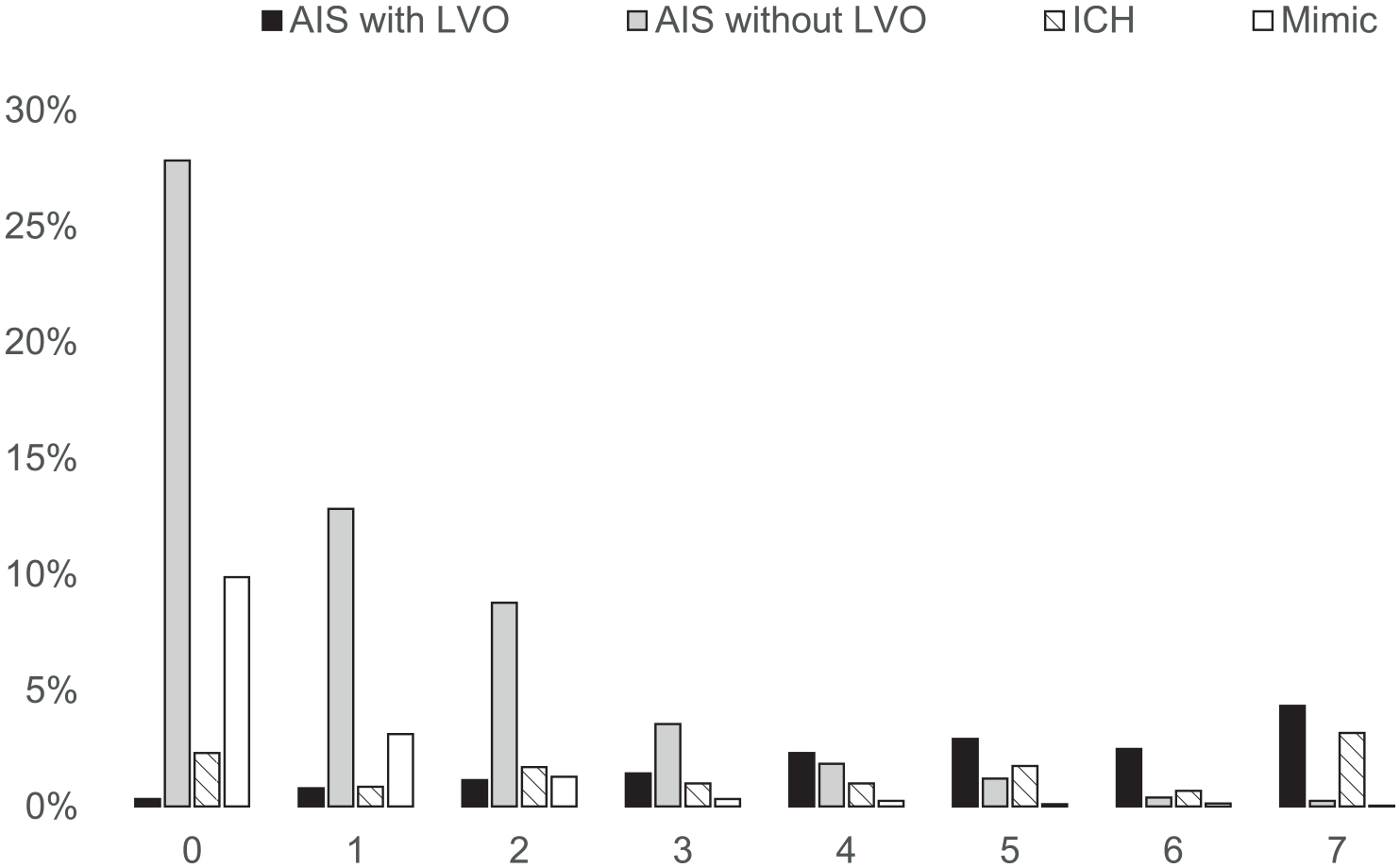
Distribution of acute ischemic stroke with large vessel occlusion (AIS with LVO; black), without LVO (AIS without LVO; grey), intracranial haemorrhage (ICH; dashed), and stroke mimics (Mimic; white) across FAST-ED App scores.
While the likelihood of ICA and M1 occlusion increased with higher scores of FAST-ED App, occlusion of the M2 segment showed a peak at a score of 5 and occlusions of the basilar artery were nearly equally distributed across the full range (compare Figure 4).

Distribution of most proximal site of large vessel occlusion across FAST-ED App scores. Internal carotid artery (ICA, black), first segment of the middle cerebral artery (M1; grey), first segment of the middle cerebral artery (M2; dashed), and basilar artery (BA; white).
By using the shortened FAST-ED App algorithm, the assessment of neglect and denial could be omitted to calculate the score in 2163 of the 2815 cases (76.8%). In 17 of these cases, the FAST-ED App score was 3 while having an original version FAST-ED score of 4: 3/439 cases with LVO-related AIS (0.7%), 10/1586 cases with AIS without LVO (0.6%), and 4/346 cases with ICH (1.1%). The 3 LVO-related AIS had occlusion of M2 (n = 2) and ICA (n = 1).
Hypothetical prehospital stroke triage model
In Table 2, we compare the following scenarios: (A) All patients with acute stroke symptoms are transferred to the referral centre first disregarding initial FAST-ED App score. Patients with LVO and patients with severe ICH (defined as ICH with FAST-ED App ⩾ 4) would receive secondary transportation to a CSC. (B) Patients with FAST-ED App ⩾ 4 are directly transferred to CSC and only those with a FAST-ED App < 4 are admitted to the referral centre. Only patients with LVO and FAST-ED App < 4 would require secondary transportation to a CSC.
Hypothetical unselected and selective admission of 100 stroke cases with and without large vessel occlusion (LVO) to primary or comprehensive stroke centres (PSC, CSC). Projected number of severe intracranial haemorrhage (ICH) is given in brackets.

CSC (first), primary admission to comprehensive stroke centre; CSC (second), secondary transportation to comprehensive stroke centre; FAST-ED, Field Assessment Stroke Triage for Emergency Destination; LVO, large vessel occlusion; PSC, primary stroke centre.
Numbers are given per 100 patients in this model: In scenario A, 23 patients would require secondary transportation (ICH = 7 and AIS with LVO = 16). In scenario B, only 4 patients would require secondary transportation. In all, 23 patients would be directly transported to CSC of whom 12 patients with LVO could immediately receive EVT. Among the remaining 11 non-LVO cases directly transported to the CSC, 7 severe ICH should be expected. The risk of secondary transportation would thus be reduced to 17% at a price of 4 patients being unnecessarily transported to the CSC without LVO or severe ICH. The number needed to screen to prevent one secondary transportation would be 5 in this scenario.
Discussion
This is the first validation study of a shortened FAST-ED assessment for smartphone-app guided prehospital stroke triage. The app-version of the FAST-ED scale in an urban setting showed very good accuracy in identifying stroke patients with LVO (sensitivity 77% and specificity 87%). FAST-ED App ⩾ 4 correctly identified 86% of patients for the presence of LVO-related AIS. Our results are in line with prior validation studies, assessing the full version of FAST-ED calculated from NIHSS at admission.10,14
The sequence for FAST-ED App omits the assessment of denial and neglect in patients who are incapable to understand the questions of these items. In addition, they are omitted if the patients do not have arm-paresis. This allowed a quicker assessment in more than ¾ of our sample and missed only 0.7% of LVO- related AIS. As additionally false positives were prevented, the prediction accuracy of FAST-ED App remained consistent in comparison to the original FAST-ED.
An advantage of our analysis is the well-documented large sample of consecutively admitted patients with suspected stroke to a large emergency department in an urban area. By inclusion of patients with ICH and stroke mimics in our analysis, we emulated a real-world urban scenario predicting the number needed to screen to recognize LVO-related AIS or severe ICH by EMS personnel. In this hypothetical prehospital stroke triage model, we assumed that LVO associated stroke and severe ICH would require special care of a CSC. Prehospital stroke triage with FAST-ED and a cut-off at 4 would decrease the rate of secondary transportation from 23% to 4%. Every fifth patient that is screened would avoid one unnecessary secondary transport to a CSC (number needed to screen). Additional transportation times for direct transportation to CSCs in urban areas like our German Ruhr area (Ruhrgebiet, Germany) are usually negligible, as referral centres and CSCs are located close by. In the city of Essen, the mean transportation time for AIS patients from the emergency site to the hospital is markedly below 10 min.
Even though the primary analysis of the recently completed cluster-randomized Spanish RACECAT trial (NCT02795962) failed to show clinical benefit when directing EVT candidates directly to CSCs in a rural area, secondary analysis indicated potential benefit for the subgroup of patients with transportation times of less than 60 min. The time-dependency of the effect is supported by results of a recently published model suggesting to transport patients with LVO-related AIS to a CSC if the additional delay-to-IVT is below 30 min in urban and 50 min in rural settings. 15
The limitations of a retrospective analysis are well known and will not be listed in detail. Our sample comes from an emergency admission of a single university CSC, but in the city of Essen patients as per local standard are mainly transported to our hospital based on the site of emergency in the city and not based on level of stroke severity. Even though it has been shown that scoring of EMS personnel and hospital stroke physicians does not substantially differ,12,16 it should be mentioned that patients could improve or deteriorate during transportation to the emergency department resulting in different results on LVO recognition scales. Most EMS-suspected stroke with final diagnosis other than stroke (i.e. stroke mimics) were never considered as strokes upon first evaluation in the emergency admission by the stroke neurologist in charge. Therefore, NIHSS scoring was not performed by the stroke neurologist upon admission and needed to be retrospectively calculated according to a previously validated algorithm 9 for this analysis.
Thus, our next step is a prospective in-field validation study for the smartphone-app guided assessment of patients with suspected stroke by EMS personnel using FAST-ED. This study started in March 2019 and has been registered as NCT04404504 at ClinicalTrials.gov.
Conclusion
These first results of a retrospective validation study for EMS stroke-triage showed equal quality for a shorter version of FAST-ED to identify patients with LVO. This encourages for further evaluation of this score in a prehospital prospective setting.
Supplemental Material
sj-docx-1-tan-10.1177_17562864211057639 – Supplemental material for Validation of a shortened FAST-ED algorithm for smartphone app guided stroke triage
Supplemental material, sj-docx-1-tan-10.1177_17562864211057639 for Validation of a shortened FAST-ED algorithm for smartphone app guided stroke triage by Benedikt Frank, Felix Fabian, Bastian Brune, Bessime Bozkurt, Cornelius Deuschl, Raul G. Nogueira, Christoph Kleinschnitz and Martin Köhrmann in Therapeutic Advances in Neurological Disorders
Footnotes
Author contributions
B.F. and M.K. conceived the original idea, supervised the project, performed the analysis and drafted the manuscript with input from all authors; all authors had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. All authors provided critical feedback and helped shape the research, analysis and manuscript.
Conflict of interest statement
No authors received any payments for work on the submitted manuscript. BF and MK report modest fees for advisory roles with Allm Inc. RGN reports consulting fees for advisory roles with Anaconda, Biogen, Cerenovus, Genentech, Imperative Care, Medtronic, Phenox, Prolong Pharmaceuticals, Stryker Neurovascular and stock options for advisory roles with Astrocyte, Brainomix, Cerebrotech, Ceretrieve, Corindus Vascular Robotics, Vesalio, Viz-AI, and Perfuze. The other authors refer no conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
