Abstract
Objective
To review the characteristics and outcomes of pediatric patients on a ketogenic diet (KD), an established treatment option for individuals with intractable epilepsy, in a tertiary epilepsy center.
Methods
This retrospective study included pediatric patients diagnosed with intractable epilepsy who had experienced no benefits from at least two appropriately chosen antiseizure medications. All patients were hospitalized, started a KD without fasting, and were observed for complications and tolerance. The etiology of epilepsy, side effects, and KD efficacy on seizure outcomes were also examined.
Results
Of 16 children included in the study, nine (56%) experienced significant seizure improvement, with three becoming seizure-free during the KD. Ten patients were fed orally, and six were fed through gastrostomy feeding tubes. Most were on a 3:1 ratio, and nine reached ketosis within the first three days of KD initiation. Initial recurrent hypoglycemia was documented in four patients, and four experienced vomiting and acidosis. Most families complied with the diet, and all of the children gained weight during the study period.
Conclusion
Ketogenic diets are an established and effective treatment for childhood epilepsy, with reversible mild adverse effects. A non-fasting KD protocol is a safe and effective option for children with intractable epilepsy.
Keywords
Introduction
Despite the availability of multiple antiseizure medications, approximately one-third of individuals with epilepsy are diagnosed with intractable epilepsy. 1 Ketogenic diets (KDs) are high-fat, adequate-protein, low-carbohydrate diets that mimic the metabolic pathway for fasting, and have been a well-recognized treatment option for epilepsy since 1920. Ketone body creation has been postulated to exert antiseizure effects on certain brain receptors. 2 Fat-to-protein and carbohydrate (grams) ratios ranging between 2.5:1 and 4:1 are commonly used regimens. 3 Moreover, medium-chain triglyceride oil is a major fat source that may be added, which allows for more carbohydrate consumption. Other modifications, including KD-centric modified Atkins diets and low glycemic index treatments, are also available. 4 In many KD centers worldwide, a KD is started with an initial fasting period to stimulate ketosis. 5 However, fasting prior to KD initiation in children may pose nausea, vomiting, anorexia, hypoglycemia, dehydration, and seizure risks. Starting a KD without fasting is an alternative and safe option. 6 Some centers initiate KDs in outpatient settings, though hospitalization to start a KD may provide a better opportunity for educational sessions, monitoring of side effects, and close follow-ups. 7
Few KD studies have been conducted in the Middle East, including Saudi Arabia.8,9 The present study aim was to retrospectively review the characteristics of pediatric patients who were prescribed a non-fasting KD, and KD treatment outcomes, at King Faisal Specialist Hospital and Research Center (KFSH & RC) in Jeddah, Saudi Arabia.
Patients and methods
Study population
This retrospective study included pediatric patients (aged 1–14 years) with intractable epilepsy who were treated with a non-fasting KD, and followed for more than 12 months in clinics at KFSH & RC, Jeddah, Saudi Arabia. Inclusion criteria were: (1) hospital admission for non-fasting KD initiation between June 2013 and July 2018; and (2) a diagnosis of refractory epilepsy (after experiencing no benefits from at least two appropriately selected antiseizure medications). Exclusion criteria were: (1) failure to comply with the KD protocol during diet initiation; (2) swallowing difficulties in children not on feeding tubes; (3) gastroesophageal reflux disease that did not responded to medical management; (4) food allergies manifesting as diarrhea during diet initiation; (5) failure at diet induction; and (6) poor family compliance (e.g., due to social circumstance, parents with difficulties in preparing KD, parents with many children who could not comply with the KD recommendations of the treating team).
The KFSH & RC institutional review board approved this single-center study (IRB No. 2019-18) and the reporting of the study results conforms to STROBE guidelines. 10 Due to the retrospective study design and de-identification of patient data, informed consent from patient’s parents or legal proxies was not required.
Treatment and follow-up
The study included children with intractable epilepsy, aged ≤14 years, who were admitted for KD initiation following a detailed clinical history and examination, and who fulfilled the inclusion criteria. Initial investigations before KD initiation comprised: complete blood count; renal function test and electrolytes (serum sodium, potassium, chloride, urea, creatinine, calcium, magnesium, phosphate and bicarbonate, vitamin D, zinc and selenium); serum liver function test (total protein, albumin, aspartate aminotransferase [AST], alanine transaminase [ALT] and γ-glutamyl transferase [GGT]); fasting lipid profile; serum acylcarnitine profile; anticonvulsant drug levels (if applicable); and metabolic tests (if indicated), including tandem mass spectrometry, urine organic acids, serum amino acids, electroencephalography (EEG), brain magnetic resonance imaging (MRI), genetic work-up (if indicated) and renal ultrasound (annually and if indicated). KD explanations and counseling were started in the pediatric neurology clinic. Clinical dietitians conducted a detailed nutritional assessment in which the patient’s food intake, baseline weight and height, and ideal weight for stature were reviewed. Food records, allergies, and preferences were then reviewed with the family, and the clinical dietitians also explained the purpose, ratio, and composition of the KD.
During admission to the pediatric ward, the KD was initiated without the need for fasting. In infants, regular bottle milk was switched to KetoCal® ketogenic milk (Nutrica®; Macquarie Park, NSW, Australia). Infants were partially weaned from KetoCal® milk, i.e., weaning with ketogenic food according to the KD menu, while keeping KetoCal® milk as part of the daily recipe. Regular blood sugar was monitored daily, and urine ketone levels were monitored every morning. The KD was initiated gradually at a 1:1 ratio (i.e., fat-to-carbohydrate grams, with protein grams rolled into carbohydrate grams) as tolerated. If patients developed lethargy, sweating, jitteriness, or tachycardia-suggestive hypoglycemia (blood glucose level < 2.8 mmol/L), 30 ml of orange juice was administered as a drink, otherwise, 60 ml of 5% dextrose was intravenously injected. Other complications, e.g., intolerance, were managed through slower ketosis introduction. Dextrose-free i.v. fluids were used to manage dehydration. Antiseizure medications were continued using a carbohydrate-free formula along with carbohydrate-free multivitamins, calcium, and vitamin D. Upon discharge, patients were supplied with formulas, food recipes, a glucometer, a weighing scale, and ketone strips.
After discharge, patients were initially followed-up in the clinic after 2–4 weeks. The families were instructed to measure urine ketone levels in the morning, and any urgently needed diet adjustments in ratio or calories were discussed and followed through phone calls. Follow-up investigations were performed on a regular schedule. Complete blood counts, and liver and renal function tests, were conducted. Changes in anthropometric parameters and fasting lipids were also monitored and recorded. Children on the KD received a face-to-face appointment with a pediatric epileptologist and dietitian every three months, to review their progress and monitor their response, and to address their concerns. Every patient was followed for at least 12 months.
Study parameters
The following data were extracted from medical records: demographic data, including patient age and sex; detailed history and examination; side effects; time until any improvement in seizures; category of seizure improvement; electroencephalography (EEG) results; and brain magnetic resonance imaging (MRI) findings. Seizure improvement (change in frequency and duration of seizures versus baseline) was categorized according to the following scale: no improvement; <50% improvement; >50% improvement; and >90% improvement. Data were extracted from regular clinical records obtained every 2–4 weeks. Data regarding time elapsed until improvement and any side effects were also extracted.
Statistical analyses
Data are presented as mean ± SD or n (%) prevalence, and were analyzed using SPSS software, version 21.0 (IBM Corp., Armonk, NY, USA) for Windows. Changes in continuous parameters over time were analyzed using paired Student’s t-test. A P-value <0.05 was viewed as statistically significant.
Results
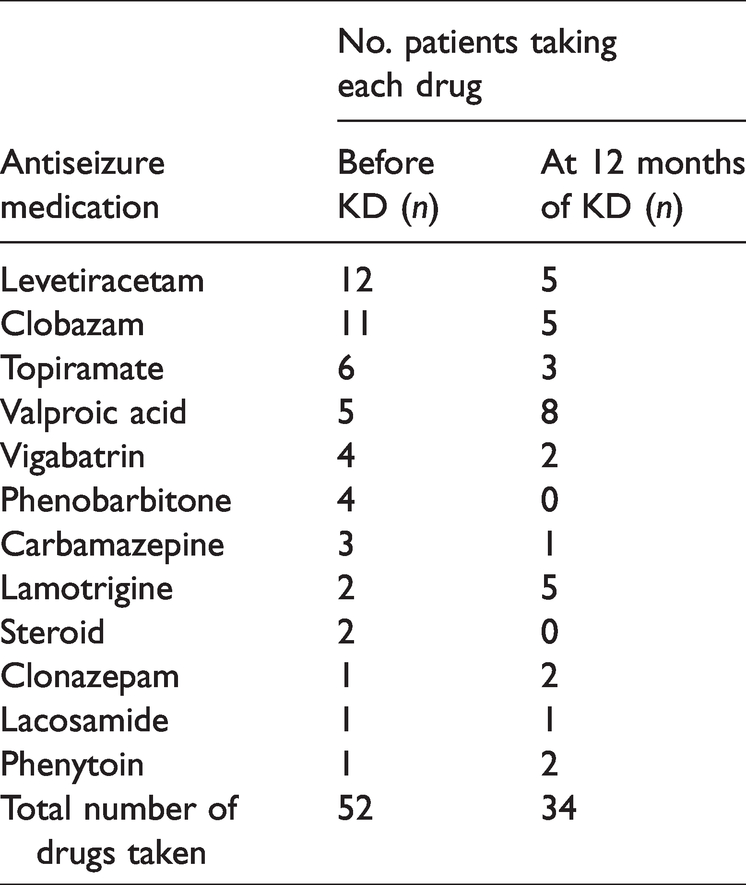
A total of 16 patients were enrolled, comprising eight males and eight females (overall age range, 2–14 years). Fifteen patients had not experienced benefits from at least two appropriately selected and tolerated antiseizure medications, and one patient was diagnosed clinically with GLUT1 deficiency. Clinical and demographic characteristics of the 16 studied patients are summarized in Table 1. The mean number of different antiseizure medications used by each patient was 3.3 before starting the KD, and 2.1 at 12 months of KD treatment. A total of 52 antiseizure medications were used by the whole study population at baseline, and this dropped to a total of 34 medications at 12 months of the KD (Table 2).
Clinical and demographic characteristics of 16 pediatric patients (aged 2–14 years) with intractable epilepsy treated with a ketogenic diet.
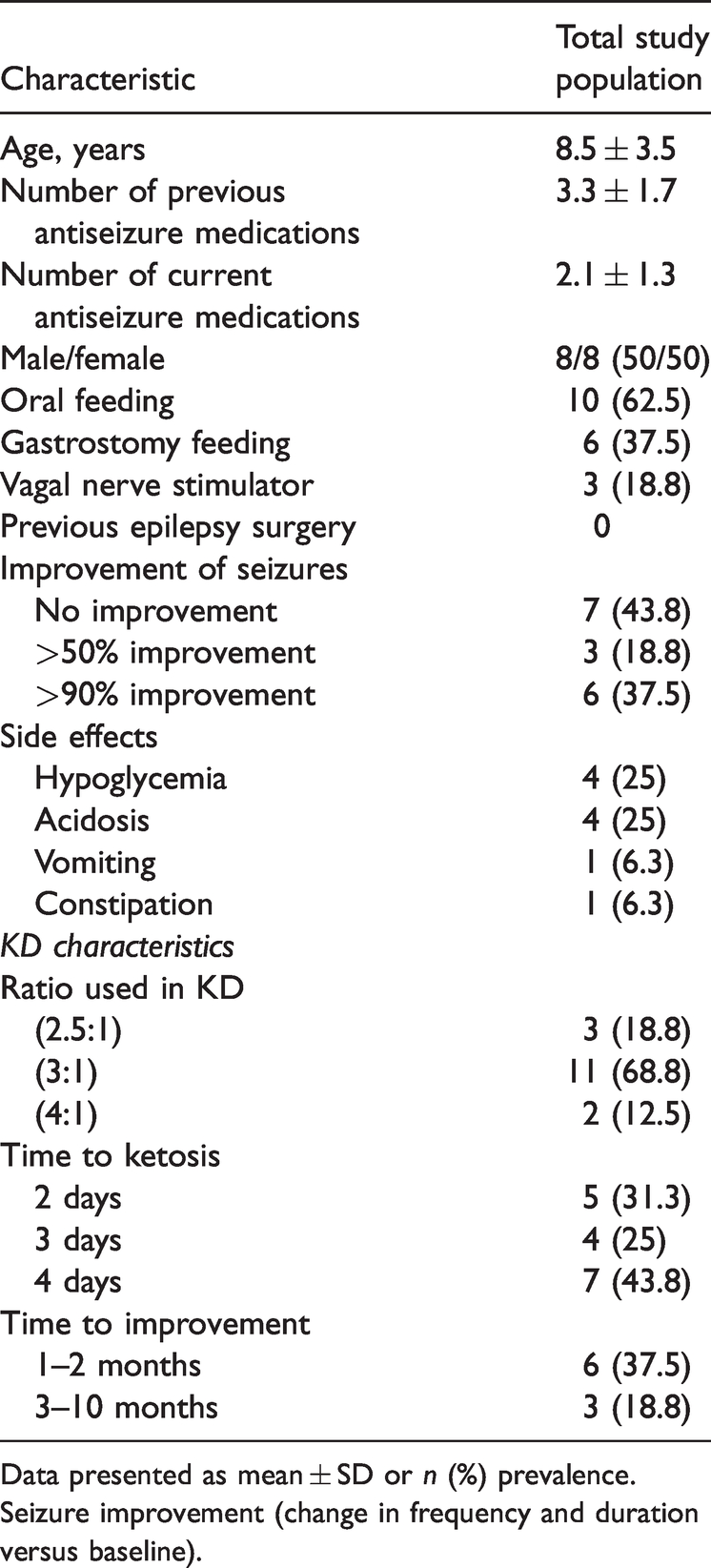
Data presented as mean ± SD or n (%) prevalence. Seizure improvement (change in frequency and duration versus baseline).
Proportion of pediatric patients (aged 2–14 years) with intractable epilepsy taking antiseizure medications prior to, and at 12 months of, treatment with a ketogenic diet (KD).
More than 60% of the cohort (10/16 patients) were consuming the KD by mouth, while the other six patients received the KD via gastrostomy feeding tubes. After starting the KD, nine patients (56%) reported >50% seizure improvement, and out of those, six experienced >90% improvement in seizure frequency, three became seizure-free after the KD, and three of the seven who did not improve had vagal nerve stimulators implanted. Changes in anthropometric parameters and fasting lipids for each patient are shown in Figure 1.
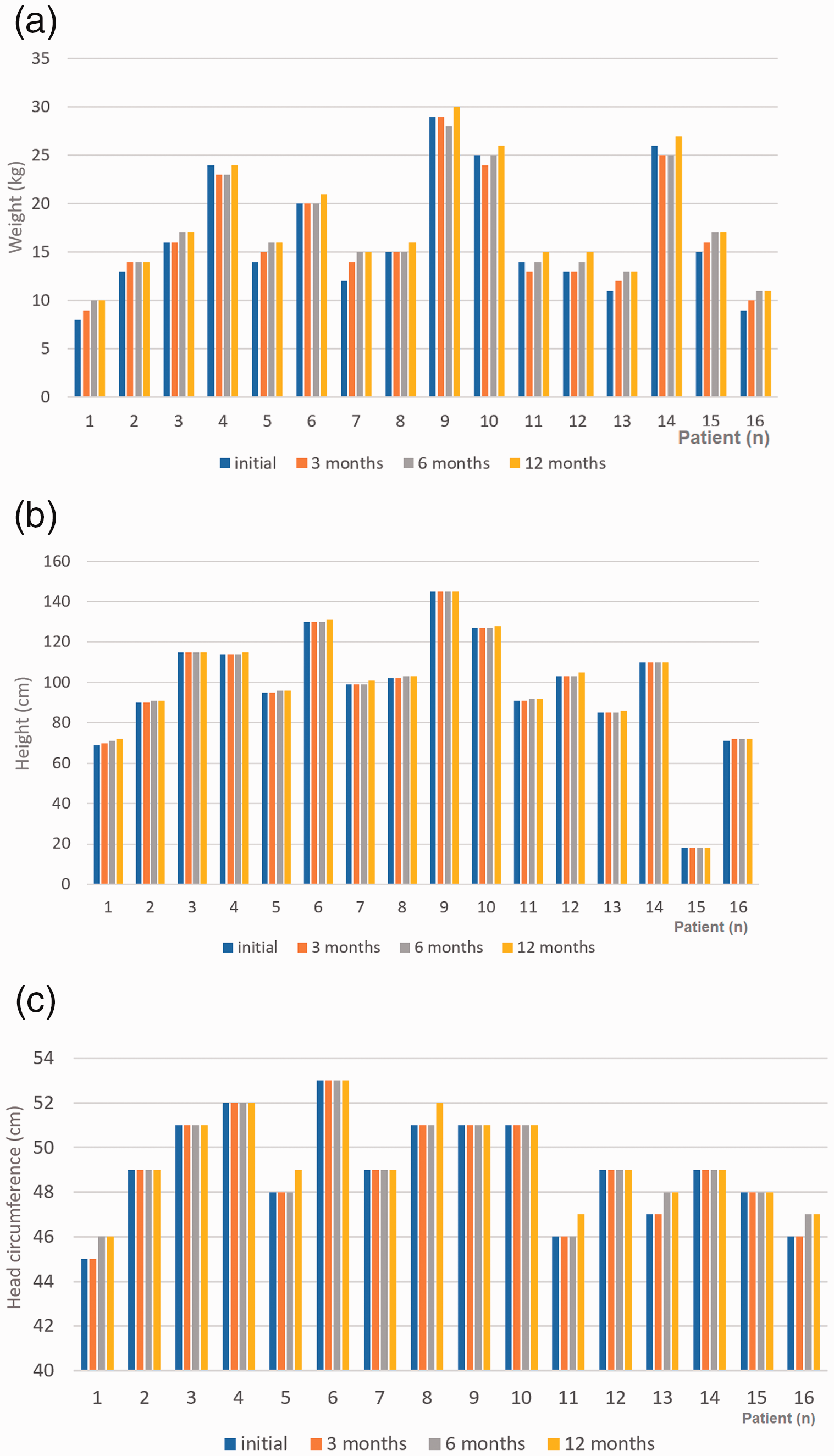
Initial and follow-up anthropometric and clinical data for each of 16 pediatric patients (aged 2–14 years) with intractable epilepsy treated with a ketogenic diet: (a) Weight; (b) height; (c) head circumference; (d) cholesterol; (e) triglycerides; (f) low-density lipoprotein; (g) high-density lipoprotein.

Continued.
A KD ratio of 3:1 was used in 11 children with epilepsy, with the KD introduced slowly to ensure that it was well-tolerated without major complications. Three patients developed ketosis on a 2.5:1 ratio of fat-to-carb and protein grams. The remaining two cases developed urine ketones with a 4:1 ratio. Most of the patients developed ketosis within three days, with the remainder needing up to one week to develop ketosis. Six of the children on the KD improved within the first two months after beginning the diet, with the remaining three patients taking up to 10 months to show meaningful improvement in seizures. Parents also reported improvements in behavior and attention.
The non-fasting KD protocol was associated with a few adverse effects, as four children developed hypoglycemia on one or more occasions. Glucose supply was adjusted as indicated in the treatment protocol to elicit slower initiation of the diet. Mild acidosis was also observed in four patients, and was corrected by lowering the KD ratio and sodium bicarbonate levels if needed. One patient was vomiting initially, then improved, while another had constipation, but experienced relief through laxative use. All patients gained weight upon clinic follow-up, with the gain particularly statistically significant (P < 0.05) soon after diet initiation (Table 3). Height also proportionally increased for age (P < 0.05). Cholesterol and triglyceride levels were higher at 12 months than at the start of KD treatment (P < 0.05). Other lipid parameters did not show statistically significant changes (Table 3).
Changes in anthropometric and laboratory data between study time-points in 16 pediatric patients (aged 2–14 years) with intractable epilepsy treated with a ketogenic diet.

Data presented as mean change (SD change).
Statistically significant change in value (P < 0.05; Paired Student’s t-test).
Four families were not properly compliant with the diet, and their children failed to respond to the KD.
All 16 children showed abnormal generalized epileptiform discharges on their EEGs, and seizure types were classified according to the International League Against Epilepsy. Six patients showed normal results for brain MRI. Various MRI brain abnormalities were observed, such as asphyxia and migrational disorders. Four children registered brain atrophy on their brain MRI scans. Cortical migration malformations caused epilepsy was observed in two patients, and dysgenesis of the corpus collosum was observed in two patients. The youngest patient was 2 years old and was diagnosed with GLUT1 deficiency, while another patient registered Chromosome 9 deletion on a microarray.
Discussion
King Faisal Specialist Hospital and Research Center in Jeddah is one of the few national centers in Saudi Arabia that offers KD for children with epilepsy. This single-center retrospective study reviewed the demographic and clinical data of pediatric patients with intractable epilepsy and the effectiveness of KD on these patients using a non-fasting KD protocol. Overall, the non-fasting KD protocol was reported to be associated with fewer and milder adverse effects compared with a fasting protocol, 11 including among children. 12 Some centers have suggested using fasting protocols to initiate KD due to faster response and ketosis, 13 however, fasting in children is more likely to cause hypoglycemia, lethargy, and acidosis. 14 Furthermore, fewer side effects (e.g., hypoglycemia and gastrointestinal intolerance) were reported among children put on gradual non-fasting KD protocols, 15 and seizure improvement outcomes were reported to be similar. 16 In the present study, minimal and reversible side effects were reported from a non-fasting protocol, in line with other published studies.16–19 Efficacy regarding seizure outcomes and duration until seizures were under control were also similar to previously reported studies.20,21 A study by Van der Louw et al., 7 suggested avoiding fasting, particularly in children younger than 2 years old, due to potential side effects.
Decreasing the number of antiseizure medications is one of the secondary potential benefits of a KD. The medication total prior to starting the KD was 52, compared with 34 antiseizure medications at 12 months of KD use (Table 2). Multiple extant studies reported similar decreases in antiseizure medication totals after KD initiation, owing to improvements in seizure burden.22,23 Eventually, a lower number of medications will lead to fewer side effects.
Overall, the KD demonstrated strong efficacy, with three patients (19%) showing more than 50% improvement, and six (37%) showing more than 90% improvement. The remaining seven patients did not show meaningful seizure reduction. The efficacy results shown in the present study were similar to that of other published studies.24–26
Adherence to the diet was reported as a limiting factor in some studies.27–30 In the present cohort, families complied with the KD without major obstacles. This may be attributed to careful selection of patients and families before initiating the KD. Also, the KD team conducted several preparatory sessions with the 16 subjects, including one-on-one sessions and handing out educational literature about the KD. Moreover, our clinical dietitians were available for phone calls or chats on the online WhatsApp® social media application if they needed to ask any urgent questions and triage patients to a pediatric neurologist or emergency room if needed.
Increments in anthropometric measures were observed in the present cohort. This is similar to other studies showing that KD is a safe nutritional option in this age group.31,32 A modest increase in cholesterol and lipid profiles can be seen as a long-term protentional side effect of KD. In the present study, cholesterol levels were elevated during follow-up compared with other parameters, which has also been observed in other cohorts.33,34
The results of the present study may be limited by the relatively small number of patients investigated and the single-center setting. Another limitation, similar to other KD studies, was the retrospective observational study design. Future randomized controlled studies with larger study populations should be conducted to further verify the present results.
Conclusion
Ketogenic diets are a safe and effective alternative to medications in the treatment of epilepsy. A non-fasting protocol was associated with few side effects and might be better tolerated, particularly in pediatric patients. Few KD studies have been conducted in Saudi Arabia, and more are required to verify the present results. The present study results support the safety and efficacy of non-fasting KDs, suggesting that they may be used as a safe alternative to fasting to elicit ketosis.
Footnotes
Author contributions
HA, OM, NA, HA HK, AE, and AT wrote and reviewed the manuscript, and helped in data collection.
Acknowledgment
We acknowledge and appreciate the children on a ketogenic diet and their families.
Declaration of conflicting interest
The Authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
