Abstract
Susac syndrome (SuS) is a rare autoimmune endotheliopathy leading to hearing loss, branch retinal artery occlusions and encephalopathy. Young females are more frequently affected than males, making counselling for family planning an important issue. We reviewed published cases on SuS during pregnancy or in the postpartum period, and selected 27 reports describing the details of 33 patients with SuS. Treatment options and implications for pregnancy and breastfeeding are discussed. We propose new areas for research and suggest a management strategy.
Keywords
Introduction
Susac syndrome (SuS) is named after John Susac, who was the first to describe the syndrome of encephalopathy, hearing loss and branch retinal artery occlusions (BRAO).1,2 It is a rare disease, with just over 500 cases described worldwide. 3 Diagnostic criteria were proposed by the European Susac Consortium in 2016. 4 The pathophysiology of this neuroinflammatory disease, which affects the endothelial cells of microvessels in the brain, cochlea and retina, remains poorly understood. Activated cytotoxic CD8+ T-cells contribute to inflammatory damage of the endothelium. Anti-endothelial cell antibodies are present in 25% of patients, but their role in SuS pathogenesis is not clear.5–7 Treatment is based on expert opinion and case-series as clinical trials are non-existent in this rare disease. A practical treatment guideline for SuS based on a single expert opinion has been proposed recently, offering different therapeutic regimens for milder to more severe forms of the disease. 8 Less aggressive treatment recommendations have been made by others. 9
SuS affects young women more frequently than men, with a female:male ratio of 3.5:1. 10 It is not surprising that, in the age category affected, family planning is often not completed, making counselling necessary. Moreover, SuS can present for the first time, or relapse after a period of disease remission, during pregnancy or in the postpartum period. 10
In this article, we review published cases of SuS during pregnancy and the postpartum period, discuss issues in family planning in SuS patients, suggest areas for further research and propose a management strategy.
Review of published cases
We searched the literature (Pubmed) and internet (Google) for published case reports, case series and review articles for descriptions of SuS patients during pregnancy, postpartum or after termination of pregnancy (search terms: Susac, pregnancy, postpartum; search until August 2020) and selected 27 reports describing a total of 33 SuS patients.11–37 All cases are listed in Table 1. The mean age at pregnancy was 28.6 years. In 21 patients, the disease was diagnosed during pregnancy (five in the first trimester, seven in the second trimester, eight in the third trimester and one not reported) and in eight patients there were relapses during pregnancy or in the postpartum period. In two patients, the first symptoms of SuS presented shortly after abortion (one spontaneous, one induced). Only two patients with SuS completed a pregnancy without relapses. In one patient, pregnancy was discovered when she was treated with cyclophosphamide (CYC) and the pregnancy was terminated for this reason. Pregnancy was terminated in six cases to allow treatment with potential foetotoxic drugs like CYC. Delivery of a healthy baby (at term or preterm) was described in 22 cases. One stillbirth and one spontaneous abortion were reported. Notably, six patients had one or more pregnancies, without symptoms of SuS, before the index pregnancy when SuS was diagnosed. Treatment during pregnancy consisted most frequently of steroids, anticoagulant or antiplatelet therapy, with add-on intravenous immunoglobulins (IVIG) in six cases and plasma exchange (PLEX) in two cases. One patient started CYC at 28 weeks gestational age due to ongoing relapses and she delivered a healthy baby. 30 Most patients improved on therapy, but residual cognitive, visual and/or hearing impairments were present in most patients. Complete recovery was rare. A few patients had a history of symptoms, compatible with SuS.
Case reports, case series and review articles for descriptions of SuS patients during pregnancy, postpartum or after termination of pregnancy.

AZA, azathioprin; BRAO, branch retinal artery occlusions; CSF, cerebrospinal fluid; CYC, cyclophosphamide; FA, fluorescein angiography; GA, gestational age; IVIG, intravenous immunoglobulins; MMF, mycophenolate mofetil; MRI, magnetic resonance imaging; OCB, oligoclonal bands; SNHL, sensorineural hearing loss; SuS, Susac syndrome; wbc, white blood cells.
Pregnancy planning: general
Fertility
While no specific reports have been published on fertility in patients with SuS, this topic is of importance. Indeed, treatment with CYC may induce infertility in young female patients who have not yet completed their family. The risk depends on the patient’s age at treatment and the cumulative dose. 38 Consulting a fertility specialist before the start of this treatment is recommended. As SuS is more frequent in females than in males, a role for hormones in pathophysiology may be suspected. A case of a SuS relapse after starting oestrogen replacement therapy, in a patient who had been in remission for 18 years, has been reported, suggesting the possibility of a role for hormones in triggering late relapse. 39 However, recently a female-to-male transgender patient developing SuS under treatment with testosterone was described, challenging the hypothesis of (female) sex hormones as important players. 40 Alternatively, this coincidence may not be associated with hormonal treatment at all, as men and women both can be affected. Whether oral contraceptive pills and hormonal treatments used during in vitro fertilisation (IVF) procedures may increase the risk of SuS or SuS relapse remains to be elucidated. A first case of SuS remaining in remission after a successful IVF procedure was published recently . 35 Usually patients with SuS are advised to change their systemic contraception to a local method because of the unknown impact of hormonal treatment, 41 but the evidence to support this strategy is scarce and is in part taken from the concept that hormonal treatments are considered prothrombogenic.
Genetics and heritability
To our knowledge, no cases of familial SuS have been published. No studies on (immuno)genetics have been reported to date. In a study with 14 patients, all but one SuS patient who was homozygous for HLA C*04, expressed HLA-C*06 and/or HLA-C*07. Comparing the peptide binding motifs of these HLA-C allotypes revealed that the binding motifs of HLA-C*06:02 and HLA-C*07:02 are almost identical. 5 SuS is considered to be a non-hereditary disease. However, as in other autoimmune diseases, such as multiple sclerosis (MS), genetic risk factors likely play a role in the development of the disease. This field is still open to research.
Disease activity monitoring
For patients who have a known diagnosis of SuS, regular clinical monitoring during pregnancy and the postpartum period is advisable, to detect disease relapse or recurrence at an early stage. Indeed, the reported cases demonstrate that there is a risk of disease onset and recurrence in these periods. During pregnancy, brain magnetic resonance imaging (MRI) is generally considered to be safe, especially when benefits outweigh potential risks. Gadolinium contrast is not administered during pregnancy, due to slightly increased risk of neonatal death. Moreover, T2 hyperintensities and diffusion weighted imaging, which can show the typical callosal lesions, may be a worthwhile alternative to gadolinium-enhanced MRI. 42 For patients who are diagnosed during pregnancy, a fluorescein angiogram may add important diagnostic information. However, fluorescein may cross the placenta and enter the amniotic fluid. There are no teratogenic risks in animals. Safety information in humans is limited and therefore the decision to perform a fluorescein angiography should be made on a case-by-case basis and be performed only when the benefits outweigh the potential risks. 43
Timing of pregnancy
SuS patients attempting pregnancy should preferably be free from disease activity and stable without therapy or stable on a treatment that is compatible with pregnancy. Advance pregnancy planning and counselling is therefore highly recommended in this patient group. It is generally accepted now that women with autoimmune diseases like systemic lupus erythematosus (SLE) and vasculitis may attempt pregnancy during quiescent periods of their disease, maintaining a compatible therapy during the preconception, pregnancy and postpartum periods.44,45 In our opinion, the same advice may be applied to SuS patients. Attempting pregnancy when the disease is not (temporarily) in remission should be advised against, because of the risks to the mother when SuS flares up. Disease remission for a duration of at least 6 months seems prudent before attempting pregnancy. This advice is in accordance with recommendations for patients with SLE who wish to become pregnant. 44 However, disease remission for 6 months is no guarantee of no relapse during pregnancy, as disease recurrence has been described 23 years after initial symptoms, potentially elicited by pregnancy. 25 In conclusion, timing of a pregnancy should be a shared decision between patient and clinician, and patients should be informed of the risk of disease relapse during or after pregnancy.
Compatibility of commonly used treatments for SuS with pregnancy and breastfeeding
Recommendations on treatment of SuS have been published recently and are based on expert opinion. 8 There are no guidelines on treatment of SuS during pregnancy, where potential foetal toxicity of treatments needs to be taken into consideration. In the reported cases from patients with SuS during pregnancy, mainly steroids, IVIG and PLEX have been utilized during pregnancy, whereas cyclophosphamide and rituximab were kept for severe and refractory cases, after delivery (see Table 1). However, in one severe case, CYC was started in the 28th week of pregnancy because of ongoing relapses, without foetal toxicity. 30 It is important to note that all treatments described for SuS are off-label use.
Corticosteroids
Corticosteroids are used to treat disease flares, both intravenously in a high-dose pulse and orally in tapering schedules. Risk monitoring during pregnancy consists of following glycemia and blood pressure. Corticosteroids should be avoided in the first trimester, if possible, especially between 8th and 11th gestational week to reduce the slightly elevated risk of cleft lip and palate, but data are scarce. 46 One single pulse seems to be safe, while repeated or continued administration of corticosteroids may lead to growth retardation or preterm birth. Others state that prednisolone and methylprednisolone use is safe even in the first trimester. 47 Methylprednisolone and prednisolone should be preferred over dexamethasone, because penetration of the placental barrier is only 10%.
Intravenous immunoglobulins
It is important to assess the serostatus of the patient before starting IVIG, as administration of IVIG may lead to false positive serologic results. Indeed, serologic testing will detect endogenous IgG, produced by the patient, as well as administered IgG. 48 IVIG will cross the placenta. IVIG are used widely in the treatment of SuS: many case series and case reports describe amelioration of symptoms, and expert opinion recommends IVIG or subcutaneous IG (scIG). IVIG are safe in pregnancy and breastfeeding.47,49
Plasma exchange
PLEX seems to be safe in pregnancy and has been used as a rescue therapy in different neuroimmunological diseases, such as MS, antiphospholipid syndrome, thrombotic thrombocytopenic purpura, neuromyelitis optica spectrum disorders (NMOSD) or myasthenia gravis (MG).50,51 In SuS, PLEX seems to be useful in acute episodes. 21 There are no reports of immunoadsorption in SuS.
Mycophenolate mofetil
Mycophenolate mofetil (MMF) is teratotoxic (pregnancy loss, congenital malformations) and should be avoided in pregnancy. Men and women should use effective contraceptives strictly during the treatment period, and women additionally for at least another 6 weeks. No information is available on the excretion and effects of MMF in breast milk; expert recommendation is to avoid breastfeeding with MMF [United States Food and Drug Administration (FDA)]. 47
Azathioprin
Data on azathioprin (AZA) in other immunological diseases do not show any teratogenic effect, but there are hints of premature births and low birth weight. Whether this is due to the underlying disease, to the drug itself or other drugs used in combination, needs to be resolved. Cases of infants with bone marrow depression after maternal AZA use have been described. These side effects seem to be rare and should be weighed against potential relapses when discontinuing the drug if the mother is stable.47,50 Thus, in treatment-naive pregnant women with SuS onset, AZA should not be the first line treatment. However, AZA can be continued during pregnancy after risk/benefit evaluation. Regular monitoring of leucocytes and thrombocytes is advisable. During lactation, AZA is probably safe, as drug levels in breastmilk remain very low, especially 4 h after intake. 52
Methotrexate
Methotrexate (MTX) is contraindicated in pregnant women because of the teratogenic effects. It should be stopped at least 3 months before attempting conception. 47 Data on excretion in breastmilk are scarce and lactation should therefore be avoided during MTX use.53,54
Cyclophosphamide
CYC is contraindicated in pregnant women because of the teratogenic effects. However, there is some preliminary evidence in the field of cancer treatment that chemotherapy could be administered during the second and third trimester, with low risk of severe problems for the foetus.47,55,56 In selected cases, treatment with CYC during pregnancy after the first trimester can be considered, in a centre that has experience with management of complicated pregnancies with a multidisciplinary team of at least a gynaecologist, a neurologist and a neonatologist. CYC is excreted in breastmilk, may suppress the infants bone marrow and should be avoided during lactation.57,58
Tumour necrosis factor alpha inhibitors
Tumour necrosis factor alpha (TNF-α) inhibitors are contraindicated in patients with demyelinating disease as these therapies may increase inflammation and induce relapses, underlining the importance of an optimal differential diagnosis. In patients with SuS, TNF-α inhibitors seem to be helpful in case reports and case series in patients with relapses with classic immunotherapies. 59 Based on sparse data from case series and case reports, TNF-α inhibitors do not appear to be associated with a high risk of teratogenicity, but a harmful effect cannot be ruled out definitively. In rheumatological diseases, TNF-α inhibitor use may be associated with a higher rate of preterm delivery, but this may be due to disease activity. TNF-αa inhibitor should be discontinued around the third trimester when transfer across the placenta is greatest.47,60 The decision to use TNF-α inhibitors as an off-label medication in pregnant women with SuS should be reserved for very severe or life-threatening disease. Breastfeeding is compatible with TNF-α inhibitors. 61
Rituximab
Information on rituximab (RTX) in pregnancy is based on case reports of women with immunological and malignant diseases. The monoclonal antibody can pass the blood–placenta barrier. The average half-life of RTX is 20–31 days. RTX seems to be associated with a higher risk of premature births, with consideration of the potential harmful effect of the underlying disease as a concurrent cause. B cells will be depleted in newborns; thus, measuring B lymphocytes in foetuses is recommended if RTX has been administered after the 20th week of pregnancy. In NMOSD, RTX administration is recommended close to the time of conception to have a long-term protective effect during pregnancy.47,50 RTX is transferred to breast milk in minimal amounts.62,63 Moreover, in breastmilk-fed infants from mothers treated with anti-CD20 therapies, no negative impact on health of the infants up to the age of 1 year was detected. 64 To summarise, careful evaluation of the risks and benefits of stopping or the continuation of RTX treatment is necessary. In patients with severe autoimmune disease, it is acceptable to attempt pregnancy closely after the last RTX dose and to consider redosing of RTX if relapses occur during pregnancy. 65
Natalizumab
Natalizumab (NAT) is registered as treatment for relapsing remitting MS (RRMS). Its mechanism of action is interesting, because it inhibits lymphocyte adhesion and thus migration through the blood–brain barrier, by blocking alpha4-integrin. In one case, NAT was reported to exacerbate SuS. 66 However, in an animal model and in four SuS patients, disease improvement was seen. 5 One advantage of NAT is that it has been used during pregnancy in RRMS patients and seems relatively safe in clinical practice. However, insufficient data are available to draw firm conclusions.67–69 Another issue is the risk of progressive multifocal leukoencephalopathy in patients who are likely immunosuppressed by other treatments received prior or concomitantly. From MS, it is known that a rebound of disease activity may occur after cessation of treatment with NAT. When used only in patients with high disease activity, or when alternative treatment options are lacking, treatment with NAT might be continued under careful and frequent control and consideration of all the risks and benefits in pregnant women with RRMS.64,69 The last dose should be administered before the 30–34th week. During lactation, current data for administration of NAT are limited, but reassuring.63,67,69 In conclusion, the mechanism of action of NAT and clinical experience suggest that this agent may be of interest in SuS patients.
Calcineurin inhibitors
Cyclosporin A (CSA) and tacrolimus (TAC) are used in NMOSD, MG and SLE, and sometimes in SuS.51,61,70–72 TAC and CSA should not be started, but can be continued relatively safe in pregnancy. However, strict drug level monitoring is required to limit toxicities. Metabolites of CSA and TAC pass the placental barrier. No major malformations have been reported with CSA or TAC. Premature birth and low birth weight have been reported in humans (FDA). Most data have been derived from patients receiving organ transplantation. Caution in the use of these therapies during pregnancy in SuS is therefore warranted. Limited data suggest that the excreted levels of TAC and CSA in breastmilk are low and unlikely to negatively affect the infant. TAC and CSA are considered probably safe during breastfeeding.73–75 However, caution is warranted and monitoring of drug levels in the infants blood may be necessary, as even with low amounts of CSA excreted in breastmilk, infant levels may have therapeutic concentrations in the blood. 76
Acetyl salicylic acid
High-dose acetyl salicylic acid (ASA) should be used with caution in pregnancy. Low dose ASA (81mg) preconception has not been associated with increased risk of major adverse events when used throughout pregnancy. 77 Epidemiologic studies describe increased risk of miscarriage, cardiac malformations, and gastroschisis under ASA in early pregnancy; the absolute risk of cardiovascular malformations increased from less than 1% to up to approximately 1.5%. The risk is believed to increase with dose and duration of therapy (FDA). For secondary stroke prevention, low dose ASA during pregnancy is reasonable, and breastfeeding can be considered during intake of low dose ASA. 78 In most patients with SuS, ASA is added to reduce the risk of vessel occlusion based on expert opinions; however, evidence is lacking. Luminal occlusion in SuS is caused by hypertrophied and reactive endothelial cells.79,80 Whether ASA effectively reduces endothelial inflammation in SuS remains to be proven.
Nimodipine
Nimodipine is a calcium antagonist that leads to vasodilatation. It is lipophilic and can pass the blood–brain barrier. It has been used in SuS in the past, but the immunopathogenesis does not support the use of nimodipine.
Discussion
We have summarised more than 30 cases of SuS, with description of disease course and treatment during pregnancy or postpartum period. Strikingly, approximately two out of three patients of these cases were diagnosed during pregnancy. One likely explanation is that there is a publication bias towards new diagnosed cases in pregnancy, while pregnancies in SuS patients who are in remission and have a normal course are not reported. A prospective, international registry for patients with SuS, containing specific pregnancy forms, could be a solution to solve this potential reporting bias. Patients who are in remission and have pregnancies without relapse or complications, as well as their treating physicians, should be encouraged to share their data and participate in these registries. Patient-driven or active patient-participation in these registries may help to collect the necessary data. Another potential explanation of SuS relapse during pregnancy is the role of hormones and changes in the immune system. It is well-known that the course of several autoimmune diseases changes during pregnancy. Th1-related diseases such as rheumatoid arthritis or MS tend to stabilise, while Th2-related diseases like SLE or vasculitis carry a risk of exacerbation during pregnancy. 45 Systematically studying the immunology of SuS before, during and after pregnancy may lead to better knowledge on pathophysiological mechanisms involved in disease relapse and remission.
Due to the rarity of this disease, there are no randomized controlled trials to guide treatment, and therapy is based on expert opinions and is, in part, based on knowledge of other immunological diseases. We propose a period of at least 6 months disease remission before attempting pregnancy (see Figure 1). This seems a reasonable approach in SuS and is in accordance with recommendations for patients with SLE. 44 In our opinion, and based on the risk profile of the drugs, for maintenance treatment during pregnancy, first choices are low dose oral (methyl)prednisolone and monthly IVIG. AZA, CSA and TAC may be considered as maintenance treatment during pregnancy, in patients who are known with SuS, but are not a first choice to start during pregnancy. MMF and MTX should be stopped before attempting conception and should not be started during pregnancy or lactation. RTX, with a last dose not too long before conception, may be a treatment option in patients who had severe disease and who wish to lower the risk of disease exacerbation during pregnancy as much as possible, in analogy with NMOSD management. 65 To treat SuS exacerbations during pregnancy and lactation, high-dose IV methylprednisolone can be considered, either alone or in combination with IVIG and/or PLEX. Adding ASA can be considered safe. In severe cases, RTX might be started and NAT might be continued during pregnancy, in analogy with treatment of severe SLE or RRMS. When treatment-refractory, very severe relapses occur, in the second or third trimester of pregnancy, after careful consideration, CYC can be regarded as a rescue therapy option, in analogy to other life-threatening autoimmune disease (see Box 1). During lactation, only small amounts of monoclonal antibodies are excreted into breastmilk. Therefore, TNF-α inhibitors, RTX and NAT may be relatively safe and considered to administer while breastfeeding.62,64,69,81 Also, AZA, TAC and CSA may be safe during lactation. Finally, these pregnancies should be considered as high-risk pregnancies and follow up by or consultation with experts in the field of neuroimmunology is a prerequisite.
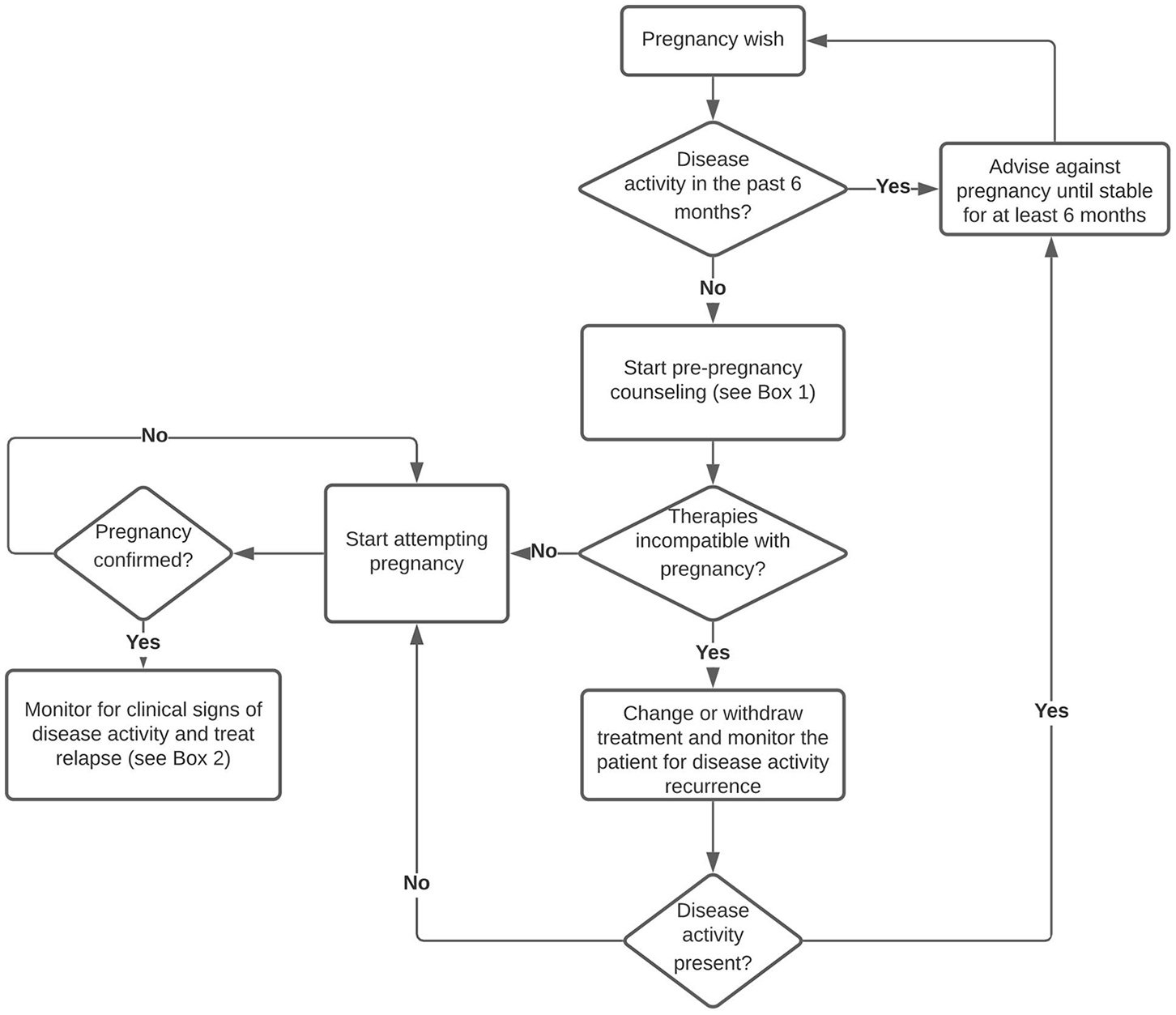
Management of pregnancy in SuS.
Recommendations on management of SuS patients before, during and after pregnancy.

AZA, azathioprin; CSA, cyclosporin A; CYC, cyclophosphamide; IVIG, intravenous immunoglobulins; MMF, mycophenolate mofetil; MRI, magnetic resonance imaging; MTX, methotrexate; NAT, natalizumab; PLEX, plasma exchange; RTX, rituximab; TAC, tacrolimus; TNF-α, tumor necrosis factor alpha; SuS, Susac syndrome.
Footnotes
Funding
BW is supported by a clinical PhD fellowship 1701919N and travel grant K219819N from the Research Foundation Flanders
Conflict of interest statement
The institution of BW received travel support to attend meetings, fees for participation in advisory boards, speaker honoraria and grants for research and/or patient support and/or education from Biogen, Roche, Merck, Novartis, Genzyme, Celgene. IK received travel expenses for attending meetings from Pfizer and CSL Behring. IK received speaker honoraria from Daiichi Sankyo.
