Abstract
Background:
The objective of this study was to evaluate monthly intramuscular adrenocorticotropic hormone (ACTH) gel versus intravenous methylprednisolone (IVMP) add-on therapy to interferon β for breakthrough disease in patients with relapsing forms of multiple sclerosis.
Methods:
This was a prospective, open-label, examiner-blinded, 15-month pilot study evaluating patients with Expanded Disability Status Scale (EDSS) score 3.0–6.5 and at least one clinical relapse or new T2 or gadolinium-enhanced lesion in the previous year. Twenty-three patients were randomized to ACTH (n = 12) or IVMP (n = 11) and completed the study. The primary outcome measure was the cumulative number of relapses. Secondary outcomes included EDSS, Mental Health Inventory (MHI), plasma cytokines, MS Functional Composite (MSFC), Quality-of-Life (MS-QOL) score, bone mineral density (BMD), and new or worsened psychiatric symptoms per month. Brain magnetic resonance imaging was analyzed post hoc. This was a preliminary and small-scale study.
Results:
Relapse rates differed significantly [ACTH 0.08, 95% confidence interval (CI) 0.01–0.54 versus IVMP 0.80, 95% CI 0.36–1.75; rate ratio, IVMP versus ACTH: 9.56, 95% CI 1.23–74.6; p = 0.03]. ACTH improved (p = 0.03) MHI (slope 0.95 ± 0.38 points/month; p = 0.02 versus slope −0.38 ± 0.43 points/month; p = 0.39). On-study decreases (all p < 0.05) in eight cytokine levels occurred only in the ACTH group. However, on-study EDSS, MSFC, MS-QOL, BMD, and MRI lesion changes were not significant between groups. Psychiatric symptoms per patient were greater with IVMP than ACTH (0.55, 95% CI 0.12–2.6 versus 0; p < 0.0001). Other common adverse events were insomnia and urinary tract infections (IVMP, seven events each) and fatigue or flu symptoms (ACTH, five events each).
Conclusions:
This study provided class II evidence that ACTH produced better examiner-assessed cumulative rates of relapses per patient than IVMP in the adjunctive treatment of breakthrough disease in multiple sclerosis.
Introduction
Although several effective disease-modifying therapies (DMTs) are presently available for long-term management of relapsing forms of multiple sclerosis (MS), breakthrough disease and progressive deterioration remain a clinical challenge. Data to guide MS treatment decisions, particularly for breakthrough disease, have been actively accumulating over the last several years.
Historically, periodic or monthly administration of systemic corticosteroids, such as intravenous methylprednisolone (IVMP) pulse therapy, was one of the first strategies for management of worsening or breakthrough MS and is still employed to enhance the effects of DMTs for MS [Myhr and Mellgren, 2009; Sorensen et al. 2009; Lienert et al. 2013; Zivadinov et al. 2008, 2012; Piri Cinar et al. 2013; Mezei et al. 2013; Shaygannejad et al. 2013]. This approach developed after systemic corticosteroids were approved for MS relapse treatment. Corticosteroids soon became broadly used for this indication and show efficacy in addressing acute inflammatory states in MS [Myhr and Mellgren, 2009; Sorensen et al. 2009].
The use of systemic corticosteroids as maintenance therapy is not novel in the management of autoimmune conditions, and several publications appeared in the 1990s and early 2000s highlighting this therapy as a maintenance approach [Filippini et al. 2000, 2013]. The initial high scientific interest in corticosteroids for this potential indication has diminished as newer highly effective DMTs have emerged in the past several years for first-line or escalation therapy [Hoepner et al. 2014; La Mantia et al. 2016; Miller, 2015; Gross and Krieger, 2015; Linker and Haghikia, 2016; D’Amico et al. 2015]. These DMTs have entered clinical practice with the potential to adequately address the problem of breakthrough disease.
Even as new treatment options have emerged with impressive efficacy data for MS, monthly steroid administration remains an ordinarily used off-label treatment option. However, despite the overall reliability of corticosteroids, they are not always effective or well tolerated [Myhr and Mellgren, 2009; Sorensen et al. 2009; Lienert et al. 2013; Zivadinov et al. 2008; Shaygannejad et al. 2013; Filippini et al. 2000]. Common adverse reactions, both with short- and long-term use, include osteoporosis, steroid-induced diabetes, hypertension, gastritis and gastrointestinal ulcer, weight gain, edema, and psychological or psychiatric impairments. In addition, regular administration of corticosteroids may suppress the functioning of adrenal glands.
Adrenocorticotropic hormone (ACTH) gel (H.P. Acthar Gel, repository corticotropin injection; Mallinckrodt ARD Inc., Hazelwood, MO, USA) is a long-acting formulation of the full-sequence ACTH (ACTH1–39; 80 units/ml) [Acthar for Healthcare Professionals, 2015]. ACTH1–39 includes pro-opiomelanocortin peptides and has been approved by the US Food and Drug Administration (FDA) for acute exacerbations of MS [Berkovich, 2013] since 1978, immediately prior to the same approval for IVMP in 1979. To date, ACTH and IVMP remain the only FDA-approved treatment options for MS relapses [Stuart, 2004].
Unlike pulse corticosteroids, which clinical trials have demonstrated to be an option when clinical activity becomes more frequent or severe in spite of treatment with a platform drug such as interferon β-1b (IFNβ-1b), IFNβ-1a, or glatiramer acetate [Catania, 2008], long-term ACTH pulse therapy for MS has not been evaluated in clinical trials. Although, historically, the mechanism of ACTH action has been considered to be due to stimulation of corticosteroid release, accumulating preclinical evidence suggests that ACTH has direct corticosteroid-independent (extra-adrenal) effects on the immune and central nervous systems (CNS). These effects may be relevant to MS and other neuroinflammatory conditions [Catania, 2007; Catania et al. 2004; Arnason et al. 2013; Gong, 2011]. Extra-adrenal anti-inflammatory and potentially neuroprotective effects of melanocortins, including ACTH, act at the cellular level (i.e. as actions on immunocytes) and at the systemic level through stimulation of all five melanocortin receptors [Bomback and Radhakrishnan, 2011; Benjamins et al. 2014; Catania et al. 2010].
ACTH and other melanocortins inhibit activation of nuclear factor κB (NFκB), which leads to inhibition of the production of a wide range of inflammatory mediators [Brod and Hood, 2011]. Orally administered ACTH exerts anti-inflammatory effects in mice via modulation of regulatory T (Treg) cells [Szabo et al. 1989]. Both ACTH1–39 and ACTH1–24 activate anti-inflammatory signals by potentiating sympathetic/adrenergic pathways [Gothert, 1984; Brzoska et al. 2008], which play a role in the efferent anti-inflammatory cholinergic pathway [Giuliani et al. 2010]. Neurotrophic effects were suggested in spinal cord injury and protective effects in ischemic brain injury [Catania et al. 2004; Gong, 2011]. We hypothesize that these immunomodulatory and direct anti-inflammatory effects may confer benefit when ACTH is given as pulse therapy for breakthrough MS. This study was designed to evaluate pulse therapy with ACTH gel as an add-on therapy to IFNβ in patients with relapsing forms of MS experiencing breakthrough disease. This is a preliminary and small-scale study.
Methods
Study participants
This single-center, randomized, examiner-blinded, 15-month pilot study [ClinicalTrials.gov NCT01049451] was conducted from November 2009 (recruiting began) to July 2013 (database closed). All patients were from the University of Southern California (USC) MS Comprehensive Care Center and Los Angeles County MS Clinic. All patients provided written, informed consent prior to participation and were free to discontinue at any time. Patients were subject to appropriate safety monitoring by the principal investigator and a safety committee consisting of physicians not directly involved in the study, including an endocrinologist, a neurologist who specializes in MS, and a neurologist with clinical experience in the use of ACTH gel. The study was conducted in accordance with the International Conference on Harmonization guidelines and the Declaration of Helsinki. The study protocol was reviewed and approved by the USC Health Science Campus Institutional Review Board.
Specific inclusion criteria included patients with relapsing forms of MS aged 18–65 years receiving ongoing IFNβ treatment for at least 6 months. They also had to have Expanded Disability Status Scale (EDSS) scores of 3.0–6.5 and at least one clinical relapse or at least one new lesion with high signal on T2-weighted images or gadolinium (Gd) enhancement on brain magnetic resonance imaging (MRI) scans within the previous year. At study entry, in the IVMP group, 5 of 11 patients had MRI activity, 2 of whom also had relapse activity; 6 patients had relapse activity only. In the ACTH group, 6 of 12 patients had MRI activity, 2 of whom also had relapse activity; 6 patients had relapse activity only.
Patients were excluded if they had received natalizumab, mitoxantrone, rituximab, or other chemotherapy treatments in the past. Patients who had received corticosteroid treatment up to 90 days before screening, had started new medication within 30 days of screening, had a history of drug or alcohol abuse within 1 year of screening, and those with any clinically significant medical condition that would interfere with trial participation were also excluded.
Study participants were randomized in a 1:1 ratio via stratified permuted block randomization to treatment with monthly pulses of ACTH (80 units intramuscularly once daily for three consecutive days) or IVMP (1 dose of 1 g intravenously) for 12 months. Two patients in the ACTH group for a period of time were allowed to administer the drug 1 ml every 10 days instead of three consecutive days per month. After 12 months, both study drugs were stopped and patients were evaluated one more time at month 15. Using the combination of gender and three age categories (18–29, 30–49, 50–65), six strata were created. To ensure balanced distribution between the treatment arms, the smallest permute block was chosen and every effort was undertaken to distribute participants equally based on current IFN drug and MS duration. Only evaluators were blinded. Neurologic examinations were performed every 3 months for 15 months on each patient by neurologists blinded to the treatment groups. Blood draws for cytokines were uniformly timed in each patient at baseline, after month 6, and after month 12, with the on-study draws performed at the trough (i.e. for baseline and month 6, obtained on the morning of the day of the next scheduled monthly cycle of IVMP or ACTH, before dosing).
Outcome measures
For each outcome measure, we describe the level of evidence (class) based on established scheme requirements for therapeutic questions [French and Gonsenth 2008]. The primary outcome measure was cumulative number of relapses per patient assessed by the blinded neurologists (class II evidence). Secondary outcome assessments included EDSS, Mental Health Inventory (MHI), MS Functional Composite (MSFC), MS Quality-of-Life (MS-QOL) scale score, and bone mineral density (BMD) (class II). The subjects were required to report any adverse events (AEs) they experienced. The psychiatric symptoms (class IV) recorded were irritability/anxiety, depression, nightmare/sleep-talking, anger, and panic attacks. Immunologic studies (class II) assessing plasma cytokine profiles by multiplex cytokine assay were also performed. Because this was a small pilot study, some outcomes not specified in the exploratory protocol were added before patient data were collected. These include some outcome measures (BMD, new or worsened psychiatric symptoms, cumulative rate of new or worsened psychiatric symptoms) and a post hoc analysis of clinically obtained brain MRI findings (see below) in order to assess MRI changes post treatment (all class IV).
Multiplex cytokine assays
The immunologic assays were conducted blind to treatment assignment, clinical data, and sequence in time. Levels of plasma cytokines (class II) were measured at the Human Immune Monitoring Center at Stanford University. Human 51-plex (Luminex, Affymetrix, Inc., Santa Clara, CA, USA) was used according to manufacturer’s recommendations, with modifications as described below. Briefly, samples were mixed with antibody-linked polystyrene beads on 96-well filter-bottom plates and incubated at room temperature for 2 h, followed by overnight incubation at 4°C. Room temperature incubation steps were performed on an orbital shaker at 500–600 rpm. Plates were vacuum filtered and washed twice with wash buffer, then incubated with biotinylated detection antibody for 2 h at room temperature. Samples were then filtered and washed twice as above and resuspended in streptavidin phycoerythrin. After incubation for 40 min at room temperature, two additional vacuum washes were performed, and samples resuspended in Reading Buffer (Affymetrix, Inc.). Each sample was measured in duplicate. Plates were read using a Luminex 200 instrument (Luminex Corp., Austin, TX, USA) with a lower bound of 100 beads/sample/cytokine.
MRI
MRI scans were obtained on a clinical basis with no prescribed or consistently enforced acquisition protocol (class IV). All patients had baseline brain MRI available to determine eligibility for the study. However, regarding quantitative MRI post processing and quantification to explore treatment effects, this was performed retrospectively using only the most consistently obtained scan acquisitions. Patients were also required to have had MRI at baseline and approximately 15 months later. A subset of the overall cohort, 15 patients (ACTH, n = 9; IVMP, n = 6), underwent consistent T1-weighted spin-echo scan acquisitions pre and post Gd on a 1.5 T scanner and were included in the consideration of counts of T1 hypointense (‘black holes’) and Gd-enhanced lesions. The following parameters comprised T1 acquisition protocol: TR/TE 500–760/9–17 ms, slice thickness 3.0–5.0 mm. These images were used to calculate the number and volume of Gd-enhanced lesions, number of T1 hypointense lesions, and number of baseline Gd-enhanced lesions that evolved to T1 black holes at follow up.
One patient in the ACTH group had a technically insufficiently Gd-enhanced image and was excluded from calculation of Gd-enhanced lesion volume. Three patients were excluded due to other within-patient deviations in scan acquisition, leaving 12 (ACTH, n = 7; IVMP, n = 5) for analysis of total brain volume of T1 hypointense lesions (T1LV). A smaller subset, seven patients (ACTH, n = 4; IVMP, n = 3), underwent consistent T2-weighted fluid-attenuated inversion recovery (FLAIR) scan acquisition on the same 1.5 T scanner and thus were included for consideration of brain T2 hyperintense lesions. The FLAIR acquisition protocol comprised the following parameters: TR/TE 8800/150 ms, TI 2200 ms, slice thickness 3 mm. Four aspects of T2 lesions were assessed: total brain volume of T2 lesions (T2LV); number of new T2 lesions at follow up; number of enlarging T2 lesions at follow up; and number of new or enlarging T2 lesions at follow up. All image analysis was performed at the Laboratory for Neuroimaging Research at the Brigham and Women’s Hospital under the supervision of Dr Bakshi using Jim (v.5, Xinapse Systems Ltd, West Bergholt, UK) by observers who were unaware of clinical information or treatment group assignment. Lesions were identified by consensus of two experienced observers (ST, MN), and verified by an experienced observer (RBa). Volumetric analysis was performed by one of the observers (ST) using an edge-finding tool with manual adjustments as needed.
Statistical analysis
To compare relapse rates between treatment groups, the risk ratio was estimated by mean cumulative function with robust estimator of covariance for recurrent events. Mixed-effect modeling was used to compare the trajectory of EDSS scores, psychometrics, and plasma cytokine assessments over time. Baseline plasma cytokine measures in patients with ACTH and IVMP were assessed using a Wilcoxon rank sum test. Longitudinal cytokine measures in each group were compared by assessing the differences in slope by mixed-effect modeling to compare trajectory. No interim analyses of results were performed.
Results
Baseline demographics
Patient characteristics are shown in Table 1. Twenty-three patients with relapsing forms of MS were randomized to ACTH (n = 12) or IVMP (n = 11) and completed the study (Figure 1). Mean age (40.1 ± 13.1 versus 39.4 ± 12.3 years), baseline EDSS score (4.6 ± 1.5 versus 4.6 ± 1.3), and IFNβ therapy duration (11.3 ± 2.6 versus 10.6 ± 3.3 months) were not statistically different between the treatment groups.
Patients with relapsing forms of multiple sclerosis: demographics by treatment group.

All patients had relapsing–remitting MS, except two patients in each group who had relapsing–progressive MS [Lublin and Reingold, 1996].
Wilcoxon rank sum test for age, EDSS, and dose, χ2 test for sex.
Comparison of the distribution of male and female subjects in ACTH versus IVMP group.
Mean ± SD, median (Q1–Q3) [Kurtzke, 1983].

Study design and patient flow diagram for the primary endpoint (cumulative relapse rate). ACTH, adrenocorticotropic hormone; IVMP, intravenous methylprednisolone.
Primary outcome: relapse rates
Over 15 months, the cumulative number of relapses per patient (Figure 2) was 0.08 with ACTH [95% confidence interval (CI) 0.01–0.54] versus 0.80 with IVMP (95% CI 0.36–1.75); risk ratio IVMP versus ACTH: 9.56 (95% CI 1.23–74.6; p = 0.03). Among the IVMP group, one relapse occurred between baseline and month 3, two between months 3 and 6, one between months 6 and 9, zero between months 9 and 12, and four between months 12 and 15. However, in the ACTH group, only one relapse occurred between months 3 and 6.
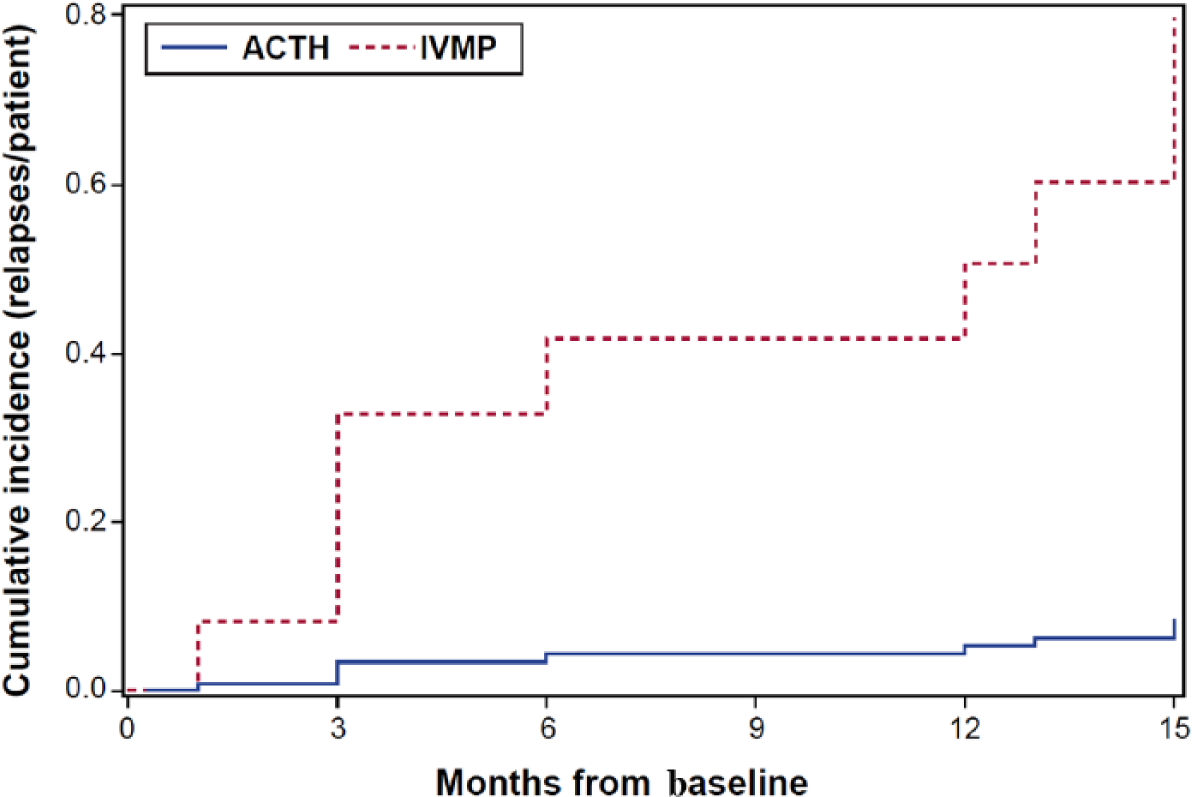
Cumulative incidence of relapses over 15 months by treatment group. Over 15 months, the cumulative number of relapses per patient was 0.08 with adrenocorticotropic hormone (ACTH) [95% confidence interval (CI) 0.01–0.54] versus 0.80 with intravenous methylprednisolone (IVMP) (95% CI 0.36–1.75); risk ratio IVMP versus ACTH: 9.56 (95% CI 1.23–74.6; p = 0.03). Among the IVMP group, one relapse occurred between baseline and month 3, two between months 3 and 6, one between months 6 and 9, zero between months 9 and 12, and four between months 12 and 15. However, in the ACTH group, only one relapse occurred between months 3 and 6.
Secondary outcomes
EDSS, MSFC, MS-QOL scale score, and BMD
Mixed-effect modeling showed no difference between groups in trajectory slopes of EDSS over time, but significantly stronger (p = 0.03) improvement in MHI for ACTH (slope: 0.95 ± 0.38 points/month; p = 0.02) compared with IVMP (slope: −0.38 ± 0.43 points/month; p = 0.39) (Table 2). There were no statistically significant between-group differences in MSFC, MS-QOL scale score, or BMD.
Trajectory slope and expected value by month after baseline and follow up, by treatment group.

p value time and group interaction test from mixed-effect model.
p value for testing whether group specific slope = 0 from mixed-effect model.
ACTH, adrenocorticotropic hormone; IVMP, intravenous methylprednisolone; MOS, Medical Outcomes Survey.
Plasma cytokines
Levels of 51 plasma cytokines from patients at baseline were compared with those obtained after 6 and 12 months. Of the 51 analytes measured, TNF-related apoptosis-inducing ligand (TRAIL), macrophage colony-stimulating factor (M-CSF), monokine induced by IFNγ (MIG), tumor necrosis factor β (TNFβ), macrophage inflammatory protein 1α (MIP1α), nerve growth factor (NGF), granulocyte colony-stimulating factor (G-CSF), and IFNα had significant differences in the slope calculated between baseline through 6 and 12 months in levels of these cytokines between treatment groups (Figure 3). Decreasing levels of these cytokines were observed in the ACTH group but not the IVMP group. Baseline levels of NGF were statistically higher (p ⩽ 0.05) in the ACTH than the IVMP arm, but this measurement was not used as part of the randomization process, as the measurements were performed in bulk after the trial was completed.

On-study change in plasma cytokine levels in the ACTH versus IVMP groups. Relative changes were estimated from a linear mixed model and rescaled by minus baseline and plus 100. p values were determined using interaction term in linear mixed modeling to compare trajectory slopes. ACTH, adrenocorticotropic hormone; G-CSF, granulocyte colony-stimulating factor; IFNα, interferon α; IVMP, intravenous methylprednisolone; M-CSF, macrophage colony-stimulating factor; MIG, monokine induced by interferon γ; MIP1α, macrophage inflammatory protein 1α; NGF, nerve growth factor; TNFβ, tumor necrosis factor β; TRAIL, TNF-related apoptosis-inducing ligand.
MRI assessment of lesions
There were no significant between-group differences at baseline (Table 3), and out of nine MRI measures assessed at the end of the study, none were significantly different at that time point. Five MRI comparisons favored IVMP and four favored ACTH, though none reached statistical significance. There was internal inconsistency in the results (e.g. T2 lesion volume favored ACTH, but counts of T2 lesions favored IVMP).
Baseline MRI measures by treatment group.
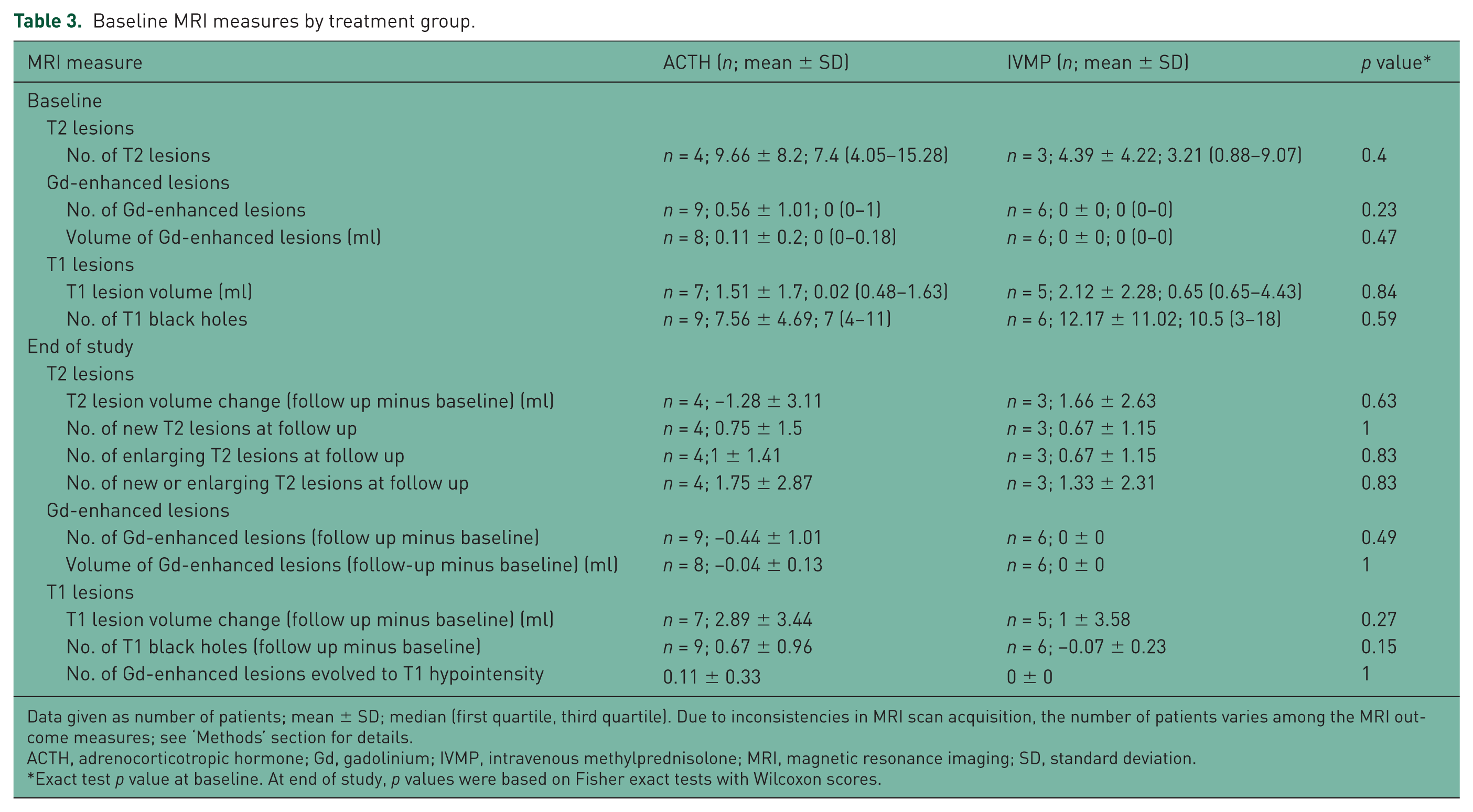
Data given as number of patients; mean ± SD; median (first quartile, third quartile). Due to inconsistencies in MRI scan acquisition, the number of patients varies among the MRI outcome measures; see ‘Methods’ section for details.
ACTH, adrenocorticotropic hormone; Gd, gadolinium; IVMP, intravenous methylprednisolone; MRI, magnetic resonance imaging; SD, standard deviation.
Exact test p value at baseline. At end of study, p values were based on Fisher exact tests with Wilcoxon scores.
Adverse events and psychiatric symptoms
The number of AEs occurring in each group are listed in Table 4. The following AEs that are not listed in Table 4 occurred once in the IVMP but not the ACTH group: balance problems, fecal incontinence, speech difficulty, indigestion/decreased appetite, bacteriuria, mouth tingling, urinary hesitancy, vaginal discharge, nightmare/sleep talking, upper lip sores, and arm pain. One of each of the following AEs not listed in Table 4 occurred in the ACTH but not the IVMP group: foot numbness, shingles, headache, cellulitis, abdominal pain, ear infection, heat intolerance, shortness of breath, hand weakness, melasma, and fasciitis that required foot surgery. The cumulative number of new or worsened psychiatric symptoms per patient was greater with IVMP (0.55, 95% CI 0.12–2.6) than ACTH (0; p < 0.0001) (Figure 4).
Number of adverse events by treatment group.

The number of events across all patients is shown.
Includes two patients who experienced depression (two events total); other psychiatric events included agitation, anxiety, emotional lability, panic attacks. The cumulative number of new or worsened psychiatric symptoms per patient was greater with IVMP (0.55, 95% CI 0.12–2.6) than ACTH (0; p < 0.0001) (see also Figure 4).
ACTH, adrenocorticotropic hormone; CI, confidence interval; IVMP, intravenous methylprednisolone.

New or worsened psychiatric symptoms on study. The cumulative number of new or worsened psychiatric symptoms per patient on study was greater with intravenous methylprednisolone (IVMP) (0.55, 95% CI 0.12–2.6) than adrenocorticotropic hormone (ACTH) (0; p < 0.0001
Discussion
Systemic corticosteroids are known to be general immune suppressants [Myhr and Mellgren, 2009]. While corticosteroids effectively reduce inflammation, long-term use supplements the immune-modulating effects of DMTs. For example, a 3-year study by Ravnborg and colleagues in patients with relapsing–remitting MS (n = 341) found that the combination of methylprednisolone (500 mg orally in monthly ‘pulses’; three doses over 3 days) and IFNβ-1a reduced the rate of relapses by 38% compared with IFN plus placebo, and patients in the combination arm also had improved MSFC metrics and MRI outcomes [Ravnborg et al. 2010]. Thus monthly steroids remain in the list of options for second-line treatment for MS. Pulse steroids as an add-on to the first-line DMT is arguably the only combination therapy which is practically used for MS management today [Sorensen et al. 2009; Kutz, 2016].
ACTH is the only treatment other than systemic steroids that is FDA approved for acute exacerbations of MS and as such is being used in cases of corticosteroid intolerance, diabetes, or suboptimal response [Stuart, 2004; Gong, 2011; Kutz, 2016]. Unlike pulse corticosteroids, long-term ACTH pulse therapy for MS has not been previously evaluated in clinical trials until this study.
Both the clinical and immunologic results indicate there is significant differentiation between the ACTH and IVMP arms. Clinical outcomes indicated the cumulative rate of relapse per patient over 12 and 15 months and the risk ratio for relapse over 15 months favored the ACTH arm. The ACTH group also had significantly stronger improvement in MHI than the IVMP group. No significant between-group differences were noted in the disability metric.
Levels of TRAIL, M-CSF, MIG, TNFβ, MIP1α, NGF, G-CSF, and IFNα were significantly decreased during the 12-month treatment period of ACTH but not IVMP. ACTH-associated reduction in the proinflammatory cytokines that relate to astrocytic (TNFβ) [Plant et al. 2005], macrophage (M-CSF, MIP1α) [Hamilton, 2008], neutrophil (G-CSF) [Rust et al. 2016] and T-cell (MIG) [Balashov et al. 1999] function is consistent with the observed decreased disease activity in these patients. It is also of interest that TRAIL, NGF, and IFNα were reduced by ACTH treatment. Increased blood TRAIL levels have been correlated with remission in MS [Moreno et al. 2013]; NGF has been shown to be protective in MS animal models [Arredondo et al. 2001]; and, finally, IFNα, a type I IFN similar to IFNβ, has well known protective effects in MS [Durelli et al. 1995]. Therefore, our results suggest that the mechanism of action of ACTH treatment is not mediated through TRAIL, type IFN signaling, or NGF.
The steroid-independent, anti-inflammatory effects of melanocortins, including ACTH, occur on both cellular and systemic levels through stimulation of all five melanocortin receptors, which inhibit production of a wide range of inflammatory mediators and activate anti-inflammatory signals [Catania, 2007; Gong, 2011; Giuliani et al. 2010]. Because this study included only patients with MS on IFNβ (to maintain a similar immunologic background for all study participants), the differences in immunologic outcomes between ACTH and IVMP lend support to the hypothesis that ACTH effects could be mediated by extra-adrenal steroid-independent mechanisms.
Add-on or combination therapy regimens typically increase the risks of AEs. In our study, the most common side effects included psychiatric symptoms, infections, insomnia, fatigue, and flu symptoms. Some of the AEs (e.g. fatigue and flu symptoms) were observed more frequently in the ACTH group and others (e.g. psychiatric symptoms, infections, insomnia) were more frequent in the IVMP group. New or worsened psychiatric symptoms only occurred in the IVMP group (although psychiatric side effects have been reported with ACTH; Berkovich, 2013), and that may potentially indicate a specific patient population is more likely to benefit from this particular option.
Our study has several limitations to be considered. There was a discordance between the clinical and MRI results between treatment groups. The IVMP group showed significantly higher relapse rates, but no increases in MRI lesion activity, compared with the ACTH group. However, it should be noted that, while T2 hyperintense and Gd-enhanced lesions are standard, validated outcome measures in MS [Zivadinov and Bakshi, 2004; Filippi et al. 2006], there is often a divergence between clinical and MRI findings [Zivadinov and Bakshi, 2004]. For example, the relationship between cerebral Gd-enhanced lesions and current, future, or previous clinical relapses is usually weak [Zivadinov and Bakshi, 2004]. Moreover, there was an imbalance in our study between the clinical and MRI dataset in that several patients who completed the clinical measures of the study did not have adequate MRI data for quantitative analysis. The MRI metrics were assessed post hoc and were not among the protocol prespecified outcomes of this study; these metrics were thus limited by the MRI undersampling of patients and the retrospective nature of the MRI analysis. The MRI measures assessed did not show a significant difference between groups, which limits the strength of our findings. Future studies should include prospectively designed MRI outcome measures, which are the standard in MS therapeutic trials [Filippi et al. 2006; Greenberg et al. 2016]. Other limitations are the lack of a placebo group, and that the principal investigator was not blinded. In addition, the small sample size does not allow the results to be generalizable to larger populations. We would emphasize that, given these limitations, our observations should be considered preliminary, warranting further evaluations on larger numbers of patients.
Currently, the FDA has not approved the use of corticosteroids or ACTH as long-term therapy for MS. Prolonged courses of corticosteroids or ACTH could lead to serious complications and therefore require careful monitoring. In this study, there were no substantial safety issues in either group. The potential need for adjunctive pulse ACTH therapy to limit future relapses and disease progression should be balanced against the availability in recent years of numerous approved first-line or escalation therapies for relapsing forms of MS [Salhofer-Polanyi and Leutmezer, 2014]. The value of this preliminary study in today’s treatment landscape is uncertain because a failure of β IFN (with new lesions and relapses) might require directly proceeding to one of the approved second-line therapies, and not to adding corticosteroids or ACTH. In spite of this important limitation, our results may have merit for certain clinical settings. Even in the ‘real world’ current landscape of such a wide array of approved treatment options, the goal of achieving ‘no evidence of disease activity’ remains elusive [Rotstein et al. 2015], driving the continuing search for novel combination therapies [Greenberg et al. 2016].
Conclusion
ACTH pulse therapy was more effective than IVMP in decreasing the rate of relapses without negative effects on psychiatric symptoms in patients with relapsing forms of MS experiencing breakthrough disease. Clinical outcomes reached in this study were supported by the analysis of plasma cytokines. Although this is a pilot study that included a relatively small population, the findings suggest the potential benefit of adjunctive ACTH pulse therapy in relapsing forms of MS.
Footnotes
Acknowledgements
The authors thank Wendy Gilmore, PhD, and Brett Lund, PhD, for their input to protocol development and Maura Fernandez and Dawood Subhani for study coordination. The authors would like to acknowledge the members of the independent Safety Committee, Leslie Weiner, MD, Jorge Mestman, MD, and Arthur Partikian, MD, for their time, and expertise in reviewing every clinical event and side effect throughout the time of the clinical study and its extension, an activity that required monthly live meetings for more than 2 years. The authors also thank Ruth K. Grossman, CMPP, Frank Beebe, PharmD, and Aric Fader, PhD, of MedVal Scientific Information Services for editorial assistance in preparing initial drafts of this manuscript, which was funded by Mallinckrodt ARD Inc. The final draft was edited by, approved by, and fully under the control of the coauthors.
Portions of this work were presented at the Consortium of Multiple Sclerosis Centers 2010 Annual Meeting [Berkovich et al., Int J MS Care 2010; 12(Suppl. 1): 81]; the 2011 Joint Triennial Congress of the European and Americas Committees for Treatment and Research in Multiple Sclerosis (Berkovich et al., Multi Scler J 2011; 17: S249); the American Academy of Neurology 2013 Annual Meeting (Berkovich et al., Neurology 2013; 80: e205–206); and the 2013 European Committee for Treatment and Research in Multiple Sclerosis Conference on Rehabilitation [Amezcua et al., Multi Scler J 2013; 19(Suppl. 1): 514–515].
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Study funding was supplied by Mallinckrodt ARD Inc. (formerly Questcor Pharmaceuticals), who provided a medical accuracy review.
Conflict of interest statement
R. Berkovich: served on advisory boards for Acorda, Avanir, Bayer, Biogen, Genzyme, Teva, and Questcor (now Mallinckrodt ARD); served as speaker for Acorda, Avanir, Bayer, Biogen, Genzyme, and Teva; received financial support for investigator-initiated studies from National MS Society, Questcor, and Teva. R. Bakshi: received consulting fees from AbbVie, Biogen, EMD-Serono, Genentech, Novartis, and Questcor (now Mallinckrodt ARD); received research support from Biogen, EMD-Serono, Novartis, Questcor, Sanofi-Genzyme, and Teva. L Amezcua: received consulting fees from Acorda, Biogen, Genzyme, Novartis, and Questcor (now Mallinckrodt ARD); received research support from Acorda, Biogen, and Novartis. L Steinman: received consulting and lecturing fees from Biogen, Merck-Serono, Novartis, Celgene, Receptos, and Teva. The other authors have no disclosures to report.
