Abstract
Background:
Dimethyl fumarate (DMF) was approved by the US Food and Drug Administration (FDA) for treatment of relapsing–remitting multiple sclerosis (RRMS) based on two phase III randomized clinical trials (RCTs). There were not enough non-White patients enrolled in these RCTs to allow for subgroup analysis based on race. Efficacy and tolerability of DMF therapy across various racial groups is unknown.
Methods:
Retrospective chart review was performed on all patients with RRMS who were started on DMF in two tertiary multiple sclerosis (MS) clinics. Efficacy and tolerability of DMF was compared across three self-identified racial groups: White-American (WA), African-American (AA) and Hispanic-American (HA).
Results:
A total of 390 RRMS patients were included in the study: 261 (66.9%) WA, 69 (17.7%) AA and 52 (13.3%) HA. When comparing ‘pre-DMF’ (1 year) and ‘on DMF’ (mean follow up of 14 months) periods, statistically significant reduction in rates of annualized relapses (WA from 0.44 to 0.19, AA from 0.39 to 0.15, and HA from 0.39 to 0.14; no differences between groups), new T2 lesions (WA from 45% to 23%, AA from 39% to 23%, HA from 52% to 26%; no difference between groups), and Gd+ lesions (WA from 25% to 13%, AA from 24% to 7%, HA from 23% to 12%; no difference between groups) were seen. DMF was relatively well tolerated across all groups, with an overall discontinuation rate of 20% (no difference between the three groups).
Conclusion:
Efficacy of DMF in our clinic population did not differ across three major ethnic groups, WA, AA and HA, and was comparable with results observed in the pivotal studies. These ‘real-life’ data suggest that race is not a factor that needs to be taken into account when initiating DMF.
Background
Dimethyl fumarate (DMF) demonstrated significant clinical and radiologic efficacy in the two phase III double-blind, randomized clinical trials in patients with relapsing–remitting multiple sclerosis (RRMS), DEFINE and CONFIRM [Fox et al. 2012; Hutchinson et al. 2013; Bar-Or et al. 2013; Gold et al. 2012]. However, none of the studies included race or ethnicity in their subgroup analysis due to very low non-White patient participation, with 83–85% of patients being white in CONFIRM and 78–79% in DEFINE. This is a well recognized limitation of multiple sclerosis (MS) clinical trials to date [Avasarala, 2014].
Efficacy and tolerability of DMF therapy across various racial groups may differ, since clinically important differences in MS course and response to therapies exist between patients of different races. For example, African-Americans (AAs) tend to have more frequent relapses, worse post relapse recoveries, faster transition from relapsing–remitting to secondary-progressive MS and more rapid accumulation of ambulatory disability compared with White-Americans (WAs) [Cree et al. 2004; Kister et al. 2010; Kaufman et al. 2003; Naismith et al. 2006; Marrie et al. 2006]. Importantly, AA patients tend to have poorer response to interferon-b (IFNb) therapy compared with WA [Cree et al. 2005], which could be due to differences in the natural history of the disease (e.g. more severe course in AA), intrinsically different response to IFNb, or a combination of these factors [Klineova et al. 2012; Cree et al.2009]. On the other hand, natalizumab has been shown to significantly improve the relapse rate and accumulation of brain lesions in AA.
MS is also increasingly being recognized in Hispanic-Americans (HAs), with studies demonstrating younger age at onset and higher frequency of optic nerve and spinal cord involvement; but an overall rate of disability progression that is comparable with that of WA [Amezcua et al. 2011; Buchanan et al. 2010; Hadjixenofontos et al. 2015; Langer-Gould et al. 2013]. Post hoc analysis of patients included in the clinical trials for fingolimod demonstrated the drug to be effacaious and well tolerated [Chinea Martinez et al. 2014]. However, data on Hispanic patients’ response to other therapies is limited.
New York University (NYU) Multiple Sclerosis Comprehensive Care Center in New York City, and Barnabas MS Care Center in Livingston, New Jersey, serve large, ethnically diverse patient populations. In this work, we sought to compare ‘real-life’ efficacy and tolerability of DMF in MS across the three major racial groups represented in our centers: WAs, AAs and HAs.
Methods
Retrospective chart review was performed on all patients with relapsing–remitting MS (2010 Revised McDonald Criteria), who were age 18 years old or older and were started on DMF between March 2013, when DMF was approved for clinical use in the US, and July 2014. All patients were followed for routine neurologic care at NYU or Barnabas MS Care Centers. Institutional review board approval was obtained in both institutions. The entire record for each patient (including all MRI reports) was reviewed, starting from 1 year prior to DMF initiation and up to 1 February 2015. From each chart, we extracted demographic and disease-related data, including age, gender, race, disease type, duration of disease at commencement of DMF, and prior disease-modifying-therapy history. The number of neurologist-determined relapses, new or enlarging T2 lesions, and new T1 contrast-enhancing lesions (Gd+) were recorded for brain and spinal cord (when available) spine MRI performed prior to and during DMF therapy through 1 February 2015. Efficacy and tolerability of DMF was compared across the three self-identified racial groups: WAs, AAs and HAs. The discontinuation rates of DMF in the three groups were calculated utilizing the entire cohort, but clinical and radiographic outcomes were assessed in patients who remained on the drug for at least 1 month. Prevalence of side effects was determined based on chart review of physician and nurses’ notes.
Statistical methods
For all continuous variables, the two-sample t-test and Wilcoxon rank sum test were used to detect statistical difference between AA and HA versus WA. For all categorical variables, the Chi-square test was used. Unadjusted annualized relapse rate (ARR) for pre- and post-DMF was estimated by repeated Poisson regression model. As a sensitivity analysis of ARR, rates of subjects’ relapsed pre- and post-DMF were compared by McNemar test. Rates of subjects with new T2 with or without Gd+ lesions pre- and post-DMF were also compared by McNemar test.
ARR, rate ratio, and 95% confidence intervals (CI) were estimated by a negative binomial regression model. Hazard ratios and 95% CI were generated using a Cox proportional hazards model. Models were adjusted for age, sex, and number of relapses 1 year prior to DMF. Lesion mean ratio for patients with new T2 lesions and 95% CI were estimated by a negative binomial regression model adjusted for age, sex, and baseline new T2 lesion counts. Odds ratio for patients with Gd+ lesions and 95% CI were estimated by the ordinal logistic regression model adjusted for age, sex, and baseline Gd+ lesion counts.
Results
Demographics
A total of 390 RRMS subjects fulfilled our inclusion criteria: 261 (66.9%) WA, 69 (17.7%) AA and 52 (13.3%) HA. Baseline demographic characteristics of the three groups are shown in Table 1. The mean age at DMF start was highest in WA, 47.4 years, lowest in HA, 42.5 years (p = 0.005 compared with WA), and intermediate in AA, 44.5 years (p = 0.095 compared with WA). HA patients were also diagnosed at an earlier mean age of 32.2 years (p = 0.009) as compared with WA (36.4 years) and AA (33.5 years, p = 0.05 compared with WA). Duration of disease, however, was similar across the three groups: 13.2 years in WA, 12.9 years in AA (p = 0.90 compared with WA) and 12.3 years in HA (p = 0.84 compared with WA). The highest proportion of women was in the AA group (83%, p = 0.044 compared with WA), second highest in the HA group (81%, p = 0.13) and lowest in WA (71%). The percentage of disease modifying therapy (DMT)-naïve patients was similar in all three groups (Table 1). Prior DMT therapy included natalizumab, 13%; fingolimod, 12%; teriflunomide, 2%; interferon beta-1a (IM), 18%; interferon beta-1b, 7%; glatiramer acetate, 23%; interferon beta-1a (SQ), 13%; and other, 2%. Six patients received treatment for less than 1 month (one WA, one AA and four HA), and were therefore excluded from the outcomes analysis.
Demographics.

Comparisons made versus Whites (Wilcoxon test). SD, standard deviation; MS, multiple sclerosis; DMT, disease modifying therapy; DMF, dimethyl fumarate.
Relapses
Among WAs, unadjusted ARR pre-DMF was 0.439, and ‘on DMF’, 0.196 (p <0.0001, compared with pre-DMF rate); in AA, pre-DMF relapse rate was 0.410, and 0.162 on DMF (p = 0.0064, compared with pre-DMF rate); in HA, the relapse rate decreased from 0.42 to 0.16 (p = 0.0050, compared with pre-DMF rate) (Figure 1). Overall, 18% of patients experienced a relapse while on DMF.

Pre- and post - dimethyl fumarate: annualized relapse rate.
The adjusted ARR on DMF was similar across the three ethnic groups: 0.20 in WA, 0.17 in AA, and 0.17 in HA, with no statistically significant difference in adjusted hazard ratio (aHR): in AA, aHR = 0.762, (95% CI, 0.392 and 1.478;p = 0.42, compared with WA), and in HA, aHR = 0.775 (95% CI, 0.376 and 1.598; p = 0.49, compared with WA) (Table 2).
Disease activity on dimethyl fumarate.

ARR, adjusted annualized relapse rate; aLMR, adjusted lesion mean ratio; aOR: adjusted odds ratio, DMF, dimethyl fumarate.
Comparisons made versus American.
Based on negative binomial regression model, adjusted for age, sex, and number of relapse 1 year prior to DMF.
Based on Cox model, adjusted for age, sex, and number or relapse 1 year prior to DMF.
Based on negative binomial regression model, adjusted for age, sex, and baseline new T2 lesion counts.
Based on ordinal logistic regression model, adjusted for age, sex, and baseline Gd+ lesion counts.
New magnetic resonance imaging T2 and gadolinium-enhanced lesions
Among WAs, 45% of patients had new T2 lesions within 1 year before DMF commencement and 23% had new lesions after starting DMF (p < 0.0001). Among AAs, there were 40% of patients with new lesions pre-DMF, with a drop to 23% of patients post-DMF (p = 0.029). Among HAs, 52% had new lesions pre-DMF and 25% (p = 0.02) of patients after starting DMF (Figure 2). Overall, 23% of patients demonstrated new T2 lesions while on DMF. Adjusted mean counts of new T2 lesions while on DMF were 0.39 in the WA group, 0.30 in AA and 0.35 in HA, with no statistically significant differences seen between WA and AA groups [lesion mean ratio of 0.77 (95% CI, 0.41 and 1.42; p = 0.39)] compared with WA, and between the WA and HA groups [lesion mean ratio of 0.89 (95% CI, 0.46 and 1.70, p = 0.88)] (Table 2).

Pre- and post-dimethyl fumarate analysis: % patients with new T2 lesions.
Among WA, 25% of patients demonstrated Gd+ lesions 1 year prior to start of DMF, and 13% post-DMF (p = 0.0003); in AA pre-DMF, 25% had Gd+ lesions and 7% post-DMF (p = 0.01); and in HA pre-DMF, 23% had Gd+ lesions and 13% post-DMF (p = 0.08) (Figure 3). Overall, 12% of patients demonstrated Gd+ enhancing lesions on MRI while on DMF. Comparing WA with AA, adjusted odds ratio of Gd+ lesion reduction was 0.36 (95% CI, 0.129 and 0.984;p = 0.05) and comparing HA to WA, adjusted odds ratio was 0.61 (95% CI, 0.23 and 1.57;p = 0.30) (Table 2).
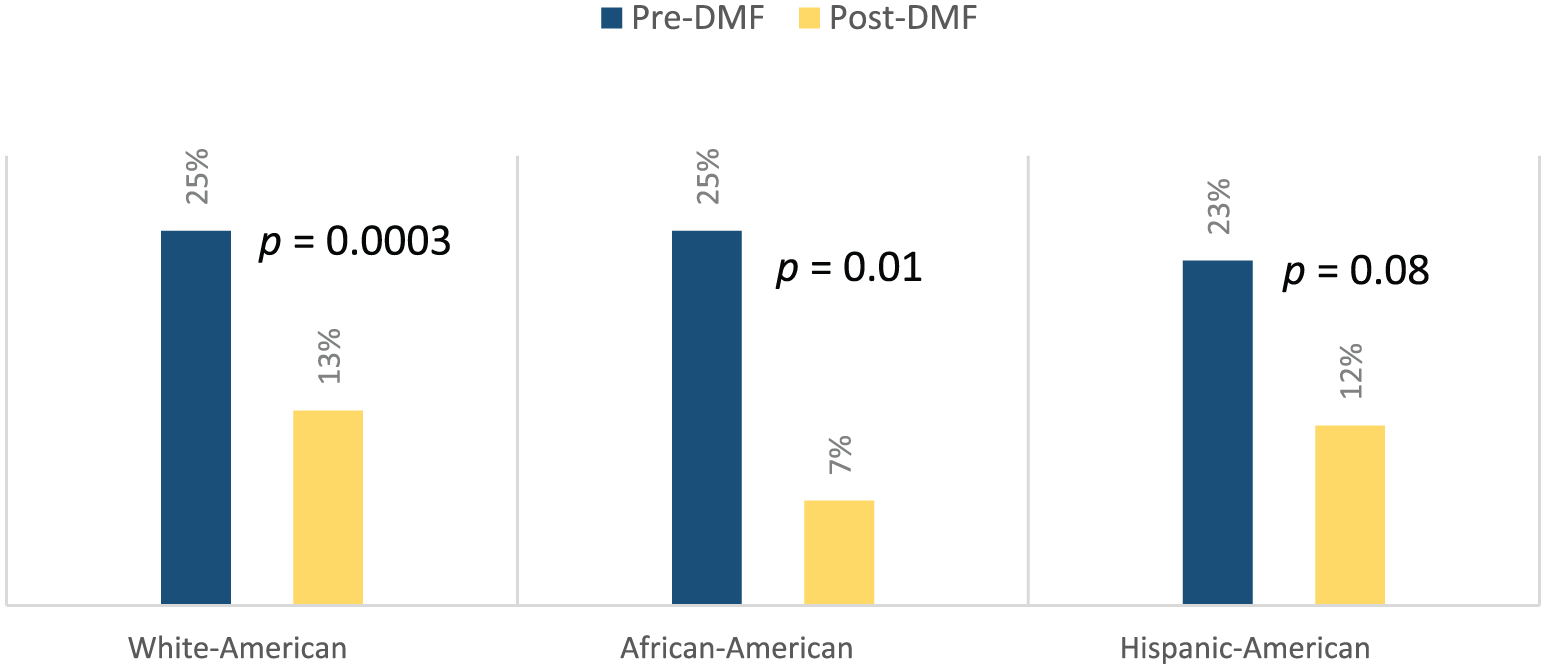
Pre- and post-dimethyl fumarate analysis: % of patients with GD+ lesions.
Discontinuation rates and adverse events
The mean duration of treatment with DMF was 14 months, with no statistical difference in treatment duration between the groups: WA 14.3 months; AA 14 months (p = 0.57 compared with WA); and HA 13 months (p = 0.55 compared with WA). Percentage of patients on DMF for less than 12 months was 28% in WA; 31% in AA; and 36% in HA. Overall DMF discontinuation rate was 20%, with no statistical significant difference between groups: 19% for WA, 15% for AA (p = 0.40, compared with WA) and 29% for HA (p = 0.12, compared with WA) (Table 1). Average time on DMF prior to discontinuation was 8 months. Main reasons of discontinuation including gastrointestinal (GI) side effects (25%); flushing (16%); clinical or radiographic relapse (26%); lymphopenia (9%); and other reasons, such as pregnancy and insurance loss (33%). Overall, 65% of patients experienced one or more side effects. AA reported the lowest rate of side effects: 51% of patients (p = 0.004 compared with WA), and the highest in HA, 71% (p = 0.78 compared with WA), Table 3. The most common side effects were GI related: nausea (22%), vomiting (10%), diarrhea (19%), abdominal pain (20%) and reflux (8%). Facial flushing was reported by 66% of patients, with 72% of WAs reporting this symptom as compared with 54% AAs and 51% HAs. The AA and HA groups had higher rates of itching (31% for AA and 21% for HA) as compared with WA (10%), Table 3. No statistical significant difference was seen n comparing individual side effect frequencies in the WA versus AA and HA groups.
DMF Side effects.

Comparisons made versus Whites.
Review of the absolute lymphocyte counts (ALC) in the groups revealed a statistically significant drop in ALC (p < 0.0001) in the WA group; a trend to significance in AA (p = 0.051); and a nonsignificant decrease in HA (p = 0.15) after commencement of DMF (Figure 4). Significantly more WA patients developed grade II or higher lymphopenia (ALC < 800/µl) compared with AA (p = 0.04, Fisher exact test), and HA (p = 0.007, Fisher exact test). No specific DMT use prior to DMF commencement was identified as a risk for developing leukopenia. Mean time to first lymphopenic result was 404 days in WA, 324 days AA and 396 days in HA, with no significant differences noted between the three groups. Patients who developed grade II or higher lymphopenia (ALC < 800) were significantly older than patients who did not develop lymphopenia (p<0.001 for each of the three groups, with average age of lymphopenic patients being 51.2 years for WA, 52.0 years for AA, and 52.3 years for HA.
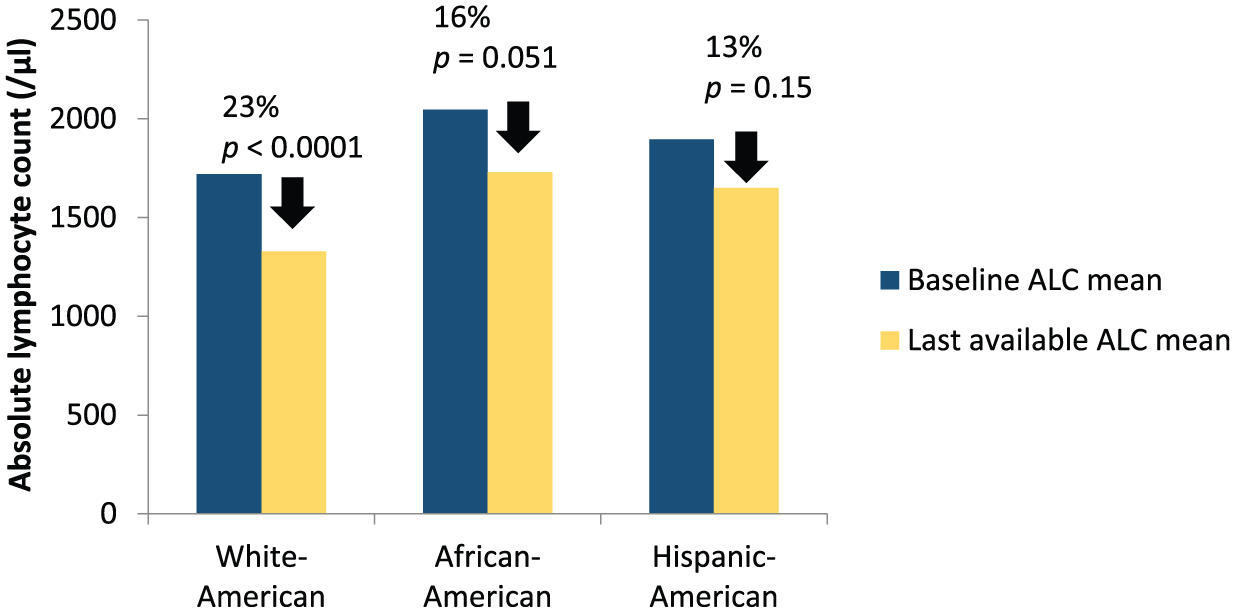
Lymphocyte count changes on dimethyl fumarate.
Discussion
Patients of three major racial subgroups represented in our clinics, WA, AA and HA, experienced similar relapse rate of 0.20–0.16 during mean on DMF follow up of 14 months on DMF. These results are consistent with ARR reported in clinical trials (CONFIRM, 0.22) [Fox et al. 2012] and (DEFINE, 0.17) [Gold et al. 2012], though such comparisons should be interpreted with caution given that patient selection for DMF in a ‘real-life’ setting is different from that of clinical trials. Importantly, our study showed DMF to be equally efficacious in reducing relapses and MRI activity among AA and HA with RRMS, the two patient groups that were under-represented in pivotal clinical trials. Reductions in rates of annualized relapses, new T2 lesions, and Gd+ lesions seen, when comparing pre-DMF (1 year) and on-DMF treatment period (mean follow up of 14 months), were of similar magnitude in all three groups. (Although, regression to the mean may be partly responsible for the absolute magnitude of the decrease in rates of relapses or new lesions on treatment, this effect would not be expected to differentially affect reduction in rates among the three groups, which all had similar pre-DMF disease activity.) Furthermore, statistically significant reductions in rates of annualized relapses and new T2 lesions were seen in three groups, although Gd+ lesion reduction was not seen in HA, presumably related to a lower number of HA patients in the study (n = 52) as compared with WA (n = 261) and AA (n = 69). Statistically significant decreases of Gd+ lesions were seen in the WA and AA groups.
DMF was relatively well tolerated in our clinic with 20% discontinuation rate. The discontinuation rate was lower than what was seen in clinical trials (CONFIRM, 29%) [Fox et al. 2012] and (DEFINE, 31%) [Gold et al. 2012]. These data suggest that, in ‘real life’, lower discontinuation rates than what have been seen in clinical trials is achievable. Importantly, discontinuation rates did not differ across races, contrary to the well documented reports of racial disparities in compliance with therapy [Saha et al. 2008]. The relatively low discontinuation rate may be related shorter follow up period as compared to clinical trials, or to extensive patient education by clinicians and nurses at our centers, and a slower and more flexible titration protocol than in the pivotal trials. The titration protocol typical recommended for our patients is 120 mg daily for 14 days, 240 mg daily for 14 days, followed by the full dose of 240 mg twice a day. GI symptoms and facial flushing were the most common side effects of DMF in our patients, in line with the known adverse-event profile of the drug. Although no statistical significant difference was seen in comparing individual side effects in the WA versus AA and HA groups, AA did report increased itching and less facial flushing as compared with WA and HA. Perhaps a darker skin pigment makes the AA less aware of facial flushing, but leads them to be more aware of the pruritis.
Lymphopenia was listed as a cause for DMF discontinuation for 9% of our cohort. Presumably, clinicians stopped DMF in these patients because of concerns about DMF-induced progressive multifocal leukoencephalopathy (PML) [Rosenkranz et al. 2015] in patients with persistent lymphopenia [Khatri et al. 2015]. ALC drop was seen in all three groups, with statistical significance seen in WA, with AA trending towards significance. A small number of patients in the AA and HA groups may be a limiting factor in our ability to detect significance. Older age does appear to be a risk factor for developing lymphopenia on DMF therapy, a finding previously reported [Longbrake and Cross, 2015; Foley et al 2016].
The main limitations of this study are the retrospective design and lack of a prespecified definition of relapse, which was left to the treating physician’s discretion. Furthermore, MRI analysis was based on radiologist reports at our institutions, and no centralized review of MRI was utilized. Nevertheless, the lack of formalized relapse definition or a central MRI reader would not be expected to introduce any systematic biases when comparing efficacy of the drug across ethnic groups. Another limitation is that disability was not systematically collected in our study.
In conclusion, efficacy of DMF in our clinic population was comparable with the reports of pivotal studies and, importantly, was similar in magnitude across WA, AA and HA groups. DMF was well tolerated in all groups, with overall drug survival rate of ~80% over a 14-month period. This ‘real life’ data provide preliminary support that race is not a factor that needs to be taken into account when considering DMF in routine practice.
Footnotes
Acknowledgements
Y. C. Ma from PharmStats carried out on the statistical analysis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was supported through Investigator Initiated Trial Grant from Biogen Idec. Sponsor was not involved in data collection, analysis, writing or editing this manuscript.
Conflict of interest statement
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LZR has received research support from Biogen Idec. She has received compensation for advisory board and speaker activities for Biogen Idec and Teva. KP has received speaker and consulting fees from Acorda , TEVA, and Biogen Idec. CS has received compensation for advisory board and speaker activities for Biogen Idec. IK has served on scientific advisory board for Biogen Idec and Genentech. Received research support from Guthy-Jackson Charitable Foundation, National Multiple Sclerosis Society, Biogen-Idec, Serono, and Novartis. The other authors have no conflicts of interest to declare.
