Abstract
Dimethyl fumarate (DMF) is a widely used oral disease-modifying therapy for multiple sclerosis (MS). Its efficacy and safety profiles are supported by over a decade of experience. Differences exist between Asia and Europe/United States in the prevalence and characteristics of MS; most data for DMF are derived from populations outside Asia. DMF was recently (2021) approved for use in China. The objectives of this review were to evaluate the evidence for DMF’s profile, to provide an update to healthcare providers on current knowledge surrounding its use and to assess the relevance of existing data to use in China. This study used a modified Delphi method based on the insights of a scientific Steering Committee (SC), with a structured literature review conducted to assess the data of DMF. The literature review covered all papers in English (from 01 January 2011 to 21 February 2022) that include ‘dimethyl fumarate’ and ‘multiple sclerosis’, and their MeSH terms, on PubMed, supplemented by EMBASE and Citeline searches. Papers were categorized by topic and assessed for relevance and quality, before being used to formulate statements summarizing the literature on each subject. SC members voted on/revised statements, requiring ⩾80% agreement and ⩽10% disagreement for inclusion. Statements not reaching this level were discussed further until agreement was reached or until there was agreement to remove the statement. A total of 1030 papers were retrieved and used to formulate the statements and evidence summaries considered by the SC members. A total of 45 statements were agreed by the SC members. The findings support the positive efficacy and safety profile of DMF in treating patients with MS. Limited Chinese patient data are an ongoing consideration; however, based on current evidence, the statements are considered applicable to both the global and Chinese populations. DMF is a valuable addition to address unmet MS treatment needs in China.
Introduction
Differences in the incidence, prevalence, presentation and prognosis of multiple sclerosis (MS) have been observed between East Asian populations and the rest of the world.1,2,3,4,5,6,7,8 For example, the prevalence of MS is estimated as 2.39/100,000 in China, 8.62/100,000 in Southeast Asia, 142.81/100,000 in Europe and 43.95/100,000 globally. 9
Dimethyl fumarate (DMF) is a fumaric acid ester used to treat MS.10,11,12,13 DMF (Tecfidera®; Biogen-Idec, Cambridge, MA, USA) was approved for treatment of MS in the United States in March 201314,15 and the European Union (EU) in January 2014 (starting with 120 mg oral capsules, administered twice daily for 7 days, followed by a twice daily 240 mg maintenance dose).10,15 In Asia, DMF was approved in South Korea in July 2016 and in Japan in December 2016.16,17 DMF has been approved in other Asian locations including, but not limited to, India (February 2015), Hong Kong (March 2016), Singapore (July 2016) and Thailand (March 2020).18,19,20,21 DMF is a widely used oral disease-modifying therapy (DMT) for MS, 22 having been prescribed to approximately 580,500 persons globally in the clinical trial and post-market settings, corresponding to 1,267,327 person-years of exposure through 30 June 2022 (data on file; published 2021 data), 23 and was approved for use in the People’s Republic of China in 2021. 3
Although there are more than 13 years of DMF data, including long-term follow-up from its pivotal clinical trials (DEFINE, CONFIRM) and the long-term extension study (ENDORSE),24,25,26,27,28 comparatively little of this data has been derived from East Asia. 25 It is therefore relevant to consider whether DMF performs similarly in treating these populations, as elsewhere in the world.
Early treatment of MS with DMT is part of the strategy to improve patient prognosis. It is important to continually assess what is known about different DMTs in the light of additional clinical trials and post-marketing studies and real-world experience, to evolve clinical practice. For example, MS organizations and working groups have demonstrated the value of updating practice through agreed statements on the use of DMTs during COVID-19. 29 With the recent approval of DMF in China, it is an opportune time to review global and Asian DMF practice. Therefore, the objective of this work was to use a modified Delphi method to assess the current use of DMF in clinical practice and identify considerations relevant for its use going forward, in China and other countries, so that these serve as a valuable resource to sharing up-to-date evaluation of evidence supporting good clinical practice for treatment of MS.
Methods
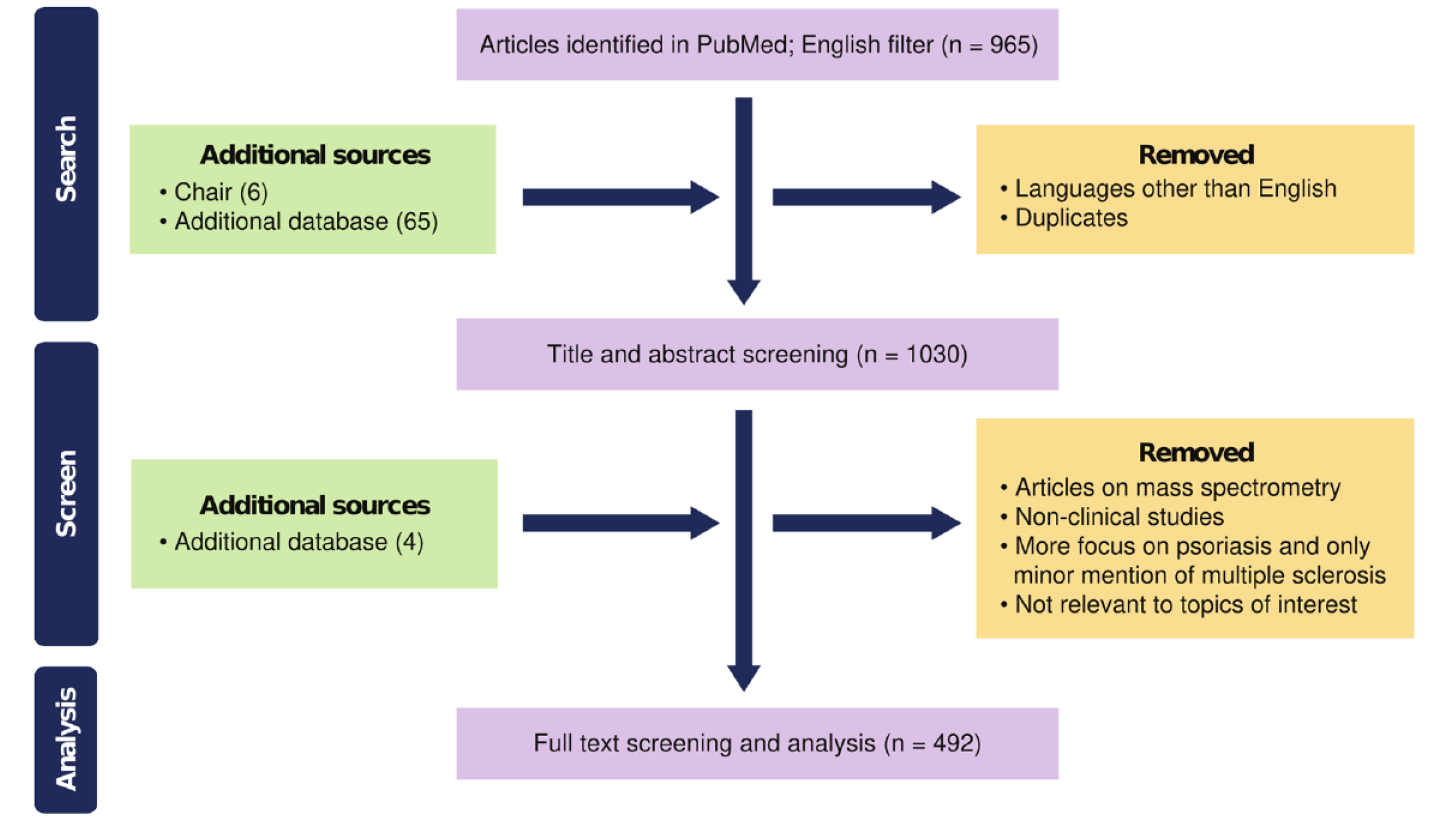
This study used a modified Delphi method (Figure 1) based on the insights of a scientific Steering Committee (SC). First, a structured literature review was performed to identify relevant evidence addressing the question, ‘How is DMF used in the treatment of MS?’ The literature review considered results from a PubMed search (from 01 January 2011 to 21 February 2022) including the core search terms ‘dimethyl fumarate’ AND ‘multiple sclerosis’ and their MeSH terms (search strings are detailed in Supplemental material; literature search strategy in Figure 2). Additional terms were used to categorize results into relevant topics.

Study flow for this modified Delphi process.
Literature search strategy.
The main search was supplemented by additional searches of EMBASE and Citeline. The topics of interest were categorized as: efficacy, safety, mechanism of action, pharmacokinetics/pharmacodynamics (PK/PD), treatment sequencing, adverse event (AE) management, infection, progressive multifocal leukoencephalopathy (PML) risk, effects on haematological parameters, effects on organ systems, drug-drug interactions, pregnancy/lactation, COVID-19 and paediatric populations.
The literature was first screened by title and abstract by editorial support staff from MIMS Limited for relevance and quality. Due to the known level of variation in the formulation of compounded products, 30 only papers using commercially produced DMF were included. Quality was based on the totality of several factors: type of publication (guidelines > systematic reviews > meta-analyses > opinion pieces > case reports), study design (randomized controlled trials (RCTs) > observational or cohort studies > case studies; and direct comparisons were considered superior evidence to indirect comparisons) and study size (more than 500 participants > fewer than 500 participants). Funding sources were also noted. The culmination of these factors was reflected in an overall evidence rating (high, medium high, moderate, medium low and low). Statements were developed based on the evidence and reviewed by SC members along with the supporting evidence and evidence rating.
The SC comprised 14 expert members (neurologists) representing clinical expertise from Germany (n = 1), United Kingdom (n = 1), Australia (n = 1), Spain (n = 2), Japan (n = 1), Switzerland (n = 1) and China (n = 7). SC members were selected based on their clinical experience with MS, publication record and any previous contributions to MS treatment guidelines. For practicality, topics were split into two sets: Set 1 topics, for Statements 8–24, 29–33 and 40–41, were discussed at the first meeting and Set 2 topics, for Statements 1–7, 25–28, 34–39 and 42–46, were discussed at the second meeting (Figures 1 and 3a, 3b, 3c). Each topic set underwent two or more anonymous voting rounds with thresholds for agreement of ⩾80% and disagreement of ⩽10%. Statements not meeting the criteria were discussed at one of two online SC meetings for revision (Figure 1).



Summary of voting responses and guiding statements on clinical use of dimethyl fumarate in treating multiple sclerosis.
Voting administration, collation of responses, collating meeting minutes and statement revision were conducted by the editorial support team to negate bias. In addition to ‘Agree’ and ‘Disagree’, SC members could indicate agreement ‘with revision’, with suggestions provided as free-text entries, which were collated for discussion with highlighting of the incorporated updates (examples given in Figure 1). Statements discussed during meetings were revised and re-circulated for a second round of voting. For statements with a response exceeding the disagreement threshold, the second round had a voting option for statement deletion. After two voting rounds for each statement set, statements requiring higher levels of agreement were revised and voted on in the same round until agreement was reached on either a revised wording or deletion of the statement. Where the level of disagreement was below the threshold, the statement was included, as such statements reflect majority opinion, although not necessarily unanimity. In situations where practice or evidence differed significantly between regions, statements about global and Chinese or East Asian populations are presented separately.
Results
A total of 965 papers were retrieved with the search terms and a further 70 distinct references (one duplicate was removed) were used to supplement the search from additional sources. The number of participants voting in each round and the levels of agreement attained are summarized in Figures 1 and 3a, 3b, 3c, respectively.
In the first round of voting on Set 1 statements (expressed as per cent responses on total number of votes received on all statements), there was 81% agreement, 13% agreement with revisions, 3% requesting further discussion at the meeting and 3% disagreement. The level of overall agreement increased to 88% by the second vote, with a further 6% agreeing with revisions (4% disagreement and 2% suggested deletions).
For the Set 2 statements, at round 1 there was 70% overall agreement, 17% agreement with revision and 12% disagreement. After the second vote there was 94% overall agreement on the statements, 5% agreement with revision and 1% disagreement.
The most common reason for disagreement in both sets was a perceived lack of supporting evidence. Agreement with revision was typically for the correction of language-related issues such as simplifying wording. In total, 45 statements were finally included (Figure 3a, 3b, 3c).
Mechanism of action
Two statements were proposed on the mechanism of action of DMF. DMF’s effects on the immune system are complex and multi-faceted (Figure 3a, 3b, 3c, Statements 1 and 2); DMF influences both B cell and T cell populations and pro-inflammatory cytokine production to reduce inflammatory activity.31,32,33,34,35,36,37 One of the main pathways of activity of DMF is via nuclear factor erythroid 2–related factor 2 (Nrf2), which has anti-oxidative and anti-inflammatory effects.13,38,39,40,41,42 The anti-oxidative and anti-inflammatory effects of the Nrf2 pathway may contribute to the cytoprotective abilities of DMF.38,39 DMF may prevent demyelination and axonal loss through reduction in pro-inflammatory reactive astrocytes. 43 Changes in immune cell populations, including a reduction in pro-inflammatory Th17 cells and increase in Th2 naïve/anti-inflammatory cells, induced by DMF produce a net shift away from a pro-inflammatory immune state. 44 Reductions in memory T and B cells similarly reduce overactive immune responses and the resulting neuronal damage in MS.36,44
PK/PD and dosing
Upon ingestion, DMF is rapidly metabolized to monomethyl fumarate (MMF), which can cross the blood-brain barrier to exert its effects (Figure 3a, 3b, 3c, Statements 3–6).39,40,45 MMF does not accumulate in the body, owing to a short terminal half-life of ~1 h. 45 A high-calorie and fat-rich meal delays Tmax by hours and reduces Cmax by approximately 40%. 45 However, area under the curve is not affected by taking DMF with a meal, which enables DMF to maintain efficacy, while reducing side effects.10,45 The impacts of having a meal on Tmax and Cmax reduce both gastrointestinal (GI) AEs and flushing, thereby alleviating DMF’s most common undesired effects.46,47 Patients with severe renal or hepatic dysfunction were excluded from the pivotal clinical trials of DMF. However, neither condition is expected to influence the level of exposure to MMF.10,12
Situation in China/East Asia
The studies in East Asian patients used the same dose regimen and reported a consistent AE profile for DMF, as the pivotal trials, which largely included Caucasian patients.16,48,49,50 No change in dosing is required for this population.
Efficacy statements
Three efficacy statements were included (Figure 3a, 3b, 3c, Statements 7–9). The overall level of evidence for the efficacy of DMF was considered high, albeit with varying ratings for individual parameters. For relapse rate/annualized relapse rates (ARRs), radiological evidence of disease activity and disability progression, the data are derived from several large RCTs, meta-analyses and systematic reviews,25,26,27,28,49,51,52,53 and have been incorporated into guidelines from all major MS organizations, including in Japan.16,54,55,56 The pivotal clinical trials most convincingly demonstrated improvements in relapse rate/ARR with DMF treatment.25,26,27,28 Long-term data support that DMF treatment results in reductions in disability progression, with few overall relapses, but members of the SC considered this to have less strong support than the stated effects on ARR. 57 Radiological evidence of disease activity is perhaps the least well-supported parameter of this set, given more variability in magnetic resonance imaging (MRI) assessment across centres and studies and therefore a lower quality of evidence. 51 Despite the varying level of evidence for different aspects of efficacy, the overall position of the SC is that data exist for improvements on each of these parameters.
The importance of initiating a DMT soon after diagnosis is highlighted in several international guidelines54,55 and is supported by convincing evidence from both individual trials (including DMF specifically) and reviews.58,59,60 The SC noted the strong evidence for this statement and encourages early, appropriate initiation of a DMT.
Serum neurofilament light chains (sNFLs) are a marker of neuroinflammation and are thought to represent a proxy measure for axonal damage in MS. 61 sNFL is reduced by DMTs in MS, 62 and lower baseline sNFL levels in a small (n = 80) observational trial predicted a greater response to DMF treatment, assessed by achieving no evidence of disease activity (NEDA) and reducing effector immune cells. 61 The study concords with a slightly larger trial (n = 127) which showed that DMF reduced NFL in the blood and cerebrospinal fluid (CSF) of treatment-naïve patients and could normalize the CSF levels in 73% of participants. 63 As a comparatively new outcome measure with limited use in clinical practice, sNFL relevance for prognosis or disease outcomes requires further research. Nonetheless, DMF has shown some effects on this marker, and it may be a potential proxy measure for treatment response or prognosis.
Situation in China/East Asia
Evidence for efficacy of DMF in the East Asian population comes primarily from Japan, which contributed the majority (80%) of the East Asian population (n = 142) in the APEX study, followed by South Korea (14%).48,49 A post hoc sub-analysis of Asian data from Fox et al. 2 has also been performed, confirming treatment efficacy and positive benefit-risk balance. In general, treatment results are similar between East Asian and Caucasian/rest-of-the-world populations, although there is lower incidence of MS but potentially poorer prognosis in Asian populations, possibly due to increased frequency of spinal lesions in this population and incomplete resolution of first attacks. There is no predicted difference for Asian populations which would lead to preferring later treatment initiation, and considering a higher potential first-year relapse rate and poorer prognosis,2,64 it may be high priority to initiate treatment of Asian patients early in the clinical course. Many patients with MS in China do not initiate a DMT at the earliest opportunity 4 for reasons of cost, 3 perception of the condition as ‘mild’ and the advice of neurologists and other healthcare professionals.3,4 The treatment pattern in China continues to evolve, with early diagnosis and sustained use of DMTs during periods of remission becoming more common. 3
Haematologic parameters and infection risk
Seven statements on haematological parameters and infection risk achieved agreement (Figure 3a, 3b, 3c, Statements 10–17). In general, the risk of lymphopenia with DMF was recognized – but also that this did not translate to a substantial increase in risk of infections. Very rare occurrences of PML warrant clinical vigilance.
Lymphopenia
DMF studies consistently show a ~30% decline in mean absolute lymphocyte count in treated patients, which typically stabilizes within 6–12 months of treatment initiation.48,65,66,67 Lymphopenia is a risk factor for (serious) opportunistic infections and associated mortality in the general population.68,69 For this reason, it is important to monitor lymphocyte counts in patients receiving a therapy which may induce lymphopenia, such as DMF.70,71 However, the degree of lymphopenia induced by DMF has not been associated with increased incidence of serious infections,44,65,72 despite some case reports and database review findings of opportunistic infections.69,73,74,75,76,77,78,79,80 Degree of lymphopenia has also been proposed by some to predict better responses to DMF, as a proxy measure for a less overactive immune response and pro-tolerogenic profile,44,70,71,81,82 but most of the data have come from relatively small studies. The data are equivocal, as some studies (e.g. Longbrake et al. 31 ) including larger controlled trials have found no relationship between lymphopenia and therapeutic efficacy of DMF.31,44,65 It has also been determined that CD4+ and CD8+ T cell counts correlate with absolute lymphocyte counts, suggesting that separate monitoring is not required. 67 After treatment discontinuation, most patients with lymphopenia experience recovery of lymphocyte count; the extent and duration of lymphopenia influence the time required to attain this. 83 In the opinion of the SC, the data are insufficient to conclusively link lymphopenia with therapeutic efficacy, and lymphopenia is not recommended for use as a prognostic factor.
Vaccination response
Potential suppression of the immune system by DMTs is a risk factor not only for infections but also for a blunted response to vaccination, resulting from a reduced humoral and/or cellular response.29,84,85 The PROCLAIM study assessed immunoglobulin levels in patients initiating DMF and found no impact of DMF on immunoglobulin levels. 44 Similarly, immune response to vaccination was found to be similar between DMF and interferons.86,87 The SC therefore supports the conclusion that DMF does not interfere with vaccination.
Tuberculosis
Active tuberculosis infections need to be excluded before starting DMF treatment. In the EU/European Economic Area (EEA), tuberculosis has been estimated to occur at approximately 9.6 cases per 100,000 population (2019). 88 In comparison, the rates in China are estimated to be as high as 58 per 100,000 population, with substantially higher rates of latent infection (2019). 89 In general, screening for latent tuberculosis may be considered before initiating DMF;90,91,92 however, DMF is not one of the DMTs for which screening is mandatory.90,91,92,93 The risk for reactivation of latent tuberculosis infection appears to be low,91,93,94 and the decisions regarding testing ultimately lie with the treating physician.
Progressive multifocal leukoencephalopathy
Several DMTs have been associated with PML, caused by reactivation and viral replication of John Cunningham Virus (JCV) in glial cells, which then causes demyelination.23,95,96 PML is very rare and, in the cases that exist, occurs more commonly in immunocompromised patients. 95 As of 1 September 2022, there have been 12 cases of PML reported in approximately 580,500 patients treated with DMF, a rate of ~1/50,000 patients (0.9/100,000 patient years of DMF exposure)23,95 (data shown: on file; previous data referenced). 23
Prolonged Grade 3 lymphopenia is a risk factor for PML and a reason to discontinue DMF therapy.29,97 Based on reported trial data, it is estimated that around 5–10% of patients treated with DMF will experience Grade 3 lymphopenia, and 2.2% of patients will have prolonged (⩾6 months) Grade 3 lymphopenia.65,98 However, only a few cases have been reported of patients experiencing complications from prolonged severe lymphopenia.65,97,99,100 Age ⩾50 years is another risk factor for PML; however, neither age ⩾50 years nor severe prolonged lymphopenia are absolute identifiers of those at increased risk of PML, with recorded case reports in patients without these characteristics.95,101,102,103
Closer monitoring of patients with a higher risk of PML is recommended. This includes monitoring for lymphopenia23,29 and regular (every 6–12 months) MRI scans.16,103 Discontinuing the suspected causative agent is recommended when patients develop PML. 104
Situation in China/East Asia
The high incidence of latent tuberculosis infection in China89,105 and infrastructural challenges in the availability of testing pose a practical obstacle to implementing routine screening before initiating DMF. However, as the risk of reactivation is considered very low,91,93,94 the recommendation for China is that screening for latent infections is not mandated.
Through the risk stratification process for PML with natalizumab, clinicians in China may be aware of the use of the JCV antibody index. Based on high JCV antibody positivity detected in MS patients from Hong Kong, JCV seropositivity is expected to be high in China. 106 However, JCV seropositivity does not play a role in risk stratification for DMF. For clinical practice in China, it is important to note that JCV index testing is not required with DMF; age and lymphopenia are sufficient criteria for determining PML monitoring.16,29,95 Clinical vigilance for this condition remains very important, despite its rarity.
Comorbidities, organ function and cancer risk
Four statements on comorbidities and organ function were approved, mostly with a focus on cardiovascular and hepatic safety (Figure 3a, 3b, 3c, Statements 18–22). As MS causes progressive disability, patients are encouraged to maintain a healthy lifestyle, including a balanced diet combined with an exercise programme at a tolerable level. 107 Patients with MS are at higher risk of several forms of cardiovascular disease, including myocardial infarction and heart failure. 108 DMF is not associated with cardiovascular AEs such as arrhythmia or hypertension and is unlikely to contribute to cardiovascular morbidity in patients with MS.109,110
Drug-induced liver injury is a recognized risk for patients receiving DMTs for MS. 111 Alanine transaminase (ALT)/aspartate aminotransferase (AST) elevations have been reported in major trials of most DMTs, including DMF.25,26,48,50,112,113 With DMF, persistent or severe ALT/AST derangement or discontinuation due to liver toxicity occurred in <1–2% of clinical trial participants.28,112 Post-marketing evidence has identified ALT/AST elevation as relatively common; however, outright liver injury/hepatotoxicity has been recorded in very few DMF-treated patients (n = 4–14).109,112 In light of the low risk of liver toxicity with DMF, periodic laboratory testing (up to regular 6-monthly testing) is sufficient to monitor for liver toxicity.29,112 To date, the number of reported cases of hepatotoxicity with DMF is limited to 14, plus one case of acute hepatitis E virus infection.112,114,115 Of those 14 cases, the earliest elevations of ALT/AST were recorded shortly after treatment initiation; liver injury was mild (six patients) or moderate/severe (eight patients) and there were no cases of liver failure. 115
In patients with liver disease, DMF may be used with caution, at its normal dosage, as it is metabolized independently of the cytochrome P450 system. 112 Any risk of exacerbation or reactivation of hepatitis B virus (HBV)/hepatitis C virus (HCV) infection is mitigated in part by screening for these infections before initiation of most DMTs. 112
Situation in China/East Asia
HBV and HCV are more common in China than in the EU, 116 meaning that the requirement to screen for these viruses may yield positive results more frequently. With positive results, antiviral therapy and adjustments to the initiation of DMT should be considered; specialist assistance from hepatology or infectious disease colleagues is required to determine the appropriate course of action. 112
Cancer risk
A single statement was proposed for cancer risk (Figure 3a, 3b, 3c, Statement 23). An increased risk of malignancy in patients with MS has been investigated repeatedly, including in those with long-term use of DMTs. 117 Overall, the malignancy rate in patients treated with DMF is similar to age- and sex-matched individuals without MS. 118 Duration of DMF treatment was concluded as a risk protective factor against neoplasms, in a small Spanish study. 119 Recent database analyses have produced conflicting results, with one finding no evidence of an increased cancer risk 120 and the other being the first publication to find an increased risk of neoplasm with DMF. 121 Consequently, it may be prudent to be vigilant in checking for cancer in DMF-treated patients, but this does not yet support a malignancy rate higher than the general population. 118
Treatment initiation and decision-making
Location and licencing determine the permitted indications for DMF. If permitted by the local label, DMF can be used in both patients with relapsing-remitting multiple sclerosis (RRMS) and patients with clinically isolated syndrome (CIS). 12 The efficacy and tolerability of DMF are well balanced, and it is suitable as an initial therapy.27,28,122,123,124,125,126,127,128,129,130,131,132 The SC proposed four statements on this topic (Figure 3a, 3b, 3c, Statements 24–27).
Early-in-treatment challenges with tolerability are the largest reason for the discontinuation of DMF.133,134 To mitigate the risk of AEs, a more gradual up-titration to the target dose may be considered for up to 4 weeks.10,135,136,137 It is important to note that the full dose is required for efficacy. Positive relationships between healthcare providers and patients are an important part of goal setting, partly through enhancing adherence. Self-management by patients may be improved by participation in a shared decision-making process. 138 Treatment selection will partly depend on the goals of the patient.138,139,140,141 These aspects of positive patient relationships may help to enhance tolerability when starting a new therapy. As treatments for MS have advanced, the achievable treatment goals/targets have evolved. NEDA-3 (no relapses, no new radiological evidence of disease activity, no disability progression) is the current benchmark proxy for best disease control.142,143,144,145 It may be beneficial to explain this as a target to patients as part of the goal-setting process.
Treatment switching
Five treatment switching statements were approved (Figure 3a, 3b, 3c, Statements 28–32). Evidence for the profile of DMF was initially established in comparison with both placebo and glatiramer acetate in the DEFINE, CONFIRM and ENDORSE trials.25,26,27,28,113 ENDORSE re-randomized patients previously treated with glatiramer acetate to DMF, with similar efficacy and tolerability results to those reported in treatment-naïve patients. 28 Outside pivotal clinical trials, these findings are further supported by observational studies of patients switching from other first-line therapies to DMF.122,124,132,146,147,148,149,150,151 Based on these results, horizontal switching, especially from injectable therapies, is a viable use of DMF.
Early (versus later) initiation of an effective DMT is linked to better long-term outcomes for patients with MS (see efficacy statements, Figure 3a, 3b, 3c, Statements 7–9), exemplified by achieving NEDA-3 with DMF.49,54,55,58,151 Treatment inertia may delay switching, and latency to initiation of an effective therapy may result in poorer outcomes, meaning that in addition to initiating DMTs early, switches should be considered if a therapy has proved intolerable or has shown a lack of efficacy in the judgement of the treatment team.29,142
When switching to a new therapy it is important to consider its characteristics, to properly prepare the patient for the switch. 56 The most common AEs of DMF treatment are GI symptoms and flushing.26,46,152 Discontinuation of DMF is more commonly attributable to tolerability issues (68% of discontinuing patients cited this as the main reason for discontinuation) than to a lack of efficacy (15% of patients).134,153 However, a study of 886 patients in Spain reported similar rates of DMF discontinuation as a consequence of AEs and lack of efficacy (13.2% and 13.5% of patients, respectively).133,150 Side effects resulting in discontinuation most commonly occur early in treatment,153,154 suggesting that particular attention can be paid to mitigating AEs and promoting tolerability in the early period of treatment. Rarely, discontinuation may be due to lymphopenia. As a general principle, if a therapy is discontinued for a particular AE, switching to a therapy with a similar AE profile may not be the most appropriate choice. With DMF, limited evidence from a small observational study has suggested that lymphocyte counts may not recover in patients who discontinued therapy and switched to other lymphodepleting agents, because of lymphopenia. 155 Out of caution, such switches should be avoided if possible. A United States study reports that most patients who discontinued DMF with lymphopenia experienced lymphocyte count reconstitution. 83
Situation in China/East Asia
In China, as many as 58% of patients do not initiate DMTs upon their diagnosis of MS. 3 The reasons include cost, a historical lack of available DMTs and low patient acceptance of therapy for what they may erroneously perceive as a ‘mild’ condition. 4 Many patients will therefore be initiating DMF treatment while DMT-naïve. There are no contraindications to initiating therapy with DMF based on prior steroid or other therapies,10,12 but the timing of initiation may depend on satisfactory condition of the patient, as judged by the neurologist, for example without lymphopenia from corticosteroid or azathioprine use.
Adverse event management and tolerability
As with treatment initiation, important considerations for AE management and tolerability are counselling, shared decision-making and mitigation strategies (Figure 3a, 3b, 3c, Statements 33–36). Forward planning, counselling and expectation setting all assist adherence with DMF by improving tolerability.46,136,137,156,157 Involving patients in goal setting and integrating their preferences into therapy management is beneficial for initiating DMTs in MS.136,156
Mitigation of AEs primarily involves either GI symptoms or flushing. 46 Symptomatic therapies for GI AEs include proton pump inhibitors, antihistamines and assorted other agents for nausea such as metoclopramide, domperidone, diphenhydramine and dimenhydrinate.46,135 Gradual up-titration (over 4 weeks) or temporary reductions in dose when an AE is encountered help to mitigate the risk of discontinuation.136,137 The goal is still to achieve the full dose, consistently, as it is required for efficacy. 46 Taking DMF with food continues to be recommended to mitigate GI AEs.136,157,158
For flushing, aspirin (75–325 mg) daily, or every second day, can be used typically as pre-treatment 30 min before DMF.11,46,136,159,160 Antihistamines are only rarely required but can also be used.136,137 Metoprolol, acetaminophen, ibuprofen, cosmetics and counselling have been used for flushing by at least one respondent in a consensus statement. 46
There have been few reports of hypersensitivity in the literature, despite large numbers of patients treated with DMF.161,162,163,164,165 Where they have occurred, management has shown a response to steroid and antihistamine treatment.
Concomitant therapies
Two statements summarize DMF use with symptomatic treatment (Figure 3a, 3b, 3c, Statements 37 and 38). Corticosteroids are commonly used in the treatment of acute relapses in patients with MS.16,54,145 While DMTs aim to avoid relapses, if they occur it is important that the DMT does not prevent acute or symptomatic therapy from being undertaken. DMF has no known drug-drug interactions,10,11,12,166 including with single doses of interferon beta-1a (IFN-b1a) and glatiramer acetate, or aspirin. The former characteristic facilitates switching, whereas the latter facilitates symptomatic mitigation of flushing symptoms. 46 GI adverse effects and flushing are the most common side effects with DMF. 46 Symptomatic therapies such as antihistamines (flushing), proton pump inhibitors (GI) and aspirin (flushing), among others, can be used to mitigate these AEs and improve tolerability.46,137,152 Aspirin mitigates flushing without increasing GI AEs. 159
In total, the lack of drug-drug interactions means that AEs and relapses may both be managed without concern for the patient’s DMF use.
COVID-19 and vaccinations
The use of DMTs during the COVID-19 pandemic has been an important and vibrant topic of clinical consideration. The SC considered two statements on this topic and was aligned with recommendations from numerous societies worldwide (Figure 3a, 3b, 3c, Statements 39 and 40). First, patients treated with DMF have not shown an increased risk of severe COVID-19 infection. 167 It is recommended that DMT selection should continue to be based on the activity and severity of the MS 29 and that the risks from MS outweigh those of COVID-19 in most cases.85,168,169 Patients with MS may experience a diminished response to vaccines, depending on the DMT which they are receiving and its mechanism of action.29,87,170,171 The patient response to vaccination, including for COVID-19, has been found to be intact during treatment with DMF, and the AE profiles of the influenza and COVID-19 vaccines have not been shown to be influenced by DMF.29,87,172,173 DMF does not interact with vaccines for COVID-19 and patients taking DMF should continue therapy, independent of vaccination timing.29,167 Data attained with influenza vaccination suggest that vaccine use does not increase the risk of subclinical disease activity in DMF-treated patients. 87
Pregnancy/lactation
Overall, the lack of accumulation of DMF/MMF makes it suitable for use in proximity to pregnancy, although it should still be discontinued during pregnancy (Figure 3a, 3b, 3c, Statements 41–44).
DMF has shown some developmental toxicity at clinically relevant doses in animal studies but not in human populations.174,175,176,177,178,179,180 In the United States, DMF is pregnancy category C, meaning it should be discontinued when a patient wants to become pregnant.10,11,12,55,175 In humans, there is no evidence of reduced fertility or increased risk of congenital malformation or miscarriage rates with inadvertent exposure to DMF in pregnant patients within the first trimester.174,175,176,177,178,179,180 Clinical evidence is limited, meaning that the characteristics of DMF’s formulation contribute to its recommended use. Namely, DMF does not accumulate, so no washout period is required before patients who have been treated with DMF wish to attempt pregnancy. 181
There are very few published works considering the issue of DMF during breastfeeding. Any potential use of DMF during breastfeeding will need to balance the benefits and potential risks.10,11,12,182 The primary active metabolite of DMF (MMF) has been detected in small quantities in breast milk in a small case study of two lactating patients. 183 The authors concluded the risk to the infant was likely to be low, considering the low concentration levels, but there is too little evidence to be confident in this regard. 183
Aside from pregnancy and breastfeeding, avoiding pregnancy via contraception may be influenced by DMT choice. Norgestimate/ethinyl estradiol oral contraceptives have been studied in a small RCT in combination with DMF, without impact on the efficacy or tolerability of either class of agent. 166 These study findings accord with the DMF prescribing information, which indicates no drug-drug interactions.10,11,12 A United Kingdom consensus on MS and pregnancy also advises that DMF does not reduce the effectiveness of hormonal contraception. 179
Situation in China/East Asia
The DMF prescribing information in China notes that there are no adequate data on developmental risk in pregnant women. 10 The evidence above suggests that therapy should not be continued during pregnancy and that discontinuation of DMF can occur close to pregnancy confirmation and does not need to be stopped (washed out) for a long period in advance of attempts to become pregnant. 181 For breastfeeding, the Chinese label agrees that the risks and benefits of treatment on the mother and the breastfed infant should be considered when making treatment decisions. Family planning and fertility are very important considerations for female MS patients in China, but no differences in practice with DMF are likely based on the generated data.
Paediatrics
DMF has only limited data in paediatric populations, with trial data generated only from the FOCUS and CONNECTED studies of ~20 paediatric patients. In these trials, DMF performed similarly as in adult populations in terms of both efficacy and tolerability (Figure 3a, 3b, 3c, Statement 45).184,185 In the FOCUS trial, AEs that were common with DMF in paediatric patients included headache (n = 4, 18%), dysmenorrhea (n = 2, 9%), GI disorders (n = 12, 55%) and respiratory, thoracic and mediastinal symptoms such as cough (n = 3, 14%) and upper respiratory tract infection (n = 2, 9%). 184 Data from a limited number of case reports, cohort studies and retrospective observational studies support the similarity of DMF performance in paediatric and adult populations (Figure 3a, 3b, 3c, Statement 45).186,187,188,189 The CONNECT study, a phase III open-label randomized clinical trial of DMF (n = 78) against IFN-b1a (n = 72), has recently shown that DMF was well tolerated, with favourable MRI and adjusted ARR for DMF-treated paediatric patients; 190 noting it was published after this article’s evidence review and it will be considered more completely in future work.
Discussion
More than half a million patients have been treated with DMF since its launch in 2013 (data on file). 23 The statements agreed in this article were generated by reviewing over a decade of literature on DMF, spanning the original pivotal clinical trials through to the most recent post-marketing data. The data for DMF have remained largely consistent over time. 26 In terms of safety, no new signals have emerged since existing reviews. A small number of PML cases have accumulated, but the condition remains very rare (data on file). 95 The expert opinion of the SC, therefore, supports the positive efficacy and safety profile of DMF as a first-line and switch therapy agent in patients with RRMS and, if permitted by the local label, CIS.
The most recent estimates of MS prevalence in China are 2.39 per 100,000, meaning a potential MS population within the country of more than 34,000. 3 This represents a large new population of patients who could potentially be exposed to and/or benefit from DMF.
Despite over a decade of experience with DMF, data from ethnically Chinese populations comprise a small fraction of the existing data, 3 which presents challenges for understanding how DMF will be used in practice in this country. 48 Post-marketing data from within China will be valuable for further understanding the characteristics of DMF in this population. Important differences within China may include the background prevalence of relevant diseases such as hepatitis and tuberculosis,89,116 differences in the MS treatment armamentarium available, prevailing practices on patient acceptance of DMTs and healthcare professional attitudes towards MS management and its relative importance as a rare disease. 3 Monitoring infrastructure for serial (follow-up) neuroimaging and/or serology for hepatitis and tuberculosis may also be impacted by regional differences in availability and implementation priority.
The possibility of drug interactions with as-yet-unidentified Chinese medicine products cannot be categorically excluded, and the real-world use patterns of DMF along with the Chinese diet and cultural practices regarding traditional medicine will be a source of new data following its increased use in the country. Nonetheless, with the evidence generated to date, it is anticipated that DMF will behave similarly in the Chinese population as with the rest-of-the-world population. This is consistent with the confidence of the Chinese regulatory agencies in approving DMF for use in MS treatment, 10 based on similar predictions of maintained safety and efficacy in the population.
Obstacles to rational uptake in China will be confusion regarding the monitoring and testing requirements; for example, about whether JCV and latent tuberculosis testing need to be conducted. It should be made clear that neither JCV nor latent tuberculosis testing needs to be performed. Despite this, it is important that clinicians remain vigilant for PML risk alongside any other AEs.
The panel considered additional statements on the role of the gut microbiome in the aetiology of MS and potential influence on DMF efficacy but found insufficient evidence to proceed. This will be a topic of interest in the future as more evidence accumulates.191,192,193 Similarly, data from paediatric populations and in patients who are pursuing pregnancy or who are breastfeeding are also sparse; it is hoped that more data will be generated in these groups.
Thus, the limitations of the study include the sparsity of data in Asian patients as well as the level and quality of evidence in some areas, making it difficult to make conclusive recommendations on those topics. As more patients in China are treated with DMF and more experience is gathered, a repeated analysis and update of the information presented here should be conducted.
This project revealed a consistency of data suggesting DMF remains a valuable part of the MS treatment armamentarium. This evidence summary hopes to clarify all relevant aspects for more effective use in China.
Conclusion
A decade of experience with DMF continues to support its positive efficacy and safety profile in treating patients with MS. Although there is a paucity of high-quality evidence from China itself, the limited data that exist from East Asian populations support a similar profile and suggest that DMF will be a valuable addition to the MS therapeutic armamentarium and will help address unmet treatment needs in China. This review of the most recent and extensive previous literature to form conclusive summary statements is aimed to provide a relevant overview to all clinicians treating patients with MS, with the aims of optimizing care and patient outcomes.
Supplemental Material
sj-docx-1-tan-10.1177_17562864231180734 – Supplemental material for Clinical use of dimethyl fumarate in multiple sclerosis treatment: an update to include China, using a modified Delphi method
Supplemental material, sj-docx-1-tan-10.1177_17562864231180734 for Clinical use of dimethyl fumarate in multiple sclerosis treatment: an update to include China, using a modified Delphi method by Ralf Gold, Michael Barnett, Andrew Chan, Huiyu Feng, Kazuo Fujihara, Gavin Giovannoni, Xavier Montalbán, Fu-Dong Shi, Mar Tintoré, Qun Xue, Chunsheng Yang and Hongyu Zhou in Therapeutic Advances in Neurological Disorders
Footnotes
Acknowledgements
The Steering Committee authors acknowledge and thank all experts who contributed to the voting and discussion, refining the draft statements. Jeffrey Martin and Rachael Profit (MIMS, Hong Kong and Pte Limited) contributed to the literature search, project administration, visualization of the voting data and medical writing assistance to the manuscript preparation.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
