Abstract
Background:
Patients with relapsing–remitting multiple sclerosis (RRMS) may experience breakthrough disease despite effective interferon beta (IFNβ) therapy. Fludarabine (FLU) is a chemotherapeutic agent used in lymphoproliferative disorders that may be synergistic when combined with immunomodulatory therapy to control active multiple sclerosis (MS).
Objective:
The objective of this study was to explore the safety and tolerability of FLU versus monthly methylprednisolone (MP) in IFNβ-treated RRMS patients with breakthrough disease. Clinical and MRI effects of IFNβ-1a plus FLU were evaluated.
Methods:
Eighteen patients with breakthrough disease [⩾2 relapses over the prior year and ⩾1.0-point increase in Expanded Disability Status Scale (EDSS) score sustained for ⩾3 months] after >1 year of IFNβ therapy were enrolled in this prospective, open-label, randomized, proof-of-concept, pilot study. Patients received intravenous (IV) MP 1 g daily for 3 days and then were randomized to receive 3 monthly IV infusions of FLU 25 mg/m2 daily for 5 consecutive days (n = 10) or MP 1 g (n = 8). All patients maintained their intramuscular IFNβ-1a treatment throughout the study. Analyses explored safety signals and directional trends; this preliminary study was not powered to detect clinically meaningful differences.
Results:
Both combination treatments were safe and well tolerated, with all adverse events mild. Patients treated with IFNβ-1a plus FLU had similar relapse rates, EDSS scores, and MS Functional Composite scores, but significantly less acute corticosteroid use for on-study relapses and better responses on some MRI outcomes, versus patients treated with IFNβ-1a plus MP.
Conclusions:
Further study of FLU for breakthrough disease in patients with RRMS is warranted.
Keywords
Introduction
Interferon beta (IFNβ) is an immunomodulatory agent that suppresses acute inflammatory events occurring in the early stages of multiple sclerosis (MS). Reductions in relapse rate and lesions observed on MRI scans, including gadolinium-enhanced (Gd+) lesions and T2-hyperintense lesions, as well as delayed disability progression have been observed in patients with relapsing–remitting MS (RRMS) treated with IFNβ in clinical trials [IFNB Multiple Sclerosis Study Group, 1993; Jacobs et al. 1996; PRISMS Study Group, 1998; European Study Group, 1998]. However, despite effective therapy with IFNβ, some patients experience breakthrough disease in which they develop resurgence in the frequency of relapses and active MRI disease [Bermel et al. 2013].
To attempt to reduce relapses and delay the progression of disability in MS patients experiencing breakthrough disease, a range of novel treatment strategies have emerged in the past few years for monotherapy of RRMS, including oral and intravenous therapies, to serve as alternatives to long established self-injectable medications [Salhofer-Polanyi and Leutmezer, 2014]. In addition, combination therapies, such as platform therapies with the addition of azathioprine, methotrexate, or methylprednisolone (MP) have been studied and are sometimes used in clinical practice, although benefits for each vary with regard to dosage, schedule and duration of treatment and safety/tolerability [Okuda, 2014; Calabresi et al. 2002; Conway and Cohen, 2010; Fernández et al. 2002; Ticha et al. 2012; Ravnborg et al. 2010; Lublin et al. 2013; Weinstock-Guttman and Bakshi, 2004]. The goal of achieving freedom from breakthrough disease remains difficult in patients with RRMS despite receiving the currently available disease modifying therapies, indicating that a ‘treatment gap’ continues to exist [Rotstein et al. 2015].
Fludarabine (FLU) is a purine nucleoside analog prodrug that upon phosphorylation is toxic to dividing and quiescent lymphocytes and monocytes, exerting its effects through DNA synthesis interference and apoptosis [Tseng et al. 1982; Zinzani et al. 1994]. This agent has emerged as an effective treatment for patients with lymphoproliferative disorders and hematologic malignancies [Robak and Robak, 2013; Lukenbill and Kalaycio 2013; Kluin-Nelemans and Doorduijn, 2011]. Considering the immunosuppressive nature of FLU and its effectiveness in treating disorders involving immune dysregulation, we studied the use of FLU in patients with RRMS. In this prospective, proof-of-concept, pilot study, the safety and tolerability of FLU and IFNβ-1a combination therapy was compared with IFNβ-1a plus MP in a small cohort of RRMS patients who had received long-term IFNβ and experienced breakthrough disease. MP was chosen as the comparator because it is one of the most common interventions for MS [Goodin, 2014].
Patients and methods
Patients
Men and women aged 18–65 years with a diagnosis of MS [Poser et al. 1983] and an Expanded Disability Status Scale (EDSS) score [Kurtzke, 1983] of 2.0–6.5 were eligible for enrollment. Patients were required to have RRMS defined and distinguished from other MS clinical subtypes according to the standardized nomenclature developed by consensus [Lublin and Reingold, 1996]. All RRMS patients who entered this study had received uninterrupted treatment with intramuscular IFNβ-1a (Avonex®: Biogen, Inc., Cambridge, MA) 30 µg weekly for >1 year and experienced breakthrough disease defined as two or more relapses over the prior year and disability progression equivalent to ⩾1.0 unit of deterioration in EDSS score sustained for ⩾3 months. All patients were required to provide written informed consent. This study was approved by the local ethics committee of the University at Buffalo.
Patients with a history of adverse events (AEs) or intolerability to IFN-β or corticosteroids were excluded, as were patients with progressive forms of MS. Other exclusion criteria included abnormal baseline values: alanine transaminase or aspartate transaminase levels more than two times the upper limit of normal (ULN), white blood cell counts (WBC) <2300 cells/mm3, platelet counts <80,000 cells/mm3, serum creatinine levels more than two times the ULN, and prothrombin time greater than the ULN; treatment with intravenous (IV) gamma globulin, plasmapheresis, or methotrexate within 3 months of enrollment, treatment with glatiramer acetate within one year of enrollment, or prior treatment with immunosuppressive agents (i.e. azathioprine, cyclosporine, chlorambucil, cyclophosphamide, mitoxantrone, DNA analogs), FLU, cladribine, total lymphoid irradiation, or any investigational drug for MS; and history of intolerance to acetaminophen, ibuprofen, naproxen, and aspirin. Female patients who were pregnant or breast-feeding also were ineligible.
Study design
In this small, prospective, randomized, open-label, parallel-group, phase II study, all patients initially received IV MP 1 g once daily for 3 consecutive days (days −9 to −7). Thereafter, patients were randomized 1 week later to receive (1) FLU 25 mg/m2 IV once daily for 5 consecutive days per 4-week cycle for 3 consecutive cycles or (2) MP 1 g IV one day per 4-week cycle for 3 consecutive cycles. In addition, patients continued to receive weekly of IM IFNβ-1a, which was administered without interruption throughout the study in both treatment groups. Evaluators were not blinded to treatment assignment. FLU-treated patients were eligible to receive granulocyte–macrophage colony-stimulating factor in the event of neutropenia (<1000 cells/mm2). FLU- and MP-treated patients were eligible to receive additional infusions of MP for relapse or persistent inflammation as dictated by their clinician. The 3-month treatment period was followed by an observation period of 9 months.
Clinic visits occurred during screening, at study entry (days −28 to −10), during MP induction (days −9 to −7), at day 1, and at months 1, 2, 3, 6, 9, and 12. At study entry, at day 1, and at months 3, 6, 9, and 12, patients were given a general neurologic evaluation, including the MS Functional Composite (MSFC) [Fischer et al. 1999] and determination of the Ambulation Index (AI) [Hauser et al. 1983], Modified Functional System and EDSS score [Kurtzke, 1983]. Laboratory assessments (i.e. hematology, blood chemistry, urinalysis) were performed at study entry, day 1, and months 1, 2, 3, 6, 9, and 12. In addition, for each patient randomized to the FLU treatment group, a complete blood count and differential were obtained prior to the initiation of each FLU infusion.
MRI
MRI of the brain was performed at study entry and at month 12 using the same scanning protocol on a General Electric 4x/Lx 1.5-T scanner (Milwaukee, WI, USA). The MRI protocol that was relevant for quantitative analysis consisted of axial fluid-attenuated inversion recovery (FLAIR) and axial T1-weighted conventional spin-echo pre-/post-gadolinium (Gd). IV injection of 0.1 mmol/kg Gd preceded postcontrast imaging by 5 min. The detailed parameters for FLAIR were field of view (FOV) 24 × 24, matrix 192 × 256, 28 slices, 5-mm thickness, TR 8002, TE 128, inversion time 2000 ms, echo train length 22, NSA 1, and scan time 4:16. For T1 spin-echo, the parameters were FOV 24 × 18, 192 × 256, 24 slices, 5-mm thickness, TR 600, TE 9, NSA 2, and scan time 2:56. There were no interslice gaps in any of the sequences.
MRI analysis was performed on a Unix workstation (Sun Microsystems, Santa Clara, CA, USA) using the image analysis software package Jim (version 3.0, Xinapse Systems Ltd, Northants, UK, http://www.xinapse.com). A blinded trained technician who was supervised by a blinded experienced observer performed the analyses. Hypointense lesions on noncontrast T1-weighted images, defined as having reduced signal compared with white matter, nonenhanced on post-Gd images, and being at least partially hyperintense on FLAIR images, were counted manually and their volume segmented using an edge-finding tool. Gd+ lesions were identified manually, and their volume was segmented using an edge-finding tool. FLAIR hyperintense lesion volume was determined using a semiautomated thresholding procedure [Bermel et al. 2003]. To assess whole-brain atrophy, a normalized measure of whole-brain volume, brain parenchymal fraction (BPF), was obtained from the axial noncontrast T1-weighted images based on our previously validated semiautomated technique [Bermel et al. 2003; Sharma et al. 2004]. BPF was defined as the ratio of brain parenchymal volume (tissue compartment) to the total intracranial volume.
Clinical assessment
Clinical relapses were documented throughout the course of the study. Relapses were defined as symptoms of neurologic dysfunction with objective confirmation that (1) lasted at least 48 h after a period of symptomatic stability of at least 29 days and (2) occurred in the absence of a febrile illness or steroid withdrawal. Disability progression was defined as a 1.0- point worsening from a baseline EDSS score between 2.0 and 4.5 or a 0.5-point worsening from a baseline EDSS score of 5.0 (confirmed by the 3-month follow-up visit). Improvement in EDSS status was defined as a 1.0-point decrease from baseline EDSS score, confirmed by the 3-month follow-up visit.
Safety and tolerability
Adverse event monitoring occurred throughout the study. All events, regardless of severity or relationship to investigational drug, were reported.
Study end points
The primary study end point was safety and tolerability as assessed by neurologic examinations, laboratory assessments, and AEs. The primary safety and tolerability end points were measured over the entire 12-month study period. The safety parameters were (1) proportion of subjects developing grade 3 or 4 Common Terminology Criteria for Adverse Events (CTCAE) toxicity on the following hematology and liver function parameters: hemoglobin level, WBC, absolute neutrophil count (ANC), platelets, CD4 count, alanine transaminase (ALT), aspartate transaminase (AST), and bilirubin; (2) proportion of subjects developing infections and opportunistic infections; (3) median time to first grade 3 or 4 hematologic toxicity or liver toxicity; (4) mean changes in lymphocytes, hemoglobin level, WBC, ANC, platelets, CD4 count, CD8+ count, CD19+ count, ALT, AST, and bilirubin from baseline to one year; and (5) the development of suspected unexpected serious adverse reaction (SUSAR). The established mechanism of action of FLU involves preferential lymphocyte depletion. Therefore, lymphopenia in the absence of a clinical associated event or lymphopenia that did not cause treatment discontinuation was not reported as an AE. Secondary study end points included clinical relapse rate; stabilization of neurologic disease as defined by EDSS score, MSFC, and AI; brain MRI parameters; and independence from nonstudy planned corticosteroid interventions.
Statistical analysis
Statistical analysis was performed using the Statistical Package for the Social Sciences (SPSS) version 13.0. Data analyses were performed on the intention-to-treat (ITT) study population unless noted otherwise. The number of relapses and scores on the multiple neurologic clinical exams were plotted against time for every patient. For brain MRI parameters, the mean change in number and volume of Gd+, T1 hypointense, and volume of FLAIR hyperintense lesions from baseline to month 12 were calculated for each patient. Differences between the two treatment arms were compared using a nonparametric Friedman test or an analysis of variance as appropriate. Kaplan–Meier time-to-event curves were constructed to quantify the time to lack of efficacy. The significance of within-patient change in MRI measurements over time from baseline was analyzed using the Wilcoxon signed-rank sum test. This proof-of-concept trial and post hoc analyses were performed to detect directional trends. A pilot study, this was exploratory to generate preliminary results and was not based on any formal power calculations regarding safety and tolerability. All p values, provided for comparative purposes, were based on two-tailed tests, and significance was set at p = 0.05. Subgroup sizes were not powered to detect clinically meaningful differences nor were results corrected for multiple comparisons.
Results
Patients
A total of 18 patients were enrolled, 10 randomized to receive FLU and eight to receive MP. In addition, all patients continued treatment with weekly IM IFNβ-1a. Of these patients, seven (70%) in the IFNβ-1a/FLU group and seven (87.5%) in the IFNβ-1a/MP group completed the study per protocol. Two patients withdrew from study because of lack of efficacy before completing all 3 months of treatment with study medication, one in the IFNβ-1a/FLU group and one in the IFNβ-1a/MP group.
At baseline, two patients in the IFNβ-1a/FLU group had an extraordinarily high number of Gd+ lesions (Table 1); one patient presented with 21 Gd+ lesions and the other with 17. The patient with 21 Gd+ lesions was aged 35 years, had a disease duration of 5.7 years, a baseline EDSS score of 2.0, and experienced two relapses in the year before study entry; the patient with 21 Gd+ lesions was aged 43 years, had a disease duration of 7.1 years, a baseline EDSS score of 4.5, and experienced two relapses in the year before study entry. Therefore, two populations of the IFNβ-1a/FLU group were examined: for the safety outcomes, the IFNβ-1a/FLU-ITT group (n = 10) included all randomized patients; for the efficacy outcomes, the IFNβ-1a/FLU-modified ITT (IFNβ-1a/FLU-mITT) group excluded the two baseline Gd+ lesion outliers (n = 8).
Demographic and baseline disease characteristics by treatment arm and ITT or mITT classification.

Values in the table are mean (SD) unless otherwise indicated. ITT groups included all treated patients.
FLU, fludarabine; ITT, intention-to-treat; mITT, modified intention-to-treat; MP, methylprednisolone; SD, standard deviation; IFNβ, interferon beta; EDSS, Expanded Disability Status Scale; MSFC, Multiple Sclerosis Functional Composite; PASAT, 3-Second Paced Auditory Serial Addition Test; MRI, magnetic resonance imaging; Gd+, gadolinium-enhanced.
All p values >0.05 except as indicated.
Modified ITT population excluded two patients with very high Gd+ lesion numbers (21 and 17 lesions) at baseline.
p = 0.035 versus FLU-ITT (Levene’s test).
p = 0.001 versus FLU-ITT (Levene’s test).
Therefore, the clinical and MRI results of the two patients with high numbers of Gd+ lesions were reported separately. Overall, patients in the IFNβ-1a/FLU and IFNβ-1a/MP groups appeared well matched with regard to demographic and baseline disease characteristics, including number of prior relapses, EDSS, AI, and MSFC scores (except for the 25-foot timed walk), and baseline MRI parameters (except for FLAIR lesion volume). Table 1 presents the baseline data of the ITT and modified ITT (mITT) treatment populations.
Safety and tolerability
Treatment with IFNβ-1a/FLU generally was well tolerated, and no significant differences were observed between treatment groups in the overall frequency of treatment-emergent AEs (p = 0.057). Infections were the most common AE (Table 2), occurring in three subjects in the IFNβ-1a/FLU group and two subjects in the IFNβ-1a/MP group. Infections consisted of urinary tract infection, cough, and in one case herpes zoster, which was dermatomal in nature. All infectious AEs were considered mild and resolved without sequelae.
Subjects experiencing adverse events.

Values in the table are number of events (%). ITT groups included all treated patients.
FLU, fludarabine; ITT, intention-to-treat; MP, methylprednisolone.
Cough, urinary tract infection, or development of recurrent varicella zoster (shingles).
Unresolved grade 2 or 3 leukopenia that had not normalized at 4 weeks post-treatment.
Transient elevations in liver function test results.
No significant differences were observed between treatment groups with regard to severity of AEs; no AE was reported as life-threatening and no severe AEs were reported. Three cases of unresolved leukopenia, defined as a CTCAE laboratory grade 3 or grade 2 leukopenia that had not normalized within 28 days after treatment with study medication (grade 1: absolute leukocyte count < lower limit of normal −3.0 × 109/l; grade 2: < 3.0–2.0 × 109/l; grade 3: < 2.0–1.0 × 109/l), were found in the IFNβ-1a/FLU group and one case was found in the IFNβ- 1a/MP. There were no cases of grade 4 leukopenia. Patients treated with IFNβ-1a/FLU had a significantly longer duration of leukopenia than those who received IFNβ-1a/MP (3.75 versus 0.17 months; p < 0.001); however, no patient developed severe leukopenia (grade 4; absolute leukocyte count < 1000/μl) in either treatment group. No moderate (grade 3) or severe (grade 4) neutropenias, thrombocytopenias, reductions in hemoglobin levels, or deaths occurred during the study.
Relapses
Overall, patients in the IFNβ-1a/FLU group experienced fewer relapses throughout the course of the study than did patients in the IFNβ-1a/MP group (mean ± SD: 0.5 ± 0.8 versus 0.8 ± 0.5, p = 0.414; Table 3). The addition of FLU to IFNβ-1a therapy slightly delayed the median time to first relapse compared with the median time to first relapse in the IFNβ-1a/MP treatment group (10.5 versus 8.5 months, p = 0.464; Table 3 and Figure 1). Both treatments resulted in a ⩾50% decrease in mean relapse rate compared with baseline. The reduction from prestudy to on-study relapse rate was greater for patients in the IFNβ-1a/FLU group than for those in the IFNβ-1a/MP group (80% versus 60%, respectively). Patients in the IFNβ-1a/FLU group used fewer cycles of corticosteroid (0.5 ± 0.8) than those in the IFNβ-1a/MP group (0.8 ± 0.5, p = 0.015; Table 3). In the mITT population, the total amount of corticosteroids used during the study was lower in patients who received IFNβ-1a/FLU than in those who received IFNβ-1a/MP (4.4 ± 2.7 versus 8.5 ± 1.6, p = 0.002).
On study relapses, corticosteroid use and EDSS change.

Values in the table are mean (SD) unless otherwise indicated.
IFNβ, interferon beta; FLU, fludarabine; mITT, modified intention-to-treat; MP, methylprednisolone; ITT, intention-to-treat; SD, standard deviation; EDSS, Expanded Disability Status Scale.
The IFNβ-1a/FLU-mITT group excludes two patients with very high Gd+ lesion numbers (21 and 17 lesions) at baseline.
n=6.
p=0.015.

Kaplan–Meier plot of the probability of relapse in patients treated with interferon beta-1a fludarabine (IFNβ-1a/FLU) or interferon beta-1a methylprednisone (IFNβ-1a/MP) (modified intention-to-treat population). The addition of FLU to IFNβ-1a therapy slightly delayed the median time to first relapse compared with the median time to first relapse in the IFNβ-1a/MP treatment group (10.5 versus 8.5 months, p = 0.464).
During the study period, the patient with 17 baseline Gd+ lesions experienced only one relapse at the month 8 follow-up examination, which required corticosteroid treatment. The patient with 21 baseline Gd+ lesions did not experience relapse activity during the study period.
Functional outcomes
EDSS scores from baseline to months 3, 6, 9, and 12 of the study in the IFNβ-1a/FLU-mITT and IFNβ-1a/MP-ITT populations are shown in Figure 2. At 12 months, the EDSS scores had decreased for patients in the IFNβ-1a/FLU-mITT group but increased in the IFNβ-1a/MP group (mean absolute change from baseline: −0.2 ± 1.4 versus +0.5 ± 1.1, p = 0.432; Table 3). EDSS scores decreased in the IFNβ-1a/FLU-mITT group but increased in the IFNβ-1a/MP-ITT group (mean absolute change from baseline: −0.4 ± 0.4 versus +0.14 ± 1.1, p = 0.03) over months 6–12 of the study. A total of three patients reached confirmed sustained progression: one in the IFNβ-1a/FLU-mITT group after 3 months of treatment and two in the IFNβ-1a/MP-ITT group after 9 months of treatment. Four patients in the IFNβ-1a/FLU-mITT group and two patients in the IFNβ-1a/MP-ITT group improved by 1.0 point in their EDSS status (p = 0.302). This improvement was sustained by the 3-month follow-up visit. Over 12 months of follow-up, the patient with 17 baseline Gd+ lesions experienced a 0.5-point EDSS improvement whereas the patient with 21 baseline Gd+ lesions experienced a 0.5-point EDSS deterioration.

Mean and standard deviation absolute Expanded Disability Status Scale (EDSS) scores at baseline and at 3, 6, 9, and 12 months of study by treatment group. IFNβ, interferon beta; FLU-mITT, fludarabine modified intent to treat; MP-ITT, methylprednisone intent to treat. EDSS scores decreased in the IFNβ-1a/FLU-mITT group but increased in the IFNβ-1a/MP-ITT group (mean absolute change from baseline: −0.4 ± 0.4 versus +0.14 ± 1.1, p = 0.03) over months 6–12 of the study. At 12 months, the EDSS scores had decreased for patients in the IFNβ-1a/FLU-mITT group but increased in the IFNβ-1a/MP group (mean absolute change from baseline: −0.2 ± 1.4 versus +0.5 ± 1.1, p = 0.432).
Changes in MSFC absolute z score from baseline to months 3, 6, 9, and 12 of the study are shown in Figure 3; although no differences were observed between the two treatment groups, patients who received IFNβ-1a/FLU showed consistent incremental improvement over the study period. Patients in the IFNβ-1a/FLU-mITT group demonstrated improvement in total MSFC z score (mean change: +0.37, p = 0.046). Specifically, the 3-s Paced Auditory Serial Addition Test (PASAT) score (i.e. number of correct responses) increased from 30.9 ± 12.0 at baseline to 44.2 ± 13.8 at month 12 (p = 0.034). Only a trend toward improvement was seen in the timed 9-hole peg test (from 31.2 ± 12.7 s at baseline to 28.7 ± 13.4 s at month 12) and in the timed 25-foot walk score (from 8.8 ± 6.2 s at baseline to 6.3 ± 3.0 s at month 12); these changes were not statistically significant. Patients in the MP group also demonstrated an improvement only in the PASAT component of the MSFC over the course of study (from 39.0 ± 8.2 at baseline to 49.0 ± 11.1 at month 12, p = 0.046). Both patients with high numbers of baseline Gd+ lesions experienced improvements in their total MSFC z score (mean change: +0.29).
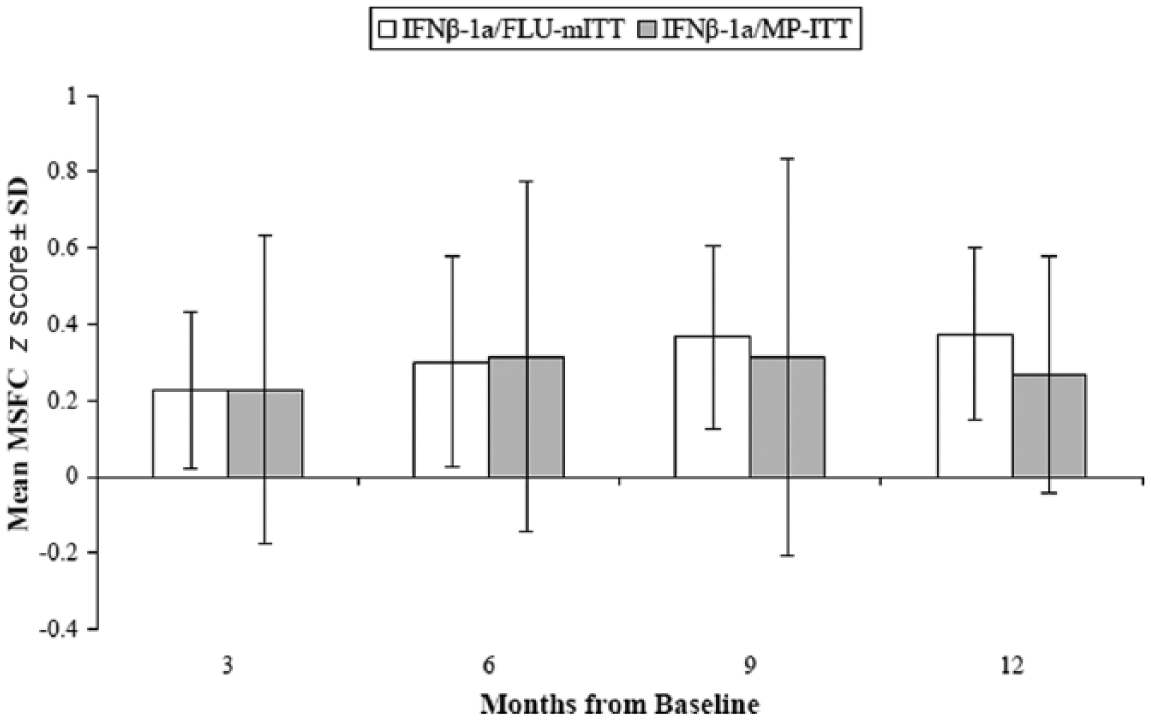
Mean and standard deviation of the absolute change in Multiple Sclerosis Functional Composite (MSFC) z score at 3, 6, 9, and 12 months of study by treatment group. Although no differences were observed between the two treatment groups, patients in the IFNβ-1a/FLU-mITT group demonstrated improvement in total MSFC z score (mean change: +0.37, p = 0.046). Specifically, the 3-s Paced Auditory Serial Addition Test (PASAT) score (i.e. number of correct responses) increased from 30.9 ± 12.0 at baseline to 44.2 ± 13.8 at month 12 (p = 0.034). Only a trend toward improvement was seen in the timed 9-hole peg test (from 31.2 ± 12.7 s at baseline to 28.7 ± 13.4 s at month 12) and in the timed 25-foot walk score (from 8.8 ± 6.2 s at baseline to 6.3 ± 3.0 s at month 12); these changes were not statistically significant. Patients in the MP group also demonstrated an improvement only in the PASAT component of the MSFC over the course of study (from 39.0 ± 18.2 at baseline to 49.0 ± 11.1 at month 12, p = 0.046).
MRI outcomes
Reductions from baseline in absolute Gd+ lesion number (MP, from 0.83 ± 0.98 to 0.50 ± 0.84, p = 0.175 for two-tailed paired samples test; FLU, from 1.00 ± 1.29 to 0.29 ± 0.49, p = 0.182 for two-tailed paired samples test) and volume (MP, from 122.2 ± 194.0 mm3 to 54.2 ± 100.8 mm3, p = 0.147; FLU, from 136.8 ± 229.4 mm3 to 19.0 ± 33.4 mm3, p = 0.208) were observed in both treatment groups, whereas minimal change was observed in T1 hypointense lesion volume (MP, from 1243.3 ± 1455.2 mm3 to 1178.9 ± 1573.6 mm3, p = 0.462; FLU, from 2292.9 ± 2610.0 mm3 to 2332.1 ± 3055.3 mm3, p = 0.908) and BPF (MP, from 0.847 ± 0.032 to 0.854 ± 0.026, p = 0.157; FLU, from 0.848 ± 0.038 to 0.847 ± 0.048, p = 0.806) from baseline to month 12 for both treatment groups. Patients in the IFNβ-1a/FLU-mITT group showed reductions in FLAIR lesion volume (from 21,812.0 ± 23,546.1 mm3 to 20,033.0 ± 21,105.1 mm3, p = 0.220), whereas patients in the IFNβ-1a/MP group showed an increase in FLAIR lesion volume (from 15,395.3 ± 13,616.9 mm3 to 16,593.1 ± 13,442.1 mm3, p = 0.210). In the IFNβ- 1a/FLU-mITT group, Gd+ lesion volume was reduced by 97.2% from baseline, Gd+ lesion number by 88.9%, and FLAIR lesion volume by 12.7%. In the IFNβ-1a-MP group, Gd+ lesion volume was reduced by 71.7%, Gd+ lesion number was reduced by 50%, and FLAIR lesion volume increased by 12% (Table 4). In the IFNβ-1a/FLU-ITT group, significant reductions were seen in Gd+ lesion volume (–98.3%, p = 0.046) and number (–93.3%, p = 0.044) and nearly significant reductions in FLAIR lesion volume (–14.2%, p = 0.066) compared with baseline measures.
MRI outcomes at 12 months.
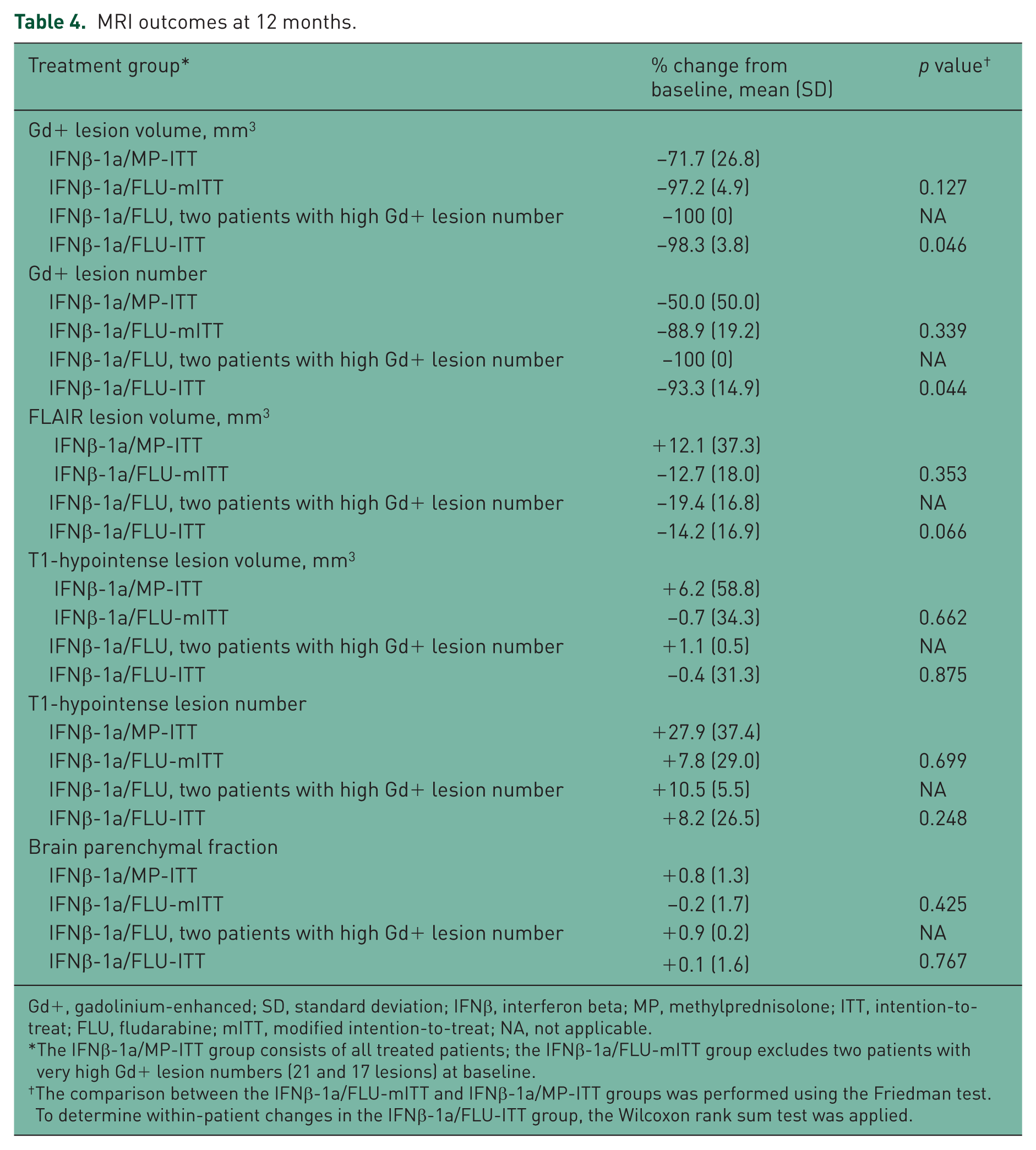
Gd+, gadolinium-enhanced; SD, standard deviation; IFNβ, interferon beta; MP, methylprednisolone; ITT, intention-to-treat; FLU, fludarabine; mITT, modified intention-to-treat; NA, not applicable.
The IFNβ-1a/MP-ITT group consists of all treated patients; the IFNβ-1a/FLU-mITT group excludes two patients with very high Gd+ lesion numbers (21 and 17 lesions) at baseline.
The comparison between the IFNβ-1a/FLU-mITT and IFNβ-1a/MP-ITT groups was performed using the Friedman test. To determine within-patient changes in the IFNβ-1a/FLU-ITT group, the Wilcoxon rank sum test was applied.
The MRI changes in the two patients with a high number of baseline Gd+ lesions are presented in Table 4. These patients experienced 100% mean reductions in Gd+ lesion number and volume and a 19.4% mean reduction in FLAIR lesion volume. Furthermore, mean changes in T1 lesion volume and BPF were minimal (+1.1% and +0.9%, respectively). The mean increase in T1 lesion number for these patients was +10.5%.
Discussion
The results of this randomized, open-label, pilot study demonstrated that IFNβ-treated patients with RRMS with breakthrough disease who received adjunct therapy with FLU or MP had similar, favorable AE profiles. To the best of the authors’ knowledge, this study is the first to examine the use of FLU as an adjunct therapy to IFNβ in MS. In our study, infections were reported in 30% of patients in the IFNβ-1a/FLU group and 25% of patients in the IFNβ-1a/MP group, which is not unexpected given the immunomodulating actions of the treatments [Zinzani et al. 1994; Fauci et al. 1976; Kupersmith et al. 1994] and the common occurrence of bladder dysfunction and urinary tract infection in this patient population. Mild leukopenia was observed in more patients receiving IFNβ-1a/FLU than those receiving IFNβ-1a/MP in our study. Likewise, of the use of FLU in lymphoid malignancies has showed mild leukopenia to be a commonly reported treatment-associated AE [Robak and Robak, 2013; Lukenbill and Kalaycio, 2013; Kluin-Nelemans and Doorduijn, 2011]. No serious or severe AEs resulted from adjunct treatment with FLU. Thus, our preliminary results suggest that FLU as an adjunct to IM IFNβ-1a appears to be safe and well tolerated over 1 year in patients with RRMS.
Although safety was the primary study end point, we also performed exploratory analyses of relapses, functional outcomes, and MRI outcomes with FLU and MP as adjunct therapy to IFNβ-1a. These analyses were conducted to determine directional trends because the treatment group sizes were not powered to detect clinically meaningful differences and the results were not corrected for multiplicity. Therefore, the efficacy outcomes presented here must be considered preliminary and interpreted within the context of the limitations of the clinical trial design. In addition, given that two patients originally randomized to the IFNβ-1a/FLU group had an extraordinarily high number of Gd+ lesions at baseline compared with the rest of the study population, these patients were not included in the efficacy analysis comparing FLU and MP adjunct therapies. Instead, efficacy results for a mITT population of IFNβ-1a/FLU-treated patients, which excluded the two outliers, were compared with those of the ITT population of IFNβ-1a/MP-treated patients; results for the two outlier patients were reported separately.
Over the course of the study, the mean number of relapses was low and was reduced by ⩾50% from baseline in both treatment groups. However, because of a natural tendency for MS attacks to diminish over time [Patzold and Pocklington, 1982], this effect cannot simply be attributable to either treatment without a proper comparator group. The IFNβ-1a/FLU-mITT group used significantly less corticosteroid treatment to manage relapses (p = 0.002). At the end of the study, EDSS scores were not found to have changed dramatically, with a mean absolute change of −0.2 in the IFNβ-1/FLU-mITT group and +0.5 in the IFNβ-1/MP group. Only three patients reached confirmed sustained progression: one in the IFNβ-1a/FLU-mITT group and two in the IFNβ-1a/MP-ITT group. Nevertheless, four patients in the IFNβ-1a/FLU-mITT group and two in the IFNβ-1a/MP-ITT group improved by 1.0 point in their EDSS status, and this improvement was sustained by the 3-month follow-up visit. In a previous 5-year phase II trial in patients (n = 88) with RRMS, single-agent pulsed MP (1 g/day for 5 days followed by oral prednisone taper) given every 4 months for 3 years resulted in significant improvement in EDSS scores compared with MP given at relapse only [Zivadinov et al. 2001]. Also, the MP regimen of the current study (1 g IV 1 day per 4-week cycle for three consecutive cycles) differed from that of the previous study. In the present study, FLU-treated patients showed a slight, nonsignificant decrease in EDSS scores at 12 months compared with the MP-treated patients. In addition, although changes in overall MSFC z score from baseline to 12 months were not significant in either study group, the IFNβ-1a/FLU-mITT group appeared to show an incremental improvement over the study course. Given that the treatment period of the present study was only 3 months, it is possible that a greater treatment effect on EDSS score may be achieved with a longer-term study or repeated cycles of FLU and MP therapy.
Although a number of MRI findings in our study were interesting, firm conclusions should be tempered by the small sample size. IFNβ-1a/FLU (ITT group) reduced Gd+ lesion volume by 98.3% from baseline and number of Gd+ lesions by 93.3% at 12 months (p = 0.046 and p = 0.044, respectively), with a nearly significant percentage reduction in FLAIR lesion volume (p = 0.066). In addition, treatment with IFNβ-1a/FLU resulted in a greater reduction in Gd+ lesion volume and number compared with IFNβ-1a/MP. These findings are important given that Gd+ lesions are predictive of clinical relapses [Bakshi et al. 2008].
In addition, treatment with IFNβ-1a/FLU led to a reduction in FLAIR lesion volume whereas treatment with IFNβ-1a/MP did not prevent volume accumulation. BPF percentage change from baseline, a measure of brain atrophy, did not differ between the two groups, although patients in the IFNβ-1a/FLU-mITT group experienced a greater decrease in BPF than those in the IFNβ-1a/MP-ITT group. These findings may be explained by a greater anti-inflammatory effect of FLU compared with MP over a short-term period, which could have attributed to a more rapid decrease in brain volume. This transitional effect recently has been described as ‘pseudoatrophy’ and is likely related to the magnitude of the anti-inflammatory effect of various disease-modifying therapies [Khoury and Bakshi, 2010].
Both patients with a high number of baseline Gd+ lesions experienced improvements in many clinical outcomes with combination treatment with IFNβ-1a and FLU. The patient with 17 baseline Gd+ lesions experienced only one relapse during the study, whereas the patients with 21 baseline lesions had no on-study relapses. Both patients experienced an improvement in MSFC z score (mean: +0.29), reductions in FLAIR lesion volume (mean: –19.4%), Gd+ lesion number (mean: –100%) and volume (mean: –100%), and minimal change in T1 lesion volume (mean: +1.1%) and FLAIR lesion volume (mean: +0.9%).
However, it is important to note that this study was exploratory and not based on any formal power calculations regarding safety and tolerability of FLU. A 3-month treatment plus 9-month observation period (combined with the small sample size) is not sufficient to make any strong claims. Moreover, we have no data on long-term effects of FLU beyond 1 year on either the 3-month dosing in 1 year, or redosing in subsequent years. It should also be noted that the intent of our study was to test FLU therapy as adjunctive, perhaps to delay or avoid escalating the patient to agents that have greater safety risks. This would be a potential option for a patient who cannot be switched to one of the newer oral or injectable therapies due to patient preference or safety/tolerability issues. Clearly, the long term safety of repetitive use has not been established by this study, which is a critical consideration for a chronic disease like MS. We emphasize that our results should be considered preliminary and would await confirmation in a larger scale trial.
In conclusion, an unmet medical need exists for therapeutic options in the treatment of patients with RRMS who experience disease activity despite long-term therapy with IFNβ. Our preliminary results suggest that IFNβ-1a/FLU treatment was safe and well tolerated over one year by patients with RRMS who experienced breakthrough disease on IFNβ-1a therapy. A number of findings in this study suggest disease-modifying benefit with IFNβ-1a/FLU versus IFNβ-1a/MP, including reductions in corticosteroid requirements to manage relapses and reduced MRI progression. Thus, short-term FLU therapy may provide rapid-onset sustained immunosuppression to control breakthrough disease while maintaining patients on IFNβ therapy.
Footnotes
Acknowledgements
The authors thank Jillian Licata, Sabrina Maurer, and Matthew Hasson of Scientific Connexions for editorial assistance in preparing this manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by an investigator-initiated study grant provided by Biogen. Berlex Lab-oratories, Montville, NJ, USA, provided fludarabine study medication.
Conflict of interest statement
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Greenberg is an employee of Abbvie, Inc. Dr Zivandinov has received speaking and consultant fees from Teva, Biogen, EMD Serono, Novartis, Claret Medical, and Genzyme and research support from Biogen, Teva, Novartis, Claret Medical, and Genzyme. Dr Bakshi has received consulting fees from Alkermes, Biogen, Novartis, and Questcor and research support from Biogen, EMD Serono, Novartis, Questcor, Sanofi-Genzyme, and Teva. The other authors have nothing to disclose.
