Abstract
Interferon beta preparations are the most widely used initial therapies prescribed for patients with relapsing–remitting multiple sclerosis. Phase III studies have demonstrated comparable efficacy on clinical measures of disease activity, variable benefits on radiological measures, and good overall tolerability. Subsequent clinical studies have attempted to compare directly the three available interferon beta preparations, reporting both safety and efficacy data. We review the literature on studies evaluating interferon beta therapy for patients with relapsing–remitting multiple sclerosis, discuss reasons for discrepant findings, and assess the utility of interferon beta-based combination regimens as the focus of future studies in the increasingly complex multiple sclerosis therapy landscape.
Introduction
For more than 15 years, interferon beta (IFNβ) therapy has been a first-line treatment for relapsing–remitting multiple sclerosis (RRMS). During this time, clinicians’ experience and evidence-based clinical data have indicated that patients are benefiting from these treatment regimens. Recent advances in our understanding of the underlying disease heterogeneity of multiple sclerosis (MS), the variable responses of patients to different treatments, and the introduction of new classes of pharmacotherapies have raised new questions regarding the optimal treatment regimen for patients with MS. As more therapeutic options for inflammatory forms of MS become available, treatment decisions for a given patient will become increasingly complex. On the cusp of these revolutions in MS therapy, we have reviewed the literature to date, comparing the three currently available IFNβ formulations at different doses and as proven platforms for new optimal therapeutic combinations.
Three IFNβ formulations are currently approved for the treatment of RRMS: subcutaneous (SC) IFNβ-1b (Betaferon®/Betaseron®, Extavia®), intramuscular (IM) IFNβ-1a (Avonex®), and SC IFNβ-1a (Rebif®; available in both 22 µg and 44 µg formulations). Although they used different primary endpoints, the respective pivotal phase III studies of each IFNβ therapy over a 2-year period demonstrated comparable efficacy with respect to relapse-related measures and varying degrees of benefit with respect to inflammatory measures, such as gadolinium enhancement [PRISMS Study Group, 1998; Jacobs et al. 1996; IFNB Multiple Sclerosis Study Group, 1993]. Both IFNβ-1a products also have been shown to slow disability progression as assessed by sustained change on the Expanded Disability Status Scale (EDSS) [PRISMS Study Group, 1998; Jacobs et al. 1996. These benefits were maintained in blinded studies of patients observed for up to 4 years [Clanet et al. 2002; PRISMS Study Group and University of British Columbia MS/MRI Analysis Group, 2001]. All IFNβ formulations are generally well tolerated, and improvements in injection technique, side effect management, and patient education have contributed to greater compliance with treatment regimens [Munschauer and Kinkel, 1997]. Regular testing for elevated hepatic enzyme levels and changes in leukocyte counts is recommended for all IFN products [Bayer Healthcare Pharmaceuticals, 2008; Biogen Idec, 2008; EMD Serono, 2008]. As a protein-based therapy, immune responses directed at IFNs have been documented. Antigenicity varies among these products, with SC IFNβ-1b as the highest, then SC IFNβ-1a, and IM IFNβ-1a as the lowest, due to differences in formulation, production technique and additives, method of administration, pH, and in vivo aggregation of the product. Several studies have demonstrated that the risk of developing neutralizing antibodies to IFN within the first 9 to 18 months of treatment can compromise clinical efficacy beginning 18–24 months after treatment initiation [IFNβ MS study group and the University of British Columbia MS/MRI Analysis group 1996; Bertolotto et al. 2002, Francis et al. 2005]. Thus, understanding the similarities and differences among the three IFNβ formulations is essential for optimizing outcomes for patients with MS.
Summary of comparative studies of interferon beta (IFNβ) products for relapsing–remitting multiple sclerosis.

CU, combined unique; EDSS, Expanded Disability Status Scale; GA, glatiramer acetate; IFNβ, interferon beta; IM, intramuscular; IVIg, intravenous immunoglobulin; MRI, magnetic resonance imaging; SC, subcutaneous.
Significant differences in favor of SC IFNβ-1b were observed for percentage reduction in relapse frequency, although the baseline imbalances between the 2 treatment groups prohibit any direct comparisons. At baseline, SC IFNβ-1b–treated patients had more severe disease than IM IFNβ-1a–treated patients.
Study included patients with relapsing–remitting multiple sclerosis and secondary progressive multiple sclerosis.
Direct comparative studies
Levels of evidence of efficacy in clinical trials.

Comparison of relapse rates among IFNβ products
Study 1
The largest study conducted thus far, Quality Assessment in Multiple Sclerosis Therapy (QUASIMS), retrospectively enrolled more than 4700 patients with RRMS from 510 sites in the German-speaking countries of Europe [Limmroth et al. 2007] and was extended to over 7000 patients worldwide in seven other countries [Limmroth et al. 2005]. The initial cohort represented 16% of all patients who had been treated with an IFNβ preparation and approximately 27% of all patients in Germany, Austria, and Switzerland who received an IFNβ preparation for 2 or more years [Limmroth et al. 2007]. QUASIMS was conducted according to a defined study protocol, and analyses were performed by an independent contract research organization. All patients meeting the inclusion criteria (RRMS, minimum treatment duration of 2 years, complete relapse history, documented EDSS scores) were included in the investigation. Patients receiving 2 years of uninterrupted treatment with one of the IFNβ therapies or those who were treatment-naïve were considered the initial treatment (IT) group. Patients who started a different disease-modifying therapy (DMT) after initial treatment were considered the follow-up therapy (FT) group [Limmroth et al. 2007].
The results of QUASIMS suggest that there is comparable efficacy during 2 years of treatment among the available IFNβ preparations in this population (approximately 50% of patients were progression free over 2 years) using these documented treatment outcomes. A greater percentage of patients who received IFNβ therapies initially (the IT group) remained relapse-free compared with patients who received IFNβ as a follow-up therapy (the FT group). Annualized relapse rates over 2 years were not significantly different in either group regardless of specific IFN treatment. For the IT group, the relapse rate was 0.51 for IM IFNβ-1a, 0.52 for IFNβ-1b, 0.53 for SC IFNβ-1a 22 µg, and 0.63 for SC IFNβ-1a 44 µg [Limmroth et al. 2007]
A subset analysis comparing patients who were switched from IM IFNβ-1a to SC IFNβ-1b with patients who were switched from SC IFNβ-1b to IM IFNβ-1a did not reveal any differences in the course of the disease at 2-year follow up [Limmroth and Putzki, 2005]. When comparing patients who changed to different IFNβ preparations due to side effects, QUASIMS confirmed data from earlier studies showing that patients treated with an SC formulation were more likely to discontinue treatment than those treated with the IM formulation, largely because of injection-site reactions [Durelli et al. 2002; Panitch et al. 2002]. Only 0.7% of patients discontinued IFNβ treatment because of central nervous system adverse events (e.g. depression). Despite the limitations of the QUASIMS trial, such as lack of MRI measurements and neutralizing antibody data, QUASIMS provided important data demonstrating similar efficacy of the different IFNβ preparations in a large cohort of patients.
Study 2
Another study involving 65 centers in northern Italy compared 834 patients with RRMS treated with SC IFNβ-1b (250 µg every other day) with 647 patients treated with IM IFNβ-1a (30 µg once weekly) [Milanese et al. 2003]. Mean treatment duration was 1.8 years in the SC IFNβ-1b group and 1 year in the IM IFNβ-1a group. The change in EDSS scores and percentage of relapse-free patients was comparable for both groups. Significant differences in favor of SC IFNβ-1b versus IM IFNβ-1a were noted for percentage reduction in relapse frequency at 6 months (68% vs. 55%, p < 0.001), 12 months (60.4% vs. 55%, p = 0.01), 18 months (63.7% vs. 57.1%, p = 0.01), and 24 months (64% vs. 56%, p = 0.02); however, patients inthe SC IFNβ-1b group had greater disease severity at baseline. In addition, there were significantly more dropouts seen in the SC IFNβ-1b group than in the IM IFNβ-1a group (41.1% vs. 15.3% over 3 years, p < 0.001), including withdrawals due to adverse events (15.3% vs. 3.8%) and disability progression (7.7% vs. 3.2%). The SC IFNβ-1b formulation was associated with a higher incidence of local skin reactions (33% vs. 8%) and leukopenia (17% vs. 6%). The results of this study suggested that both formulations were effective, but differences in baseline characteristics of patients in each treatment arm precluded an accurate comparison.
Study 3
A population-based surveillance study of 1033 patients with RRMS was conducted in 15 centers in Italy [Trojano et al. 2003]. Patients were treated with SC IFNβ-1b 250 µg every other day, IM IFNβ-1a 30 µg once weekly, or SC IFNβ-1a 22 µg three times weekly. Results were reported at 12-month follow up for SC IFNβ-1b (n = 234), IM IFNβ-1a (n = 217), and SC IFNβ-1a 22 µg (n = 89) and at 24-month follow up for SC IFNβ-1b (n = 209) and IM IFNβ-1a (n = 169). No differences were observed in the proportion of relapse-free patients among the three treatment groups (53.7% for SC IFNβ-1b, 54.4% for IM IFNβ-1a, 49% for SC IFNβ-1a; p = 0.10) after 12 months of treatment or between SC IFNβ-1b (37.5%) and IM IFNβ-1a (33%) after 24 months of treatment. Each of the IFNβ products produced a significant reduction in relapse rate at the 12- and 24-month time points (p < 0.001). In addition, no significant differences were seen in mean changes in relapse rate or EDSS scores at 12 or 24 months. These results are consistent with the controlled phase III studies of each IFNβ formulation [PRISMS Study Group, 1998; Jacobs et al. 1996; IFNB Multiple Sclerosis Study Group, 1993].
Study 4
A single-center observational study evaluated 469 patients with RRMS who were observed for at least 2 years during therapy with SC IFNβ-1b (n = 185), IM IFNβ-1a (n = 148), or SC IFNβ-1a (n = 136) [Río et al. 2006]. The relapse rate was significantly reduced for all three formulations (70% for SC IFNβ-1b, 64% for IM IFNβ-1a, and 74% for SC IFNβ-1a). Sustained progression of disability at 2 and 4 years was 17% and 23% for the IM IFNβ-1a group, 11% and 24% for the SC IFNβ-1b group, and 19% and 35% for the SC IFNβ-1a group. There were no reports of unexpected adverse events with any of the treatments; however, efficacy data in this patient population was inconsistent with the previous phase III study results. Overall, however, there were no distinct differences among the three IFNβ formulations.
Study 5
Romero-Lopez and colleagues evaluated IFNβ preparations for the treatment of 313 patients with RRMS and secondary progressive MS (SPMS) in Galicia, Spain [Romero-Lopez et al. 2003]. IM IFNβ-1a was administered to 26% of the patients, SC IFNβ-1b to 52.4%, and SC IFNβ-1a to 21.6% for a mean of 2.1 years [Romero-López et al. 2003]. Relapse rates were reduced by 73.3% in patients receiving IM IFNβ-1a, by 68.8% in patients receiving SC IFNβ-1b, and by 35.7% in patients receiving SC IFNβ-1a. Of patients receiving IM IFNβ-1a, 60.5% remained relapse free, compared with 33% of patients receiving SC IFNβ-1b and 54.5% of patients receiving SC IFNβ-1a. Despite differences in relapse rates among the three IFNβ formulations, the results of this study demonstrated that each was tolerable and effective in the daily clinical setting.
Study 6
In another retrospective, open-label study, patients with RRMS treated with IM IFNβ-1a (n = 79), SC IFNβ-1b (n = 77), SC IFNβ-1a 22 µg (n = 48), or glatiramer acetate (GA; n = 79) were evaluated over a period of 24 months [Haas and Firzlaff, 2005]. Baseline characteristics were comparable across treatment groups. Compared with baseline, the mean relapse rate for all treatments decreased significantly at 24 months (0.8 vs. 1.09 for IM IFNβ-1a, 0.69 vs. 1.20 for SC IFNβ-1b, 0.66 vs. 1.16 for SC IFNβ-1a, 0.36 vs. 1.16 for GA; all p < 0.05). There were no significant differences among the IFNβ preparations, but the reduction in relapse rate for patients treated with GA was significantly higher than those in patients receiving any of the other therapies (p < 0.001). Percentages of patients who were free from relapse ranged from 35.4% to 58.2% (p = 0.22) and percentages of patients who were progression free (no increase in EDSS by ≥1 point) ranged from 71.7% to 87.5 % (p = 0.13), with no statistically significant differences among the therapies. The progression index (final EDSS divided by disease duration) at 24 months, however, was significantly lower (p < 0.05; indicating less progression) in patients treated with GA and SC IFNβ-1a compared with SC IFNβ-1b. Despite small sample size and potential selection bias in treatment arms in this retrospective surveillance study, results indicated that these first-line therapies for patients with MS have consistent efficacies.
Study 7
A Danish group investigated the efficacy of SC IFNβ-1a 22 µg once weekly (n = 143) and SC IFNβ-1b 250 µg every other day (n = 158) in a prospective, multicenter, randomized, controlled, open-label, 2-year study [Koch-Henriksen et al. 2006]. The primary endpoints were annualized relapse rate, time to first relapse, and NAb formation. Annualized relapse rates (0.70 and 0.71, respectively) and time to sustained disability progression (a secondary endpoint) were similar for both groups. Development of NAbs at 2-year follow-up did not affect efficacy of either formulation. The authors in this study acknowledged that its unblinded design could have masked a true difference between the two formulations. Even though the results were not robust due to a small sample size and a large amount of missing data, a favorable trend toward SC IFNβ-1b was noted in the MRI results.
Study 8
The Prospective and Retrospective Long-Term Observational Study of Avonex and Rebif (PROOF) was a phase IV, multicenter study that combined retrospective and prospective data from patients with RRMS [Minagar and Murray, 2008]. PROOF was intended to collect up to 5 years of comparative efficacy and tolerability data using combined chart review and prospective observational data. Owing to slow enrollment, however, the study was halted after collecting 6 months of prospective data. In total, 18–30 months of data were reported for PROOF (12–24 months retrospective data plus 6 months prospective data). Treatment consisted of IM IFNβ-1a 30 µg once weekly or SC IFNβ-1a 44 µg 3 times weekly. At study termination, the median treatment duration was 19.9 months for IM IFNβ-1a (n = 69) and 16.2 months for SC IFNβ-1a (n = 67, p = 0.003). No significant differences were seen between treatment groups on stratified EDSS scores at any time point or on the proportion of patients with sustained disability progression (25.8% for IM IFNβ-1a vs. 26.7% for SC IFNβ-1a); on annualized relapse rate during last 6 months of treatment (0.35 vs. 0.48); or on changes in MRI endpoints. However, 19% of SC IFNβ-1a-treated patients were positive for NAbs at study enrollment and again 6 months later compared with none of the patients treated with IM IFNβ-1a. Although no differences were observed between NAb-positive and NAb-negative patients on any of the clinical endpoints, NAb positivity was associated with disease activity on MRI. For example, more NAb-positive than NAb-negative patients had new or enlarging T2 lesions (63.6% vs. 41.7%, p = 0.003) and larger gadolinium-enhanced (Gd+) lesions at enrollment (305.5 mm3 vs. 51.6 mm3, p = 0.015). Owing to the combination of the retrospective and prospective design and the shortened time of the prospective phase, this study was not powered to detect statistical differences between the two IFNβ-1a formulations.
Study 9
In the randomized, prospective, open-label Evidence of Interferon Dose Response: European–North American Comparative Efficacy (EVIDENCE) study, patients with RRMS were randomized to IM IFNβ-1a 30 µg once weekly (n = 338) or SC IFNβ-1a 44 µg three times weekly (n = 339) for 48 weeks [Panitch et al. 2002]. The study was conducted at 56 centers in Canada, Europe, and the United States. Patients and treating physicians were not blinded to treatment, whereas evaluating physicians and radiologists were blinded.
The primary endpoint of EVIDENCE was the proportion of relapse-free patients at 24 weeks. A total of 75% of patients in the SC IFNβ-1a group were relapse free at 24 weeks compared with 63% of patients in the IM IFNβ-1a group (odds ratio, 1.9; 95% confidence interval, 1.3–2.6; p = 0.0005). Results at 24 weeks also showed a lower mean number of combined unique (CU) active lesions in the SC IFNβ-1a group (0.8) than in the IM IFNβ-1a group (1.2). However, the mean number of CU active lesions at baseline also was numerically lower in the SC IFNβ-1a group than in the IM IFNβ-1a group (2.4 vs. 2.9). Also, MRI outcomes for T1 and T2 lesions favored SC IFNβ-1a [Panitch et al. 2002].
Although the study duration applied to the primary endpoint was only 24 weeks, the comparative phase of EVIDENCE lasted 48 weeks. Final results of the comparative phase (with an average study participation of 64 weeks) showed that the difference in the percentage of relapse-free patients had decreased but was still significant between SC IFNβ-1a (56%) and IM IFNβ-1a (48%, p = 0.023) [Panitch et al. 2005]. NAbs (titer ≥20) were more frequent in the SC IFNβ-1a group than in the IM IFNβ-1a group (25% vs. 2%, respectively; p < 0.001), and presence of NAbs were associated with increased MRI disease activity on some measures at 48 weeks in SC IFNβ-1a-treated patients. The percentage of relapse-free patients in EVIDENCE was also higher than the percentage reported in the pivotal phase III PRISMS study [PRISMS Study Group, 1998] suggesting differences in baseline disease activity. The corresponding relapse-free data for IM IFNβ-1a were nearly identical between EVIDENCE (63%) and the pivotal phase III study of IM IFNβ-1a (60%) [Jacobs et al. 1996].
In summary, the results of EVIDENCE suggest a dose-dependent effect of IFNβ-1a therapy on short-term relapse and measures of MRI activity. The mid- to long-term implications of the EVIDENCE study remain unclear. No firm conclusions regarding potential differences between the 2 IFNβ-1a preparations on disease progression can be drawn.
Study 10
The Independent Comparisons of Interferon (INCOMIN) was a prospective, randomized, multicenter, 2-year study comparing the clinical and MRI benefits of IM IFNβ-1a (n = 92) and SC IFNβ-1b (n = 96) administered over a 2-year period in patients with RRMS [Durelli et al. 2002]. Investigators evaluated clinical outcome measures in an open-label manner, whereas blinded readers evaluated brain MRI scans. The proportion of relapse-free patients and of patients free from new T2 hyperintense lesions comprised the primary outcome measures.
For the primary endpoint, 51% of SC IFNβ-1b-treated patients and 36% of IM IFNβ-1a-treated patients were relapse free over 2 years (p = 0.03) [Durelli et al. 2002]. In the pivotal phase III study of IFNβ-1b conducted by the IFNB Multiple Sclerosis Study Group, 31% of SC IFNβ-1b-treated patients were relapse free after 2 years [IFNB Multiple Sclerosis Study Group, 1993] versus the 51% in INCOMIN [Durelli et al. 2002]. For IM IFNβ-1a, 38% of patients in the phase 3 study were relapse free [Jacobs et al. 1996] compared with the 36% in INCOMIN. The MRI results of INCOMIN corresponded to the clinical findings: 55% of SC IFNβ-1b-treated and 26% of IM IFNβ-1a-treated patients were free from new T2 lesions (p < 0.001) [Durelli et al. 2002].
With regard to methodology, clinicians in this study were unblinded, which would establish it as class III evidence. In addition, baseline characteristics such as more T2 lesions, a longer disease duration, and more advanced age at the time of MS diagnosis in the IM IFNβ-1a group compared with the SC IFNβ-1b group may have contributed to more positive outcomes for SC IFNβ-1b–treated patients.
Study 11
A prospective study by Khan and colleagues also suggested superiority of SC IFNβ-1b over once-weekly IM IFNβ-1a [Khan et al. 2001]. The study compared the effect of the two interferons and also GA on relapse rates in 156 patients. In the IFN-treated patients, the mean annualized relapse rate in previously treatment-naïve patients decreased in SC IFNβ-1b-treated patients (0.55, p = 0.001) but not in IM IFNβ-1a-treated patients (0.81, p = 0.106) 18 months after treatment initiation. Owing to the open-label, unblinded design of this study, it qualifies as class III evidence.
Overall, the findings from comparative studies have been inconclusive regarding potential efficacy differences among first-line IFNβ therapies. Whereas large retrospective studies including QUASIMS suggest no major differences in efficacy among these therapies, some prospective open-label studies suggest differences in efficacy between SC IFNβ-1b or SC IFNβ-1a and IM IFNβ-1a.
Dose comparison trials: does dose matter?
The optimal dose of IFNβ is pertinent to improve outcomes in patients with a lifelong disease such as RRMS. Three controlled studies represent class I evidence regarding the dose–response relationship for the commercially available IFNβ formulations.
In the European IFNβ-1a Dose Comparison Study, 802 patients with relapsing MS were treated with IM IFNβ-1a 30 µg or 60 µg once weekly for 36 months [Clanet et al. 2002]. No significant dose-related differences were seen on measures of sustained disability progression, change in EDSS score, or relapse rate. An extension (up to year 4) of the same study also demonstrated similar efficacy between the two doses [Clanet et al. 2004].
Contrasting results were reported in the randomized, double-blind, placebo-controlled PRISMS study, which compared the two doses of SC IFNβ-1a (22 µg vs. 44 µg). PRISMS demonstrated a trend toward more favorable outcomes in patients treated with the higher dose compared with the lower dose in all endpoints studied [PRISMS Study Group, 1998]. The dose effect favoring the 44 µg formulation over the 22-µg formulation was also apparent in a subsequent 4-year extension study (risk ratio, 0.88; p = 0.069), but it did not quite reach statistical significance [PRISMS Study Group and University of British Columbia MS/MRI Analysis Group, 2001].
The placebo-crossover design of the PRISMS study allowed for a follow-up prospective study that compared pretreatment data from years 1 and 2 with posttreatment data from years 3 and 4 in patients who changed from placebo to active treatment at year 2 [Oger et al. 2005]. Patients in both the 22 µg (n = 85) and 44 µg (n = 87) groups had a significant decrease in annualized relapse rate, disease progression, and number of T2 active lesions after switching to active treatment in years 3 and 4 [Oger et al. 2005]. No significant differences were observed between the two doses in any of these measures [Oger et al. 2005].
For IFNβ-1b, the pivotal study demonstrated superior efficacy of the dose that is currently licensed (Betaseron® 250 µg every other day) over a lower dose [IFNB Multiple Sclerosis Study Group, 1993]. A more recent study assessed whether a further twofold dose increase would have an acceptable tolerability profile and might provide additional benefits. Two formulations of IFNβ-1b were evaluated in a multicenter, randomized, double-blind, dose-comparison study [Hurwitz et al. 2008]. Patients received either 250 or 500 µg of SC IFNβ-1b for ≥12 weeks. The primary endpoint was percentage of patients experiencing adverse events. MRI evaluation was included to monitor for new disease activity. A dose-response effect associated with the 500-µg dose was detected in the more common adverse events, and no differences in MRI outcomes were observed.
Results of the Betaferon Efficacy Yielding Outcomes of a New Dose (BEYOND) study compared the efficacy and tolerability of a 250 µg dose of SC IFNβ-1b with a 500 µg dose in a randomized study of patients with RRMS [O’Connor et al. 2009] who met the diagnostic criteria for MS recommended in 2001 [McDonald et al. 2001]. BEYOND was a prospective, multicenter study that included 2244 patients assigned to 250 or 500 µg of SC IFNβ-1b taken every other day, or 20 mg of SC GA taken daily.
No additional benefit was observed in patients treated with the 500 µg dose of SC IFNβ-1b compared with patients receiving the 250 µg dose with respect to the primary endpoint, relapse risk [O’Connor et al. 2009], or for any of the secondary outcomes, including EDSS progression, change in T1 hypointense lesion volume, or change in normalized brain volume. Tolerability profiles were generally similar between the treatment groups, although flu-like symptoms were significantly more common in patients taking SC IFNβ-1b compared with patients on GA (p < 0.0001), and injection-site reactions were more common in patients on GA compared with those on SC IFNβ-1a (p = 0.0005) Taken together, these data demonstrate a dose ceiling effect for IFNβ-1b therapy in RRMS and suggest the efficacy of both doses of SC IFNβ-1b is similar to that of GA.
Combination therapy with IFNβ
The use of two or more therapeutic interventions with different mechanisms of action underlies the rationale for combination therapy in any disease state. To date, few studies have evaluated combination therapy with IFNβ and agents from a different therapeutic class in patients with RRMS.
The Avonex Combination Trial (ACT) was a randomized, placebo-controlled evaluation of the efficacy and safety of the combination of IM IFNβ-1a and methotrexate in patients with RRMS [Cohen et al. 2008] who met the 2001 diagnostic criteria [McDonald et al. 2001]. ACT was designed originally as a 2-year study that would enroll 900 patients. However, because of slow patient recruitment, the study was redesigned with a smaller enrollment target and shorter duration of follow up. In the redesigned study, 313 eligible patients entered the study on stable doses of IM IFNβ-1a (30 µg once weekly) and were randomized to a 12-month course of adjunctive therapy with placebo (group 1), oral methotrexate 20 mg once weekly (group 2), intravenous methylprednisolone 1000 mg/day for 3 consecutive days every other month (group 3), or oral methotrexate and intravenous methylprednisolone at the same doses as in groups 2 and 3 (group 4) [Cohen et al. 2009]. The primary endpoint was new or enlarged T2-hyperintense lesions, and secondary endpoints were EDSS scores, Multiple Sclerosis Functional Composite (MSFC) scores, and relapses. Results from the redesigned study did not suggest any significant benefit for either combination therapy; however, there was a reduction in neutralizing antibody titers with methylprednisolone.
Also using the 2001 diagnostic criteria for MS, the Nordic trial of oral methylprednisolone as add-on therapy to IFNβ-1a for treatment of RRMS (NORMIMS) was a randomized, placebo-controlled study that evaluated the efficacy of oral methylprednisolone combined with SC IFNβ-1a in 170 Scandinavian patients who were refractory to the higher (44 µg) dose of SC IFNβ-1a [Sorensen et al. 2009]. A significant reduction in annual relapse rate (62%) was reported for patients on oral methylprednisolone and SC IFNβ-1a compared with patients on SC IFNβ-1a plus placebo. Despite the more favorable outcomes for patients on combination therapy, the study had a high rate of discontinuation. A total of 26% of patients on methylprednisolone and 17% of patients on placebo discontinued by the end of the study. The high rate of discontinuation combined with the small number of patients is a notable limitation of the study, and further evaluation in larger populations is required to confirm the efficacy of the addition of oral methylprednisolone to SC IFNβ-1a. Similar results were reported in another recent randomized, double-blind 3-year study (n = 341) in patients treated with IM IFNβ-1a combined with oral methylprednisolone [Ravnborg et al. 2009]. Patients who received methylprednisolone/IM IFNβ-1a had a significantly reduced relapse rate and significantly better MRI outcomes compared with patients receiving IM IFNβ-1a/placebo. Favorable outcomes also were reported for the combination group on the MSFC and MS Impairment Scale. As in the NORMIMS study, a large proportion of patients receiving combination therapy discontinued treatment in the first year of the study due to adverse events.
The combination of IFNβ-1a and the immunosuppressant azathioprine has been studied in several small, open-label studies that suggested stabilization on MRI or improvement in neurological disability [Pulicken et al. 2005]. In a larger, double-blind, placebo-controlled study, 181 patients were randomized to IM IFNβ-1a monotherapy, IM IFNβ-1a plus azathioprine or IM IFNβ-1a plus azathioprine and prednisone [Havrdova et al. 2009]. Patients were treated under double-blind conditions for 2 years, with the option of a 3-year, open-label extension phase. No differences were observed at 2 or 5 years between the monotherapy and combination therapy groups in annualized relapse rate, mean EDSS scores, or cumulative probability of sustained disability progression [Havrdova et al. 2009]. The percentage change in T2 lesion volume at 2 years was lower for patients receiving IFNβ-1a plus azathioprine and prednisone compared with monotherapy (+14.5% vs. +30.3%, p = 0.015). Median change from baseline in T2 lesion volume at 2 years was also lower for triple combination therapy compared with IFNβ-1a and azathioprine (+585.0 mm3 vs. +2011.5 mm3, p < 0.05) and IFNβ-1a alone (+585.0 mm3 vs. +1532.5 mm3, p < 0.01) [Havrdova et al. 2009]. These changes, however, were not sustained over the 3-year extension period, suggesting that this combination offers no additional benefit over IFNβ-1a alone [Havrdova et al. 2009].
The addition of statins to IFNβ-1b therapy seemed promising based on experimental data showing that atorvastatin, an HMG-CoA reductase inhibitor used to lower cholesterol, reduced the rate and severity of relapses in an animal model of MS, alone and in combination with other MS therapies [Stuve et al. 2006]. However, results from two clinical studies did not show any additional benefit from combining statins with IFNβ. Birnbaum and colleagues conducted a small, randomized, placebo-controlled study to test this hypothesis in patients with RRMS [Birnbaum et al. 2008]. A total of 26 patients who were on SC IFNβ-1a therapy (44 µg three times weekly) were randomized to receive combination therapy with atorvastatin, either 40 mg or 80 mg, or placebo daily for 6 months. In contrast to expectations, relapses and disease activity on MRI were more common among patients treated with atorvastatin (10/17 patients; 59%) than placebo (1/9 patients; 11%) (p = 0.22). Relapses occurred in some patients who had been stable for years, and some relapses required treatment with glucocorticoids. Based on these findings, the authors conclude that caution and careful monitoring are needed when patients with MS are treated with high-dose IFNβ and high-dose statins. The small cohort size and nonsignificant statistical findings make this trial difficult to interpret. Results from a double-blind, randomized, placebo-controlled study adding another statin, simvastatin, to IFNβ-1a therapy for patients with treatment-naïve RRMS revealed no additional benefit with the combination, and perhaps even negative effects of statins on IFNβ-1a activity [Sorensen et al. 2010].
The randomized, placebo-controlled, phase III Safety and Efficacy of Natalizumab in Combination with Interferon Beta-1a in Patients with Relapsing Remitting Multiple Sclerosis (SENTINEL) study assessed the efficacy of IM IFNβ-1a in combination with natalizumab in patients (n = 1171) with RRMS who experienced at least one relapse while on IFN therapy. Compared with IM IFNβ-1a/placebo, patients receiving IM IFNβ-1a/natalizumab demonstrated a greater reduction in the risk of sustained disability progression, a lower annualized relapse rate, and improved MRI outcomes. However, two cases of progressive multifocal leukoencephalopathy (PML), one of them fatal, were reported in the combination therapy group [Rudick et al. 2006]. Natalizumab is indicated as monotherapy for the treatment of patients with relapsing forms of multiple sclerosis to delay the accumulation of physical disability and reduce the frequency of clinical exacerbations.
Data from a small study (n = 55) involving patients with aggressive RRMS (≥2 relapses with incomplete recovery in prior year and Gd+ lesions on MRI) showed that 6 months of mitoxantrone induction therapy followed by IFNβ-1b resulted in a significantly longer time to disability progression (i.e. a ≥1-point deterioration in EDSS score) compared with IFNβ-1b/methylprednisolone [LePage and Edan, 2009].
In addition, the combination of IFNβ and GA, both first-line therapies, is being evaluated to improve efficacy in patients with MS. The putative principal mechanism of action of IFNβ (nonselective blockade of T-cell migration into the central nervous system) and GA (increased production of anti-inflammatory TH2 cells that migrate to the central nervous system) are not overlapping, and might have potential synergistic or complementary additive effects when combined. Results from studies in animal models of MS in conjunction with somewhat equivocal results from a small observational study suggest that the combination of IFNβ and GA could be investigated further [Gold, 2008; Ytterberg et al. 2007]. In a study using peripheral blood lymphocytes from patients with MS, Zang and colleagues observed potentially antagonistic effects on the regulatory function of GA (previously referred to as copolymer 1 [COP-1]) and IFNβ-1a [Zang et al. 2003]. Based on this preclinical evidence, a multicenter, randomized, phase III National Institutes of Health (NIH)-funded study (COMBI-RX) is evaluating the combination of GA and IM IFNβ-1a versus either agent alone in patients with RRMS in the United States [Gold, 2008].
Overall, current evidence from clinical studies is insufficient to support the broad use of combination therapies for long-term therapy of RRMS patients. Additional, well-designed studies are needed to identify combination treatment strategies that not only provide significantly greater long-term benefits than first-line monotherapies but also are associated with favorable tolerability and minimal additional risk.
IFN safety and tolerability
Common adverse events associated with interferon beta (IFNβ) therapies in patients with multiple sclerosis (MS). (Adapted with permission from Moses and Brandes [2008]).
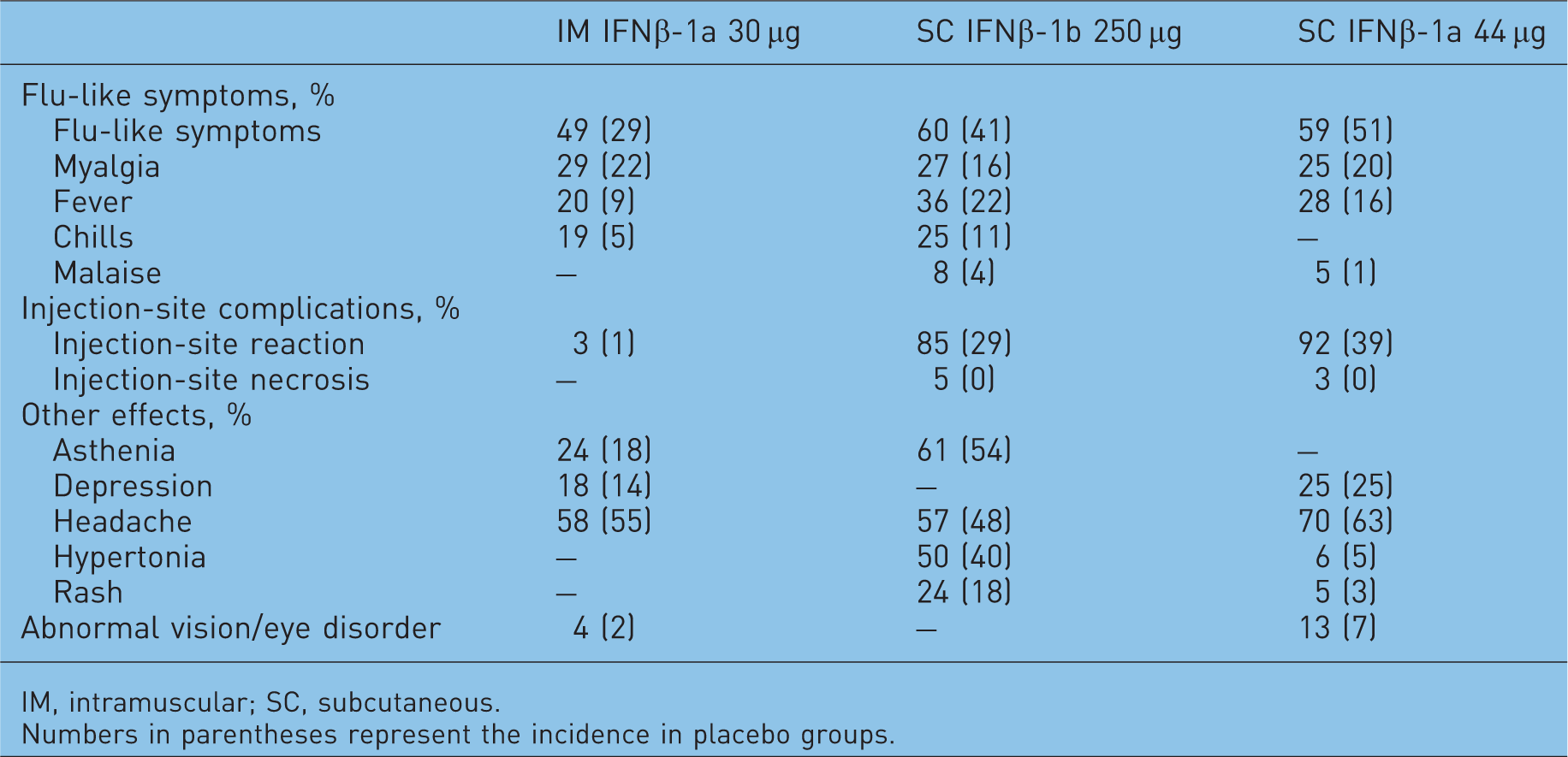
IM, intramuscular; SC, subcutaneous.
Numbers in parentheses represent the incidence in placebo groups.
Although it has been suggested that a higher incidence of depression occurs among patients taking IFNs, results from all of the phase III studies, along with QUASIMS and other studies, did not report a definitive association between patients taking IFN and the development or worsening of depressive symptoms [Moses and Brandes, 2008; Limmroth et al. 2007; PRISMS Study Group, 1998; Jacobs et al. 1996; IFNB Multiple Sclerosis Study Group and The University of British Columbia MS/MRI Analysis Group, 1995]. Depression, however, is more common among patients with MS than among those with other chronic illnesses and more common than in the general population [Patten et al. 2003]. As a result, patients with MS should be routinely monitored for signs of depression regardless of therapy [Moses and Brandes, 2008].
Since MS can develop during a woman’s childbearing years, it is important to understand whether use of IFN therapy can impact pregnancy outcomes. A retrospective longitudinal cohort study comparing pregnancy outcomes in patients on IM IFNβ-1a, SC IFNβ-1a, or IFNβ-1b therapy versus healthy controls reported higher rates of miscarriage, stillbirths, and lower mean birth weight in women who became pregnant while on IFN therapy [Boskovic et al. 2005]. In contrast, one retrospective study on pregnancy outcomes in patients participating in clinical studies of SC IFNβ-1a and IM IFNβ-1a suggested that the rate of spontaneous pregnancy loss in women on therapy at the time of conception, or who discontinued within 2 weeks prior to conception, was similar to that of the general population [Sandberg-Wollheim et al. 2005]. A recent report from the AVONEX Pregnancy Exposure Registry of women with MS who were exposed to IM IFNβ-1a within 1 week of conception or in their first trimester did not suggest any evidence of increased rate of miscarriages or birth defects [Foulds et al. 2010].
Conclusion
The postmarketing experience with the IFNs as documented in the literature, mostly as comparator open-label studies, demonstrates a significant reduction of disease activity in the treatment of RRMS with all currently available IFNβ therapies. The effects seen in the majority of the studies were comparable to the results of the pivotal studies. Most of these studies (all class III/IV evidence) suggested that there are no significant differences in efficacy among the three available IFNβ preparations.
Aside from drawbacks in methodology, the primary criticisms for all of the trials discussed here include short treatment duration, unblinded treatment regimens, and some imbalances in demographic parameters or baseline disease activity between the treatment groups. Potential selection bias resulting from unbalanced demographics, in particular, can be problematic when interpreting results. This was demonstrated in a study published by Sørensen and colleagues that used the Danish nationwide database (which contains data all patients with MS receiving immunomodulatory therapy, including all IFN preparations and GA) to evaluate effects of immunomodulatory therapy use in patients with RRMS [Sørensen et al. 2006]. While the study applied Cox regression analyses to account for differences in baseline characteristics, the authors concluded any differences in therapeutic effect were likely due to selection bias rather than to differences in the preparations themselves. The study also provided evidence that large, open-label studies are unsuitable for evaluating comparison of treatment effects between different immunomodulatory therapies.
One of the challenges inherent in comparing these many studies is that the variation in specific endpoints can limit the ability to make comparisons across studies. Furthermore, because the comparative IFNβ preparations are delivered by different routes, doses, and frequencies, none of these studies can definitively address whether any differences in efficacy are related to these different variables. Although documenting the actual relapse rates of the baseline and posttreatment groups for each of these studies was not practical for this review, it must be noted that, although the effect was similar in these reports to that of the phase III trials, the phase IV study participants typically have had larger reductions in relapse rates in every group, often double the effect seen in the original studies, for reasons which have not yet been definitively established. Entry criteria, selection bias, and ascertainment bias in evaluating and documenting sentinel events may have caused our process of studying these cohorts to ‘evolve’ over the years. It is important to note that MS trial design and execution is largely based on the pivotal clinical studies of the three IFN preparations on the market, and in many ways, the MS research community has drawn many of its practices from them.
Future design of clinical trials for patients with RRMS should capitalize on increased understanding of the pathogenesis and molecular basis of the disease. More robust surrogate markers of disease activity may be available in the near future. MRI-based technology examining volumetric changes in tissue damage and subtle inflammation, the amount and location of cortical disease, tract losses, and spinal disease in all of its aspects, may force a re-evaluation of the clinical relevance of the endpoints used to establish the basis of IFN therapy. We must also recognize that the increasingly strong evidence for immunopathologic heterogeneity in MS, in spite of the phenotypic convergence as expressed in the clinical presentations, may establish better indicators of those patients who will respond to IFNs, and those who do not. Testing of IFN-induced gene expression using RNA or protein measures may soon help in establishing likely responders to treatment [Weinstock-Guttman et al. 2008]. These measures may also contribute to identification of confounding occult variables in all of our clinical trials and may force re-examination of the results, in light of imbalances of treatment groups that they reveal.
Although a need exists for well-designed, long-term studies to answer the question of the absolute and relative efficacy of IFNβ drugs in the treatment of RRMS, it is likely that the expenditures required for such studies will be reasonably applied to the development of new therapies. It is clear that issues such as NAbs, safety, tolerability, and efficacy (or lack thereof) in combination with agents having different mechanisms of action will be critical determinants of the continued first-line role of IFNβ in the MS armamentarium.
A new oral preparation (fingolimod) has been approved recently and other oral preparations are currently being evaluated in phase III studies (e.g. cladribine, teriflunomide, BG-12, laquinomod, and firategrast). In addition, parenteral drugs with innovative therapeutic targets (such as rituximab, ocrelizumab, alemtuzumab, immune ablation/stem cell reconstitution regimens, and various vaccine strategies) are becoming promising alternatives to IFNβ therapy. Neural and glial reconstitution and regeneration strategies, as they are tested, will increasingly challenge all of these interventions to demonstrate positive synergistic effects with them on repair of the damaged nervous system.
As these potential new therapeutics become available, there will be considerable advantage to establishing risk–benefit for individuals across the disability spectrum with respect to treatment decisions at critical points in their disease course. This will necessarily lead to a transition from protocols that focus only on monotherapies used until patients become refractory to combination treatment plans. These combinations could exploit therapies with different mechanisms of action and/or toxicity and utilize models such as pulse+maintenance, sequential regimens, and simultaneous positive synergy regimens. The mechanics of designing and performing such trials, especially the decision on whether to re-treat using drugs with high potential for toxicity, have not yet been established. In any case, it is likely that IFNs will continue to play an important role as the platform therapy for patients with MS.
Footnotes
Acknowledgments
The authors acknowledge Sabrina Maurer and Matthew Hasson, Scientific Connexions, Newtown, PA, USA, for editorial and technical assistance in preparing this manuscript for submission. This support was funded by Biogen Idec.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
V. Limmroth has received research funding, compensation, travel grants, and/or honoraria from Almirall, Bayer-Schering, Biogen Idec, EMD Serono, GSK, Merck, Novartis, Sanofi-Aventis, and Teva Neuroscience. N. Putzki has received research funding, compensation, travel grants, and/or honoraria from Allergan, Almirall, Bayer Schering, Biogen Idec, GlaxoSmithKline, Ipsen, Merck-Serono, Novartis, Sanofi-Aventis, Teva Neuroscience, and Wyeth and is now and employee of Biogen Idec. M. Bertelmann declares that she has no conflicts of interest. N.J. Kachuck has received research funding, compensation, travel grants, and/or honoraria from Bayer-Schering, Bayhill Pharmaceuticals, Biogen Idec, EMD Serono, the National Multiple Sclerosis Society, Pepgen, and Teva Neuroscience.
