Abstract
Neurostimulation techniques for the treatment of primary headache syndromes, particularly of chronic cluster headache, have received much interest in recent years. Occipital nerve stimulation (ONS) has yielded favourable clinical results and, despite the limited numbers of published cases, is becoming a routine treatment for refractory chronic cluster headache in specialized centres. Meanwhile, other promising techniques such as spinal cord stimulation (SCS) or sphenopalate ganglion stimulation have emerged. In this article the current state of clinical research for neurostimulation techniques for chronic cluster headache is reviewed.
Keywords
Cluster headache: definition, clinical characteristics and standard therapy
Cluster headache (CH) is an extremely severe and debilitating trigemino-autonomic primary headache syndrome. Clinically CH is characterized by attacks of severe pain localized orbitally, supraorbitally or temporally, lasting for 15–180 minutes and occurring from once every other day to eight times daily. The attacks are associated with one or more of the following symptoms: conjunctival injection, lacrimation, nasal congestion, rhinorrhea, forehead and facial sweating, miosis, ptosis and eyelid oedema [Silberstein et al. 2005]. Attacks occur in series (so-called cluster periods or bouts) that are usually separated by remission periods lasting for months or years. In chronic cluster headache (CCH) substantial remission periods are missing. Cluster headache has a 1-year prevalence of about 0.1% [Sjaastad and Bakketeig, 2003] and about 10% of this group have chronic cluster headache [Lance and Goadsby, 2005].
Usually treatment is based on drug therapy. Agents used for acute therapy are inhalation of high-flow oxygen, sumatriptan subcutaneous injection and zolmitriptan nasal spray. For transitional or short-term prophylaxis, corticosteroids and ergotamine derivatives are used. The cornerstone of maintenance prophylaxis is verapamil, but lithium and methysergide may also be used. Some patients respond to melatonin and topiramate [Dodick and Capobianco, 2001].
In addition, under optimal medical therapy a number of patients do not experience sufficient pain relief [Silberstein et al. 2005].
Recently, guidelines for the definition of therapy refractory CCH have been proposed [Goadsby et al. 2006], but the exact proportion of these patients is still unknown. However, the number of published lesional and surgical approaches for CCH points towards the considerable size of the problem.
Lesional treatments
A variety of lesional treatments such as glycerol injection [Ekbom et al. 1987], radiofrequency lesioning [Sweet, 1988; Mathew and Hurt, 1988] or gamma knife lesioning of the trigeminal nerve [Donnet et al. 2005, 2006; McClelland et al. 2006, 2007], trigeminal tractotomy [Green, 2003], trigeminal sensory nerve section [Jarrar et al. 2003; Kirkpatrick et al. 1993], surgical section [Rowed, 1990] or radiofrequency [Sweet, 1988] of the nervus intermedius, combinations of nerve sections [Morgenlander and Wilkins, 1990], decompression of the facial nerve [Solomon and Apfelbaum, 1986] and endoscopic block [Felisati et al. 2006], radiofrequency treatment [Sanders and Zuurmond, 1997] or gamma knife lesioning [Lad et al. 2007] of the sphenopalatine ganglion have been proposed.
In a study of microvascular decompression of the trigeminal nerve, alone or in combination with section and/or microvascular decompression of the nervus intermedius, the short-term success rate (defined as 50% relief or greater) was 73% but dropped to 46% in long-term follow up. Initially, half of the patients had a relief of more than 90%.
Risks associated with these surgical procedures include death, permanent neurological deficits, corneal anaesthesia and visual loss. Some of these procedures require a craniotomy. Also, a switch of the CCH to other side in the further course is possible.
Deep brain stimulation
The results of posteromedial hypothalamotomy [Sano et al. 1970] and the identification of a hypothalamic activation during cluster attacks [May et al. 1998; Sprenger et al. 2004] led to the use of deep brain stimulation (DBS) for refractory CCH. A first series of 16 patients showed excellent results with 13 patients pain free or nearly pain free and three patients improved [Franzini et al. 2003; Leone, 2006]. Later studies followed consensus criteria for patient selection [Leone et al. 2004]. In 2008 a review summarized the results of hypothalamic DBS in 38 refractory CCH patients. With a follow up of between 1 and 4 years, 23 patients (61%) were pain free or almost pain free [Leone et al. 2008].
Schoenen and colleagues reported a fatal haemorrhage following DBS implantation due to a previously unnoted cerebral aneurysm. Moreover, in their series of six patients, in another patient the procedure had to be stopped due to panic attacks with autonomic disturbances [Schoenen et al. 2005].
In DBS pain relief can emerge with a delay of up to 3 months. In a prospective, randomized crossover study of 11 patients receiving DBS electrodes, no difference between active and sham stimulation was observed during the blinded crossover phase, however in the open phase six of 11 patients responded to stimulation (decrease in weekly attack frequency of >50%) [Fontaine et al. 2010].
Recently it has been questioned whether stereotactic intervention in these disorders has been targeted at the appropriate locus, and whether this may account for the approximately 40% of patients with a poor response to DBS [Matharu and Zrinzo, 2011], since the target data for DBS are derived from positron emission tomography (PET) studies with limited spatial resolution and functional magnetic resonance imaging (fMRI) data which have a better spatial resolution hint at a locus of activation antero-superior to that derived from PET studies [Matharu and Zrinzo, 2010].
Occipital nerve stimulation
Occipital nerve stimulation (ONS) had been proposed as a treatment for refractory migraine [Matharu et al. 2004; Popeney and Alo, 2003], occipital neuralgia [Johnstone and Sundaraj, 2006; Kapural et al. 2005; Slavin et al. 2006] and other intractable headache disorders [Weiner and Reed, 1999].
Also the use of a small unilateral completely implantable miniaturized device known as ‘bion’ has yielded favourable results in patients with hemicrania continua [Burns et al. 2008].
The role of occipital stimulation in CH was first examined by Burns and colleagues. They published a pilot study of 8 patients [Burns et al. 2007] and a follow-up study of 14 patients [Burns et al. 2009] on ONS for CCH. In the pilot study 6 of 8 patients and in the follow-up study 10 of 14 patients experienced a reduction in attack frequency. The reduction of attack frequency was more than 40% in 6 of 8 patients in the pilot study and six of 14 patients in the follow-up study. In a study by Magis and colleagues seven of eight patients had a decrease in attack frequency of more than 40% [Magis et al. 2007]. Mean attack frequency was decreased by 19%, 29% and 80% in the two studies of Burns and colleagues and the study of Magis coworkers, respectively. In the latter study, on average the ONS to baseline attack ratio per month was 0.65 during the whole follow up (mean 15.1 months).
Spinal cord stimulation
Spinal cord stimulation (SCS) has for a long time been successfully employed in a variety of neuropathic pain syndromes as well as in vasogenic pain such as angina pectoris or peripheral vascular disease. There are recent reports on stimulation of the craniocervical junction for several types of head and facial pain [Balzer et al. 2011; Tomycz et al. 2011]. The latter may be regarded as a kind of very high cervical SCS.
Seven years ago we treated a patient with medication refractory cluster headache. We decided to perform a test of cervical SCS electrode as compassionate treatment. The clinical results in this patient in the postoperative course and in the long-term follow up were quite encouraging [Wolter et al. 2008] and we decided to offer this treatment option to other patients with refractory CCH. By now we have implanted eight patients and the clinical results of the first seven patients were published recently [Wolter et al. 2011]. All in all, SCS in our study showed clinical effects comparable to or better than ONS (Table 1). The promising clinical results, however, were shadowed by a high rate of lead breakages and dislocations. These complications, however, were benign and easy to handle.
Overview of results from different neurostimulation studies for chronic cluster headache.
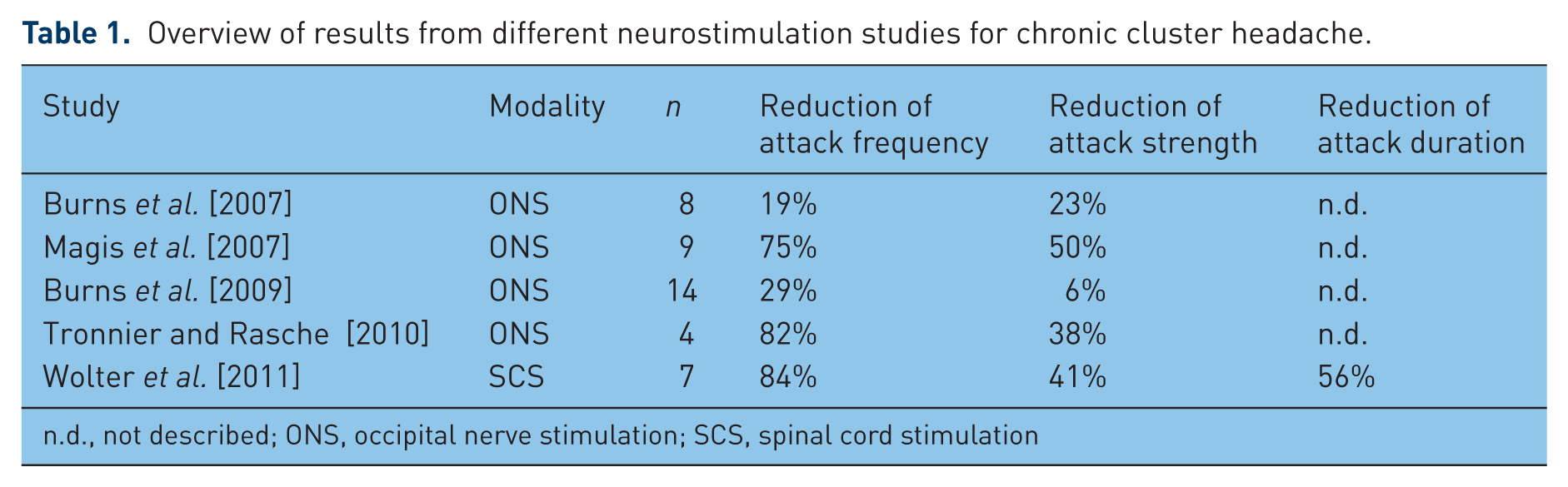
n.d., not described; ONS, occipital nerve stimulation; SCS, spinal cord stimulation
SCS in contrast to ONS acts immediately. All of our patients felt at least some effect from SCS from the operation day onward. This allows for a test phase, prior to implantation of the impulse generator. Electrodes can also be implanted bilaterally, in the case of side switch of CCH. In two of our patients this was the case: both received a second contralateral electrode, and now they are able to control head pain on both sides separately.
Although being slightly more invasive than ONS, in SCS the risks of the intervention are minimal. In our view it is still too early to decide whether SCS might become a first-line treatment for therapy refractory CCH. However, it can be used as a reserve option in the case of insufficient effects of ONS.
Ganglion sphenopalatinum stimulation
The sphenopalatine (pterygopalatine) ganglion (SPG) receives input from the maxillary branch of the trigeminal nerve, parasympathetic fibres originating from the superior salivatory nucleus in the brainstem and sympathetic fibres form the carotid plexus (via the deep petrosal nerve). In the SPG there is a tight anatomical and physiological relationship of sympathetic, parasympathetic and trigeminal fibres. The SPG plays a pivotal role in driving the parasympathetic features and in sterile meningeal inflammation as substrate for trigeminally mediated head pain, as well as in pain transmission in CH. Consequently the SPG has been targeted in a couple of lesional treatments [Felisati et al. 2006; Lad et al. 2007; Sanders and Zuurmond, 1997; Solomon and Apfelbaum, 1986]. Recently a case of long-term neurostimulation of the SPG has been reported with excellent and long-lasting pain relief [Ibarra, 2007]. Moreover, a new study showed that stimulation of the SPG can be effective as an acute treatment for cluster attacks. In six patients, 18 attacks were treated and a complete resolution of symptoms was seen in 11 attacks, a partial resolution of symptoms in three attacks and minimal or no relief in four attacks [Ansarinia et al. 2010]. Similar results have been reported after SPG stimulation as acute treatment for migraine [Tepper et al. 2009]. SPG stimulation becomes particularly attractive, because it may, in contrast to ONS and SCS, offer the possibility to abort an ongoing attack. Therefore, future studies of stimulation of the SPG are urgently warranted.
Recommendations
Neurostimulation has opened promising perspectives for the treatment of refractory CCH. ONS and SCS seem to have a stronger pain relieving effect than DBS. Therefore, apart from the inherent risks of DBS, this option should not be performed as a first-line treatment for refractory CCH. To the best of the authors’ knowledge, there are no more ongoing trials of DBS for CCH in Germany. Whether SCS should be used as a first-line therapy or as a reserve therapy cannot be decided on the basis of the present data.
SPG stimulation might become an attractive alternative to ONS and SCS but on the basis of the present data it cannot yet be recommended, except in the context of a clinical study.
Future perspectives
Further studies will show whether there is a place for SPG stimulation in the future. Irrespective from the final lead location, in the future new programming parameters (i.e. burst stimulation [De Ridder et al. 2010] ) or high-frequency stimulation may deliver improved pain relief in our patients.
Footnotes
T. Wolter has received a single travel grant from Boston Scientific in 2010.
H. Kaube was member of the advisory board of MSD. He received honoraria for lectures from Boehringer, Pfizer, Medtronic, Boston Scientific and ANS St Jude.
